Abstract
Asynchronous calibration could allow opportunistic screening based on routine CT for early osteoporosis detection. In this phantom study, a bone mineral density (BMD) calibration phantom and multi-energy CT (MECT) phantom were imaged on eight different CT scanners with multiple tube voltages (80–150 kVp) and image reconstruction settings (e.g. soft/hard kernel). Reference values for asynchronous BMD estimation were calculated from the BMD-phantom and validated with six calcium composite inserts of the MECT-phantom with known ground truth. Relative errors/changes in estimated BMD were calculated and investigated for influence of tube voltage, CT scanner and reconstruction setting. Reference values for 282 acquisitions were determined, resulting in an average relative error between calculated BMD and ground truth of − 9.2% ± 14.0% with a strong correlation (R2 = 0.99; p < 0.0001). Tube voltage and CT scanner had a significant effect on calculated BMD (p < 0.0001), with relative differences in BMD of 3.8% ± 28.2% when adapting reference values for tube voltage, − 5.6% ± 9.2% for CT scanner and 0.2% ± 0.2% for reconstruction setting, respectively. Differences in BMD were small when using reference values from a different CT scanner of the same model (0.0% ± 1.4%). Asynchronous phantom-based calibration is feasible for opportunistic BMD assessment based on CT images with reference values adapted for tube voltage and CT scanner model.
Similar content being viewed by others
Introduction
Osteoporosis is defined by systemic bone mineral density (BMD) loss and microarchitectural deterioration, resulting in increased skeletal fragility and consequently a higher fracture risk1. Early detection of osteoporosis is of utmost importance, before an undetected progression of BMD-loss can lead to irreversible complications such as chronic pain, height loss with kyphosis causing posture impairment and eventually paraplegia resulting in lifelong physical disability2,3. Additionally, osteoporosis is a comorbidity of a multitude of medical conditions, e.g. chronic pulmonary disease, and osteoporosis itself has a negative effect on the outcome of other diseases. Osteoporosis affects about 500 million people and is one of the leading chronic diseases worldwide4. Pharmaceutic treatment of osteoporosis is limited in that it is not curative and may only prevent further fractures and disease progression. However, since the early stages of osteoporosis are silent, diagnosis is often only made when fractures have already occurred5. With current increases in overall life expectancy, osteoporosis can only be expected to increase in relevance in the future, as it is more prevalent in old age.
The reference standard to diagnose osteoporosis is dual-energy X-ray absorptiometry (DEXA)6. An alternative to DEXA are quantitative computed tomography (QCT) measurements, which allow for the detailed analysis of e.g. vertebrae and avoiding measurements of fractured bone, based on the three-dimensional nature of the data7,8. While these exams are justified for patients with a clinical suspicion of osteoporosis, opportunistic screening would allow to measure BMD in routine CT examinations. This could become a preventive approach without additional patient radiation exposure and without the need for specific screening programs9. Prospective BMD measurements with QCT rely on the use of a calibration phantom that is scanned simultaneously below the patient, whereas opportunistic screening attempts to estimate the BMD without the simultaneous use of calibration phantoms9,10,11. Some studies have previously investigated the potential for retrospective BMD measurements from routine CT. For example, Jang et al. published average CT numbers in the L1 vertebra obtained from more than 20,000 CT examinations, which can be used as a reference to identify patients with low BMD12. Nevertheless, this method does not allow exact BMD quantification and correlation to gold standard DEXA or QCT measurements. Another approach is to rely on asynchronous calibration measurements, where calibration data is obtained by intermittent QCT phantom scans, independently from the patient scans13,14. This approach allows for the retrospective assessment of BMD, as the calibration measurements can be performed and applied after the patient data have already been acquired. However, current studies on this topic are limited to specific acquisition protocols on specific CT scanners13,14. For widespread retrospective BMD assessment, calibration data is needed for each acquisition protocol on every CT scanner. Furthermore, studies have investigated the influence of some study parameters on BMD quantification, including the accuracy of retrospective BMD quantification depending on CT scanner, acquisition parameters and reconstruction settings. However, simultaneous changes of these parameters over a large parameter range have not been investigated in detail yet11. The aim of this phantom study was to evaluate the accuracy of BMD measurements with asynchronous phantom calibration for different CT scanners, acquisition parameters and reconstruction settings. Additionally, the influence of the CT scanners, acquisition parameters and reconstruction setting on the accuracy of the BMD measurements was assessed.
