Abstract
This case–control study aimed to identify the clinical characteristics and explore the risk factors for liver fibrosis in metabolic associated fatty liver disease (MAFLD) patients with hepatitis B virus (HBV) infection. The patients were grouped into MAFLD + HBV and MAFLD (without HBV infection). Propensity score matching (PSM) was used to match baseline features between the groups. We included 401 patients with biopsy-proven MAFLD, 179 of whom had HBV infection. A total of 83 pairs were successfully matched via PSM, and steatosis scores and ballooning in the MAFLD + HBV group were lower than those in the MAFLD group, while the inflammation scores and liver fibrosis stages were higher. After adjusted for confounding factors, HBV infection was associated with a higher risk of significant liver fibrosis in patients with MAFLD [odds ratio (OR): 3.140, P = 0.003]. Overall, 43.58% (78/179) of patients in the MAFLD + HBV group had significant liver fibrosis. Further multivariate regression analysis, hypertension (OR: 2.640; P = 0.031), type 2 diabetes (OR: 4.939; P = 0.035), and elevated glutamyl-transferase levels (OR: 3.980; P = 0.001) were risk factors for liver fibrosis in the MAFLD + HBV group. This suggests metabolic rather than viral factors are more closely associated with liver fibrosis in MAFLD patients with HBV infection.
Similar content being viewed by others
Introduction
Over the past 20 years, under the influence of overnutrition and sedentary lifestyle in modern society, non-alcoholic fatty liver disease (NAFLD) has increased rapidly, posing a major health and economic burden to all societies1,2. In 2020, the International Fatty Liver Expert Group announced that NAFLD was renamed as metabolic dysfunction-associated fatty liver disease (MAFLD). Both metabolic disorders and hepatic steatosis are necessary for the diagnosis of MAFLD3, but unlike NAFLD, does not require the exclusion of other defined etiologies of chronic liver disease, such as viral infection or excessive alcohol intake4,5.
Hepatitis B virus (HBV) infections are common in Asia. The prevalence of HBV infection is approximately 5%-6% in the general Chinese population6. Therefore, a large population is potentially at risk of developing MAFLD with concurrent HBV infection, forming an important subtype of MAFLD. The coexistence of metabolic dysfunction and viral infection is a striking feature of MAFLD complicated by HBV infection, which may act synergistically to significantly increase the risk of cirrhosis, hepatocellular carcinoma (HCC), and liver-related deaths7,8,9,10.
The progression of liver fibrosis has been generally accepted as a reliable factor for predicting the overall or liver-related death rate among MAFLD cases11,12,13,14. However, the clinical characteristics and risk factors for liver fibrosis in MAFLD patients infected with HBV remain unclear, and whether metabolic or viral factors were more closely related to liver fibrosis is also currently unknown. To address these issues, we compared the metabolic, etiological, and histological features of MAFLD patients with and without HBV infection in a large biopsy-proven cohort, and then explored the risk factors of liver fibrosis in MAFLD patients infected with HBV.
Methods
Study subjects and design
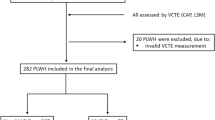
This cross-sectional study included all patients with liver stiffness measurements greater than 7 kPa as determined by Fibroscan prior to liver biopsy between 2011 and 2021 at the Affiliated Hospital of Hangzhou Normal University (Hangzhou, China). Individuals with the following conditions were excluded (Fig. 1): (1) evidence of chronic liver diseases such as chronic viral hepatitis C, alcoholic liver disease, autoimmune liver disease; (2) history of malignancy or missing data concerning weight, heigh, or etiological markers, including hepatitis B surface antigen (HBsAg), hepatitis B e-Antigen (HBeAg), and hepatitis B virus deoxyribonucleic acid (HBV DNA); (3) use of hypoglycemic or antilipidemic drugs exerting potential effects on liver fibrosis; (4) patients with hepatitis B receiving antiviral therapy.
