Abstract
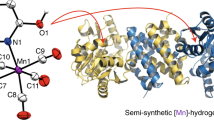
Hydrogenases are promising templates for constructing new H2-based catalysts. [Fe]-hydrogenase, which features an iron-guanylylpyridinol (FeGP) cofactor, catalyses a reversible hydride transfer from H2 to methenyl-tetrahydromethanopterin (methenyl-H4MPT+, a C1 carrier in methanogens). Here, we present a detailed mechanistic scenario of this reaction based on the 1.06 Å resolution structure of [Fe]-hydrogenase in a closed active form, in which the Fe of the FeGP cofactor is positioned near the hydride-accepting C14a of a remarkably distorted methenyl-H4MPT+. The open-to-closed transition generates an unsaturated pentacoordinated Fe on expulsion of a water ligand. Quantum mechanics/molecular mechanics computations based on experimental models indicate that a deprotonated 2-OH group on the FeGP cofactor acts as a catalytic base and provides a fairly complete picture of H2 activation: H2 binding on the empty Fe site was found to be nearly thermo-neutral while H2 cleavage and hydride transfer proceed smoothly. The overall reaction involves a repositioning and relaxation of the distorted methenyl-H4MPT+.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data that support the plots within this paper and other findings of this study are available from the corresponding authors on reasonable request. X-ray crystallographic data are available in the RCSB-Protein Data Bank under accession numbers 6HAC (open conformation), 6HAV (closed conformation form A) and 6HAE (closed conformation form B).
References
Fontecilla-Camps, J. C., Volbeda, A., Cavazza, C. & Nicolet, Y. Structure/function relationships of [NiFe]- and [FeFe]-hydrogenases. Chem. Rev. 107, 4273–4303 (2007).
Lubitz, W., Ogata, H., Rudiger, O. & Reijerse, E. Hydrogenases. Chem. Rev. 114, 4081–4148 (2014).
Thauer, R. K. et al. Hydrogenases from methanogenic archaea, nickel, a novel cofactor and H2 storage. Annu. Rev. Biochem. 79, 507–536 (2010).
Vignais, P. M. & Billoud, B. Occurrence, classification and biological function of hydrogenases: an overview. Chem. Rev. 107, 4206–4272 (2007).
Zirngibl, C. et al. H2-forming methylenetetrahydromethanopterin dehydrogenase, a novel type of hydrogenase without iron–sulfur clusters in methanogenic archaea. Eur. J. Biochem. 208, 511–520 (1992).
Shima, S. & Ermler, U. Structure and function of [Fe]-hydrogenase and its iron-guanylylpyridinol (FeGP) cofactor. Eur. J. Inorg. Chem. 2011, 963–972 (2011).
Vogt, S., Lyon, E. J., Shima, S. & Thauer, R. K. The exchange activities of [Fe] hydrogenase (iron–sulfur-cluster-free hydrogenase) from methanogenic archaea in comparison with the exchange activities of [FeFe] and [NiFe] hydrogenases. J. Biol. Inorg. Chem. 13, 97–106 (2008).
Hartmann, G. C., Santamaria, E., Fernández, V. M. & Thauer, R. K. Studies on the catalytic mechanism of H2-forming methylenetetrahydromethanopterin dehydrogenase: para-ortho H2 conversion rates in H2O and D2O. J. Biol. Inorg. Chem. 1, 446–450 (1996).
Thauer, R. K., Klein, A. R. & Hartmann, G. C. Reactions with molecular hydrogen in microorganisms: evidence for a purely organic hydrogenation catalyst. Chem. Rev. 96, 3031–3042 (1996).
Schleucher, J., Schwörer, B., Thauer, R. K. & Griesinger, C. Elucidation of the stereochemical course of chemical reactions by magnetic labeling. J. Am. Chem. Soc. 117, 2941–2942 (1995).
Pilak, O. et al. The crystal structure of the apoenzyme of the iron–sulfur cluster-free hydrogenase. J. Mol. Biol. 358, 798–809 (2006).
Shima, S. et al. The crystal structure of [Fe]-hydrogenase reveals the geometry of the active site. Science 321, 572–575 (2008).
Hiromoto, T. et al. The crystal structure of C176A mutated [Fe]-hydrogenase suggests an acyl–iron ligation in the active site iron complex. FEBS Lett. 583, 585–590 (2009).
Hiromoto, T., Warkentin, E., Moll, J., Ermler, U. & Shima, S. The crystal structure of an [Fe]-hydrogenase-substrate complex reveals the framework for H2 activation. Angew. Chem. Int. Ed. 48, 6457–6460 (2009).
Lyon, E. J. et al. Carbon monoxide as an intrinsic ligand to iron in the active site of the iron–sulfur-cluster-free hydrogenase H2-forming methylenetetrahydromethanopterin dehydrogenase as revealed by infrared spectroscopy. J. Am. Chem. Soc. 126, 14239–14248 (2004).
Tamura, H. et al. Crystal structures of [Fe]-hydrogenase in complex with inhibitory isocyanides: implications for the H2-activation site. Angew. Chem. Int. Ed. 52, 9656–9659 (2013).
Shima, S. & Ataka, K. Isocyanides inhibit [Fe]-hydrogenase with very high affinity. FEBS Lett. 585, 353–356 (2011).
Dey, A. Density functional theory calculations on the mononuclear non-heme iron active site of Hmd hydrogenase: role of the internal ligands in tuning external ligand binding and driving H2 heterolysis. J. Am. Chem. Soc. 132, 13892–13901 (2010).
