Abstract
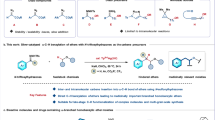
Carbon–hydrogen bond functionalization methods are important tools for efficiently upgrading simple precursors to more valuable compounds. The ubiquity of amines in pharmaceuticals and natural products has led to considerable interest in strategies for the selective amidation of C–H bonds in a tunable manner. An ongoing challenge involves achieving control in situations where targeted bonds have varying bond strengths or similar steric/electronic environments. Herein, we report two complementary silver catalysts that are capable of selecting between β- or γ-C–H bonds that reside in similar steric/electronic environments, overriding a reaction at a weaker C–H bond in favour of a stronger one and activating primary C–H bonds. The mild conditions, low cost of silver, good yields and easy purification make this approach ideal for late-stage functionalizations to furnish valuable 1,2- and 1,3-aminoalcohols from easily prepared carbamate esters.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data supporting the findings of this study are available within the manuscript and its Supplementary Information, or available from the author on reasonable request. Experimental details and full spectroscopic characterization data for all new compounds and copies of proton and carbon NMR spectra are provided in the Supplementary Information. The X-ray crystallographic coordinates for [(Py5Me2)AgOTf]2 in this study have previously been reported and have been deposited at the Cambridge Crystallographic Data Centre under deposition no. 1491194. These data can be obtained free of charge from the Cambridge Crystallographic Data Centre via www.ccdc.cam.ac.uk/data_request/cif. Furthermore, the corresponding author can be contacted directly.
References
Krise, J. P. & Oliyai, R. in Biotechnology: Pharmaceutical Aspects (eds Stella, V. J. et al.) Vol. 5 (Springer, 2007).
Klingler, F. D. Asymmetric hydrogenation of prochiral amino ketones to amino alcohols for pharmaceutical use. Acc. Chem. Res. 40, 1367–1376 (2007).
Gallou, I. & Senanayake, C. H. Cis-1-amino-2-indanol in drug design and applications to asymmetric processes. Chem. Rev. 106, 2843–2874 (2006).
Corey, E. J. & Zhang, F. re- and si-Face-selective nitroaldol reactions catalyzed by a rigid chiral quaternary ammonium salt: a highly stereoselective synthesis of the HIV protease inhibitor Amprenavir (Vertex 478). Angew. Chem. Int. Ed. 38, 1931–1934 (1999).
O’Brien, P. Sharpless asymmetric aminohydroxylation: scope, limitations, and use in synthesis. Angew. Chem. Int. Ed. 38, 326–329 (1999).
McGrath, N., Brichacek, M. & Njardarson, J. T. A graphical journey of innovative organic architectures that have improved our lives. J. Chem. Ed. 87, 1348–1349 (2010).
Hong, S. Y. et al. Selective formation of γ-lactams via C–H amidation enabled by tailored iridium catalysts. Science 359, 1016–1021 (2018).
Roizen, J. L., Harvey, M. E. & Du Bois, J. Metal-catalyzed nitrogen-atom transfer methods for the oxidation of aliphatic C–H bonds. Acc. Chem. Res. 45, 911–922 (2012).
Alderson, J. M., Corbin, J. R. & Schomaker, J. M. Tunable, chemo- and site- selective nitrene transfer reactions through the rational design of silver(i) catalysts. Acc. Chem. Res. 50, 2147–2158 (2017).
Che, C.-M., Le, V. K.-Y. & Zhou, C.-Y. in Comprehensive Organic Synthesis (eds Knochel, P. & Molander, G. A.) 2nd edn (Elsevier, 2014).
Lu, H. & Xhang, X. P. Catalytic C–H functionalization by metalloporphyrins: recent developments and future directions. Chem. Soc. Rev. 40, 1899–1909 (2011).
Díaz-Requejo, M. M. & Pérez, P. J. Coinage metal catalyzed C–H bond functionalization of hydrocarbons. Chem. Rev. 108, 3379–3394 (2008).
Dequirez, G., Pons, V. & Dauban, P. Nitrene chemistry in organic synthesis: still in its infancy? Angew. Chem. Int. Ed. 51, 7384–7395 (2012).
Hazelard, D., Nocquet, P.-A. & Compain, P. Catalytic C–H amination at its limits: challenges and solutions. Org. Chem. Front. 4, 2500–2521 (2017).
Müller, P. & Fruit, C. Enantioselective catalytic aziridinations and asymmetric nitrene insertions into C–H bonds. Chem. Rev. 103, 2905–2920 (2003).
