Abstract
Endothelial cell malignancies are rare in the Western world and range from intermediate grade hemangioendothelioma to Kaposi sarcoma to aggressive high-grade angiosarcoma that metastasize early and have a high rate of mortality. These malignancies are associated with dysregulation of normal endothelial cell signaling pathways, including the vascular endothelial growth factor, angiopoietin, and Notch pathways. Discoveries over the past two decades related to mechanisms of angiogenesis have led to the development of many drugs that intuitively would be promising therapeutic candidates for these endothelial-derived tumors. However, clinical efficacy of such drugs has been limited. New insights into the mechanisms that lead to dysregulated angiogenesis such as mutation or amplification in known angiogenesis related genes, viral infection, and chromosomal translocations have improved our understanding of the pathogenesis of endothelial malignancies and how they evade anti-angiogenesis drugs. In this review, we describe the major molecular alterations in endothelial cell malignancies and consider emerging opportunities for improving therapeutic efficacy against these rare but deadly tumors.
Similar content being viewed by others
KeyPoints
-
Intermediate- and high-grade endothelial cell malignancies are rare, but can be associated with substantial morbidity and mortality.
-
HIV and KSHV viral proteins directly interact with Notch pathway and VEGF pathway proteins, contributing to the tumorigenesis of Kaposi sarcoma.
-
Angiosarcomas contain multiple abnormalities that may lead to primary resistance to VEGF/VEGFR inhibitors. These include VEGFR2 (KDR) and PLCG1 mutations, loss-of-function PTPRB mutations, and amplification of c-MYC and VEGFR3 (FLT4).
-
Treating chemotherapy-resistant endothelial malignancies with VEGF-targeted drugs has had limited success. Therefore, more effective therapies are needed.
-
Understanding the deregulated signaling pathways in endothelial cell neoplasms may reveal insights into driver and drug-resistance mechanisms.
Introduction
Endothelial cells are known to line established blood vessels and initiate the establishment of new blood and lymph channels in vascular development. Malignancies arising from endothelial cells are rare in developed countries; however, the tumors that do arise tend to be highly aggressive and difficult to treat. While high-grade endothelial malignancies respond well to traditional chemotherapy agents such as taxanes (Fig. 1), their durability is poor and the tumors acquire drug resistance rapidly.1 Targeted therapies such as anti-angiogenic agents that would be intuitive for these malignancies have had limited success in the clinic. Therefore, a renewed effort aimed at identifying the mechanisms by which endothelial cell malignancies evade currently available angiogenesis inhibitors is required to improve outcomes of patients with these rare diagnoses. New insights from the laboratory include identification of potential drivers of endothelial malignancies that can function as new targets for future clinical development.
The clinical and pathological features of endothelial cell malignancies have been reviewed previously.2,3,4,5 These tumors generally occur in adults and are classified as having intermediate or high malignant potential. Intermediate grade vascular neoplasms include epithelioid, spindle cell, psuedomyogenic, and malignant endovascular papillary hemangioendotheliomas. Each of these entities is classically differentiated by histological appearance, but recent evidence suggests that at least some of the intermediate grade hemangioendotheliomas are driven by chromosomal translocations (Table 1). Compared to other intermediate types, epithelioid hemangioendotheliomas (EHEs) of visceral origin tend to be multifocal with propensity for metastasis despite their slow proliferative rate. Histopathologically, these tumors are characterized by an epithelioid appearance with disorganized vascular channels and are sometimes mistaken for carcinomas (e.g., lung adenocarcinoma).6 The diagnosis is often made by the presence of one of several characteristic chromosomal translocations.
Endothelial cell malignancies of high malignant potential include angiosarcomas and, in immunocompromised hosts, Kaposi sarcoma (KS). Angiosarcomas belong to the high-grade end of the spectrum, with an aggressive clinical course characterized by a high propensity for both local recurrence and distant metastasis. Clinically, there are two distinct subtypes. Primary angiosarcomas can occur anywhere in the body; the more common sites include the scalp, breast, liver, spleen, bone, and heart.2 Secondary angiosarcomas arise from chronic lymphedema in the extremities or from radiation exposure to the chest wall following breast cancer treatment and are often molecularly associated with amplification of c-MYC.7 KS has a viral etiology and is caused by KS-associated herpes virus (KSHV; also known as HHV8). Originally described in elderly men of Mediterranean descent, it is also associated with the human immunodeficiency virus (HIV) and immune suppression8 and is now one of the most common malignancies in Sub-Saharan Africa.9
A common feature of endothelial cell malignancies is the dysregulation of normal endothelial cell signaling pathways (Fig. 2). Multiple mechanisms contribute to the observed dysregulation, including viral oncoproteins, chromosomal rearrangements, and paracrine signaling in the microenvironment. To date, targeting angiogenesis pathways has had varying degrees of success (Table 2). In this review, we summarize the existing clinical, molecular, and biological knowledge to frame a path toward a greater understanding of the pathophysiology of endothelial cell malignancies and improved clinical outcomes.
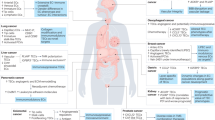
Key pathways in endothelial cell malignancies. Alterations in angiosarcoma (orange stars) include 1. Activating mutations in VEGFR2 (KDR), and amplification of VEGFR3 (FLT4), 2. A recurrent activating R707Q mutation in PLCG1, 3. Loss of function mutations in PTPRB, removing the inhibitory signal on Tie2, 4. Mutations in K-, H-, and N-RAS, BRAF, and MAPK1, and amplification of B- and C- RAF, and MAPK1. 5. Mice with loss of Tsc1 have constitutive activation of mTOR signaling and develop angiosarcoma. 6. C-MYC amplification is associated with radiation or lymphedema-induced angiosarcoma. The KSHV-derived LANA protein stabilizes HEY (6) leading to c-MYC transcription in KS cells, and stabilizes the Notch intracellular domain (NICD) (7), leading to increased Notch-mediated signaling. The HIV-1 protein Tat binds to alpha-5-beta-1 and alpha-v-beta-3 integrin receptors and stimulates migration and invasion. Tat also stimulates the release of preformed, extracellularly bound bFGF into a soluble form that can induce vascular cell growth and prevent apoptosis, and Tat directly interacts with VEGFR2 leading to ligand independent activation of its downstream effectors (8). Expression of the lytic phase KSHV viral G-protein coupled receptor (vGPCR) leads to activation of the MAPK and PI3K/mTOR pathways (9) which ultimately causes HIF-1a-mediated transcription of Notch-related proteins (10). Ang2 angiopoietin2, AKT protein kinase B, b-FGF basic fibroblast growth factor, BMP bone morphogenetic protein, DLL delta-like, ERK1/2 mitogen-activated protein kinase 1/2, FGFR, FAK focal adhesion kinase, FGFR fibroblast growth factor receptor, GRB2 growth factor receptor-bound protein 2, HES hairy enhancer-of-split, HEY hairy and enhancer of split related protein, HIF-1alpha hypoxia inducible factor 1 alpha subunit, LANA latency-associated nuclear antigen, MAML mastermind-like protein, MEK MAPK kinase, mTOR mammalian target of rapamycin, MYC Myc proto-oncogene, PLCG1 phospholipase C-gamma 1, PI3K phosphoinositide 3-kinase, PTPRB receptor-like protein-tyrosine phosphatase (PTP) beta, RBPJ recombinant binding protein suppressor of hairless, S6K P70-S6Kinase 1, TACE ADAM17, Tie2 TEK tyrosine kinase, endothelial, Tsc1 tuberous sclerosis 1, VEGF(R) vascular endothelial growth factor (receptor)
Physiologic angiogenesis pathways in endothelial cell malignancies
Vascular endothelial growth factor (VEGF)/VEGF receptor (VEGFR) signaling
VEGF/VEGFR2-mediated signaling is critical for tip cell selection and migration in physiological angiogenesis. VEGFR2 and VEGFR3 both promote tip cell formation, breakdown of the basement membrane, and loss of pericyte coverage, allowing for endothelial cell migration. Unsurprisingly, endothelial cell neoplasms have aberrant VEGF/VEGFR pathway signaling.
In aggressive angiosarcomas, alterations in VEGF and its receptors have been well characterized, including mutations and amplifications (Fig. 2). Angiosarcomas have high VEGF-A and VEGFR (1–3) expression, with rates ranging from 65–94% for VEGFR (1–3).10 VEGFR2 mutations have been reported in 10% of angiosarcomas11 and in 2 of 6 angiosarcomas in a smaller series,12 with mutations identified in the extracellular, transmembrane, and kinase domains. However, the prevalence of these VEGFR2 mutations in angiosarcomas remains uncertain, as no VEGFR2 mutations were revealed in other studies including whole-genome- or whole-exome sequencing.13 The functional consequence of these mutations is not fully understood, but at least some are thought to be activating mutations that act as drivers in a subset of angiosarcomas.11
Perhaps the strongest clinical correlation of VEGF/VEGFR dysregulation in angiosarcoma is the finding of VEGFR3 (FLT4) gene amplification in secondary (radiation- or lymphedema- induced) angiosarcomas.7 These amplifications are generally found in combination with other alterations such as c-MYC amplification and mutations in PLCG1 and PTPRB. Thus, although VEGFR3 drives lymphangiogenesis and induces sprouting and tip cell migration, the individual contribution of VEGFR3 amplification in these cases remains unclear. Effective targeting of the VEGF/VEGFR axis either by knocking down VEGF-A, -C, or -D, or treatment with the pan-VEGFR inhibitor axitinib in a mouse model with constitutive mammalian target of rapamycin (mTOR) activation (develop tumors consistent with lymphangiosarcoma) suggests that targeting the upstream ligand or receptor may be a therapeutic option even in cases when downstream activating mutations are identified.14 Although these alterations do not represent the primary genetic driver events, targeting VEGF/VEGFR signaling is a rational clinical approach in angiosarcoma.
