Abstract
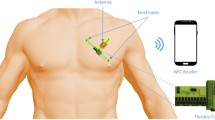
Differential amplifiers based on organic thin-film transistors (OTFTs) are attractive for monitoring human vital signs because of their signal amplification and noise elimination capabilities. However, substantial variations in OTFTs lead to the degradation of signal processing performance in circuits and restrict the development of organic differential amplifiers capable of recording weak physiological potentials. Here, we report a 2-μm-thick ultraflexible organic differential amplifier capable of processing physiological signals with high signal integrity and sensitivity. Our approach uses a circuit design and compensation technique that suppress the mismatch among OTFTs to less than a few percent. This leads to a common-mode noise attenuation factor below −12 dB, even during bending to ~50 μm. Using our flexible amplifier, we monitor electrocardiogram signals, where the power of 60 Hz electrical harmonic noise was reduced ~60 times during amplification, yielding electrocardiogram signals with a signal-to-noise ratio of 34 dB.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data that support the plots within this paper and other findings of this study are available from the corresponding author upon reasonable request.
References
Xu, S. et al. Soft microfluidic assemblies of sensors, circuits and radios for the skin. Science 344, 70–74 (2014).
Gao, W. et al. Fully integrated wearable sensor arrays for multiplexed in situ perspiration analysis. Nature 529, 509–514 (2016).
Imani, S. et al. A wearable chemical–electrophysiological hybrid biosensing system for real-time health and fitness monitoring. Nat. Commun. 7, 11650 (2016).
Jang, K.-I. et al. Self-assembled three dimensional network designs for soft electronics. Nat. Commun. 8, 15894 (2017).
Khodagholy, D. et al. In vivo recordings of brain activity using organic transistors. Nat. Commun. 4, 1575 (2013).
Kang, S. K. et al. Bioresorbable silicon electronic sensors for the brain. Nature 530, 71–76 (2016).
Lu, L. et al. Wireless optoelectronic photometers for monitoring neuronal dynamics in the deep brain. Proc. Natl Acad. Sci. USA 115, E1374–E1383 (2018).
Kim, J. et al. Stretchable silicon nanoribbon electronics for skin prosthesis. Nat. Commun. 5, 5747 (2014).
Lim, S. et al. Transparent and stretchable interactive human machine interface based on patterned graphene heterostructures. Adv. Funct. Mater. 25, 375–383 (2015).
Yeo, W. H. et al. Multifunctional epidermal electronics printed directly onto the skin. Adv. Mater. 25, 2773–2778 (2013).
Wang, X., Gu, Y., Xiong, Z., Cui, Z. & Zhang, T. Silk-molded flexible, ultrasensitive and highly stable electronic skin for monitoring human physiological signals. Adv. Mater. 26, 1336–1342 (2013).
Miyamoto, A. et al. Inflammation-free, gas-permeable, lightweight, stretchable on-skin electronics with nanomeshes. Nat. Nanotechnol. 12, 907–913 (2017).
Koo, J. H. et al. Wearable electrocardiogram monitor using carbon nanotube electronics and color-tunable organic light-emitting diodes. ACS Nano 11, 10032–10041 (2017).
Mannsfeld, S. C. B. et al. Highly sensitive flexible pressure sensors with microstructured rubber dielectric layers. Nat. Mater. 9, 859–864 (2010).
Lipomi, D. J. et al. Skin-like pressure and strain sensors based on transparent elastic films of carbon nanotubes. Nat. Nanotechnol. 6, 788–792 (2011).
Pang, C. et al. A flexible and highly sensitive strain-gauge sensor using reversible interlocking of nanofibres. Nat. Mater. 11, 795–801 (2012).
Schwartz, G. et al. Flexible polymer transistors with high pressure sensitivity for application in electronic skin and health monitoring. Nat. Commun. 4, 1859 (2013).
Gong, S. et al. A wearable and highly sensitive pressure sensor with ultrathin gold nanowires. Nat. Commun. 5, 3132 (2014).
Hua, Q. et al. Skin-inspired highly stretchable and conformable matrix networks for multifunctional sensing. Nat. Commun. 9, 244 (2018).
Lochner, C. M., Khan, Y., Pierre, A. & Arias, A. C. All-organic optoelectronic sensor for pulse oximetry. Nat. Commun. 5, 5745 (2014).
Yokota, T. et al. Ultraflexible organic photonic skin. Sci. Adv. 2, e1501856 (2016).
Bandodkar, A. J. et al. Tattoo-based potentiometric ion-selective sensors for epidermal pH monitoring. Analyst 138, 123–128 (2013).
Yazicioglu, R. F., Merken, P., Puers, R. & Hoof, Ch. V. A 60 μW 60 nV/√Hz readout front-end for portable biopotential acquisition system. IEEE J. Solid State Circuits 42, 1100–1110 (2007).
Yazicioglu, R. F. et al. A 30 μW analog signal processor ASIC for portable biopotential signal monitoring. IEEE J. Solid State Circuits 46, 209–223 (2011).
