Abstract
Paramecium bursaria is a mixotrophic ciliate species, which is common in stagnant and slow-flowing, nutrient-rich waters. It is usually found living in symbiosis with zoochlorellae (green algae) of the genera Chlorella or Micractinium. We investigated P. bursaria isolates from around the world, some of which have already been extensively studied in various laboratories, but whose morphological and genetic identity has not yet been completely clarified. Phylogenetic analyses of the SSU and ITS rDNA sequences revealed five highly supported lineages, which corresponded to the syngen and most likely to the biological species assignment. These syngens R1–R5 could also be distinguished by unique synapomorphies in the secondary structures of the SSU and the ITS. Considering these synapomorphies, we could clearly assign the existing GenBank entries of P. bursaria to specific syngens. In addition, we discovered synapomorphies at amino acids of the COI gene for the identification of the syngens. Using the metadata of these entries, most syngens showed a worldwide distribution, however, the syngens R1 and R5 were only found in Europe. From morphology, the syngens did not show any significant deviations. The investigated strains had either Chlorella variabilis, Chlorella vulgaris or Micractinium conductrix as endosymbionts.
Similar content being viewed by others
Introduction
Paramecium bursaria has been studied since decades because of its easiness to be kept and experimentally manipulated under manifold cultivation conditions. Some major aspects on this model ciliate were investigated in detail: (i) P. bursaria lives in symbiosis with coccoid green algae belonging to the genera Chlorella and Micractinium (Pröschold et al.1 and references therein). Advantages of this close relationship include nutritional aspects as the algae provide photosynthetic products and photoprotection to the ciliate2,3. Accordingly, different aspects such as the process of cell–cell recognition and the symbiont-specificity are of great interest (Fujishima4 and articles therein). (ii) Complex mating systems in P. bursaria were discovered in mating experiments during (sexual) conjugation processes. So far, six genetic varieties were originally detected by Sonneborn5 and later designated as syngens 1 to 6, which were considered as biological species6,7,8. Most syngen-types have four (syngens 1 and 3) or eight (syngens 2 and 4–6) mating types9. As the strains that Bomford9 used for his experiments were lost, Greczek-Stachura et al.10 established a new syngen system, i.e., R1-R5 in principle most likely corresponding to Bomford’s syngens and indicated by a “B” but in a different order (R1–B6, R2–B4, R3–B1, R4–B2, and R5—B3 according to Greczek-Stachura et al.10). Syngen 5 of Bomford9 was not included. However, the subdivision into syngens was accompanied by phylogenetic analyses of the ITS (internal transcribed spacer regions; partial SSU–ITS1–5.8S–ITS2–partial LSU region) rDNA, the mitochondrial COI (cytochrome oxidase I) and H4 histone genes10. The five syngens were recently described as cryptic species based on COI haplotypes and named accordingly as Paramecium primabursaria, P. bibursaria, P. tribursaria, P. tetrabursaria and, P. pentabursaria11. Unfortunately, these species were not validly described according to the International Code for Zoological Nomenclature (ICZN), which requires formal descriptions and deposition of holotype specimens to public museums.
Despite such detailed studies in respect to conjugation and endosymbiosis, the morphology and the phenotypic plasticity of P. bursaria has only been rarely investigated. Kreutz et al.12 compared the morphology and ultrastructure of one P. bursaria strain with another “green” Paramecium, i.e., Paramecium chlorelligerum. However, both species were investigated directly from field samples and the phenotypic plasticity was not studied from cultured material.
The aim of this study was the comparison of 48 P. bursaria strains using an integrative approach to answer the following questions: (i) How many phylogenetic lineages among the investigated strains can be revealed? (ii) Do they correspond to the known syngen affiliations? (iii) Does the morphology of the ciliate strains differ among the syngens? (iv) Do the different syngens show any biogeographic pattern? and, (v) Do all strains bear the same algal endosymbiont? We studied the strains both isolated from diverse geographical regions and acquired from culture collections. First, we sequenced the SSU and ITS rDNA sequences. Subsequently, from each phylogenetic clade, at least one strain was selected to study its morphology and phenotypic plasticity from living and silver-stained specimens. Finally, the green algal endosymbionts were identified both from morphology and a diagnostic PCR approach.