VEGF signaling also contributes to the development of KS. AIDS-KS spindle cells are stimulated to secrete VEGF by platelet-derived growth factor (PDGF)-beta and interleukin (IL)-1 beta, and co-injection of these two factors increases the vascularity of KS-like lesions in mice.15 VEGF acts as an autocrine growth factor in KS, which has high levels of VEGFR1, VEGFR2,16 and VEGFR317 compared with adjacent normal skin. Expression of the virally encoded vGPCR (discussed below) is sufficient to induce VEGF-mediated angiogenic phenotypic switching in mouse fibroblasts18 and immortalizes human umbilical vein endothelial cells (HUVEC) by upregulation and activation of VEGFR219 VEGF expression is less well-characterized in other malignancies of endothelial cell origin. Some studies have reported VEGF expression in a high proportion of EHEs, and associated increased VEGF staining intensity with more aggressive disease.20, 21 Additional work is needed to fully characterize the contributions of the VEGF pathway in EHE pathogenesis.
Clinical results with anti-VEGF drugs such as bevacizumab and VEGFR2-blocking tyrosine kinase inhibitors (TKIs) have been disappointing in patients with endothelial cell malignancies (Table 2). The response rate to bevacizumab monotherapy was 9% (median progression free survival (PFS) of just 3 months) in patients with angiosarcoma;22 addition of bevacizumab to paclitaxel did not provide additional benefit over paclitaxel alone.23 Sorafenib, a TKI with anti-VEGFR2 activity, had a similarly low response rate reaching 14.6% in patients with advanced angiosarcoma (median PFS of 2–5 months).24,25,26 Clinical responses to other TKIs including pazopanib27, 28 and sunitinib29, 30 have also been published in small reports, but larger phase II and III clinical trials of sunitinib31 or pazopanib32 in soft tissue sarcoma included only a limited number of patients with angiosarcomas. Similarly, some studies have reported benefit in EHE patients treated with bevacizumab, sunitinib, or pazopanib, but larger studies are needed to draw definitive conclusions about response rates. In addition, these studies lacked a control arm, making it difficult to quantify the benefit of these agents in this indolent tumor with slow progression.22, 33,34,35 Interestingly, in patients with KS, HAART plus bevacizumab resulted in an overall response rate of 31%, with three complete responses.36 Despite this finding, anti-VEGF/VEGFR therapy is not currently used routinely in KS likely due to higher response rates with liposomal doxorubicin and taxanes.
Angiopoietin/Tie2 signaling
In contrast to VEGF signaling, which stimulates early vascular development, angiopoietin (Ang)/Tie signaling promotes endothelial cell survival and stability.37 Different Tie2 ligands, notably Ang1 and Ang2, have variable effects on Tie2 signaling. Ang1 is produced by multiple cell types, whereas Ang2 is produced primarily by endothelial cells and is expressed only in tissues undergoing remodeling.38 Ang2 can function as an agonist or antagonist depending on the environment.39 Interestingly, Ang2 is upregulated in solid tumor angiogenesis, and high levels of Ang2 are associated with worse outcomes in multiple cancer types.40
A transmembrane phosphatase, receptor-like protein-tyrosine phosphatase (PTP) beta (PTPRB), dephosphorylates Tie2, rendering it inactive.41, 42 Loss-of-function mutations in PTPRB are relatively common in angiosarcoma and were found in 26% of angiosarcomas; interestingly, all these mutations were in secondary angiosarcomas.13 Normally, PTPRB dephosphorylates Tie2; thus, loss of PTPRB in angiosarcoma likely increases Ang/Tie2 signaling and may activate multiple pathways downstream from Tie2, such as the protein kinase B (AKT)/phosphoinositide 3-kinase (PI3K)/mTOR, endothelial nitric oxide synthase, focal adhesion kinase (FAK), mitogen-activated protein kinase (MAPK), and downstream of kinase-related protein/non-catalytic region of tyrosine kinase adaptor protein 1/p21-activated protein kinase (DOCK/NCK/PAK) pathways.40 Silencing PTPRB in HUVECs led to increased sprouting, even in the presence of VEGFR2 inhibitors such as sunitinib, demonstrating that PTPRB loss represents a mechanism of canonical activation of Tie2 signaling in angiosarcoma.13
Ang2 is highly expressed in angiosarcoma, and higher Ang2 secretion is correlated with more advanced angiosarcoma stage; in contrast, Ang1 seems to have a minimal role in angiosarcoma43 and higher Ang1 expression correlates with improved survival.44 Ang2, Tie1, and Tie2 are strongly expressed in both angiosarcoma and KS samples.44, 45 The correlation of increased Ang2 expression with worse outcomes and that of increased Ang1 expression with improved survival suggests that these ligands may be promising therapeutic targets. However, in a phase II study of trebananib, a peptibody against both Ang1 and Ang2, no responses were seen in angiosarcoma patients. Trebananib increased Ang1 and Ang2 levels after treatment, likely contributing to the lack of response.46 Additionally, antagonizing Ang1 may diminish its seemingly beneficial effect. Given the differential effects of Ang1 and Ang2, a better approach may be to target Ang2 independently with a specific agent.
Notch signaling
In physiological vascular development, Notch signaling is active at endothelial tip cells, where increased DLL4 expression stimulates Notch signaling in neighboring stalk cells and inhibits them from migrating, thus promoting organized vascular branching.47 Upon ligand binding, Notch receptors are cleaved by regulated proteolysis and the cleaved intracellular component travels to the nucleus and interacts with hairy and enhancer of split related-2 (HERP-2/Hey-1) and hairy enhancer-of-split (HES) proteins, leading to transcription (Fig. 2).48 The Notch ligand Jagged1 has a pro-angiogenic effect, whereas DLL4 counters the proliferative effects of VEGF49 and Jagged1.50
The observation of endothelial neoplasms forming after DLL4 inhibition in mouse models implicated Notch signaling in the development of proliferative vascular tumors, especially in the liver.51 Conditional knockout of Notch1 in the liver in a mouse model resulted in hepatic angiosarcomas in 86% of mice by 50 weeks.52 Studies in a separate mouse model confirmed that loss of Notch1 heterozygosity leads to endothelial cell neoplasms of varying histological grade with approximately the same penetrance; interestingly, the liver was the primary site.53 However, few, if any, significant Notch abnormalities have been identified in the sequencing efforts of human angiosarcoma samples, and the clinical relevance of these observations remains to be determined. Notch pathway targeted therapies such as DLL4 inhibitors and Notch receptor antagonists have effects that would mechanistically lead to disorganized vascularization as seen in the mouse models, and are, therefore, not being tested in angiosarcoma. In other cancer types, Notch has oncogenic or tumor suppressor roles depending on context, and it similarly can have pro- or anti--angiogenic effects in physiological tissue remodeling. The potential effect of nonspecific Notch inhibition with gamma-secretase inhibitors on hemangioendothelioma and angiosarcoma remains to be determined. A pathogenic role for the Notch pathway, with druggable targets for therapeutic development, is more established in KS tumorigenesis primarily due to the effects of KSHV proteins on endothelial cells. This is described in more detail below. While these observations reflect the importance of this pathway in deregulation of angiogenesis, targeting Notch signaling is complicated by several unique features of this pathway. Effects of Notch are remarkably context dependent and the signal itself has dose-dependent effects downstream. Furthermore, Notch signal tends to have a very short intracellular half-life and sustained inhibition may not be needed. To utilize Notch inhibitors in treatment of angiosarcoma, it would be important to identify the optimal level and timing of inhibition for disease control without excessive toxicity.
Other angiogenesis-related pathways in endothelial cell malignancies
Platelet-derived growth factor
Paracrine signaling between endothelial cells and perivascular cells is mediated, in part, by PDGF signaling. Endothelial cells secrete PDGF-BB, which increases pericyte coverage and maintains the integrity of the endothelial cell basement membrane.54 At least five different PDGF isoforms interact with two different PDGF receptors (PDGFRs).55 PDGFR activation results in autophosphorylation of the receptor, which in turn activates phospholipase C-gamma (PLCG).56 As described below, PLCG1 activation by VEGFR2 acts as a driver for a subset of angiosarcomas and leads to resistance to VEGF/VEGFR targeted therapies. Treatment with dasatinib or imatinib, which inhibit PDGFR as well as other kinases, decreased cell viability in vitro and decreased tumor growth in vivo in a canine xenograft model of hemangiosarcoma.57 Isolated responses to imatinib, a PDGFR inhibitor, have been noted in patients with angiosarcoma.58, 59 A recently completed clinical trial of olaratumab, a monoclonal antibody against PDGFR-alpha, combined with doxorubicin (NCT01185964) for soft tissue sarcoma showed promising results including an improvement in overall survival; olaratumab has not been evaluated for treatment of angiosarcoma. Endothelial cell malignancies contain disorganized endothelium, and have not been proven to be associated with pericytes in the same way as normal endothelial cells.
Angiostatin and endostatin
Angiostatin and endostatin are protein fragments that suppress tumor growth and angiogenesis.60, 61 Endostatin has been used to treat angiosarcoma, but its effectiveness could not be determined because it was given in combination with cytotoxic chemotherapy.62 The vast majority (>90%) of endothelial cell neoplasms including benign hemangiomas, EHEs, and angiosarcomas express annexin II, an angiostatin receptor.63 Angiostatin inhibits hemangioendothelioma growth in vivo, but does not affect proliferation or induce apoptosis in vitro.64 Interestingly, endostatin paradoxically stimulates hemangioma-derived endothelial progenitor cells in an in vitro migration assay, a phenomenon not observed in hemangioendothelioma cells.65, 66 Although angiostatin and endostatin are sometimes used in patients with endothelial cell malignancies, more evidence is needed to fully assess their potential benefits.