This study conformed to the ethical guidelines of the Declaration of Helsinki and was approved by the Ethics Committee of the Affiliated Hospital of Hangzhou Normal University (Approval Number/ID: 2020 (02)-KS-022). As this was an observational retrospective study, the requirement for informed consent was waived by the Ethics Committee.
Clinical examination, biochemical analyses, and biopsy assessment
Diastolic blood pressure (DBP), systolic blood pressure (SBP), height, and body weight were measured by professional physicians in accordance with standard protocols. Body mass index (BMI) was calculated as weight (kg) divided by height (m) squared (kg/m2). Blood samples were obtained after 8 h of fasting, and routine blood and biochemical tests were conducted to assess the following: fasting plasma glucose (FPG), triglycerides (TG), total cholesterol (TC), high/low-density lipoprotein cholesterol (HDL-c/LDL-c), alanine aminotransferase (ALT), gamma-glutamyl-transferase (GGT), aspartate aminotransferase (AST), albumin (ALB), glycated hemoglobin (HbA1c), and serum uric acid (SUA). The biochemical tests were performed using an automated biochemical analyzer in accordance with the manufacturer’s instructions (Model 7180; Hitachi, Tokyo, Japan). Serological markers of HBV infection, including HBsAg, HBcAb, HBeAg, and hepatitis B e-Antibody (anti-HBe), were obtained using commercially available enzyme immunoassays. Serum HBV DNA levels were quantified using a commercially available real-time polymerase chain reaction assay in accordance with the manufacturer’s instructions, with a linear dynamic detection range of 3 × 101–10 × 109 IU/ml.
All liver biopsies were reassessed by three experienced histopathologists blinded to participant details. The steatosis score (positive if > 5%, according to the Brunt classification), stage of fibrosis (based on a meta-analysis of histological data for viral hepatitis score), ballooning, and degree of inflammation were evaluated 15,16,17. Fibrosis stage ≥ 2, degree of inflammation ≥ 2, and steatosis score ≥ 2 was defined as significant liver fibrosis, active inflammation, and severe steatosis, respectively.
Diagnosis
The diagnosis of MAFLD was based on evidence of hepatic steatosis, which was determined according to the results of liver biopsy in patients with BMI values ≥ 25, those with type 2 diabetes mellitus (T2DM), or those with at least two metabolic risk abnormalities despite BMI values lower than 25 5. Abnormalities indicative of metabolic risk included the following: (1) TG ≥ 1.70 mmol/L or use of certain medications; (2) HDL-c < 1.0 for men and 1.3 mmol/L for women or use of certain medications; (3) BP ≥ 130/85 mmHg or use of certain medications; (4) prediabetes FPG 5.6–6.9 mmol/L, HbA1c 5.7–6.4%, or 2-h post-load glucose level 7.8–11.0 mmol/L; and (5) waist circumference more than 90 cm and 80 cm in men and women, respectively. Data for high-sensitivity C-reactive protein (SCRP) and homeostasis model assessment index insulin resistance (HOMA-IR) were absent. Patients with MAFLD were divided into two groups based on the results of the HBsAg test: MAFLD + HBV group (HBsAg positive) and MAFLD group (HBsAg negative). In terms of disease course, HBV infection was diagnosed by histopathologists based on the location of inflammation and immunohistochemical results.
Statistical analysis
Continuous variables were analyzed via Student’s t-test or the Mann–Whitney U-test when compared between two groups. The chi-square test was used to compare categorical variables. Propensity score matching (PSM) was used to balance age, sex, and metabolic factors between the two groups at a ratio of 1:1 and using a caliper value of 0.2. Univariate and multivariate logistic regression analyses were performed to identify factors contributing to liver fibrosis. Adjusted odds ratios (OR) and relevant 95% confidence intervals (CIs) were estimated using a parametric proportional hazards model. SPSS version 26.0 (IBM, Armonk, NY, USA) was used for statistical analyses, with P < 0.05 indicating statistical significance.
Disclosure of ethical statement
This study conformed to the ethical guidelines of the Declaration of Helsinki and was approved by the Ethics Committee of the Affiliated Hospital of Hangzhou Normal University (Approval Number/ID: 2020 (02)-KS-022).