Results
Molecular Phylogeny of Paramecium bursaria and Identification of its Endosymbionts
The SSU and ITS rDNA of the nuclear ribosomal operon were sequenced to infer the genetic variability of the investigated strains. The SSU and ITS rDNA sequences were aligned according to their secondary structure (examples are presented for the strain SAG 27.96; Fig. 1 and Supplementary Fig. 1). Additional sequences acquired from GenBank were incorporated into a dataset, which included all syngens also from references known for P. bursaria. The phylogenetic analyses revealed five highly supported lineages among the P. bursaria strains, which corresponded to their syngen assignment. As demonstrated in Fig. 2, all investigated strains belonging to the syngens R1, R2 and R5 originated from Europe, whereas the others of the syngens R3-R4 showed a worldwide distribution. The three known green algal endosymbionts, i.e., Chlorella variabilis (Cvar), Chlorella vulgaris (Cvul) and Micractinium conductrix (Mcon) showed no or only little affiliation to specific syngens.
Molecular phylogeny of the Paramecium bursaria species complex based on SSU and ITS rDNA sequence comparisons. The phylogenetic tree shown was inferred using the maximum likelihood method based on the datasets (2197 aligned positions of 19 taxa) using the computer program PAUP 4.0a169. For the analyses, the best model was calculated by PAUP 4.0a169. The setting of the best model was given as follows: TVM + I (base frequencies: A 0.2983, C 0.1840, G 0.2271, T 0.2906; rate matrix A–C 2.6501, A–G 8.6851, A–U 5.3270, C–G 0.91732, C–U 8.6851, G–U 1.0000) with the proportion of invariable sites (I = 0.9544). The branches in bold are highly supported in all bootstrap analyses (bootstrap values > 50% calculated with PAUP using the maximum likelihood, neighbour—joining, and maximum parsimony). The clades are named after the syngens (color‐coded) proposed by Greczek‐Stachura et al.10 and Bomford9 in brackets. The accession numbers are given after the strain numbers. The endosymbiotic green algae identified are highlighted (Mcon—Micractinium conductrix, Cvar—Chlorella variabilis and Cvul—Chlorella vulgaris) after the origin of the P. bursaria strains. The reference strain of each syngen is marked with an asterisk. The strains used for morphological comparisons are marked with a green dot next to the strain number.
Synapomorphies of the Paramecium bursaria Syngens
As demonstrated in Fig. 2, the subdivision of the P. bursaria strains into syngens is supported by the phylogenetic analyses of the SSU and ITS rDNA sequences. To figure out if these splits were also supported by characteristic molecular signatures, we studied the secondary structures of both SSU and ITS of all available sequences. We discovered 30, respectively 23 variable positions among the SSU and ITS sequences (numbers of these positions in the respective alignments are given in Fig. 3). All syngens showed characteristic patterns among the SSU and ITS. Only the syngens R1 and R2 could not be distinguished using the SSU only, however, in combination with the ITS, each syngen is characterized by unique synapomorphies as highlighted in yellow (Fig. 3). In addition, few variable base positions within syngens (marked in blue in Fig. 3) have been recognized in the ITS regions. For comparison with literature data, we also analyzed all available sequences of the mitochondrial COI gene to find synapomorphies for the five syngens. Within this gene, only 18 variable positions at the amino acid level could be discovered of which 13 are diagnostic for the five syngens (Fig. 3).
