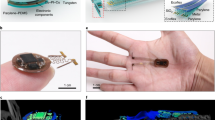
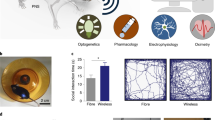
Abstract
Recently developed ultrasmall, fully implantable devices for optogenetic neuromodulation eliminate the physical tethers associated with conventional set-ups and avoid the bulky head-stages and batteries found in alternative wireless technologies. The resulting systems allow behavioural studies without motion constraints and enable experiments in a range of environments and contexts, such as social interactions. However, these devices are purely passive in their electronic design, thereby precluding any form of active control or programmability; independent operation of multiple devices, or of multiple active components in a single device, is, in particular, impossible. Here we report optoelectronic systems that, through developments in integrated circuit and antenna design, provide low-power operation, and position- and angle-independent wireless power harvesting, with full user-programmability over individual devices and collections of them. Furthermore, these integrated platforms have sizes and weights that are not significantly larger than those of previous, passive systems. Our results qualitatively expand options in output stabilization, intensity control and multimodal operation, with broad potential applications in neuroscience research and, in particular, the precise dissection of neural circuit function during unconstrained behavioural studies.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The data that support the plots within this paper and other findings of this study are available from the corresponding author upon reasonable request.
References
Boyden, E. S., Zhang, F., Bamberg, E., Nagel, G. & Deisseroth, K. Millisecond-timescale, genetically targeted optical control of neural activity. Nat. Neurosci. 8, 1263–1268 (2005).
Deisseroth, K. Optogenetics. Nat. Methods 8, 26–29 (2011).
Park, S. I. et al. Soft, stretchable, fully implantable miniaturized optoelectronic systems for wireless optogenetics. Nat. Biotechnol. 33, 1280–1286 (2015).
Pisanello, F. et al. Multipoint-emitting optical fibers for spatially addressable in vivo optogenetics. Neuron 82, 1245–1254 (2014).
Yizhar, O., Fenno, L. E., Davidson, T. J., Mogri, M. & Deisseroth, K. Optogenetics in neural systems. Neuron 71, 9–34 (2011).
Chen, R., Canales, A. & Anikeeva, P. Neural recording and modulation technologies. Nat. Rev. Mater. 2, 16093 (2017).
Shin, G. et al. Flexible near-field wireless optoelectronics as subdermal implants for broad applications in optogenetics. Neuron 93, 509–521 (2017). e503.
Montgomery, K. L. et al. Wirelessly powered, fully internal optogenetics for brain, spinal and peripheral circuits in mice. Nat. Methods 12, 969–974 (2015).
Park, S. I. et al. Stretchable multichannel antennas in soft wireless optoelectronic implants for optogenetics. Proc. Natl Acad. Sci. USA 113, E8169–E8177 (2016).
Ho, J. S. et al. Self-tracking energy transfer for neural stimulation in untethered mice. Phys. Rev. Appl. 4, 024001 (2015).
Gutruf, P. & Rogers, J. A. Implantable, wireless device platforms for neuroscience research. Curr. Opin. Neurobiol. 50, 42–49 (2018).
Klapoetke, N. C. et al. Independent optical excitation of distinct neural populations. Nat. Methods 11, 338–346 (2014).
Akerboom, J. et al. Genetically encoded calcium indicators for multi-color neural activity imaging and combination with optogenetics. Front. Mol. Neurosci. https://doi.org/10.3389/fnmol.2013.00002 (2013).
Harvey, C. D., Collman, F., Dombeck, D. A. & Tank, D. W. Intracellular dynamics of hippocampal place cells during virtual navigation. Nature 461, 941–946 (2009).
Tye, K. M. et al. Amygdala circuitry mediating reversible and bidirectional control of anxiety. Nature 471, 358–362 (2011).
Hight, A. E. et al. Superior temporal resolution of Chronos versus channelrhodopsin-2 in an optogenetic model of the auditory brainstem implant. Hear. Res. 322, 235–241 (2015).
Gerlai, R. A small fish with a big future: zebrafish in behavioral neuroscience. Rev. Neurosci. 22, 3–4 (2011).