Consent to participate/consent to publish
As this was an observational retrospective study, the requirement for informed consent was waived by the Ethics Committee.
Results
Establishment of the study
As shown in Fig. 1, a total of 681 patients with biopsy-proven steatosis were included in the data screening. A total of 175 individuals with steatosis were excluded since they did not meet the diagnostic criteria for MAFLD, while 105 individuals were further excluded due to incomplete data. Among the 401 patients diagnosed with MAFLD, 256 patients were overweight/obese, 64 patients had T2DM, and 81 patients with normal or lean weight had metabolic disorders. The average age of the included patients was 43.14 ± 11.31 years; 44.64% (179/401) of patients had MAFLD with HBV infection (MAFLD + HBV group), and 55.36% (222/401) of patients had MAFLD without HBV infection (MAFLD group).
The proportion of male patients was higher in the MAFLD + HBV group than in the MAFLD group (88.82% vs. 72.52%, P < 0.001). No significant differences in age, SBP, DBP, BMI, FPG, HDL-c, SUA, or ALB levels were observed between the MAFLD and MAFLD + HBV groups (P > 0.05). However, the MAFLD + HBV group exhibited lower levels of liver enzymes, LDL-c, TG, and TC, as well as lower rates of obesity, hypertension, T2DM, low HDL-c, hypertriglyceridemia, hypercholesterolemia, and hyperuricemia than the MAFLD group (P < 0.05) (Table 1).
After PSM, 83 pairs were finally matched, and there were no statistically significant differences in sex, age, BMI, DBP, SBP, FPG, LDL-c, TC, liver enzyme levels, or rates of hypertension, T2DM, low HDL-c, hypertriglyceridemia, hypercholesterolemia, or hyperuricemia between the two groups (P > 0.05) (Table 1).
Comparison of histological features in MAFLD patients with and without HBV infection
After PSM, sex, age, BMI, liver enzymes, and metabolic factors were comparable between patients with and without HBV infection. Inflammation scores and liver fibrosis stages were higher in the MAFLD + HBV group than in the MAFLD group, while steatosis and ballooning scores were lower (P < 0.05) (Fig. 2). In the multivariate analysis, model 1 was adjusted for age and sex, while model 2 was adjusted for model 1 plus metabolic parameters, including BMI, T2DM, low HDL-c, hypertriglyceridemia, hypercholesterolemia, high LDL-c, hypertension, and hyperuricemia. The results indicated that HBV infection was associated with lower hepatic steatosis scores (OR: 0.251, 95% CI: 0.117–0.542, P < 0.001) and ballooning scores (OR: 0.119, 95% CI: 0.049–0.294, P < 0.001) yet higher stages of liver fibrosis (OR: 3.140, 95% CI: 1.479–6.663, P = 0.003) in patients with MAFLD (Table 2). However, no significant differences were observed in inflammation between patients with and without HBV infection after adjusting for confounders.
Comparison of histopathological characteristics between metabolic dysfunction-associated fatty liver disease with and without hepatitis B virus (HBV) infection. (A) Comparison of hepatic steatosis score segregated by HBV infection; (B) comparison of ballooning score segregated by HBV infection; (C) comparison of inflammation score segregated by HBV infection; (D) comparison of fibrosis stage segregated by HBV infection.