Methods
No ethics approval was necessary for the phantom measurements in this study. The evaluation of patient data was approved by the institutional review board of the University Hospital Heidelberg under number S-937/2020 and performed in accordance with the Declaration of Helsinki 2013. Informed consent was waived and all methods were carried out in accordance with state-of-the-art guidelines and regulations.
Data acquisition
Calibration phantom
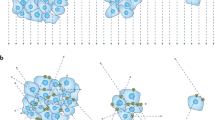
A commercially available QCT calibration phantom was used for asynchronous calibration measurements (OSTEO, Siemens Healthineers; Fig. 1a). This QCT calibration phantom consists of two phases corresponding to 0 mg/ml and 200 mg/ml hydroxylapatite15. The calibration phantom was scanned simultaneously with a commercially available image quality phantom (CATPhan 600, The Phantom Laboratory), which provided additional absorption (Fig. 1a).
Validation phantom
A commercially available multi-energy CT phantom (MECT Phantom, SunNuclear) with tissue-equivalent inserts was used for validation (Fig. 1b,c). Six inserts containing calcium composites were scanned. Ground truth equivalent hydroxylapatite concentrations were calculated based on the calcium content given by the manufacturer (Table 1). To this end, relative calcium content (Ca%) was multiplied with the density of the insert (ρ) to arrive at the calcium concentration in mg/ml. To further convert calcium concentrations to hydroxylapatite concentrations (BMDequiv), the calcium concentration of hydroxylapatite was calculated from its chemical formula: Ca5(PO4)3OH. Based on the molecular weights of the individual components of hydroxylapatite, 100 mg of hydroxylapatite contain 39.89 mg of Calcium (Ca). Inversely, we assume that an insert with a calcium concentration of 100 mg/ml is equivalent to a hydroxylapatite concentration of 251 mg/ml.
CT acquisition parameters
Eight CT scanners representing seven different scanner models from three manufacturers located at our institution and a nearby practice were included in the study (Table 2). For each scanner, all available settings for tube voltage were evaluated. Reconstruction settings were chosen to reflect the most commonly used clinical parameters on the respective scanner. These were intended to cover most of the potential patients’ data to be included in later evaluations. Both filtered-back projection and iterative reconstruction were included in the study with reconstruction kernels optimized for both soft-tissue and bone.
The following parameters were kept constant between all CT scanners for improved comparability: slice thickness 3 mm, slice increment 3 mm, rotation time 1 s, CTDIvol 15 mGy. Data acquisition was repeated with a minimum gap of 6 months for four of the eight CT scanners to investigate changes in reference values over time, e.g. because of changes in scanner performance, scanner calibration or maintenance.
Reference values
For each acquisition, reference values were determined for the two phases of the calibration phantom by means of region of interest (ROI) evaluation. Rectangular ROIs with a size of 417 mm2 were placed in the calibration phantom for all slices covered by the image quality phantom (Fig. 1a). Images showing artifacts at the phantom edges in the z-direction were excluded from the measurement. A total of 64 images could be evaluated per measurement, corresponding to a length of 192 mm. Linear regression analysis was performed for each acquisition, determining the slope and intercept of a linear function to compute bone density or hydroxylapatite values from CT numbers:
Evaluation
Validation of BMD quantification
The image data from the measurements of the validation phantom were analysed with a commercially available software tool from the phantom manufacturer (RapidCheck, SunNuclear), which automatically measures mean CT numbers of all inserts. CT numbers measured for inserts containing calcium composites were converted to bone-densities according to Eq. (2), using the previously obtained reference values. Calculated bone-densities were compared to the true equivalent hydroxylapatite concentrations of the insert (BMDTruth) as given in Table 1 (calculated with Eq. 1) to analyse measurement accuracy. Results are given in terms of relative error, e.g.