Yartsev, M. M. & Ulanovsky, N. Representation of three-dimensional space in the hippocampus of flying bats. Science 340, 367–372 (2013).
Lu, L. et al. Wireless optoelectronic photometers for monitoring neuronal dynamics in the deep brain. Proc. Natl Acad. Sci. USA 115, E1374–E1383 (2018).
Samineni, V. K. et al. Fully implantable, battery-free wireless optoelectronic devices for spinal optogenetics. Pain 158, 2108–2116 (2017).
Wang, L., Jacques, S. L. & Zheng, L. MCML—Monte Carlo modeling of light transport in multi-layered tissues. Comput. Methods Programs Biomed. 47, 131–146 (1995).
Keijzer, M., Jacques, S. L., Prahl, S. A. & Welch, A. J. Light distributions in artery tissue: Monte Carlo simulations for finite‐diameter laser beams. Lasers Surg. Med. 9, 148–154 (1989).
Yaroslavsky, A. et al. Optical properties of selected native and coagulated human brain tissues in vitro in the visible and near infrared spectral range. Phys. Med. Biol. 47, 2059 (2002).
Yona, G., Meitav, N., Kahn, I. & Shoham, S. Realistic numerical and analytical modeling of light scattering in brain tissue for optogenetic applications. eNeuro 3, ENEURO.0059-0015.2015 (2016).
Acknowledgements
We acknowledge support from the Center for Bio-Integrated Electronics at Northwestern University. C.R.H. is supported by Cancer Center Support Grant P30 CA060553 from the National Cancer Institute awarded to the Robert H. Lurie Comprehensive Cancer Center. Z.X. acknowledges support from the National Natural Science Foundation of China (grant number 11402134). Y.H. acknowledges support from the National Science Foundation (grant numbers 1400169, 1534120 and 1635443).
Author information
Authors and Affiliations
Contributions
P.G., A.V.-G., Z.X. and J.A.R. designed research. P.G., V.K., A.V-G., Z.X., A.B., C.-J.S., Y.X., C.R.H., E.A.W., I.K., S.R.K, T.R. and J.P.L. performed research. P.G., A.V.-G., Z.X., C.R.H., E.A.W., I.K., Y.H., D.C. and J.A.R. analysed data. P.G. and J.A.R. wrote the paper.
Corresponding author
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figures 1–14
Supplementary Video 1
Demonstration of output intensity modulation. The program increases intensity from OFF to full intensity and terminates with an indicator flash.
Supplementary Video 2
Demonstration of output modulation using one-way communication. Remotely selected programs include: State 1, sequential blinking of all four LEDs; State 2, alternate blinking of left and right shank; State 3, blinking of LED1; State 4, blinking of LED2; State 5, blinking of LED3; State 6, blinking of LED4 and subsequent reset to State 1.
Supplementary Video 3
Demonstration of individual control over device functionality of multiple devices in one experimental environment.
Supplementary Video 4
Freely moving mouse with constant-intensity device implanted and active.
Supplementary Video 5
Bilateral optogenetic implant operating inside a 7 tesla small animal MRI.
Rights and permissions
About this article
Cite this article
Gutruf, P., Krishnamurthi, V., Vázquez-Guardado, A. et al. Fully implantable optoelectronic systems for battery-free, multimodal operation in neuroscience research. Nat Electron 1, 652–660 (2018). https://doi.org/10.1038/s41928-018-0175-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41928-018-0175-0
This article is cited by
-
Body-conformable light-emitting materials and devices
Nature Photonics (2024)
-
Accelerated intestinal wound healing via dual electrostimulation from a soft and biodegradable electronic bandage
Nature Electronics (2024)
-
Nanofabricated high turn-density spiral coils for on-chip electromagneto-optical conversion
Microsystems & Nanoengineering (2024)
-
Study of electrical conductivity, biocompatibility and degradation effect of copper electrodes with hydrogel coatings for use in flexible electronics
Applied Physics A (2024)
-
Wireless light energy harvesting and communication in a waterproof GaN optoelectronic system
Communications Engineering (2022)