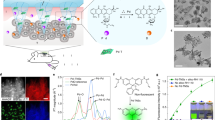
Abstract
The transformational impact of bioorthogonal chemistries has inspired new strategies for the in vivo synthesis of bioactive agents through non-natural means. Among these, Pd catalysts have played a prominent role in the growing subfield of bioorthogonal catalysis by producing xenobiotics and uncaging biomolecules in living systems. However, delivering catalysts selectively to specific cell types still lags behind catalyst development. Here, we have developed a bioartificial device comprising cancer-derived exosomes that are loaded with Pd catalysts by a method that enables the controlled assembly of Pd nanosheets directly inside the vesicles. This hybrid system mediates Pd-triggered dealkylation reactions in vitro and inside cells, and displays preferential tropism for their progenitor cells. The use of Trojan exosomes to deliver abiotic catalysts into designated cancer cells creates the opportunity for a new targeted therapy modality; that is, exosome-directed catalyst prodrug therapy, whose first steps are presented herein with the cell-specific release of the anticancer drug panobinostat.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data that support the plots within this paper and other findings of this study are available from the corresponding author on reasonable request.
References
Streu, C. & Meggers, E. Ruthenium-induced allylcarbamate cleavage in living cells. Angew. Chem. Int. Ed. 45, 5645–5648 (2006).
Li, N., Lim, R. K., Edwardraja, S. & Lin, Q. Copper-free sonogashira cross-coupling for functionalization of alkyne-encoded proteins in aqueous medium and in bacterial cells. J. Am. Chem. Soc. 133, 15316–15319 (2011).
Spicer, C. D., Triemer, T. & Davis, B. G. Palladium-mediated cell-surface labeling. J. Am. Chem. Soc. 134, 800–803 (2012).
Michel, B. W., Lippert, A. R. & Chang, C. J. A reaction-based fluorescent probe for selective imaging of carbon monoxide in living cells using a palladium-mediated carbonylation. J. Am. Chem. Soc. 134, 15668–15671 (2012).
Yang, M., Jia, S., Zhang, X. & Chen, P. Palladium-triggered deprotection chemistry for protein activation in living cells. Nat. Chem. 6, 352–361 (2014).
Mascareñas, J. L., Sánchez, M. I., Penas, C. & Vázquez, M. E. Metal-catalyzed uncaging of DNA-binding agents in living cells. Chem. Sci. 5, 1901 (2014).
Völker, T., Dempwolff, F., Graumann, P. L. & Meggers, E. Angew. Chem. Int. Ed. 53, 10536 (2014).
Wang, J. et al. Chemical remodeling of cell-surface sialic acids through a palladium-triggered bioorthogonal elimination reaction. Angew. Chem. Int. Ed. 54, 5364–5368 (2015).
Tomás-Gamasa, M., Martínez-Calvo, M., Couceiro, J. R. & Mascareñas, J. L. Transition metal catalysis in the mitochondria of living cells. Nat. Commun. 7, 12538 (2016).
Tsubokura, K. et al. In vivo gold complex catalysis within live mice. Angew. Chem. Int. Ed. 56, 3579–3584 (2017).
Destito, P., Couceiro, J. R., Faustino, H., López, F. & Mascareñas, J. L. Ruthenium‐catalyzed azide-thioalkyne cycloadditions in aqueous media: a mild, orthogonal, and biocompatible chemical ligation. Angew. Chem. Int. Ed. 56, 10766–10770 (2017).
Vidal, C., Tomás-Gamasa, M., Destito, P., López, F. & Mascareñas, J. L. Concurrent and orthogonal gold(i) and ruthenium(ii) catalysis inside living cells. Nat. Commun. 9, 1913 (2018).
Stenton, B. J., Oliveira, B. L., Matos, M. J., Sinatra, L. & Bernardes, G. J. L. A thioether-directed palladium-cleavable linker for targeted bioorthogonal drug decaging. Chem. Sci. 9, 4185–4189 (2018).
Chatterjee, A. et al. An enantioselective artificial Suzukiase based on the biotin–streptavidin technology. Chem. Sci. 7, 673–677 (2016).
Jeschek, M. et al. Directed evolution of artificial metalloenzymes for in vivo metathesis. Nature 537, 661–665 (2016).
Okamoto, Y. et al. A cell-penetrating artificial metalloenzyme regulates a gene switch in a designer mammalian cell. Nat. Commun. 9, 1943 (2018).