Here, BMDmeasured is the BMD calculated according to Eq. (2) using the reference values, while BMDTruth is the ground truth calculated according to Eq. (1) (Table 1).
Influence of reference values
As a gold standard, a reference value has to be determined for each potential combination of CT scanner, tube voltage and reconstruction setting. In a multi-center study with different CT scanners and a diverse patient collective, where varying acquisition protocols and acquisition settings are used, the number of required reference measurements can be very high.
The validation data was analyzed to determine whether reference measurements are necessary for every potential combination of acquisition parameter, reconstruction setting and CT scanner. As described below, BMD was calculated using a reference value from a different acquisition and compared to the result using the actual reference value. If the difference in BMD is small, then it might not be necessary to have different reference measurements.
For example, CT-numbers from one acquisition, e.g. at 80 kVp tube voltage, were converted to BMD (Eq. 2) by using reference values from a different acquisition, e.g. at 100 kVp tube voltage. The calculated BMD was then compared to the ground truth (Eq. 3) and to the BMD calculated with the actual reference value, i.e.:
Here, BMD80kV is the BMD calculated with the actual reference value at 80 kVp. BMD100kV vs 80 kV is calculated from the CT-numbers acquired at 80 kVp tube voltage, but converted to BMD with the reference values (i.e. slope and intercept; see Eq. 2) measured with a tube voltage of 100 kVp instead. For each scanner, every potential combination of tube voltage was considered. The analysis was performed separately for the following factors (consider also Table 2):
-
1.
CT Scanner
-
2.
Tube voltage
-
3.
Reconstruction setting
-
4.
Repeated measurements (> 6-months time difference)
Regarding the comparison of CT scanners, data were analyzed while considering all scanners, considering only same-manufacturer scanners and considering only same-model scanners. Data were matched between CT scanners with exact matches for tube voltage, but reconstruction kernels were regarded as interchangeable in the following three groups:
-
1.
Filtered back-projection with soft-tissue kernel (e.g. “B” or “B30f.”)
-
2.
Filtered back-projection with hard kernel (e.g. “YB” or “B70f.”)
-
3.
Iterative reconstruction (e.g. “IMR” or “I30f.”)
Statistical analysis
A linear correlation coefficient (Pearson’s r) was calculated to assess the correlation between calculated bone density values and ground truth.
To determine which factors influence BMD quantification, two general linear models were trained to perform analysis of covariances for both the calculated BMD (Eq. 2) as well as the relative error compared to the ground truth (Eq. 3). The following independent variables were considered for both general linear models: tube voltage, CT scanner, reconstruction setting, evaluated insert, repetition. All interactions between the independent variables were included in the model. Values from all six inserts were considered simultaneously by considering the type of insert as an independent variable. Because of the differences in material composition between the different inserts (Table 1), the evaluated insert was modeled as a categorical variable. Both models were built in a multi-step design, removing non-significant effects until all variables left had a significant effect or were part of a significant interaction effect.
Further analysis was done to determine which factors have to be considered when obtaining reference values. Analysis of variances (ANOVA) was performed to compare relative errors of BMD quantification (Eq. 3) when varying reference values. Relative errors in determined BMD when varying reference values for different influencing factors (e.g. tube voltage, etc.) were compared by Tukey’s test to the relative error for the gold standard BMD quantification, where the actual reference value was used. Additionally, differences in calculated BMD values (Eq. 4) where tested for non-zero differences with a one-tail Student’s t-test.
A p-value of 0.05 was chosen as the threshold for statistical significance. The analysis was performed using MATLAB 2021a (Mathworks) and SAS 9.4 (SAS Institute).
Experimental application
To experimentally explore the application of the determined reference values for BMD quantification, data from a single patient was retrospectively evaluated. The patient was acquired on CT Scanner #4 with two different acquisition protocols. Acquisition #1 was performed at 100 kVp tube voltage with a tube current–time product of 21 mAs and reconstructed using the I40s Kernel with a slice thickness of 0.7 mm. Acquisition #2 was performed together with the calibration phantom at 80 kVp tube voltage with a tube current–time product of 120 mAs, and reconstructed using the S80s Kernel with a slice thickness of 10 mm. BMD was calculated with the determined reference values from acquisition #1. BMD was measured with a commercial application for BMD quantification (syngo Osteo CT; Siemens Healthineers) for acquisition #2.