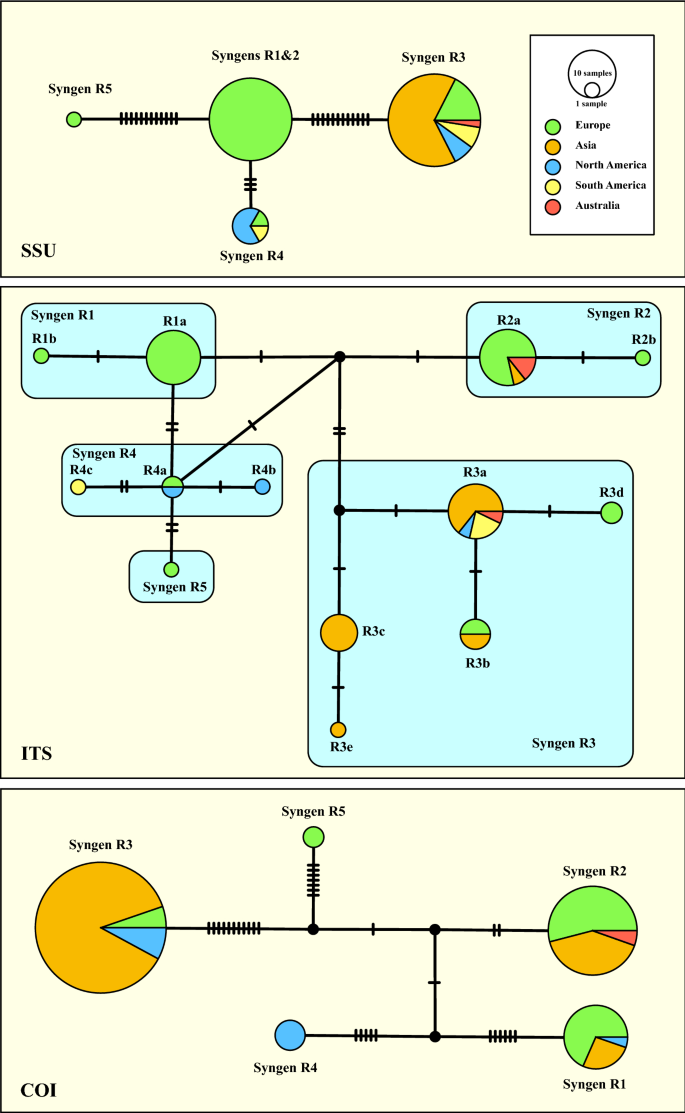
The synapomorphies discovered above were used to get insights into the geographical distribution of each P. bursaria syngen. Despite the complete SSU and ITS rDNA sequences included in the phylogeny presented in Fig. 2, records of the partial SSU or ITS rDNA sequences are available in GenBank (BLASTn search; 100% identity;13). Considering the metadata of our investigated strains and of the entries in GenBank (Supplementary Table 1), we constructed three haplotype networks using the Templeton-Crandall-Sing (TCS) approach. The SSU haplotype network (Fig. 4) containing 84 records showed that the syngens R1, R2 and R5 were only found in Europe, whereas the other three syngens have been discovered around the world. A similar distribution pattern occurred when using the ITS (101 entries in GenBank). Records of syngens R1 and R5 have only been found in Europe, whereas all other syngens were distributed around the world. The 132 COI records found in GenBank by the BLASTn search were used for the haplotype network, which also showed the similar pattern (Fig. 4).
TCS haplotype networks of the five syngens inferred from SSU, ITS rRNA, and COI sequences of the Paramecium bursaria species complex. This network was inferred using the algorithm described by Clement et al.40,41. Sequence nodes corresponding to samples collected from different geographical regions.
Ciliate Taxonomy
Considering all our findings, P. bursaria is morphologically highly variable, and obviously represents a cryptic species complex (Figs. 5, 6; Supplementary Table 2). The known five syngens most likely represent biological species according to Mayr14 and can be attributed to the cryptic species described by Greczek-Stachura et al.11. As mentioned above, the assignments of these cryptic species by Greczek-Stachura et al.11 have not been validly described according to the ICZN. In addition, the naming using a mixture of Latin prefix and Greek suffix is also not appropriate (the epithet bursa derived from the Greek word byrsa). Therefore, we describe the five syngens as new species as follows. The general morphological features of these species are summarized in Table 1.