Endoglin/transforming growth factor beta
Endoglin (CD105) is a component of the transforming growth factor beta receptor family that is expressed on endothelial cells, mesenchymal stem cells, and monocytes and has been specifically considered a drug target for novel agents designed to target tumor angiogenesis.67, 68 In physiological angiogenesis, endoglin mediates TGF-beta signaling via activin a receptor type II-like 1 (ALK1), which acts as a pro-angiogenic mediator and increases endothelial cell migration and proliferation, counteracting the potential inhibitory effect of TGF-beta on endothelial cells.69 There are two isoforms of endoglin, with S-endoglin playing a critical role in vascular senescence.70 Endoglin mutations cause hereditary hemorrhagic telangiectasia type 1, which is characterized by vascular dysplasia and hemorrhage,71 but to date no mutations in endoglin have been identified in endothelial cell neoplasms. Angiosarcomas have high expression levels of endoglin, with 95–100% staining positive.72, 73 Levels of TGF-beta pathway proteins are higher in angiosarcoma of bone than in primary angiosarcomas of soft tissue.74 However, the importance of endoglin in mediating the observed increase in TGF-beta signaling in bone angiosarcoma is not established, and the near universal expression of endoglin in angiosarcomas regardless of their site of origin makes any causative presumptions premature without additional study. An anti-endoglin antibody is currently being tested in clinical trials in combination with pazopanib for soft tissue sarcoma, with preliminary results in a phase I/II trial showing 2/2 patients with complete response, suggesting efficacy in angiosarcoma that requires further investigation (NCT01975519).75
Intracellular oncogenic signaling pathways
Mitogen-activated protein kinases
In normal endothelial cells, VEGF induced VEGFR endocytosis and regulated MAPK activation are important factors that stabilize filopodia-carrying endothelial sprouts and ensure that a transient signal allows for stability in a branching vessel.76 VEGFR2 activation leads to PLCG1 phosphorylation and transduces the activating signal of its binding ligand to ensure normal vascular function.77,78,79,80 Autophosphorylation of VEGFR2 leads to the recruitment of PLCG1, binding of PLCG1 at its N-terminal SH2 domain, and subsequent activation of PLCG1 (ref. 81). Phosphorylation at PLCG1-Y783 causes a conformational change that relieves the auto-inhibition of the C-terminal SH2 domain, and leads to downstream signaling.82, 83 Activated PLCG1 catalyzes the conversion of phosphatidylinositol 4,5-bisphosphate to diacylglycerol and inositol 1,4,5-triphosphate, leading to protein kinase C-dependent MAPK signaling.84, 85 A recurring mutation in PLCG1, R707Q, has been identified in angiosarcoma, with a prevalence of 9–30% (refs 12, 13). R707 is in the auto-inhibitory SH2 domain of PLCG1, and the missense mutation is hypothesized to destabilize this domain and reduce its auto-inhibitory effect.13 Indeed, canonical activation was demonstrated by expressing the PLCG1-R707Q in HUVECs, which resulted in the downstream activation of MAPK and NFAT-mediated signaling irrespective of PLCG1 activation by VEGFR2 (ref. 12). Interestingly, sequencing from a patient with progression following initial response to sunitinib revealed a PLCG1-R707Q mutation in the metastasis, but this mutation was not found in the primary tumor. This finding suggests a mechanism by which angiosarcomas could develop adaptive resistance to VEGFR-targeted therapy.86 In addition to PLCG1 mutations, mutations or amplifications in K-, H-, and N-RAS, B- and C-RAF, and MAPK1 were identified in angiosarcoma.87 Preclinical studies with canine angiosarcomas have demonstrated anti-tumor activity with MEK inhibition.88
Phosphoinositide 3-kinase/protein kinase B/mammalian target of rapamycin
The PI3K/AKT/mTOR signaling pathway is frequently activated in many cancer types. In normal endothelial cells, this pathway is activated through several stimuli, including VEGF, Ang/Tie2, and integrins.89 AKT is phosphorylated compared to normal adjacent endothelial cells in nearly all endothelial cell neoplasms.90, 91 Interestingly, different AKT isoforms have opposing effects. In hemangioma, hemangioendothelioma, and angiosarcoma models, AKT1 has been demonstrated to promote growth and migration, whereas AKT3 inhibits growth.90 Overexpression of AKT1 is sufficient to create proliferative neoplasms, but without the ability to metastasize.92
Downstream in the PI3K pathway, mTOR complex 1 (mTORC1) activation leads to the activation of p70 S6-kinase and S6 ribosomal protein. Angiosarcomas have increased activation of S6-kinase and S6 ribosomal protein; topical rapamycin inhibits the growth of patient-derived infantile hemangioma (IH) cells and established hemangioendothelioma and angiosarcoma cell lines in vitro and the growth of hemangioendothelioma mouse xenografts.93
Mice with a conditional knockout of tuberous sclerosis complex 1 (Tsc1), a negative regulator of mTORC1, develop paw angiosarcomas at 6 weeks that are responsive to rapamycin.94 The sustained mTORC1 signaling that results from Tsc1 loss leads to increases in HIF1-alpha and c-MYC-mediated VEGF transcription, creating an autocrine loop that is required for tumor maintenance.14 Although canine angiosarcoma xenografts in athymic nude mice showed minimal response to temsirolimus alone, temsirolimus sensitized angiosarcomas to MEK inhibition, suggesting cross-talk between the MAPK and PI3K/AKT/mTOR pathways.88 Although the PI3K/AKT/mTOR pathway is activated in angiosarcoma, this activation does not appear to be due to mutations or losses directly affecting this pathway, such as phosphatase and tensin homolog loss or PI3K mutations.95 Targeting the pathway with mTOR inhibitors remains a promising therapy and is currently used for patients with angiosarcoma in the investigational setting.
Transcriptional regulation
Myc proto-oncogene (MYC)
Dysregulation of the transcription factor MYC has been implicated in many cancer types.96 MYC amplification is well described in secondary angiosarcoma7, 97 and has also been demonstrated in primary angiosarcoma.98 In addition to MYC, Ret proto-oncogene (RET) signaling is upregulated and cyclin-dependent kinase inhibitor 2C is downregulated in secondary angiosarcoma,99 which is in agreement with the finding that N-MYC is a downstream target of Ret that downregulates cyclin-dependent kinase inhibitor p18, leading to proliferation of cultured fibroblasts.100 MYC may contribute to the aggressive angiogenic phenotype of angiosarcoma by upregulating miR-17-92 and thus downregulating thrombospondin-1 (TSP1), an angiogenesis inhibitor.101 Genomic analyses revealed that MYC-related pathways were upregulated in murine angiosarcoma cell lines isolated from primary hepatic angiosarcomas in Notch1 conditional knockout mice.102 Interestingly, mice in which Cdk6 activity is increased by rendering them insensitive to inhibition by INK4 develop angiosarcomas with a high prevalence (~50%), particularly when the Cdk6 alteration is introduced in a p53 heterozygous or null background.103 To date, Cdk4/6 inhibitors have not been evaluated in angiosarcoma, but could represent an important opportunity.
MYC also plays an important role in KS pathogenicity. The KSHV virus protein LANA stabilizes c-MYC by preventing its phosphorylation at Thr58, thus preventing apoptosis.104 In primary effusion lymphoma, another malignancy associated with KSHV, MYC is required to maintain the latency of KSHV,105 and inhibition of bromodomain and extra-terminal domain bromodomain (BET), a therapeutic strategy for targeting pathologic MYC activation,106 has had promising results in vitro in other KSHV associated tumors.107 In other models, MYC has been shown to promote neovascularization by either downregulating anti-angiogenic factors such as TSP1 and connective tissue growth factor108 or interacting with hypoxia to induce VEGF-A production.109 The latter mechanism, which used a model for dermal angiogenesis, may be particularly relevant in endothelial cell tumors that commonly arise in the skin.
HIF/hypoxia
Under normoxic conditions, the transcription factor HIF-1a is primed for degradation by the von Hippel-Lindau tumor suppressor (VHL), an E3 ubiquitin ligase. Hypoxia leads to HIF-1a-mediated pro-angiogenic signaling and consequently the recruitment of blood vessels to solid tumors. In mice, removal of VHL’s inhibition of HIF via mosaic VHL knockout led to the formation of vascular lesions ranging from hemangiomas to a single mouse that developed an angiosarcoma.110 HIF-1a and HIF-2a expression is reported in a subset of angiosarcomas,111, 112 but HIF-1a does not appear to be a notable driver of angiosarcoma growth.112
On the other hand, HIF-1a and hypoxia-related pro-angiogenic pathways play a role in the transition of KSHV-infected endothelial cells to KS.113 A G protein-coupled receptor encoded by KSHV (vGPCR) contains an activating V138D mutation, which leads to agonist-independent induction of the MAPK and p38 signaling pathways. This, in turn, leads to HIF-1a phosphorylation and a HIF-1a-dependent increase in VEGF secretion.114 In addition, vGPCR also increases mTOR complex signaling, suggesting that multiple pathways activated by vGPCR converge on HIF-1a-mediated VEGF transcription and secretion.115 HIF-1a-mediated transcription is also induced by the KSHV LANA protein, by targeting the HIF-1a suppressors VHL and p53 for degradation,116 as well as by direct protein–protein interactions between LANA and HIF-1a that stabilize HIF-1a and promote its translocation to the nucleus (Fig. 2 117).
Viral oncoproteins in KS
The discovery of KSHV led to discoveries regarding the oncogenic role for the virus (reviewed in ref. 118). Here, we focus specifically on KSHV and its direct role in co-opting physiologic angiogenesis to lead to transformation of endothelial cells to Kaposi spindle cells.