Yusop, R. M., Unciti-Broceta, A., Johansson, E. M. V., Sánchez-Martín, R. M. & Bradley, M. Palladium-mediated intracellular chemistry. Nat. Chem. 3, 239–243 (2011).
Unciti-Broceta, A., Johansson, E. M. V., Yusop, R. M., Sánchez-Martín, R. M. & Bradley, M. Synthesis of polystyrene microspheres and functionalization with Pd(0) nanoparticles to perform bioorthogonal organometallic chemistry in living cells. Nat. Protoc. 7, 1207–1218 (2012).
Tonga, G. Y. et al. Supramolecular regulation of bioorthogonal catalysis in cells using nanoparticle-embedded transition metal catalysts. Nat. Chem. 7, 597–603 (2015).
Miller, M. A. et al. Nano-palladium is a cellular catalyst for in vivo chemistry. Nat. Commun. 8, 15906–15919 (2017).
Clavadetscher, J., Indrigo, E., Chankeshwara, S. V., Lilienkampf, A. & Bradley, M. In-cell dual drug synthesis by cancer-targeting palladium catalysts. Angew. Chem. Int. Ed. 56, 6864–6868 (2017).
Hoop, M. et al. Mobile magnetic nanocatalysts for bioorthogonal targeted cancer therapy. Adv. Funct. Mater. 28, 1705920 (2018).
Liu, Y. et al. Catalytically active single-chain polymeric nanoparticles: exploring their functions in complex biological media. J. Am. Chem. Soc. 140, 3423–3433 (2018).
Wang, F., Zhang, Y., Du, Z., Ren, J. & Qu, X. Designed heterogeneous palladium catalysts for reversible light-controlled bioorthogonal catalysis in living cells. Nat. Commun. 9, 1209 (2018).
Weiss, J. T. et al. Extracellular palladium-catalysed dealkylation of 5-fluoro-1-propargyl-uracil as a bioorthogonally activated prodrug approach. Nat. Commun. 5, 3277 (2014).
Weiss, J. T. et al. Development and bioorthogonal activation of palladium-labile prodrugs of gemcitabine. J. Med. Chem. 57, 5395–5404 (2014).
Weiss, J. T., Carragher, N. O. & Unciti-Broceta, A. Palladium-mediated dealkylation of N-propargyl-floxuridine as a bioorthogonal oxygen-independent prodrug strategy. Sci. Rep. 5, 9329 (2015).
Clavadetscher, J. et al. Copper catalysis in living systems and in situ drug synthesis. Angew. Chem. Int. Ed. 55, 15662–15666 (2016).
Pérez-López, A. M. et al. Gold-triggered uncaging chemistry in living systems. Angew. Chem. Int. Ed. 56, 12548–12552 (2017).
Bray, T. L. et al. Bright insights into palladium-triggered local chemotherapy. Chem. Sci. 9, 7354–7361 (2018).
Adam, C. et al. Bioorthogonal uncaging of the active metabolite of irinotecan by palladium‐functionalized microdevices. Chem. Eur. J. 24, 16783–16790 (2018).
Eda, S. et al. Biocompatibility and therapeutic potential of glycosylated albumin artificial metalloenzymes. Nat. Catal. https://doi.org/10.1038/s41929-019-0317-4 (2019).
Valadi, H. et al. Exosome mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat. Cell Biol. 9, 654–659 (2007).
Gould, S. J., Booth, A. M. & Hildreth, J. E. The Trojan exosome hypothesis. Proc. Natl Acad. Sci. USA 100, 10592–10597 (2003).
Gourlay, J. et al. The emergent role of exosomes in glioma. J. Clin. Neurosci. 35, 13–23 (2017).
Hornick, N. I. et al. AML suppresses hematopoiesis by releasing exosomes that contain microRNAs targeting c-MYB. Sci. Signal. 9, ra88 (2016).
Huan, J. et al. Coordinate regulation of residual bone marrow function by paracrine trafficking of AML exosomes. Leukemia 29, 2285–2295 (2015).
Alvarez-Erviti, L. et al. Delivery of siRNA to the mouse brain by systemic injection of targeted exosomes. Nat. Biotechnol. 29, 341–345 (2011).
Li, X. et al. Exosomes in cancer: small transporters with big functions. Cancer Lett. 435, 55–65 (2018).