Results
A total of 473 CT acquisitions were performed, including 282 acquisitions for reference measurements and 191 acquisitions for validation. These acquisitions cover a parameter space of eight CT scanners, seven CT scanner models, three manufacturers, ten tube voltages, 22 image reconstructions, and up to two repetitions.
Validation of BMD quantification
Comparing the determined BMD values for the different inserts to the ground truth showed an average relative error of − 9.2% ± 14.0%. Correlation between calculated bone density values and ground truth was very strong with R2 = 0.99 (p < 0.0001). Considering inserts labelled as “Calcium” only (i.e. 50, 100, and 300 mg calcium; Table 1), the average relative error was − 2.0% ± 15.4%. Differences in relative error could be observed between the inserts, as shown in Fig. 2a. Further differences in relative error could be observed between CT scanners and tube voltages (Figs. 2b,c). On average, absolute values of relative error decreased with increasing tube voltage (Fig. 2c). The differences in relative error between different reconstruction kernels were small (Fig. 2d).
Relative error of computed bone mineral density compared to the calculated ground truth values over the different investigated parameters: (A) phantom insert (B) CT scanner (C) tube voltage (D) reconstruction setting. Note the differences in sample sizes for the different parameters (also Table 2).
Statistical models for BMD quantification showed a good fit and a significant influence of independent variables on both the absolute BMD values (p < 0.0001; R2 = 0.99) and the relative error to ground truth (p < 0.0001; R2 = 0.99). While tube voltage, CT scanner and evaluated insert had a significant effect in both models, the repetition was only significant as part of interaction effects. The reconstruction setting was not a significant effect in either of the models. Consequently, only tube voltage, CT scanner and ground truth BMD can be considered to have a significant influence on BMD quantification. Reported p-values for all effects included in the final model are shown in Table 3.
Influence of reference values
Comparing the results achieved when using reference values from a different acquisition to the results achieved with the actual reference values (Eq. 4) shows that the tube voltage (average relative difference: 3.8% ± 28.2%) and CT scanner (− 5.6% ± 9.2%) have a strong influence on the results (Fig. 3a). Using reference values from a different CT scanner of the same scanner model showed little impact on the results (0.0% ± 1.4%). Differences were larger when using reference values from any CT scanner of the same manufacturer (− 5.5% ± 6.6%) and largest when using any other CT scanner, regardless of manufacturer (− 5.6% ± 9.2%). The influence of the reconstruction settings was comparatively low (0.2% ± 0.2%), with a mean difference of 0.0% ± 0.1% when comparing iterative reconstruction to filtered back-projection. Comparisons between repeated calibration measurements had a mean difference of 0.1% ± 0.7%. A significant non-zero difference in calculated BMD was found for tube voltage, CT scanners (all evaluations) and reconstruction setting, but not for the repetition (p < 0.05).
(A) Relative difference between calculated bone mineral density values when varying the reference value used for the calculation. “Scanner (all)” refers to using matching reference values (i.e. same tube voltage and similar reconstruction kernel) from any of the other scanners. (B) Relative error compared to ground truth when varying the reference value used for the calculation, compared to the value calculated when using the actual reference value (Influencing factor: “None”).
Similarly, analyzing the relative error between calculated BMD and ground truth when using reference values from different acquisitions shows error levels on par with the gold standard when varying repeated measurements (− 9.5% ± 13.9%), reconstruction settings (− 8.7% ± 14.3%), or when using a different CT scanner of the same scanner model (− 10.5% ± 14.2%; Fig. 3b). Absolute values of relative errors are slightly larger comparing CT scanners of the same manufacturer (− 11.3% ± 14.2%), or any other CT scanner regardless of manufacturer (− 11.0% ± 14.7%), or using reference values acquired for a different tube voltage (− 5.9% ± 26.5%).