Brandenberg, O. F., Fasan, R. & Arnold, F. H. Exploiting and engineering hemoproteins for abiological carbene and nitrene transfer reactions. Curr. Opin. Biotech. 47, 102–111 (2017).
Espino, C. G. & Du Bois, J. A Rh-catalyzed C–H insertion reaction for the oxidative conversion of carbamates to oxazolidinones. Angew. Chem. Int. Ed. 40, 598–600 (2001).
Espino, C. G., Wehn, P. M., Chow, J. & Du Bois, J. Synthesis of 1,3-difunctionalized amine derivatives through selective C–H bond oxidation. J. Am. Chem. Soc. 123, 6935–6936 (2001).
Cui, Y. & He, C. Efficient aziridination of olefins catalyzed by a unique disilver(i) compound. J. Am. Chem. Soc. 125, 16202–16203 (2003).
Cui, Y. & He, C. A silver-catalyzed intramolecular amidation of saturated C–H bonds. Angew. Chem. Int. Ed. 43, 4210–4212 (2004).
Li, Z., Capretto, D. A., Rahaman, R. H. & He, C. Silver-catalyzed intermolecular amination of C–H groups. Angew. Chem. Int. Ed. 46, 5184–5186 (2007).
Rigoli, J. W. et al. Chemoselective allene aziridination via Ag(i) catalysis. Org. Lett. 15, 290–293 (2013).
Rigoli, J. W., Weatherly, C. D., Alderson, J. M., Vo, B. T. & Schomaker, J. M. Tunable, chemoselective amination via silver catalysis. J. Am. Chem. Soc. 135, 17238–17241 (2013).
Weatherly, C. D., Alderson, J. M., Berry, J. F., Hein, J. E. & Schomaker, J. M. Catalyst-controlled nitrene transfer by tuning metal:ligand ratios: insight into the mechanisms of chemoselectivity. Organometallics 36, 1649–1661 (2017).
Dolan, N. S., Scamp, R. J., Yang, T., Berry, J. F. & Schomaker, J. M. Catalyst-controlled and tunable, chemoselective silver-catalyzed intermolecular nitrene transfer: experimental and computational studies. J. Am. Chem. Soc. 138, 14658–14667 (2016).
Ju, M., Weatherly, C. D., Guzei, I. A. & Schomaker, J. M. Chemo- and enantioselective silver-catalyzed aziridinations. Angew. Chem. Int. Ed. 56, 9944–9948 (2017).
Scamp, R. J., Alderson, J. M., Phelps, A. M., Dolan, N. S. & Schomaker, J. M. Ligand-controlled, tunable silver-catalyzed C–H amination. J. Am. Chem. Soc. 136, 16720–16723 (2014).
Scamp, R. J., Jirak, J. G., Guzei, I. A. & Schomaker, J. M. General catalyst for site-selective C(sp 3)–H bond amination of activated secondary over tertiary alkyl C(sp 3)–H bonds. Org. Lett. 18, 3014–3017 (2016).
Huang, M., Yang, T., Paretsky, J., Berry, J. F. & Schomaker, J. M. Inverting steric effects: using ‘attractive’ non-covalent interactions to direct silver-catalyzed nitrene transfer. J. Am. Chem. Soc. 139, 17376–17386 (2017).
Mat Lani, A. S. & Schomaker, J. M. Site-selective, catalyst-controlled alkene aziridination. Synthesis 50, 4462–4470 (2018).
Alderson, J. M., Corbin, J. R. & Schomaker, J. M. Investigation of transition metal-catalyzed nitrene transfer reactions in water. Bioorg. Med. Chem. 26, 5270–5273 (2018).
Padwa, A., Flick, A. C., Leverett, C. A. & Stengel, T. Rhodium(ii)-catalyzed aziridination of allyl-substituted sulfonamides and carbamates. J. Org. Chem. 69, 6377–6386 (2004).
Hayes, C. J., Beavis, P. W. & Humphries, L. A. Rh(ii)-catalysed room temperature aziridination of homoallyl-carbamates. Chem. Commun. 4501–4502 (2006).
Maestre, L., Sameera, W. M. C., Díaz-Requejo, M. M., Maseras, F. & Pérez, P. J. A general mechanism for the copper- and silver-catalyzed olefin aziridination reactions: concomitant involvement of the singlet and triplet pathways. J. Am. Chem. Soc. 135, 1338–1348 (2013).
Fiori, K. W., Espino, C. G., Brodsky, B. H. & Du Bois, J. A mechanistic analysis of the Rh-catalyzed intramolecular C–H amination reaction. Tetrahedron 65, 3042–3051 (2009).