Meckes, D. G. Jr Exosomal communication goes viral. J. Virol. 89, 5200–5203 (2015).
Abrami, L. et al. Hijacking multivesicular bodies enables long-term and exosome-mediated long-distance action of anthrax toxin. Cell Rep. 5, 986–996 (2013).
Cross, R. Meet the exosome, the rising star in drug delivery. Chem. Eng. News 96, 22–23 (2018).
Zitvogel, L. et al. Eradication of established murine tumors using a novel cell-free vaccine: dendritic cell-derived exosomes. Nat. Med. 4, 594–600 (1998).
Munoz, J. L. et al. Delivery of functional anti-miR-9 by mesenchymal stem cell-derived exosomes to glioblastoma multiforme cells conferred chemosensitivity. Mol. Ther. Nucleic Acids 2, e126 (2013).
Smyth, T. et al. Surface functionalization of exosomes using click chemistry. Bioconjug. Chem. 25, 1777–1784 (2014).
Yang, T. et al. Exosome delivered anticancer drugs across the blood-brain barrier for brain cancer therapy in Danio rerio. Pharm. Res. 32, 2003–2014 (2015).
Kamerkar, S. et al. Exosomes facilitate therapeutic targeting of oncogenic KRAS in pancreatic cancer. Nature 546, 498–503 (2017).
György, B. et al. Rescue of hearing by gene delivery to inner-ear hair cells using exosome-associated AAV. Mol. Ther. 25, 379–391 (2017).
Kim, S. M. et al. Cancer-derived exosomes as a delivery platform of CRISPR/Cas9 confer cancer cell tropism-dependent targeting. J. Control. Release 266, 8–16 (2017).
Wang, Q. et al. ARMMs as a versatile platform for intracellular delivery of macromolecules. Nat. Commun. 9, 960 (2018).
Hood, J. L., Scott, M. J. & Wickline, S. A. Maximizing exosomes colloidal stability following electroporation. Anal. Biochem. 448, 41–49 (2014).
Alhasan, A. H., Patel, P. C., Choi, C. H. J. & Mirkin, C. A. Exosome encased spherical nucleic acid gold nanoparticle conjugates as potential microRNA regulation agents. Small 10, 186–192 (2014).
Busato, A. et al. Magnetic resonance imaging of ultrasmall superparamagnetic iron oxide-labeled exosomes from stem cells: a new method to obtain labeled exosomes. Int. J. Nanomed. 11, 2481–2490 (2016).
Illes, B. et al. Exosome-coated metal–organic framework nanoparticles: an efficient drug delivery platform. Chem. Mater. 29, 8042–8046 (2017).
Betzer, O. et al. In vivo neuroimaging of exosomes using gold nanoparticles. ACS Nano 11, 10883–10893 (2017).
Ha, D., Yang, N. & Nadithe, V. Exosomes as therapeutic drug carriers and delivery vehicles across biological membranes: current perspectives and future challenges. Acta Pharm. Sin. B. 6, 287–296 (2016).
Luan, X. et al. Engineering exosomes as refined biological nanoplatforms for drug delivery. Acta Pharmacol. Sin. 38, 754–763 (2017).
Sebastian, V., Smith, C. D. & Jensen, K. F. Shape-controlled continuous synthesis of metal nanostructures. Nanoscale 8, 7534–7543 (2016).
Herrer, L. et al. High surface coverage of a self-assembled monolayer by in situ synthesis of palladium nanodeposits. Nanoscale 9, 13281–13290 (2017).
Zhang, D. et al. An in situ TEM study of the surface oxidation of palladium nanocrystals assisted by electron irradiation. Nanoscale 9, 6327–6333 (2017).
Kikot, P., Polat, A., Achilli, E., Fernandez Lahore, M. & Grasselli, M. Immobilized palladium(ii) ion affinity chromatography for recovery of recombinant proteins with peptide tags containing histidine and cysteine. J. Mol. Recognit. 27, 659–668 (2014).
Raedler, L. A. Farydak (panobinostat): first HDAC inhibitor approved for patients with relapsed multiple myeloma. Am. Health Drug Benefits 9, 84–87 (2016).
Zheng, S. et al. Biocompatible boron-containing prodrugs of belinostat for the potential treatment of solid tumors. ACS Med. Chem. Lett. 9, 149–154 (2018).