For the analysis of the relative error depending on varying reference values, the ANOVA reported a significant difference in relative errors between the groups (p < 0.0001). Pair-wise comparison to the relative error of the gold standard BMD quantification using the actual reference value showed significant differences for tube voltage and CT scanners from the same manufacturer (p < 0.05). No significant differences were found for the reconstruction settings, repetition, CT scanners from all manufacturers or CT scanners of the same model.
Experimental application of the asynchronous calibration for opportunistic BMD measurements in a patient (Fig. 4a) showed BMD values in line with those obtained from a commercially available solution using synchronous calibration (syngo Osteo CT, Siemens Healthineers; Fig. 4b).
Image example comparing bone mineral density (BMD) measurements in terms of hydroxylapatit concentrations with synchronous and asynchronous calibration. (A) BMD measurement in the first vertebra of the lumbar spine (L1) of an 84-year-old female, showing a BMD of 102.2 mg/ml. BMD is calculated based on CT numbers of a non-contrast CT scan using the asynchronous reference values determined in this study. (B) Automated segmentation of L1 in the same 84-year-old female scanned with the reference phantom using syngo Osteo CT (Siemens Healthineers). Reported BMD of 96.2 mg/ml for L1 using synchronous calibration.
Discussion
With this study we showed that opportunistic screening of BMD is potentially feasible by using post-hoc generated, asynchronous phantom calibration for multiple CT scanners with a wide range of acquisition settings. Additionally, the influence of individual factors on the accuracy of BMD quantification was determined in this study, showing which factors should be considered for reference measurements. The results show that the acquired reference values can be used to measure BMD with an acceptable accuracy for screening purposes in a wide variety of acquisition and reconstruction settings. The results regarding the influencing factors could be used for further improvement of CT BMD measurements. While DEXA can also be influenced by factors like patient size and fat distribution16, DEXA or QCT is still warranted for follow-up to obtain a more accurate determination of individual risk for osteoporosis.
Some previous studies reported more accurate results for asynchronously calibrated BMD measurements than this study, but these studies only investigated limited acquisition and reconstruction settings over a smaller range. For example, acquisitions between 70 and 150 kVp were included in the current study, where the error was larger for small tube voltages (see Fig. 2c). While Garner et al. also analyzed tube voltages of 80 kVp, 100 kVp and 140 kVp, they focused on CT numbers instead of bone density17. In comparison, Wang et al. reported a consistent underestimation of bone density by 1.4–6.7% in their phantom study, but only investigated scans at a tube voltage of 120 kVp14. Michalski et al. also reported a 2.8% mean percent difference between asynchronous BMD measurements and regular phantom-based QCT for human cadaver scans at 120 kVp18. Woisetschläger et al. reported an average underestimation of 8–14% when comparing asynchronous measurements to those using internal CT calibration in human patients19. Despite the fact that the current study compares asynchronous measurements to synchronous phantom calibration instead of internal calibration, the results are similar. Generally, results for BMD measurements after asynchronous calibration are considered slightly less accurate than those with simultaneous calibration11. Furthermore, analysis of factors influencing the measurement accuracy shows that tube voltage has a strong influence on results, while other factors like image reconstruction and changes over time play a limited role only. In consequence, CT scans should be performed at the same tube voltage as the asynchronous calibration, or calibration has to be performed for all available tube voltages. These results are in line with those reported in a literature review by Brunnquell et al., who also cited a strong influence of tube voltage11. Furthermore, similar results were reported by Garner et al., who analyzed the influence of the tube voltage on CT numbers measured in L1 trabecular bone17. Additionally, the effect of using calibration data from a different CT scanner was investigated in this study. The results suggest that calibration data from one CT scanner might be used on different CT scanners of the same model with a moderate error, but that data from different CT scanner models should not be considered interchangeable. Even though differences between CT scanners were slightly larger when comparing different manufacturers, the difference to the ground truth was only statistically significant for CT scanners of the same manufacturer. This might be explained by the overall low number of 8 included CT scanners or the unbalanced design of the study, where most CT scanners came from a single manufacturer while also exhibiting the most technical inter-scanner variation. Consequently, further analysis seems necessary. Furthermore, specific calibration for different image reconstruction settings might not be strictly necessary, as the observed differences were small.