Comparison of clinical and histological features of patients with and without liver fibrosis in MAFLD infected with HBV
Of the 179 patients in MAFLD + HBV group, 18.44% (33/179) of patients had HBeAg-positive chronic infection, 27.37% (49/179) of patients had HBeAg-positive chronic hepatitis, 22.91% (41/179) of patients had HBeAg-negative chronic infection, and 31.28% (56/179) had HBeAg-negative chronic hepatitis. Further, 75.98% (136/179) of patients were HBV-DNA-positive and 43.58% (78/179) of patients in the MAFLD + HBV group had significant liver fibrosis (S ≥ 2). There were no statistically significant differences in sex, age, SBP, DBP, BMI, ALT, AST, LDL-c, HDL-c, TG, or TC between patients with and without liver fibrosis. In addition, rates of HBeAg-positive, HBV-DNA-positive, hypertension, low HDL-c, hypertriglyceridemia, hypercholesterolemia, and hyperuricemia did not significantly differ between patients with and without liver fibrosis (P > 0.05). Among patients in MAFLD + HBV group, those with liver fibrosis had higher levels of GGT and FPG, and a higher percentage of T2DM and obese/overweight status than those without (P < 0.05) (Table 3). For histological comparison, the proportion of patients with active inflammation (inflammation score ≥ 2) was significantly higher among those with liver fibrosis than those without liver fibrosis (85.90% vs. 37.62%, P < 0.001), whereas the proportions of severe steatosis and ballooning degeneration had no significant difference between the two groups (P > 0.05).
Risk factors for liver fibrosis in MAFLD patients with HBV infection
Univariate and multivariate logistic regression analyses were performed to further explore risk factors for liver fibrosis in MAFLD patients with HBV infection. Univariate analysis confirmed that T2DM (OR: 5.540, 95% CI: 1.72–17.63; P = 0.004) and elevated GGT levels (OR: 2.991, 95% CI: 1.612–5.550; P = 0.001) were risk factors for liver fibrosis (Table 4). Multivariate regression analysis, with sex, age, metabolic factors, viral factors, and liver enzymes taken into consideration, revealed that hypertension (OR: 2.640, 95% CI: 1.091–6.368; P = 0.031), T2DM (OR: 4.939, 95% CI: 1.121–21.796; P = 0.035), and elevated GGT levels (OR: 3.980, 95% CI: 1.735–9.132; P = 0.001) were independent risk factors for liver fibrosis in MAFLD patients with HBV infection.
Discussion
MAFLD with HBV infection is a distinct subtype of MAFLD in which metabolic and viral factors co-exist. The current results indicate that the presence of HBV is associated with lower steatosis scores and ballooning grades but a higher liver fibrosis stage in patients with MAFLD. Further risk factor analysis for liver fibrosis revealed that T2DM, hypertension, and elevated GGT levels were independent risk factors for liver fibrosis in MAFLD patients with HBV infection.
MAFLD patients with HBV infection exhibit unique histopathological characteristics (Fig. 2). Our findings are in accordance with a recent study showing that, although HBV infection is associated with a lower degree of steatosis and ballooning, it independently increases the risk of liver fibrosis in patients with MAFLD18,19. In our study, HBV infection resulted in a threefold increase in the risk of significant liver fibrosis in patients with MAFLD (Table 2). Therefore, for MAFLD patients with HBV infection, early screening and intervention for risk factors of liver fibrosis are required, as liver fibrosis has been identified as an accelerator for cirrhosis and hepatocellular carcinoma in chronic liver disease20.
Previous studies have reported that metabolic disorders such as T2DM, hypertension, dyslipidemia, and obesity are closely related to NAFLD liver fibrosis21,22,23,24,25,26. HBeAg negativity is associated with more advanced liver fibrosis in patients with chronic hepatitis B27. ALT and AST have also been identified as excellent predictors of significant liver fibrosis in patients with CHB28. The presence of both viral and metabolic factors may accelerate disease progression in MAFLD patients with HBV infection. Our study indicates that T2DM, hypertension, and elevated GGT levels are independent risk factors for significant liver fibrosis in MAFLD patients with HBV infection even after adjusting for confounding factors (Table 4). While HBeAg positivity and HBV-DNA positivity were not associated with liver fibrosis in MAFLD patients with HBV. It suggested that metabolic factors is more associated with liver fibrosis compared with viral factors in MAFLD patients with HBV infection.