Klauk, H., Zschieschang, U., Pflaum, J. & Halik, M. Ultralow-power organic complementary circuits. Nature 445, 745–748 (2007).
Gelinck, G., Heremans, P., Nomoto, K. & Anthopoulos, T. D. Organic transistors in optical displays and microelectronics applications. Adv. Mater. 22, 3778–3798 (2010).
Kaltenbrunner, M. et al. An ultra-lightweight design for imperceptible plastic electronics. Nature 499, 458–463 (2013).
Sirringhaus, H. Organic field-effect transistors—the path beyond amorphous silicon. Adv. Mater. 26, 1319–1335 (2014).
Wang, S. et al. Skin electronics from scalable fabrication of an intrinsically stretchable transistor array. Nature 555, 83–88 (2018).
Fukuda, K. et al. Fully-printed high-performance organic thin-film transistors and circuitry on one-micron-thick polymer films. Nat. Commun. 5, 4147 (2014).
Sekitani, T. et al. Ultraflexible organic amplifier with biocompatible gel electrodes. Nat. Commun. 7, 11425 (2016).
Guerin, M. et al. High-gain fully printed organic complementary circuits on flexible plastic foils. IEEE Trans. Electron Devices 58, 3587–3593 (2011).
Marien, H., Steyaert, M., Veenendaal, V. E. & Heremans, P. Analog techniques for reliable organic circuit design on foil applied to an 18 dB single-stage differential amplifier. Org. Electron. 11, 1357–1362 (2010).
Marien, H., Steyaert, M. S. J., Veenendaal, V. E. & Heremans, P. A fully integrated ΔΣ ADC in organic thin-film transistor technology on flexible plastic foil. IEEE J. Solid State Circuits 46, 276–284 (2011).
Maiellaro, G. et al. High-gain operational transconductance amplifiers in a printed complementary organic TFT technology on flexible foil. IEEE Trans. Circuits Syst. 60, 3117–3125 (2013).
Chang, J., Zhang, X., Ge, T. & Zhou, J. Fully printed electronics on flexible substrate: high gain amplifiers and DAC. Org. Electron. 15, 701–710 (2014).
Pecunia, V. et al. Trap healing for high-performance low-voltage polymer transistors and solution-based analogue amplifiers on foil. Adv. Mater. 29, 1606938 (2017).
Fuketa, H. et al. 1-μm-thickness ultra-flexible and high electrode-density surface electromyogram measurement sheet with 2 V organic transistors for prosthetic hand control. IEEE Trans. Biomed. Circuits Syst. 8, 824–833 (2014).
Kondo, M. et al. Ultraflexible and ultrathin polymeric gate insulator for 2 V organic transistor circuits. Appl. Phys. Express 9, 061602 (2016).
Acknowledgements
This work was supported by the Japan Society for the Promotion of Science (JSPS) KAKENHI grant (nos. JP16H01857, JP18H01861 and JP19J22073), the New Energy and Industrial Technology Development Organization (NEDO), the Center of Innovation Program from Japan Science and Technology Agency (JST), grant from the Commissioned Research of NICT and the Brain Mapping by Integrated Neurotechnologies for Disease Studies (Brain/MINDS) project of the Japan Agency for Medical Research and Development (AMED). Part of this work was performed at the Advanced Photonics and Biosensing Open Innovation Laboratory, National Institute of Advanced Industrial Science and Technology (AIST). This work for circuit simulation and design was supported by VLSI Design and Education Center (VDEC), the University of Tokyo, in collaboration with Synopsys and Cadence Design Systems. The authors thank Nippon Kayaku Co. for supplying DNTT and Daisan Kasei Co. for high-purity parylene (diX-SR).
Author information
Authors and Affiliations
Contributions
M.S., T.U. and T.S. conceived the concept and experiments. M.S., M.A. and S.Y. designed the circuit and conceived the process for mismatch compensation. M.S., T.U., M.K. and N.N. developed the fabrication process for OTFTs and fabricated the device. M.S. carried out all experiments for device characterization and data collection for the organic circuit, with help from T.U., M.K., M.A. and N.N. M.S. wrote the manuscript in consultation with T.U., S.Y., Y.N., T.A. and T.S. All authors commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–15 and Supplementary Tables 1–2.
Rights and permissions
About this article
Cite this article
Sugiyama, M., Uemura, T., Kondo, M. et al. An ultraflexible organic differential amplifier for recording electrocardiograms. Nat Electron 2, 351–360 (2019). https://doi.org/10.1038/s41928-019-0283-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41928-019-0283-5
This article is cited by
-
Motion artefact management for soft bioelectronics
Nature Reviews Bioengineering (2024)
-
Wearable bioelectronics fabricated in situ on skins
npj Flexible Electronics (2023)
-
270 nm ultra-thin self-adhesive conformable and long-term air-stable complimentary organic transistors and amplifiers
npj Flexible Electronics (2023)
-
Thin-film transistors for large-area electronics
Nature Electronics (2023)
-
Artificial intelligence-powered electronic skin
Nature Machine Intelligence (2023)