One limitation of this study is that a generalized MECT phantom was used for validation instead of specialized BMD phantoms like the European spine phantom20. The available inserts for the MECT phantom contain relatively high calcium concentrations, surpassing what would be observed during regular BMD measurements in clinical practice and potentially leading to photon starvation artifacts at low tube voltage. Yoganandan et al. reported an average BMD of 169.7 mg/cm3 for L3 and a maximum of 429.9 mg/cm3 over the cervical, thoracic and lumbar vertebrae in healthy males, while the maximum considered in this study was 767.21 mg/cm3 (Table 1)21. Furthermore, results show differences in relative measurement errors depending on the insert, which might be based on the differences in chemical composition of the inserts. As the evaluated phantoms use artificial reconstructions of the investigated tissues, the spectral X-ray absorption of the inserts might be most accurate for acquisitions at standard settings, i.e. 120 kVp. The wide range of tube voltages from 70 kVp up to 150 kVp considered in this study might exacerbate any inaccuracies. The results show that the relative error is reduced when considering only the phantom inserts labeled as “calcium”.
While a large number of CT scanners from three different manufacturers were included in this study, not all manufacturers could be included and results may differ for the CT scanners not investigated in this study. Additionally, only a limited number of factors was considered in this study. For example, differences in CTDIvol were not investigated, only up to one repeated measurement was performed, the patient size was not considered, and changes based on scanner calibration or maintenance were not evaluated. Only a limited number of reconstruction kernels were evaluated and quantitative kernels for post-processing available on some CT scanners were not included in the study. However, the evaluated kernels were chosen to closely match clinical practice and the results indicate that the reconstruction kernel has limited influence on the BMD quantification. Moreover, different phantom sizes were used for calibration and for validation, which might limit measurement accuracy.
Furthermore, with the increased availability of Dual-Energy CT (DECT), spectral methods for bone mineral quantification have been developed as well and should be investigated22. In the future, further validation of the technique is planned by correlating results from BMD measurements with asynchronous phantom calibration to DEXA measurements in a large patient collective.
In conclusion, the results show that opportunistic calculation of BMD from CT images using asynchronous phantom calibration is possible with a relative error of about − 9.2%. The main factors that have to be considered for calibration are the tube voltage and the scanner model.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Prevention, W. S. G. on, Osteoporosis, M. of & Organization, W. H. Prevention and management of osteoporosis: Report of a WHO scientific group. (World Health Organization: 2003).
Löffler, M. T. et al. Improved prediction of incident vertebral fractures using opportunistic QCT compared to DXA. Eur. Radiol. 29, 4980–4989 (2019).
Adachi, J. D. et al. The impact of incident vertebral and non-vertebral fractures on health related quality of life in postmenopausal women. BMC Musculoskelet. Disord. 3, 1–6 (2002).
Kanis, J. A. et al. A reference standard for the description of osteoporosis. Bone 42, 467–475 (2008).
F Cosman et al 2014 Clinician’s guide to prevention and treatment of osteoporosis Osteoporos Int 25 2359 2381
Syed, Z. & Khan, A. Bone densitometry: applications and limitations. J Obstet Gynaecol Can 24, 476–484 (2002).
Link, T. M. & Lang, T. F. Axial QCT: clinical applications and new developments. J Clin Densitometry 17, 438–448 (2014).
Engelke, K. Quantitative computed tomography—current status and new developments. J Clin Densitom 20, 309–321 (2017).
Brett, A. D. & Brown, J. K. Quantitative computed tomography and opportunistic bone density screening by dual use of computed tomography scans. J Orthop Translat 3, 178–184 (2015).
Therkildsen, J. et al. Feasibility of opportunistic screening for low thoracic bone mineral density in patients referred for routine cardiac CT. J Clin Densitom 23, 117–127 (2020).