This study highlights that T2DM, hypertension, and elevated GGT levels are closely associated with liver fibrosis in MAFLD patients with HBV infection. Liver fibrosis is the result of an excessive production of extracellular matrix (ECM) that is not adequately maintained, resulting in net accumulation. In the liver, hepatic stellate cells (HSCs) constitute the main source of ECM-producing fibroblasts in models of toxic and biliary liver disease and NAFLD29,30. Insulin resistance (IR) in T2DM is recognized as an integral component of NAFLD pathogenesis that worsens with disease progression29,31,32 and the activation of HSC by IR is largely divided into distinct direct and indirect pathways. The renin–angiotensin–aldosterone system is well recognized for its essential role in the physiological regulation of blood volume, blood pressure, and sodium homeostasis33,34. Increasing evidence demonstrates that this system is overactive at different stages of liver fibrosis33,35, which may explain the association between hypertension and liver fibrosis. As a surface enzyme, GGT can cleave extracellular glutathione (GSH), maintain the balance of GSH in vivo, and play a key role in alleviating the effects of oxidative stress36. Previous studies confirmed that elevated GGT was associated with SCRP, low adiponectin, the presence of chronic kidney disease, and hepatic steatosis. It was reported that GGT elevation was associated with hepatic steatosis, and fibrosis in patients with NAFLD37,38. In addition to the above factors, the influence of genetic factors such as PNPLA3, TM6SF2, and MBOAT7 on liver fibrosis in MAFLD patients with HBV infection should also be further studied39.
The major strength of this study is that, to the best of our knowledge, it is the first to analyze risk factors for liver biopsy-proven significant liver fibrosis in MAFLD patients with HBV infection. However, this study has some limitations, including its retrospective design. Although HOMA-IR and SCRP are mentioned in the diagnostic criteria for lean and normal-weight patients with MAFLD under the new definition, these were absent in our data, which may have caused us to miss some MAFLD cases. Second, given the cross-sectional nature of the study, we were unable to determine the causal relationship between metabolic dysfunction and significant liver fibrosis, highlighting the need for further longitudinal cohort studies to verify the effects of metabolic and viral factors on liver fibrosis in MAFLD patients with HBV infection. Furthermore, due to the gender distribution of MAFLD patients, the number of women included in this study was low; therefore, conclusions may pertain more to male patients, and further research is required to confirm these findings in females.
In conclusion, MAFLD patients with HBV infection have a higher risk of liver fibrosis than patients who have pure MAFLD. Metabolic factors, hypertension and type 2 diabetes, are closely related to liver fibrosis in MAFLD patients with HBV. These results highlight that in addition to traditional antiviral therapy, screening and early intervention of metabolic diseases are required for MAFLD patients with HBV infection. For patients with diabetes and hypertension, blood noninvasive biomarkers or transient elastography should be actively performed to further define the stage of liver fibrosis. If noninvasive screening presents a high risk of liver fibrosis, liver biopsy is recommended.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Younossi, Z. et al. Global burden of NAFLD and NASH: Trends, predictions, risk factors and prevention. Nat. Rev. Gastroenterol. Hepatol. 15, 11–20 (2018).
Sarin, S. K. et al. Liver diseases in the Asia-Pacific region: A Lancet Gastroenterology & Hepatology Commission. Lancet Gastroenterol. Hepatol. 5, 167–228 (2020).
Eslam, M. et al. A new definition for metabolic dysfunction-associated fatty liver disease: An international expert consensus statement. J. Hepatol. 73, 202–209 (2020).
Eslam, M., Sanyal, A. J., George, J. & International Consensus Panel. MAFLD: A consensus-driven proposed nomenclature for metabolic associated fatty liver disease. Gastroenterology 158, 1999–2014.e1 (2020).
European Association for the Study of the Liver (EASL), European Association for the Study of Diabetes (EASD) & European Association for the Study of Obesity (EASO). EASL-EASD-EASO Clinical Practice Guidelines for the management of non-alcoholic fatty liver disease. Diabetologia 59, 1121–1140 (2016).
Jia, J. D., Hou, J. L., Wei, L. & Zhuang, H. Highlights of the guidelines of prevention and treatment for chronic hepatitis B (2019 version). Zhonghua Gan Zang Bing Za Zhi 28, 21–23 (2020).
Mak, L. Y. et al. Diverse effects of hepatic steatosis on fibrosis progression and functional cure in virologically quiescent chronic hepatitis B. J. Hepatol. 73, 800–806 (2020).