Kaposi sarcoma-associated herpes virus
Like other herpes viruses, KSHV infection consists of two phases: the lytic phase in which the virus infects the host cell and replicates and the latent phase in which the viral DNA remains in the host cell but is not actively replicating. Viral proteins specific for both phases directly interact with components of angiogenic signaling to promote tumorigenesis. Specifically, the latency associated proteins LANA and vFLIP induce Notch ligands Jagged1, and DLL4. LANA stabilizes Hey1,119 leading to decreased Hey1 degradation and consequently increased endothelial cell proliferation.120 Furthermore, KSHV harnesses Notch signaling to induce its lytic phase by utilizing RBP-J, a key transcription factor for Notch related genes, to initiate transcription of its lytic phase genes.121 vIL-6, a lytic phase protein, induced expression of Notch4, DLL1, DLL4, and downstream targets Hey1 and Hey2, and vGPCR expression induced Notch2, Notch3, and Jagged1.122 Importantly, Notch3 is typically seen on mural or smooth muscle cells adjacent to endothelial cells but not in endothelial cells themselves.123 Induction of Notch3 suggests that KSHV induces a change in phenotype from differentiated endothelial cells. This is further supported by the observation that activation of the Notch-induced transcription factors Slug and zinc finger E-box-binding homeobox 1 after KSHV infection contributes to the endothelial-to-mesenchymal transition (EndMT) that is important for the malignant progression of infected endothelial cells independently from TGF-beta signaling,124 which regulates EndMT in non-malignant ECs.125 In vitro Notch inhibition with gamma-secretase inhibitors in KS-like cell lines induces mitotic catastrophe,126 suggesting that targeting Notch may be effective for KS. KSHV GPCR also stimulates MAPK signaling. Inhibition of the MAPK pathway in in vitro models of KS led to substantial reductions in the KSHV mediated induction of Notch pathway components, suggesting that MAPK signaling is the primary mechanism by which vGPCR induces Notch activation.122
In spite of the evidence showing the role of Notch and MAPK signaling in KS, the most clinical success to date has been by targeting the mTOR signaling. vGPCR directly leads to the overactivation of the PI3K/AKT/mTOR pathway that is necessary for KSHV-induced transformation of endothelial cells to KS spindle cells.115, 127 In addition, vGPCR activation of MTORC1 leads to secretion of pro-angiogenic factors causing paracrine signaling, which recruits and stimulates other non KSHV infected cells.127 As a result, mTOR inhibition has been used more successfully in the clinic for KS. Among kidney transplant patients with KS as a result of prolonged immunosuppression with cyclosporine, changing the drug to sirolimus resulted in rapid regression of all cutaneous KS lesions.128 Furthermore, long-term stabilization with mTOR inhibition was also seen in AIDS-related KS,129 demonstrating the importance of pathogenic KSHV viral signaling even in co-infected patients.
Human immunodeficiency virus
Overexpression of fibroblast growth factor (bFGF) in normal endothelial cells results in vascular tumors in mice.130 In endothelial cell cancers, FGF signaling plays the largest role in KS. Synergy between the HIV-1 Tat protein and bFGF promotes the development of an angiogenic malignant phenotype in a preclinical mouse model.131 Targeting bFGF with antisense oligonucleotides inhibits the growth of AIDS-KS cells in vitro and in vivo, suggesting that targeting bFGF is a potential therapeutic strategy for KS.132 Tat works by several mechanisms, including binding to alpha-5-beta-1 and alpha-v-beta-3 integrin receptors via its RGD domain and stimulating migration and invasion, and also stimulating the release of preformed, extracellularly bound bFGF into a soluble form that can induce vascular cell growth and prevent apoptosis.133,134,135 Tat can also directly bind to VEGFR2 and stimulate VEGFR2 signaling independent of VEGF-A.136 Downstream, Tat activates multiple growth promoting signaling pathways including MAPK and FAK.137
Microenvironment and intercellular interactions
The complex interplay between various components of the tumor microenvironment (e.g., immune cells, fibroblasts, endothelial cells) can have either pro- or anti-tumor effects depending on the specific circumstances. Among patients with angiosarcoma, the presence of CD8 + tumor infiltrating lymphocytes correlates with a survival advantage.138 In addition to CD8 + cells, endothelial malignancies also have infiltration of CD3 + and CD4 + lymphocytes, as well as regulatory FoxP3 lymphocytes. In EHE, high CD3 + and FoxP3 + lymphocytes but not CD4 + or CD8 + lymphocytes were noted. All of the vascular tumors had high macrophage infiltration.139 Conflicting data exist regarding the expression of programmed death-ligand 1 (PD-L1).139, 140
Macrophages, in particular tumor-associated macrophages, have long been known to have a pro-angiogenic effect that promotes tumor survival (reviewed in ref. 141). Macrophages are recruited to tumors by chemoattractants produced by tumor cells. A series of studies from Japan investigated the use of risedronate (to target macrophages) combined with cytotoxic chemotherapy in the adjuvant setting with promising results.142, 143 High levels of Foxp3 + regulatory T-cells and CD163 + inhibitory macrophages relative to the number of cytotoxic T-cells were seen in these patients with primary angiosarcoma.143 In vitro treatment of angiosarcoma derived macrophages with docetaxel and risedronate increases the expression of C-X-C motif chemokines 10 and 11 (CXCL10 and CXCL11), both chemokines that recruit cytotoxic T-cells, in the treated macrophages.144 In a mouse model of angiosarcoma, inhibition of tumor-secreted IL-6 decreased macrophage numbers and increased cytotoxic T-cell infiltration, thereby decreasing tumor growth.145 This IL-6 secretion is dependent on an autocrine/paracrine network by which inhibitor of nuclear factor kappa-B (NF-KB) kinase subunit beta (IKK-beta) leads to IL-6 production and increased Stat3 activation by NF-KB-mediated transcription of gp130 and Janus kinase 2. In addition to the direct effect of IKK-beta on angiosarcoma cells, knockout of IKK-beta in host myeloid cells decreased neutrophil-derived nitric oxide, increased IL-4, and decreased IL-12 and interferon (IFN)-gamma, thus shifting the myeloid cells to the N2/M2 phenotype and increasing angiosarcoma growth.145 Moreover, inhibition of tumor secreted IL-8 has little effect on angiosarcoma cells in vitro, but prevents engraftment in vivo.146
In addition to the immunosuppression required for HHV8 infection, the immune microenvironment itself contributes to the malignant transformation of endothelial cells into KS. For example, normal endothelial cells cultured in media conditioned with activated T-cells have a phenotype consistent with early KS and are tumorigenic in nudemice.147 Both AIDS-associated and classical KS are infiltrated by CD8 + T-cells and CD14 + /CD68 + monocytes and macrophages that produce IFN-gamma.148 IFN-gamma induces KS spindle cells with an angiogenic phenotype that are similar to early KS cells. In contrast to findings in other tumors suggesting that high levels of infiltrating CD8 + lymphocytes are associated with improved outcomes, these lymphocytes have been proposed to contribute to the development of KS by producing IFN-gamma locally in the microenvironment.
Pericytes have long been known to communicate with endothelial cells to maintain EC stability. A minority of angiosarcomas develop pericyte coverage.149 Angiosarcomas that stain positive for alpha-smooth muscle actin (alpha-SMA), a pericyte marker, tend to have positive staining around malignant non-functional vascular channels.149, 150 In physiological vascular regulation, pericytes slow endothelial growth and their loss in other cancer types correlates with increased metastasis.151 In contrast, pericytes derived from less aggressive endothelial cell tumors contribute to the pro-angiogenic microenvironment by constitutive expression of VEGF-A and decreased Ang1 secretion152 (Fig. 3). Further studies are needed to clarify the role of the observed pericyte-like cells in high-grade endothelial malignancies.
Illustration of microenvironment in endothelial malignancies. a Tissue level schematic of angiosarcoma composed of malignant endothelial cells that form non-functional channel like structures. Stromal support cells such as fibroblasts and pericytes, and immune cells such as macrophages, cytotoxic and suppressor T-cells, and non-malignant endothelial cells all interact to promote tumorigenesis and angiogenesis in the microenvironment. b A selection of autocrine and paracrine signaling networks in the microenvironment of endothelial malignancies. Pro-inflammatory cytokine interleukin-6 (IL-6) is secreted by angiosarcoma cells and is dependent on JAK/STAT and IKK-beta signaling. Interferon-gamma (IFN-gamma) is secreted by macrophages and cytotoxic T-cells, which stimulates Kaposi sarcoma (KS) tumor growth. Kaposi sarcoma herpes virus (KSHV) infection in resident fibroblasts stimulates secretion of multiple pro-angiogenic factors. Pericyte coverage is decreased compared to normal endothelium in endothelial malignancies, though the role of pericytes in directly promoting tumor growth is currently debatable. Ang2 angiopoietin2, b-FGF basic fibroblast growth factor, ECM extracellular matrix, FGFR fibroblast growth factor receptor,HIF-1alpha hypoxia inducible factor 1 alpha subunit, JAK tyrosine protein kinase, mTOR mammalian target of rapamycin, MMPs matrix metalloproteinases, MYC Myc proto-oncogene, PD1 programmed cell death protein 1, PDGF(R) platelet-derived growth factor (receptor), PDL1 programmed cell death 1 ligand, sIL6R soluble IL6 receptor, SMA smooth muscle actin, STAT signal transducer and activator of transcription, Tie2 TEK tyrosine kinase, endothelial, VEGF(R) vascular endothelial growth factor (receptor)
Finally, cancer-associated fibroblasts (CAFs) are activated stromal cells that have been shown to play a key role in promoting tumorigenesis in multiple cancer types. Interestingly, one of the primary mechanisms by which CAFs exert this effect is through VEGF153 and paracrine mediated crosstalk with endothelial cells.154 There is scant data regarding the role of fibroblasts specifically in endothelial cell malignancies, and the potential role of these cells needs to be investigated in angiosarcoma and hemangioendothelioma. The role of fibroblasts contributing to the paracrine growth signaling in KS is more established; latent infection of fibroblasts with KSHV leads the fibroblasts to secrete pro-inflammatory cytokines such as VEGF-A, IL-6, IL-8, and IL-10, as well as matrix metalloproteinases that break down the extracellular matrix and increase tumor cell invasion,155, 156 and are required by KS spindle cells for in vitro growth and to maintain their tumorigenic potential in nude mice.157
Chromosomal rearrangements
Several chromosomal translocations are associated with vascular tumors (Table 1). Two separate translocations, both involving the Hippo pathway, were recently associated with EHE. The first, t(1;3), results in the fusion of the tafazzin gene TAZ (also known as WWTR1) and the calmodulin-binding transcriptional activator 1 gene CAMTA1.158,159,160,161 The second translocation results in the fusion of the yes-associated protein 1 gene YAP1 and the transcription factor binding to IGHM gene TFE3.162 YAP and TAZ are transcription factors involved in the Hippo pathway and, in normal cells, are involved in regulating cell size. The role of YAP and TAZ in cancer was reviewed recently.163 In endothelial cells, endoglin activation leads to YAP translocation to the nucleus and induction of extracellular matrix remodeling and secretion of pro-inflammatory chemokines.164 An additional translocation involves chromosomes 10p13 and 14q24; this specific translocation may involve placental growth factor (PlGF) and serve as a driver of EHE in some patients.165
The pseudomyogenic hemangioendothelioma subtype is also associated with a balanced chromosomal translocation, t(7;19)(q22;q13), which results in a fusion of the serpin peptidase inhibitor, clade E (nexin, plasminogen activator inhibitor type 1), member 1 gene SERPINE1 and FBJ murine osteosarcoma viral oncogene homolog B gene FOSB. This fusion is believed to lead to expression of the FOSB transcription factor under the SERPINE1 promoter.166 Although VEGFR1 is generally considered a decoy receptor that sequesters VEGF from VEGFR2, PlGF was previously shown to induce FOSB transcription via VEGFR1 independent from VEGFR2.167
Most studies that looked at angiosarcoma cytogenetics identified complex cytogenetics.12, 13, 168 Aberrations included gain or loss of entire chromosomes as well as partial chromosomes; interestingly, 2 of 8 cases in one series had duplication of the region on chromosome 4q that contains KIT and VEGFR2.12 A fusion of the Nucleoporin 160 kDa gene NUP160 and Solute Carrier Family 43, Member 3 gene SLC43A3, both on chromosome 11, was recently found in both primary angiosarcoma specimens and an established angiosarcoma cell line.169
Conclusions and future directions
Endothelial cell malignancies are characterized by dysregulation in multiple pathways that are highly regulated in endothelial cells for normal vascular development, as well as by some of the more typical oncogenic pathways found in other cancers. Aberrant activation of other regulatory pathways may explain why the majority of these tumors do not respond to VEGF-targeted therapies. Patient-derived cell lines and model systems that better replicate the biology seen in human angiosarcomas and hemangioendotheliomas are urgently needed to further our understanding of these rare tumors. The currently available genetically engineered mouse models of angiosarcoma are driven by knockout of Notch pathway components or FoxO,170 or overactivation of the mTOR pathway, but these may not reflect findings in human angiosarcomas. Canine models exist, but these are not practical for large scale in vivo research.