Hedegård, E. D., Kongsted, J. & Ryde, U. Multiscale modeling of the active site of [Fe] hydrogenase: the H2 binding site in open and closed protein conformations. Angew. Chem. Int. Ed. 54, 6246–6250 (2015).
Yang, X. Z. & Hall, M. B. Monoiron hydrogenase catalysis: hydrogen activation with the formation of a dihydrogen, Fe-Hδ−–Hδ+-O, bond and methenyl-H4MPT+ triggered hydride transfer. J. Am. Chem. Soc. 131, 10901–10908 (2009).
Berkessel, A. Activation of dihydrogen without transition metals. Curr. Opin. Chem. Biol. 5, 486–490 (2001).
Berkessel, A. & Thauer, R. K. On the mechanism of catalysis by a metal-free hydrogenase from methanogenic archaea: enzymatic transformation of H2 without a metal and its analogy to the chemistry of alkanes in superacidic solution. Angew. Chem. Int. Ed. Engl. 34, 2247–2250 (1995).
Shima, S., Lyon, E. J., Thauer, R. K., Mienert, B. & Bill, E. Mössbauer studies of the iron–sulfur cluster-free hydrogenase: the electronic state of the mononuclear Fe active site. J. Am. Chem. Soc. 127, 10430–10435 (2005).
Korbas, M. et al. The iron–sulfur cluster-free hydrogenase (Hmd) is a metalloenzyme with a novel iron binding motif. J. Biol. Chem. 281, 30804–30813 (2006).
Gütlich, P., Bill, E. & Trautwein, A. X. Mössbauer Spectroscopy and Transition Metal Chemistry (Springer Verlag, 2011).
Chen, D. F., Scopelliti, R. & Hu, X. L. A five-coordinate iron center in the active site of [Fe]-hydrogenase: hints from a model study. Angew. Chem. Int. Ed. 50, 5671–5673 (2011).
Shima, S. et al. Reconstitution of [Fe]-hydrogenase using model complexes. Nat. Chem. 7, 995–1002 (2015).
Kabsch, W. XDS. Acta Crystallogr. D 66, 125–132 (2010).
Winn, M. D. et al. Overview of the CCP4 suite and current developments. Acta Crystallogr. D 67, 235–242 (2011).
McCoy, A. J. et al. Phaser crystallographic software. J. Appl. Crystallogr. 40, 658–674 (2007).
Emsley, P., Lohkamp, B., Scott, W. G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D 66, 486–501 (2010).
BUSTER version 2.10.1. (Global Phasing, 2016); https://www.globalphasing.com/buster/
Adams, P. D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D 66, 213–221 (2010).
Chen, V. B. et al. MolProbity: all-atom structure validation for macromolecular crystallography. Acta Crystallogr. D 66, 12–21 (2010).
Krebs, W. G. & Gerstein, M. The morph server: a standardized system for analyzing and visualizing macromolecular motions in a database framework. Nucleic Acids Res. 28, 1665–1675 (2000).
Chung, L. W. et al. The ONIOM method and its applications. Chem. Rev. 115, 5678–5796 (2015).
Frisch, M. J. et al. Gaussian 09, Revision D.01 (Gaussian, 2016).
Zhao, Y. & Truhlar, D. G. The M06 suite of density functionals for main group thermochemistry, thermochemical kinetics, noncovalent interactions, excited states and transition elements: two new functionals and systematic testing of four M06-class functionals and 12 other functionals. Theor. Chem. Acc. 120, 215–241 (2008).
Zhao, Y. & Truhlar, D. G. Density functionals with broad applicability in chemistry. Acc. Chem. Res. 41, 157–167 (2008).
Acknowledgements
This work was supported by grants from the Max Planck Society (to S.S., E.B. and U.E.), Deutsche Forschungsgemeinschaft (Iron-Sulfur for Life, SH 87/1-1 to S.S.) and the Swiss National Science Foundation (to X.H.). M.D.W. thanks C. Corminboeuf (École Polytechnique Fédérale de Lausanne, Switzerland) for financial support and the Laboratory for Computational Molecular Design (EPFL) for providing computing resources. The authors are grateful to H. Michel for continuous support and the staff of the PX II beamline at the Swiss Light Source (SLS) for help with data collection. The authors also thank the staff from the BM30A (FIP) beamline at the European Synchrotron Radiation Facility (ESRF). G.H. was supported by a fellowship from China Scholarship Council (CSC).
Author information
Authors and Affiliations
Contributions
S.S. directed and designed research. G.H. performed cultivation, enzyme purification and crystallization. T.W. and U.E. collected X-ray data. T.W. solved, refined and deposited the structure. M.D.W. and X.H. performed and analysed the QM/MM computations. K.A. performed infrared spectroscopy and E.B. performed Mössbauer spectroscopy. All authors contributed to writing the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Supplementary Information
Supplementary Methods, Supplementary Figs. 1–15, Supplementary Tables 1–3, Supplementary references
Supplementary Data 1
Structure 2
Supplementary Data 2
Structure 3
Supplementary Data 3
Structure 4
Supplementary Data 4
Structure 5
Supplementary Data 5
TS3,4
Supplementary Data 6
TS4,5
Supplementary Video 1
Open/closed conformational change
Rights and permissions
About this article
Cite this article
Huang, G., Wagner, T., Wodrich, M.D. et al. The atomic-resolution crystal structure of activated [Fe]-hydrogenase. Nat Catal 2, 537–543 (2019). https://doi.org/10.1038/s41929-019-0289-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-019-0289-4