Seto, W. K. et al. Association Between hepatic steatosis, measured by controlled attenuation parameter, and fibrosis burden in chronic hepatitis B. Clin. Gastroenterol. Hepatol. 16, 575-583.e2 (2018).
Lee, Y. B. et al. Association between hepatic steatosis and the development of hepatocellular carcinoma in patients with chronic hepatitis B. Clin. Mol. Hepatol. 25, 52–64 (2019).
Van Kleef, L. A. et al. Metabolic dysfunction-associated fatty liver disease increases risk of adverse outcomes in patients with chronic hepatitis B. JHEP Rep. 3, 100350 (2021).
Angulo, P. et al. Liver fibrosis, but no other histologic features, is associated With long-term outcomes of patients With nonalcoholic fatty liver disease. Gastroenterology 149, 389–97.e10 (2015).
Ekstedt, M. et al. Fibrosis stage is the strongest predictor for disease-specific mortality in NAFLD after up to 33 years of follow-up. Hepatology 61, 1547–1554 (2015).
Sohn, W., Kwon, H. J., Chang, Y., Ryu, S. & Cho, Y. K. Liver fibrosis in Asians With metabolic dysfunction-associated fatty liver disease. Clin. Gastroenterol. Hepatol. 20, e1135–e1148-e48 (2022).
Singh, S. et al. Fibrosis progression in nonalcoholic fatty liver vs nonalcoholic steatohepatitis: A systematic review and meta-analysis of paired-biopsy studies. Clin. Gastroenterol. Hepatol. 13, 643–54.e1 (2015).
Brunt, E. M., Janney, C. G., Di Bisceglie, A. M., Neuschwander-Tetri, B. A. & Bacon, B. R. Nonalcoholic steatohepatitis: A proposal for grading and staging the histological lesions. Am. J. Gastroenterol. 94, 2467–2474 (1999).
Bedossa, P. & Poynard, T. An algorithm for the grading of activity in chronic hepatitis C. The METAVIR Cooperative Study Group. Hepatology 24, 289–293 (1996).
Batts, K. P. & Ludwig, J. Chronic hepatitis. An update on terminology and reporting. Am. J. Surg. Pathol. 19, 1409–1417 (1995).
Wang, M. F. et al. Clinic-pathological features of metabolic associated fatty liver disease with hepatitis B virus infection. World J. Gastroenterol. 27, 336–344 (2021).
Su, H. J. et al. Pathologic findings of patients with nonalcoholic fatty liver disease and the impact of concurrent hepatitis B virus infection in Taiwan. J. Formos. Med. Assoc. 119, 1476–1482 (2020).
Dhar, D., Baglieri, J., Kisseleva, T. & Brenner, D. A. Mechanisms of liver fibrosis and its role in liver cancer. Exp. Biol. Med. (Maywood) 245, 96–108 (2020).
Kim, Y. et al. Metabolically healthy versus unhealthy obesity and risk of fibrosis progression in non-alcoholic fatty liver disease. Liver Int. 39, 1884–1894 (2019).
Kim, Y. et al. Obesity and weight gain are associated With progression of fibrosis in patients With nonalcoholic fatty liver disease. Clin. Gastroenterol. Hepatol. 17, 543-550.e2 (2019).
Kim, D. et al. Predictors of nonalcoholic steatohepatitis and significant fibrosis in non-obese nonalcoholic fatty liver disease. Liver Int. 39, 332–341 (2019).
Nakahara, T. et al. Type 2 diabetes mellitus is associated with the fibrosis severity in patients with nonalcoholic fatty liver disease in a large retrospective cohort of Japanese patients. J. Gastroenterol. 49, 1477–1484 (2014).
Méndez-Sánchez, N. et al. Dyslipidemia as a risk factor for liver fibrosis progression in a multicentric population with non-alcoholic steatohepatitis. F1000Res 9, 56 (2020).
Cai, S. et al. Risk factors associated with liver steatosis and fibrosis in chronic hepatitis B patient with component of metabolic syndrome. United Eur. Gastroenterol. J. 6, 558–566 (2018).