Future investigation should focus on mechanisms of adaptive resistance in those with initial responses to angiogenesis inhibitors. Understanding the mechanisms by which vascular tumors have primary or adaptive resistance to anti-angiogenic therapies can guide future research and treatment paradigms not only for malignancies of endothelial cell origin, but also for other cancers (e.g., ovarian,171 lung,172 and colon173). Future studies should also focus on rational drug combinations to block oncogenic pathways, as well as evaluating combinations of targeted therapy with conventional modalities such as chemotherapy and radiotherapy. The low incidence of these tumors limits the amount of tissue available for clinical and correlative research. Modified clinical trial designs and increased multi-institutional collaboration are needed to ensure sufficient sample sizes and to accelerate clinical studies of these rare tumors.
References
Fury, M. G., Antonescu, C. R., Van Zee, K. J., Brennan, M. F. & Maki, R. G. A 14-year retrospective review of angiosarcoma: clinical characteristics, prognostic factors, and treatment outcomes with surgery and chemotherapy. Cancer J. 11, 241–247 (2005).
Ravi, V. & Patel, S. Vascular sarcomas. Curr. Oncol. Rep. 15, 347–355 (2013).
Antonescu, C. Malignant vascular tumors--an update. Mod. Pathol. 27(Suppl 1), S30–S38 (2014).
Cioffi, A., Reichert, S., Antonescu, C. R. & Maki, R. G. Angiosarcomas and other sarcomas of endothelial origin. Hematol. Oncol. Clin. North Am. 27, 975–988 (2013).
Young, R. J., Brown, N. J., Reed, M. W., Hughes, D. & Woll, P. J. Angiosarcoma. Lancet Oncol. 11, 983–991 (2010).
Ryu, H. S., Lee, S. S., Choi, H. S., Baek, H. & Koh, J. S. A case of pulmonary malignant epithelioid hemangioendothelioma misdiagnosed as adenocarcinoma by fine needle aspiration cytology. Diagn. Cytopathol. 39, 801–807 (2011).
Guo, T. et al. Consistent MYC and FLT4 gene amplification in radiation-induced angiosarcoma but not in other radiation-associated atypical vascular lesions. Genes Chromosomes Cancer 50, 25–33 (2011).
Biggar, R. J., Chaturvedi, A. K., Goedert, J. J., Engels, E. A. & Study, H. A. C. M. AIDS-related cancer and severity of immunosuppression in persons with AIDS. J. Natl. Cancer Inst. 99, 962–972 (2007).
Parkin, D. M. et al. Part I: cancer in Indigenous Africans--burden, distribution, and trends. Lancet Oncol. 9, 683–692 (2008).
Itakura, E., Yamamoto, H., Oda, Y. & Tsuneyoshi, M. Detection and characterization of vascular endothelial growth factors and their receptors in a series of angiosarcomas. J. Surg. Oncol. 97, 74–81 (2008).
Antonescu, C. R. et al. KDR activating mutations in human angiosarcomas are sensitive to specific kinase inhibitors. Cancer Res. 69, 7175–7179 (2009).
Kunze, K. et al. A recurrent activating PLCG1 mutation in cardiac angiosarcomas increases apoptosis resistance and invasiveness of endothelial cells. Cancer Res. 74, 6173–6183 (2014).
Behjati, S. et al. Recurrent PTPRB and PLCG1 mutations in angiosarcoma. Nat. Genet. 46, 376–379 (2014).
Sun, S. G. et al. Constitutive activation of mTORC1 in endothelial cells leads to the development and progression of lymphangiosarcoma through VEGF autocrine signaling. Cancer Cell 28, 758–772 (2015).
Cornali, E. et al. Vascular endothelial growth factor regulates angiogenesis and vascular permeability in Kaposi’s sarcoma. Am. J. Pathol. 149, 1851–1869 (1996).
Masood, R. et al. Vascular endothelial growth factor/vascular permeability factor is an autocrine growth factor for AIDS-Kaposi sarcoma. Proc. Natl Acad. Sci. U.S.A. 94, 979–984 (1997).
Stallone, G. et al. ID2-VEGF-related pathways in the pathogenesis of Kaposi’s sarcoma: a link disrupted by rapamycin. Am. J. Transplant. 9, 558–566 (2009).
Bais, C. et al. G-protein-coupled receptor of Kaposi’s sarcoma-associated herpesvirus is a viral oncogene and angiogenesis activator. Nature 391, 86–89 (1998).
Bais, C. et al. Kaposi’s sarcoma associated herpesvirus G protein-coupled receptor immortalizes human endothelial cells by activation of the VEGF receptor-2/ KDR. Cancer Cell 3, 131–143 (2003).
Theurillat, J. P. et al. Morphologic changes and altered gene expression in an epithelioid hemangioendothelioma during a ten-year course of disease. Pathol. Res. Pract. 199, 165–170 (2003).
Emamaullee, J. A. et al. Vascular endothelial growth factor expression in hepatic epithelioid hemangioendothelioma: Implications for treatment and surgical management. Liver Transpl. 16, 191–197 (2010).
Agulnik, M. et al. An open-label, multicenter, phase II study of bevacizumab for the treatment of angiosarcoma and epithelioid hemangioendotheliomas. Ann. Oncol. 24, 257–263 (2013).
Ray-Coquard, I. L. et al. Paclitaxel given once per week with or without bevacizumab in patients with advanced angiosarcoma: a randomized Phase II trial. J. Clin. Oncol. 33, 2797–2802 (2015).
Maki, R. G. et al. Phase II study of sorafenib in patients with metastatic or recurrent sarcomas. J. Clin. Oncol. 27, 3133–3140 (2009).
Ray-Coquard, I. et al. Sorafenib for patients with advanced angiosarcoma: a phase II Trial from the French Sarcoma Group (GSF/GETO). Oncologist 17, 260–266 (2012).
von Mehren, M. et al. Phase 2 Southwest oncology group-directed intergroup trial (S0505) of sorafenib in advanced soft tissue sarcomas. Cancer 118, 770–776 (2012).
Tomita, H. et al. Angiosarcoma of the scalp successfully treated with pazopanib. J. Am. Acad. Dermatol. 70, e19–e21 (2014).
Miura, H. & Shirai, H. Low-dose administration of oral pazopanib for the treatment of recurrent angiosarcoma. Clin. Exp. Dermatol. 40, 575–577 (2015).
Silva, E. et al. Refractory angiosarcoma of the breast with VEGFR2 upregulation successfully treated with sunitinib. Breast J. 21, 205–207 (2015).
Lu, H. J. et al. Refractory cutaneous angiosarcoma successfully treated with sunitinib. Br. J. Dermatol. 169, 204–206 (2013).
George, S. et al. Multicenter phase II trial of sunitinib in the treatment of nongastrointestinal stromal tumor sarcomas. J. Clin. Oncol. 27, 3154–3160 (2009).
van der Graaf, W. T. et al. Pazopanib for metastatic soft-tissue sarcoma (PALETTE): a randomised, double-blind, placebo-controlled phase 3 trial. Lancet 379, 1879–1886 (2012).
Bally, O. et al. Eight years tumor control with pazopanib for a metastatic resistant epithelioid hemangioendothelioma. Clin. Sarcoma Res. 5, 12 (2015).
Trautmann, K., Bethke, A., Ehninger, G. & Folprecht, G. Bevacizumab for recurrent hemangioendothelioma. Acta Oncol. 50, 153–154 (2011).
Prochilo, T. et al. Targeting VEGF-VEGFR pathway by sunitinib in peripheral primitive neuroectodermal tumor, paraganglioma and epithelioid hemangioendothelioma: three case reports. Case Rep. Oncol. 6, 90–97 (2013).
Uldrick, T. S. et al. Phase II study of bevacizumab in patients with HIV-associated Kaposi’s sarcoma receiving antiretroviral therapy. J. Clin. Oncol. 30, 1476–1483 (2012).