Huang, M. et al. Synthesis, characterization and VT-NMR studies of silver(i) complexes for selective nitrene transfer. Inorg. Chem. 56, 6725–6733 (2017).
Mak, C. L., Bostick, B. C., Yassin, N. M. & Campbell, M. G. Argentophilic interactions in solution: an EXAFS study of silver(i) nitrene transfer catalysts. Inorg. Chem. 57, 5720–5722 (2018).
Espino, C. G., Fiori, K. W., Kim, M. & Du Bois, J. Expanding the scope of C–H amination through catalyst design. J. Am. Chem. Soc. 126, 15378–15379 (2004).
Paradine, S. M. et al. A manganese catalyst for highly reactive yet chemoselective intramolecular C(sp 3)–H amination. Nat. Chem. 7, 987–994 (2015).
Harvey, M. E., Musaev, D. G. & Du Bois, J. A diruthenium catalyst for selective, intramolecular allylic C–H amination: reaction development and mechanistic insight gained through experiment and theory. J. Am. Chem. Soc. 133, 17207–17216 (2011).
Barman, D. N. & Nicholas, K. M. Copper-catalyzed intramolecular C–H amination. Eur. J. Org. Chem. 5, 908–911 (2011).
Liang, J.-L., Yuan, S.-X., Huang, J.-S. & Che, C.-M. Intramolecular C–N bond formation reactions catalyzed by ruthenium porphyrins: amidation of sulfamate esters and aziridination of unsaturated sulfonamides. J. Org. Chem. 69, 3610–3619 (2004).
Goswami, M. et al. Characterization of porphyrin-Co(iii)-‘nitrene radical’ species relevant in catalytic nitrene transfer reactions. J. Am. Chem. Soc. 137, 5468–5479 (2015).
Varela-Álvarez, A. et al. Rh2(ii,iii) catalysts with chelating carboxylate and carboxamidate supports: electronic structure and nitrene transfer reactivity. J. Am. Chem. Soc. 138, 2327–2341 (2016).
Charton, M. Nature of the ortho effect. II. Composition of the Taft steric parameters. J. Am. Chem. Soc. 91, 615–618 (1969).
Charton, M. Steric effects. I. Esterification and acid-catalyzed hydrolysis of esters. J. Am. Chem. Soc. 97, 1552–1556 (1975).
Charton, M. Steric effects. II. Base-catalyzed ester hydrolysis. J. Am. Chem. Soc. 97, 3691–3693 (1975).
Charton, M. Steric effects. III. Bimolecular nucleophilic substitution. J. Am. Chem. Soc. 97, 3694–3697 (1975).
Acknowledgements
J.M.S. is grateful to the NSF (award no. 1664374) for financial support. The following instrumentation in the Paul Bender Chemistry Instrumentation Center was supported by Thermo Q Exactive Plus by NIH 1S10 OD020022-1; Bruker Quazar APEX2 and Bruker Avance-500 by a generous gift from P. J. Bender and M. M. Bender; Bruker Avance-600 by NIH S10 OK012245; Bruker Avance-400 by NSF CHE-1048642 and the University of Wisconsin–Madison; Varian Mercury-300 by NSF CHE-0342998. C. G. Fry and H. Hofstetter at University of Wisconsin–Madison are thanked for valuable discussions about NMR equipment and techniques. M. M. Vestling of University of Wisconsin–Madison is thanked for help with mass spectrometry characterization. D. J. Weix and K. J. Garcia are thanked for access to chiral supercritical fluid chromatography instrumentation.
Author information
Authors and Affiliations
Contributions
J.M.S., M.J. and M.H. conceived the project. M.J., M.H., L.E.V. and M.D. performed the synthetic experiments. J.M.R. performed the DFT calculations. J.M.S., M.J., M.H., L.E.V. and J.M.R. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Ju, M., Huang, M., Vine, L.E. et al. Tunable catalyst-controlled syntheses of β- and γ-amino alcohols enabled by silver-catalysed nitrene transfer. Nat Catal 2, 899–908 (2019). https://doi.org/10.1038/s41929-019-0339-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-019-0339-y
This article is cited by
-
Combining visible-light induction and copper catalysis for chemo-selective nitrene transfer for late-stage amination of natural products
Communications Chemistry (2022)
-
Nitrene transfer catalysts for enantioselective C–N bond formation
Nature Reviews Chemistry (2021)
-
Nitrene-mediated intermolecular N–N coupling for efficient synthesis of hydrazides
Nature Chemistry (2021)