Wang, J. et al. HBeAg negativity is associated With more advanced liver fibrosis in patients with chronic hepatitis B: A propensity score-matching analysis. J. Clin. Gastroenterol. 54, 826–831 (2020).
Zhang, X. et al. Prognostic value of inflammatory indicators in chronic hepatitis B patients With significant liver fibrosis: A multicenter study in China. Front. Pharmacol. 12, 653751 (2021).
Schwabe, R. F., Tabas, I. & Pajvani, U. B. Mechanisms of fibrosis development in nonalcoholic steatohepatitis. Gastroenterology 7, 1913–1928 (2020).
Tsuchida, T. & Friedman, S. L. Mechanisms of hepatic stellate cell activation. Nat. Rev. Gastroenterol. Hepatol. 7, 397–411 (2017).
Eckstein, S. S., Weigert, C. & Lehmann, R. Divergent roles of IRS (insulin receptor substrate) 1 and 2 in liver and skeletal muscle. Curr. Med. Chem. 17, 1827–1852 (2017).
Friedman, S. L., Neuschwander-Tetri, B. A., Rinella, M. & Sanyal, A. J. Mechanisms of NAFLD development and therapeutic strategies. Nat. Med. 7, 908–922 (2018).
Li, S., Zhao, W., Tao, Y. & Liu, C. Fugan Wan alleviates hepatic fibrosis by inhibiting ACE/Ang II/AT-1R signaling pathway and enhancing ACE2/Ang 1–7/Mas signaling pathway in hepatic fibrosis rat models. Am. J. Transl. Res. 12, 592–601 (2020).
AlQudah, M., Hale, T. M. & Czubryt, M. P. Targeting the renin-angiotensin-aldosterone system in fibrosis. Matrix Biol. 91–92, 92–108 (2020).
Rajapaksha, I. G. et al. Liver-targeted angiotensin converting enzyme 2 therapy inhibits chronic biliary fibrosis in multiple drug-resistant gene 2-knockout mice. Hepatol. Commun. 3, 1656–1673 (2019).
] Ndrepepa, G., Colleran, R., & Kastrati, A. Gamma-glutamyl transferase and the risk of atherosclerosis and coronary heart disease. Clin. Chim. Acta 476, 130–138 (2018).
Chen, L. W., Huang, M. S., Shyu, Y. C. & Chien, R. N. Gamma-glutamyl transpeptidase elevation is associated with metabolic syndrome, hepatic steatosis, and fibrosis in patients with nonalcoholic fatty liver disease: A community-based cross-sectional study. Kaohsiung J. Med. Sci. 9, 819–827 (2021).
Xing, Y., Chen, J., Liu, J. & Ma, H. Associations between GGT/HDL and MAFLD: A cross-sectional study. Diabetes Metab. Syndr. Obes. 15, 383–394 (2022).
Liu, Z. et al. The health impact of MAFLD, a novel disease cluster of NAFLD, is amplified by the integrated effect of fatty liver disease-related genetic variants. Clin. Gastroenterol. Hepatol. 4, e855–e875 (2022).
Acknowledgments
This work was supported by the Zhejiang Provincial Department of Health Project (grant number 2020KY715).
Funding
This work was supported by the Zhejiang Provincial Department of Health Project (grant number 2020KY715).
Author information
Authors and Affiliations
Contributions
H.F.L. and Y.M.J. curated the data, visualized, prepared the original manuscript, reviewed, and edited it. G.L.Z., S.Y.L., and D.W. contributed to the investigation and methodology design. J.W. curated the data. K.Z. developed the software used. J.L. managed the project resources, prepared the original manuscript, reviewed, and edited it. All authors have reviewed and approved the submitted manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lv, H., Jiang, Y., Zhu, G. et al. Liver fibrosis is closely related to metabolic factors in metabolic associated fatty liver disease with hepatitis B virus infection. Sci Rep 13, 1388 (2023). https://doi.org/10.1038/s41598-023-28351-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-28351-3
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.