Thurston, G. & Daly, C. The complex role of angiopoietin-2 in the angiopoietin-tie signaling pathway. Cold Spring Harb. Perspect. Med. 2, a006550 (2012).
Maisonpierre, P. C. et al. Angiopoietin-2, a natural antagonist for Tie2 that disrupts in vivo angiogenesis. Science 277, 55–60 (1997).
Yuan, H. T., Khankin, E. V., Karumanchi, S. A. & Parikh, S. M. Angiopoietin 2 is a partial agonist/antagonist of Tie2 signaling in the endothelium. Mol. Cell. Biol. 29, 2011–2022 (2009).
Huang, H., Bhat, A., Woodnutt, G. & Lappe, R. Targeting the ANGPT-TIE2 pathway in malignancy. Nat. Rev. Cancer 10, 575–585 (2010).
Li, Z. et al. Embryonic stem cell tumor model reveals role of vascular endothelial receptor tyrosine phosphatase in regulating Tie2 pathway in tumor angiogenesis. Proc. Natl Acad. Sci. U.S.A. 106, 22399–22404 (2009).
Winderlich, M. et al. VE-PTP controls blood vessel development by balancing Tie-2 activity. J. Cell Biol. 185, 657–671 (2009).
Amo, Y., Masuzawa, M., Hamada, Y. & Katsuoka, K. Observations on angiopoietin 2 in patients with angiosarcoma. Br. J. Dermatol. 150, 1028–1029 (2004).
Buehler, D. et al. Expression of angiopoietin-TIE system components in angiosarcoma. Mod. Pathol. 26, 1032–1040 (2013).
Brown, L. F., Dezube, B. J., Tognazzi, K., Dvorak, H. F. & Yancopoulos, G. D. Expression of Tie1, Tie2, and angiopoietins 1, 2, and 4 in Kaposi’s sarcoma and cutaneous angiosarcoma. Am. J. Pathol. 156, 2179–2183 (2000).
D’Angelo, S.P. et al. Alliance A091103 a phase II study of the angiopoietin 1 and 2 peptibody trebananib for the treatment of angiosarcoma. Cancer Chemother. Pharmacol. 75, 629–638 (2015).
Hellstrom, M. et al. Dll4 signalling through Notch1 regulates formation of tip cells during angiogenesis. Nature 445, 776–780 (2007).
Kopan, R. & Ilagan, M. X. The canonical Notch signaling pathway: unfolding the activation mechanism. Cell 137, 216–233 (2009).
Williams, C. K., Li, J. L., Murga, M., Harris, A. L. & Tosato, G. Up-regulation of the Notch ligand Delta-like 4 inhibits VEGF-induced endothelial cell function. Blood 107, 931–939 (2006).
Benedito, R. et al. The notch ligands Dll4 and Jagged1 have opposing effects on angiogenesis. Cell 137, 1124–1135 (2009).
Yan, M. et al. Chronic DLL4 blockade induces vascular neoplasms. Nature 463, E6–E7 (2010).
Dill, M. T. et al. Disruption of NOTCH1 induces vascular remodeling, intussusceptive angiogenesis, and angiosarcomas in livers of mice. Gastroenterology 142, 967–U464 (2012).
Liu, Z. et al. Notch1 loss of heterozygosity causes vascular tumors and lethal hemorrhage in mice. J. Clin. Invest. 121, 800–808 (2011).
Lindahl, P., Johansson, B. R., Leveen, P. & Betsholtz, C. Pericyte loss and microaneurysm formation in PDGF-B-deficient mice. Science 277, 242–245 (1997).
Tallquist, M. & Kazlauskas, A. PDGF signaling in cells and mice. Cytokine Growth Factor Rev. 15, 205–213 (2004).
Kundra, V. et al. Regulation of chemotaxis by the platelet-derived growth factor receptor-beta. Nature 367, 474–476 (1994).
Dickerson, E. B. et al. Imatinib and dasatinib inhibit hemangiosarcoma and implicate PDGFR-beta and Src in tumor growth. Transl. Oncol. 6, 158–168 (2013).
Kiesel, H., Muller, A. M., Schmitt-Graeff, A. & Veelken, H. Dramatic and durable efficacy of imatinib in an advanced angiosarcoma without detectable KIT and PDGFRA mutations. Cancer Biol. Ther. 8, 319–321 (2009).
Chugh, R. et al. Phase II multicenter trial of imatinib in 10 histologic subtypes of sarcoma using a bayesian hierarchical statistical model. J. Clin. Oncol. 27, 3148–3153 (2009).
O’Reilly, M. S. et al. Angiostatin: a novel angiogenesis inhibitor that mediates the suppression of metastases by a Lewis lung carcinoma. Cell 79, 315–328 (1994).
O’Reilly, M. S. et al. Endostatin: an endogenous inhibitor of angiogenesis and tumor growth. Cell 88, 277–285 (1997).
Ye, J., Li, X. F., Wang, Y. D. & Yuan, Y. Long-term survival of a patient with scalp angiosarcoma and multiple metastases treated using combination therapy: a case report. Oncol. Lett. 9, 1725–1728 (2015).
Syed, S. P., Martin, A. M., Haupt, H. M., Arenas-Elliot, C. P. & Brooks, J. J. Angiostatin receptor annexin II in vascular tumors including angiosarcoma. Hum. Pathol. 38, 508–513 (2007).
Lannutti, B. J., Gately, S. T., Quevedo, M. E., Soff, G. A. & Paller, A. S. Human angiostatin inhibits murine hemangioendothelioma tumor growth in vivo. Cancer Res. 57, 5277–5280 (1997).
Boye, E. et al. Clonality and altered behavior of endothelial cells from hemangiomas. J. Clin. Invest. 107, 745–752 (2001).
Khan, Z. A. et al. Endothelial progenitor cells from infantile hemangioma and umbilical cord blood display unique cellular responses to endostatin. Blood 108, 915–921 (2006).
Miller, D. W. et al. Elevated expression of endoglin, a component of the TGF-beta-receptor complex, correlates with proliferation of tumor endothelial cells. Int. J. Cancer 81, 568–572 (1999).
Wong, S. H., Hamel, L., Chevalier, S. & Philip, A. Endoglin expression on human microvascular endothelial cells association with betaglycan and formation of higher order complexes with TGF-beta signalling receptors. Eur. J. Biochem. 267, 5550–5560 (2000).
Lebrin, F. et al. Endoglin promotes endothelial cell proliferation and TGF-beta/ALK1 signal transduction. EMBO J. 23, 4018–4028 (2004).
Blanco, F. J. et al. S-endoglin expression is induced in senescent endothelial cells and contributes to vascular pathology. Circ. Res. 103, 1383–1392 (2008).
McAllister, K. A. et al. Endoglin, a TGF-beta binding protein of endothelial cells, is the gene for hereditary haemorrhagic telangiectasia type 1. Nat. Genet. 8, 345–351 (1994).
Fritchie, K., Attia, S., Okuno, S., Arndt, C. & Robinson, S. Abstract B237: CD105: A therapeutic target for sarcomas. Mol. Cancer Ther. 12, B237 (2013).
Hara, H. Endoglin (CD105) and claudin-5 expression in cutaneous angiosarcoma. Am. J. Dermatopathol. 34, 779–782 (2012).
Verbeke, S. L. et al. Active TGF-beta signaling and decreased expression of PTEN separates angiosarcoma of bone from its soft tissue counterpart. Mod. Pathol. 26, 1211–1221 (2013).
Attia, S. et al. in CTOS (Salt Lake City, UT, 2015).
Nakayama, M. et al. Spatial regulation of VEGF receptor endocytosis in angiogenesis. Nat. Cell Biol. 15, 249–260 (2013).
Bai, X. C. et al. Phospholipase C-gamma1 is required for cell survival in oxidative stress by protein kinase C. Biochem. J. 363, 395–401 (2002).
Covassin, L. D. et al. A genetic screen for vascular mutants in zebrafish reveals dynamic roles for Vegf/Plcg1 signaling during artery development. Dev. Biol. 329, 212–226 (2009).
Lawson, N. D., Mugford, J. W., Diamond, B. A. & Weinstein, B. M. Phospholipase C gamma-1 is required downstream of vascular endothelial growth factor during arterial development. Genes Dev. 17, 1346–1351 (2003).
Liao, H. J. et al. Absence of erythrogenesis and vasculogenesis in Plcg1-deficient mice. J. Biol. Chem. 277, 9335–9341 (2002).
Takahashi, T., Yamaguchi, S., Chida, K. & Shibuya, M. A single autophosphorylation site on KDR/Flk-1 is essential for VEGF-A-dependent activation of PLC-gamma and DNA synthesis in vascular endothelial cells. EMBO J. 20, 2768–2778 (2001).
Poulin, B., Sekiya, F. & Rhee, S. G. Intramolecular interaction between phosphorylated tyrosine-783 and the C-terminal Src homology 2 domain activates phospholipase C-gamma1. Proc. Natl Acad. Sci. U.S.A. 102, 4276–4281 (2005).
Bunney, T. D. et al. Structural and functional integration of the PLCgamma interaction domains critical for regulatory mechanisms and signaling deregulation. Structure 20, 2062–2075 (2012).
Park, J. B. et al. Phospholipase signalling networks in cancer. Nat. Rev. Cancer 12, 782–792 (2012).
Kadamur, G. & Ross, E. M. Mammalian phospholipase C. Annu. Rev. Physiol. 75, 127–154 (2013).
Prenen, H. et al. Phospholipase C gamma 1 (PLCG1) R707Q mutation is counterselected under targeted therapy in a patient with hepatic angiosarcoma. Oncotarget 6, 36418–36425 (2015).
Murali, R. et al. Targeted massively parallel sequencing of angiosarcomas reveals frequent activation of the mitogen activated protein kinase pathway. Oncotarget 6, 36041–36052 (2015).
Andersen, N. J., Boguslawski, E. B., Kuk, C. Y., Chambers, C. M. & Duesbery, N. S. Combined inhibition of MEK and mTOR has a synergic effect on angiosarcoma tumorgrafts. Int. J. Oncol. 47, 71–80 (2015).