Brunnquell, C. L., Winsor, C., Aaltonen, H. L. & Telfer, S. Sources of error in bone mineral density estimates from quantitative CT. Eur J Radiol 144, 110001 (2021).
Jang, S. et al. Opportunistic osteoporosis screening at routine abdominal and thoracic CT: normative L1 trabecular attenuation values in more than 20 000 adults. Radiology 291, 360–367 (2019).
Habashy, A. H., Yan, X., Brown, J. K., Xiong, X. & Kaste, S. C. Estimation of bone mineral density in children from diagnostic CT images: a comparison of methods with and without an internal calibration standard. Bone 48, 1087–1094 (2011).
Wang, L. et al. Validation of asynchronous quantitative bone densitometry of the spine: accuracy, short-term reproducibility, and a comparison with conventional quantitative computed tomography. Sci Rep 7, 1–7 (2017).
Kalender, W. A. & Suess, C. A new calibration phantom for quantitative computed tomography. Med Phys 14, 863–866 (1987).
H Bolotin 2007 DXA in vivo BMD methodology: an erroneous and misleading research and clinical gauge of bone mineral status, bone fragility, and bone remodelling Bone 41 138 154
HW Garner MM Paturzo G Gaudier PJ Pickhardt DE Wessell 2017 Variation in attenuation in L1 trabecular bone at different tube voltages: caution is warranted when screening for osteoporosis with the use of opportunistic CT AJR Am J Roentgenol 208 165 170
AS Michalski BA Besler GJ Michalak SK Boyd 2020 CT-based internal density calibration for opportunistic skeletal assessment using abdominal CT scans Med Eng Phys 78 55 63
M Woisetschläger M Hägg A Spångeus 2021 Computed tomography-based opportunistic osteoporosis assessment: a comparison of two software applications for lumbar vertebral volumetric bone mineral density measurements Quant Imaging Med Surg 11 1333
Pearson, J. et al. European semi-anthropomorphic spine phantom for the calibration of bone densitometers: Assessment of precision, stability and accuracy the European quantitation of osteoporosis study group. Osteoporos Int 5, 174–184 (1995).
Yoganandan, N. et al. Trabecular bone density of male human cervical and lumbar vertebrae. Bone 39, 336–344 (2006).
van Hamersvelt, R. W. et al. Accuracy of bone mineral density quantification using dual-layer spectral detector CT: a phantom study. Eur Radiol 27, 4351–4359 (2017).
Acknowledgements
The authors would like to thank Martin Finke and Nadine Schautberger for their support towards the study and their work regarding the phantom measurements. This work was funded by the German state of Baden-Württemberg as part of the project “Komorbidom der großen Volkskrankheiten: quantitative Bildgebung und künstliche Intelligenz”. For the publication fee we acknowledge financial support by Deutsche Forschungsgemeinschaft within the funding programme „Open Access Publikationskosten“ as well as by Heidelberg University.
Funding
Open Access funding enabled and organized by Projekt DEAL. Funding was provided by State Ministry of Baden-Wuerttemberg for Sciences, Research and Arts, Germany grant number 32-5400/58/3. This publication was supported by a research scholarship from Viktoria Palm provided by the medical faculty of the Ruprecht Karls University Heidelberg.
Author information
Authors and Affiliations
Contributions
All authors have made substantial contributions to the conception or design of the work, or acquisition, analysis, or interpretation of data, or have drafted the work and substantively revised it. All authors reviewed the manuscript. All authors have read and approved of the submitted version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
Dr. Stephan Skornitzke and Dr. Viktoria Palm have ownership interests in investment funds containing stock of healthcare companies. Otherwise, the authors of this manuscript declare no relationships with any companies, whose products or services may be related to the subject matter of the article.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Skornitzke, S., Vats, N., Kopytova, T. et al. Asynchronous calibration of quantitative computed tomography bone mineral density assessment for opportunistic osteoporosis screening: phantom-based validation and parameter influence evaluation. Sci Rep 12, 20729 (2022). https://doi.org/10.1038/s41598-022-24546-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-24546-2
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.