Rubio-Ruiz, B., Weiss, J. T. & Unciti-Broceta, A. Efficient palladium-triggered release of vorinostat from a bioorthogonal precursor. J. Med. Chem. 59, 9974–9980 (2016).
Wang, H. et al. Discovery of (2E)-3-{2-butyl-1-[2-(diethylamino)ethyl]-1H-benzimidazol-5-yl}-N-hydroxyacrylamide (SB939), an orally active histone deacetylase inhibitor with a superior preclinical profile. J. Med. Chem. 54, 4694–4720 (2011).
Sancho-Albero, M. et al. Exosome origin determines cell targeting and the transfer of therapeutic nanoparticles towards target cells. J. Nanobiotechnol. 17, 16 (2019).
Sletten, E. M. & Bertozzi, C. R. Bioorthogonal chemistry: fishing for selectivity in a sea of functionality. Angew. Chem. Int. Ed. 48, 6974–6998 (2009).
Li, J. & Chen, P. R. Development and application of bond cleavage reactions in bioorthogonal chemistry. Nat. Chem. Biol. 12, 129–137 (2016).
Devaraj, N. K. The future of bioorthogonal chemistry. ACS Cent. Sci. 4, 952–959 (2018).
Capes-Davis, A. et al. Check your cultures! A list of cross-contaminated or misidentified cell lines. Int. J. Cancer 127, 1–8 (2010).
Acknowledgements
We gratefully acknowledge financial support from EPSRC (Healthcare Technology Challenge award no. EP/N021134/1) and the ERC Advanced Grant CADENCE (grant no. ERC-2016-ADG-742684). We also thank S. Irusta at the University of Zaragoza for performing X-ray photoelectron spectroscopy measurements. M.S.-A. thanks the Spanish government for an FPU PhD research fellowship. B.R.-R. thanks the EC (grant no. H2020-MSCA-IF-2014–658833). M.A. thanks the financial support of the ERC Consolidator Grant programme (grant no. ERC-2013-CoG-614715). P.M.-D. thanks AECC Fund and Refbio-Lactodermal for financial support.
Author information
Authors and Affiliations
Contributions
M.S.-A., B.R.-R., A.M.P.-L. and V.S. prepared and characterised the materials, planned and performed the experiments, analysed the data and wrote the Methods; V.S., P.M.-D. and M.A. planned and supervised the research, analysed the data and contributed to the manuscript writing; J.S. and A.U.-B. designed, coordinated and supervised the research, analysed the data and wrote the paper. All the authors checked the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare that compound 4 is protected under patent application WO/2017/199028.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Video 1
Real-time imaging of Pd-ExoA549-mediated conversion of pro-fluorophore 1 into red fluorescent fluorophore 2. The reactions were performed in PBS at room temperature and imaged by time-lapse microscopy with a ×20 objective (Ex/Em: 560/630 nm, Leica AF6000 LX) for 24 h. The frames were taken every 15 min in differential interference contrast mode (top) and under fluorescence emission (bottom). Left (positive control): resorufin 2 alone (20 μM). Middle (negative control): probe 1 alone (20 μM). Right (activation experiment): probe 1 (20 μM) + Pd-ExoA549 (0.5 μg ml).
Rights and permissions
About this article
Cite this article
Sancho-Albero, M., Rubio-Ruiz, B., Pérez-López, A.M. et al. Cancer-derived exosomes loaded with ultrathin palladium nanosheets for targeted bioorthogonal catalysis. Nat Catal 2, 864–872 (2019). https://doi.org/10.1038/s41929-019-0333-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-019-0333-4
This article is cited by
-
Hydrogen-bonded organic framework-based bioorthogonal catalysis prevents drug metabolic inactivation
Nature Catalysis (2023)
-
Fabrication of Pd NPs/Mg–Al LDH via a facile one-step hydrothermal method as highly active and stable heterogeneous catalyst for Heck coupling reaction
Journal of Materials Science (2023)
-
Exosomes loaded with ultrasmall Pt nanoparticles: a novel low-toxicity alternative to cisplatin
Journal of Nanobiotechnology (2022)
-
The roles of small extracellular vesicles in cancer and immune regulation and translational potential in cancer therapy
Journal of Experimental & Clinical Cancer Research (2022)
-
Synthetic prodrug design enables biocatalytic activation in mice to elicit tumor growth suppression
Nature Communications (2022)