Shiojima, I. & Walsh, K. Role of Akt signaling in vascular homeostasis and angiogenesis. Circ. Res. 90, 1243–1250 (2002).
Phung, T. L. et al. Akt1 and akt3 exert opposing roles in the regulation of vascular tumor growth. Cancer Res. 75, 40–50 (2015).
Lahat, G. et al. Angiosarcoma: clinical and molecular insights. Ann. Surg. 251, 1098–1106 (2010).
Perry, B. et al. AKT1 overexpression in endothelial cells leads to the development of cutaneous vascular malformations in vivo. Arch. Dermatol. 143, 504–506 (2007).
Du, W. et al. Vascular tumors have increased p70 S6-kinase activation and are inhibited by topical rapamycin. Lab. Invest. 93, 1115–1127 (2013).
Leech, J. D. et al. A vascular model of tsc1 deficiency accelerates renal tumor formation with accompanying hemangiosarcomas. Mol. Cancer Res. 13, 548–555 (2015).
Italiano, A. et al. Alterations of the p53 and PIK3CA/AKT/mTOR pathways in angiosarcomas: a pattern distinct from other sarcomas with complex genomics. Cancer 118, 5878–5887 (2012).
Meyer, N. & Penn, L. Z. Reflecting on 25 years with MYC. Nat. Rev. Cancer 8, 976–990 (2008).
Manner, J. et al. MYC high level gene amplification is a distinctive feature of angiosarcomas after irradiation or chronic lymphedema. Am. J. Pathol. 176, 34–39 (2010).
Verbeke, S. L. et al. Array CGH analysis identifies two distinct subgroups of primary angiosarcoma of bone. Genes Chromosomes Cancer 54, 72–81 (2015).
Styring, E. et al. Key roles for MYC, KIT and RET signaling in secondary angiosarcomas. Br. J. Cancer 111, 407–412 (2014).
Kulkarni, M. V. & Franklin, D. S. N-Myc is a downstream target of RET signaling and is required for transcriptional regulation of p18(Ink4c) by the transforming mutant RET(C634R). Mol. Oncol. 5, 24–35 (2011).
Italiano, A. et al. The miR-17-92 cluster and its target THBS1 are differentially expressed in angiosarcomas dependent on MYC amplification. Genes Chromosomes Cancer 51, 569–578 (2012).
Rothweiler, S. et al. Generation of a murine hepatic angiosarcoma cell line and reproducible mouse tumor model. Lab. Invest. 95, 351–362 (2015).
Rodriguez-Diez, E. et al. Cdk4 and Cdk6 cooperate in counteracting the INK4 family of inhibitors during murine leukemogenesis. Blood 124, 2380–2390 (2014).
Liu, J., Martin, H. J., Liao, G. & Hayward, S. D. The Kaposi’s sarcoma-associated herpesvirus LANA protein stabilizes and activates c-Myc. J. Virol. 81, 10451–10459 (2007).
Li, X., Chen, S., Feng, J., Deng, H. & Sun, R. Myc is required for the maintenance of Kaposi’s sarcoma-associated herpesvirus latency. J. Virol. 84, 8945–8948 (2010).
Delmore, J. E. et al. BET bromodomain inhibition as a therapeutic strategy to target c-Myc. Cell 146, 904–917 (2011).
Tolani, B., Gopalakrishnan, R., Punj, V., Matta, H. & Chaudhary, P. M. Targeting Myc in KSHV-associated primary effusion lymphoma with BET bromodomain inhibitors. Oncogene 33, 2928–2937 (2014).
Dews, M. et al. Augmentation of tumor angiogenesis by a Myc-activated microRNA cluster. Nat. Genet. 38, 1060–1065 (2006).
Knies-Bamforth, U. E., Fox, S. B., Poulsom, R., Evan, G. I. & Harris, A. L. c-Myc interacts with hypoxia to induce angiogenesis in vivo by a vascular endothelial growth factor-dependent mechanism. Cancer Res. 64, 6563–6570 (2004).
Ma, W. et al. Hepatic vascular tumors, angiectasis in multiple organs, and impaired spermatogenesis in mice with conditional inactivation of the VHL gene. Cancer Res. 63, 5320–5328 (2003).
Rathmell, W. K., Acs, G., Simon, M. C. & Vaughn, D. J. HIF transcription factor expression and induction of hypoxic response genes in a retroperitoneal angiosarcoma. Anticancer Res. 24, 167–169 (2004).
Abedalthagafi, M. et al. Sporadic cutaneous angiosarcomas generally lack hypoxia-inducible factor 1alpha: a histologic and immunohistochemical study of 45 cases. Ann. Diagn. Pathol. 14, 15–22 (2010).
Carroll, P. A., Kenerson, H. L., Yeung, R. S. & Lagunoff, M. Latent Kaposi’s sarcoma-associated herpesvirus infection of endothelial cells activates hypoxia-induced factors. J. Virol. 80, 10802–10812 (2006).
Sodhi, A. et al. The Kaposi’s sarcoma-associated herpes virus G protein-coupled receptor up-regulates vascular endothelial growth factor expression and secretion through mitogen-activated protein kinase and p38 pathways acting on hypoxia-inducible factor 1alpha. Cancer Res. 60, 4873–4880 (2000).
Jham, B. C. et al. Amplification of the angiogenic signal through the activation of the TSC/mTOR/HIF axis by the KSHV vGPCR in Kaposi’s sarcoma. PLoS ONE 6, e19103 (2011).
Cai, Q. L., Knight, J. S., Verma, S. C., Zald, P. & Robertson, E. S. EC5S ubiquitin complex is recruited by KSHV latent antigen LANA for degradation of the VHL and p53 tumor suppressors. PLoS Pathog. 2, e116 (2006).
Cai, Q., Murakami, M., Si, H. & Robertson, E. S. A potential alpha-helix motif in the amino terminus of LANA encoded by Kaposi’s sarcoma-associated herpesvirus is critical for nuclear accumulation of HIF-1alpha in normoxia. J. Virol. 81, 10413–10423 (2007).
Mesri, E. A., Cesarman, E. & Boshoff, C. Kaposi’s sarcoma and its associated herpesvirus. Nat. Rev. Cancer 10, 707–719 (2010).
Lan, K. et al. Kaposi’s sarcoma herpesvirus-encoded latency-associated nuclear antigen stabilizes intracellular activated Notch by targeting the Sel10 protein. Proc. Natl Acad. Sci. U.S.A. 104, 16287–16292 (2007).
Wang, X. et al. Latency-associated nuclear antigen of Kaposi sarcoma-associated herpesvirus promotes angiogenesis through targeting notch signaling effector Hey1. Cancer Res. 74, 2026–2037 (2014).
Liang, Y., Chang, J., Lynch, S. J., Lukac, D. M. & Ganem, D. The lytic switch protein of KSHV activates gene expression via functional interaction with RBP-Jkappa (CSL), the target of the Notch signaling pathway. Genes Dev. 16, 1977–1989 (2002).
Liu, R. et al. KSHV-induced notch components render endothelial and mural cell characteristics and cell survival. Blood 115, 887–895 (2010).
Domenga, V. et al. Notch3 is required for arterial identity and maturation of vascular smooth muscle cells. Genes Dev. 18, 2730–2735 (2004).
Gasperini, P. et al. Kaposi sarcoma herpesvirus promotes endothelial-to-mesenchymal transition through Notch-dependent signaling. Cancer Res. 72, 1157–1169 (2012).
Medici, D. et al. Conversion of vascular endothelial cells into multipotent stem-like cells. Nat. Med. 16, 1400–1406 (2010).
Curry, C. L. et al. Notch inhibition in Kaposi’s sarcoma tumor cells leads to mitotic catastrophe through nuclear factor-kappaB signaling. Mol. Cancer Ther. 6, 1983–1992 (2007).
Sodhi, A. et al. The TSC2/mTOR pathway drives endothelial cell transformation induced by the Kaposi’s sarcoma-associated herpesvirus G protein-coupled receptor. Cancer Cell 10, 133–143 (2006).
Stallone, G. et al. Sirolimus for Kaposi’s sarcoma in renal-transplant recipients. N. Engl. J. Med. 352, 1317–1323 (2005).
Krown, S. E. et al. Rapamycin with antiretroviral therapy in AIDS-associated Kaposi sarcoma: an AIDS Malignancy Consortium study. J. Acquir. Immun. Defic. Syndr. 59, 447–454 (2012).
Sola, F. et al. Endothelial cells overexpressing basic fibroblast growth factor (FGF-2) induce vascular tumors in immunodeficient mice. Angiogenesis 1, 102–116 (1997).
Ensoli, B. et al. Synergy between basic fibroblast growth factor and HIV-1 Tat protein in induction of Kaposi’s sarcoma. Nature 371, 674–680 (1994).
Ensoli, B. et al. Block of AIDS-Kaposi’s sarcoma (KS) cell growth, angiogenesis, and lesion formation in nude mice by antisense oligonucleotide targeting basic fibroblast growth factor. A novel strategy for the therapy of KS. J. Clin. Invest. 94, 1736–1746 (1994).
Barillari, G. et al. The Tat protein of human immunodeficiency virus type-1 promotes vascular cell growth and locomotion by engaging the alpha5beta1 and alphavbeta3 integrins and by mobilizing sequestered basic fibroblast growth factor. Blood 94, 663–672 (1999).
Sgadari, C. et al. Fibroblast growth factor-2 and the HIV-1 Tat protein synergize in promoting Bcl-2 expression and preventing endothelial cell apoptosis: implications for the Pathogenesis of AIDS-Associated Kaposi’s Sarcoma. Int. J. Vasc. Med. 2011, 452729 (2011).
Samaniego, F. et al. Vascular endothelial growth factor and basic fibroblast growth factor present in Kaposi’s sarcoma (KS) are induced by inflammatory cytokines and synergize to promote vascular permeability and KS lesion development. Am. J. Pathol. 152, 1433–1443 (1998).
Albini, A. et al. The angiogenesis induced by HIV-1 tat protein is mediated by the Flk-1/KDR receptor on vascular endothelial cells. Nat. Med. 2, 1371–1375 (1996).
Ganju, R. K. et al. Human immunodeficiency virus tat modulates the Flk-1/KDR receptor, mitogen-activated protein kinases, and components of focal adhesion in Kaposi’s sarcoma cells. J. Virol. 72, 6131–6137 (1998).
Fujii, H. et al. CD8(+) tumor-infiltrating lymphocytes at primary sites as a possible prognostic factor of cutaneous angiosarcoma. Int. J. Cancer 134, 2393–2402 (2014).
D’Angelo, S. P. et al. Prevalence of tumor-infiltrating lymphocytes and PD-L1 expression in the soft tissue sarcoma microenvironment. Hum. Pathol. 46, 357–365 (2015).
Kim, J. R. et al. Tumor infiltrating PD1-positive lymphocytes and the expression of PD-L1 predict poor prognosis of soft tissue sarcomas. PLoS ONE 8, e82870 (2013).
Murdoch, C., Muthana, M., Coffelt, S. B. & Lewis, C. E. The role of myeloid cells in the promotion of tumour angiogenesis. Nat. Rev. Cancer 8, 618–631 (2008).
Ishibashi, M. et al. Successful treatment of MMP-9-expressing angiosarcoma with low-dose docetaxel and bisphosphonate. Case Rep. Dermatol. 4, 5–9 (2012).
Kambayashi, Y. et al. Comparison of immunosuppressive and cytotoxic cells in angiosarcoma: development of a possible supportive therapy for angiosarcoma. Dermatology 227, 14–20 (2013).
Fujimura, T., Kambayashi, Y., Furudate, S., Kakizaki, A. & Aiba, S. Immunomodulatory effect of bisphosphonate risedronate sodium on CD163+arginase 1+M2 macrophages: the development of a possible supportive therapy for angiosarcoma. Clin. Dev. Immunol. 2013, 325412 (2013).
Yang, J. et al. INK4a/ARF [corrected] inactivation with activation of the NF-kappaB/IL-6 pathway is sufficient to drive the development and growth of angiosarcoma. Cancer Res. 72, 4682–4695 (2012).
Kim, J. H. et al. Interleukin-8 promotes canine hemangiosarcoma growth by regulating the tumor microenvironment. Exp. Cell Res. 323, 155–164 (2014).
Fiorelli, V., Gendelman, R., Samaniego, F., Markham, P. D. & Ensoli, B. Cytokines from activated T cells induce normal endothelial cells to acquire the phenotypic and functional features of AIDS-Kaposi’s sarcoma spindle cells. J. Clin. Invest. 95, 1723–1734 (1995).
Fiorelli, V. et al. Gamma-interferon produced by CD8+T cells infiltrating Kaposi’s sarcoma induces spindle cells with angiogenic phenotype and synergy with human immunodeficiency virus-1 Tat protein: an immune response to human herpesvirus-8 infection? Blood 91, 956–967 (1998).
Meis-Kindblom, J. M. & Kindblom, L. G. Angiosarcoma of soft tissue: a study of 80 cases. Am. J. Surg. Pathol. 22, 683–697 (1998).
Hart, J. & Mandavilli, S. Epithelioid angiosarcoma: a brief diagnostic review and differential diagnosis. Arch. Pathol. Lab. Med. 135, 268–272 (2011).
Cooke, V. G. et al. Pericyte depletion results in hypoxia-associated epithelial-to-mesenchymal transition and metastasis mediated by met signaling pathway. Cancer Cell 21, 66–81 (2012).
Boscolo, E., Mulliken, J. B. & Bischoff, J. Pericytes from infantile hemangioma display proangiogenic properties and dysregulated angiopoietin-1. Arterioscler. Thromb. Vasc. Biol. 33, 501–509 (2013).
Fukumura, D. et al. Tumor induction of VEGF promoter activity in stromal cells. Cell 94, 715–725 (1998).
Orimo, A. et al. Stromal fibroblasts present in invasive human breast carcinomas promote tumor growth and angiogenesis through elevated SDF-1/CXCL12 secretion. Cell 121, 335–348 (2005).
Qin, Z., Dai, L., Slomiany, M. G., Toole, B. P. & Parsons, C. Direct activation of emmprin and associated pathogenesis by an oncogenic herpesvirus. Cancer Res. 70, 3884–3889 (2010).
Dai, L. et al. Kaposi sarcoma-associated herpesvirus (KSHV) induces a functional tumor-associated phenotype for oral fibroblasts. Cancer Lett. 318, 214–220 (2012).
Salahuddin, S. Z. et al. Angiogenic properties of Kaposi’s sarcoma-derived cells after long-term culture in vitro. Science 242, 430–433 (1988).
Tanas, M. R. et al. Identification of a disease-defining gene fusion in epithelioid hemangioendothelioma. Sci. Transl. Med. 3, 98ra82 (2011).
Anderson, T. et al. Thoracic epithelioid malignant vascular tumors: a clinicopathologic study of 52 cases with emphasis on pathologic grading and molecular studies of WWTR1-CAMTA1 fusions. Am. J. Surg. Pathol. 39, 132–139 (2015).
Errani, C. et al. A novel WWTR1-CAMTA1 gene fusion is a consistent abnormality in epithelioid hemangioendothelioma of different anatomic sites. Genes Chromosomes Cancer 50, 644–653 (2011).
Patel, N. R. et al. Molecular characterization of epithelioid haemangioendotheliomas identifies novel WWTR1-CAMTA1 fusion variants. Histopathology 67, 699–708 (2015).
Antonescu, C. R. et al. Novel YAP1-TFE3 fusion defines a distinct subset of epithelioid hemangioendothelioma. Genes Chromosomes Cancer 52, 775–784 (2013).
Moroishi, T., Hansen, C. G. & Guan, K. L. The emerging roles of YAP and TAZ in cancer. Nat. Rev. Cancer 15, 73–79 (2015).
Young, K. et al. BMP9 crosstalk with the hippo pathway regulates endothelial cell matricellular and chemokine responses. PLoS ONE 10, e0122892 (2015).
He, M., Das, K., Blacksin, M., Benevenia, J. & Hameed, M. A translocation involving the placental growth factor gene is identified in an epithelioid hemangioendothelioma. Cancer Genet. Cytogenet. 168, 150–154 (2006).
Walther, C. et al. A novel SERPINE1-FOSB fusion gene results in transcriptional up-regulation of FOSB in pseudomyogenic haemangioendothelioma. J. Pathol. 232, 534–540 (2014).
Holmes, D. I. & Zachary, I. Placental growth factor induces FosB and c-Fos gene expression via Flt-1 receptors. FEBS Lett. 557, 93–98 (2004).
Wong, K. F. et al. Sinonasal angiosarcoma with marrow involvement at presentation mimicking malignant lymphoma: cytogenetic analysis using multiple techniques. Cancer Genet. Cytogenet. 129, 64–68 (2001).
Shimozono, N. et al. NUP160-SLC43A3 is a novel recurrent fusion oncogene in angiosarcoma. Cancer Res. 75, 4458–4465 (2015).
Paik, J. H. et al. FoxOs are lineage-restricted redundant tumor suppressors and regulate endothelial cell homeostasis. Cell 128, 309–323 (2007).
Pujade-Lauraine, E. et al. Bevacizumab combined with chemotherapy for platinum-resistant recurrent ovarian cancer: The AURELIA open-label randomized phase III trial. J. Clin. Oncol. 32, 1302–1308 (2014).
Sandler, A. et al. Paclitaxel-carboplatin alone or with bevacizumab for non-small-cell lung cancer. N. Engl. J. Med. 355, 2542–2550 (2006).
Hurwitz, H. et al. Bevacizumab plus irinotecan, fluorouracil, and leucovorin for metastatic colorectal cancer. N. Engl. J. Med. 350, 2335–2342 (2004).
Cohen, E. E. et al. Phase I studies of sirolimus alone or in combination with pharmacokinetic modulators in advanced cancer patients. Clin. Cancer Res. 18, 4785–4793 (2012).
Leaute-Labreze, C. et al. Propranolol for severe hemangiomas of infancy. N. Engl. J. Med. 358, 2649–2651 (2008).
Mazereeuw-Hautier, J. et al. Efficacy of propranolol in hepatic infantile hemangiomas with diffuse neonatal hemangiomatosis. J. Pediatr. 157, 340–342 (2010).
Denoyelle, F. et al. Role of propranolol in the therapeutic strategy of infantile laryngotracheal hemangioma. Int. J. Pediatr. Otorhinolaryngol. 73, 1168–1172 (2009).
Stiles, J. M. et al. Targeting of beta adrenergic receptors results in therapeutic efficacy against models of hemangioendothelioma and angiosarcoma. PLoS ONE 8, e60021 (2013).
Drolet, B. A. et al. Consensus-derived practice standards plan for complicated Kaposiform hemangioendothelioma. J. Pediatr. 163, 285–291 (2013).
Bauman, N. M. et al. Propranolol vs prednisolone for symptomatic proliferating infantile hemangiomas: a randomized clinical trial. JAMA Otolaryngol. Head Neck Surg. 140, 323–330 (2014).
Acknowledgements
We thank Joseph Munch, Kathryn Hale, and Katelyn Werner for editorial support and apologize to colleagues whose original work could not be cited due to space limitations. M.J.W. is supported by grants CCSG CA016672 and T32CA009666, and the Quad W-AACR Fellowship in Clinical/Translational Sarcoma Research. A.K.S. is supported by The Frank McGraw Memorial Chair in Cancer Research and the American Cancer Society Research Professor Award.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interest
The authors declare no competing interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Wagner, M.J., Ravi, V., Menter, D.G. et al. Endothelial cell malignancies: new insights from the laboratory and clinic. npj Precision Onc 1, 11 (2017). https://doi.org/10.1038/s41698-017-0013-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41698-017-0013-2
This article is cited by
-
Combined VEGFR and MAPK pathway inhibition in angiosarcoma
Scientific Reports (2021)
-
Endothelial sprouting, proliferation, or senescence: tipping the balance from physiology to pathology
Cellular and Molecular Life Sciences (2021)