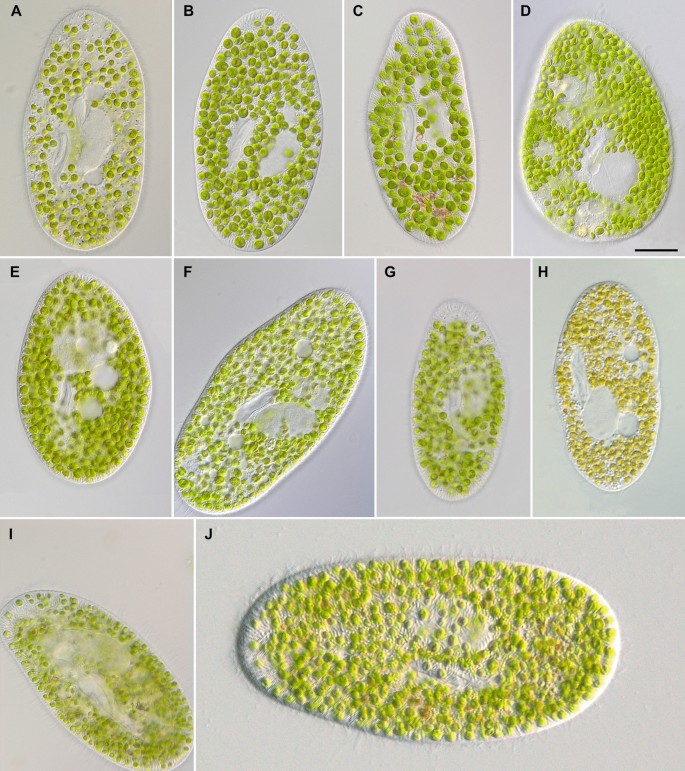
Ventral views of Paramecium bursaria morphotypes in vivo: P. protobursaria (syngen R1), i.e., strains SAG 2645 (A) and PB-25 (B); P. deuterobursaria (syngen R2), i.e., strains CCAP 1660/36 (C) and CCAP 1660/34 (D); P. tritobursaria (syngen R3), i.e., strains CCAP 1660/28 (E), CCAP 1660/26 (F) and CCAP 1660/31 (G); P. tetratobursaria (syngen R4), i.e., strains CCAP 1660/25 (H) and CCAP 1660/33 (I); P. pentobursaria (syngen R5), i.e., strain CCAP 1660/30 (J). Scale bar 20 µm.
Morphological details of the Paramecium bursaria species complex from specimens of strains PB-25 (A), CCAP 1660/30 (B), SAG 2645 (C, F, G, I, L–N), CCAP 1660/36 (D), CCAP 1660/26 (E, H), CCAP 1660/30 (J, O), CCAP 1660/16 (K) in vivo (A–F, H–O) and after silver nitrate staining (G). Adoral membranelles (A, B), endosymbiotic algae Micractinium conductrix (C), caudal and somatic cilia (D), arrows denote excretory pores of the contractile vacuoles: extruded extrusomes are shown and caudal cilia (E), ventral views showing the preoral suture and the oral opening (F), the ciliary pattern (G), arrows denote excretory pores of the contractile vacuoles (H), trichocysts and symbiotic algae underneath the pellicula (I, J), cell size variations (K), radial collecting channels (white arrows) and excretory pores (black arrows) of contractile vacuoles (L), macro- and micronucleus (M), cytopyge and characteristic rectangular pellicular pattern (N), pattern of the pellicula (O). AS anterior suture, CC caudal cilia, CP cytopyge (cell after), CV contractile vacuole, EP excretory pore of a contractile vacuole, EX extrusomes, M1–M3 membranelles 1–3, MA macronucleus, MI micronucleus, OO oral opening, S symbiotic algae, SC somatic cilia, SK somatic kineties, UM undulating membrane. Scale bars 10 µm (A, I), 20 µm (B, D–H, J, L–O), 50 µm (K).
Paramecium protobursaria sp. nov.
Synonym: Paramecium primabursaria nom. inval.
Description: The strains SAG 27.96 and PB-25 belong to syngen R1 according to Greczek-Stachura et al.10,11 and differ from other syngens by their SSU and ITS rDNA sequences (MT231333). From morphology, the cells are ellipsoidal to broadly ellipsoidal and dorso-ventrally flattened in vivo. The cells measure 70–164 × 44–65 µm; the single macronucleus is located around mid-cell and measures 25–38 × 11–22 µm; the adjacent single compact micronucleus measures 11–20 × 5–8 µm; the usually two (rarely one) contractile vacuoles, one in the anterior and one in the posterior cell portion have radial collecting channels and 1–3 excretory pores each; the number of ciliary rows/20 µm is 14–22; the length of the caudal cilia is 9–19 µm; the numerous trichocysts located in the cell cortex are 4–6 µm in length. The symbiotic algae belong to M. conductrix; the larger algae measure 4–7 × 4–7 µm; the smaller algal cells measure 2–5 × 2–5 µm.
Geographic distribution: The investigated strains of syngen R1 were found in Europe: Göttingen, Germany; Lake Mondsee, Austria. In addition, this species has been reported from different places in Europe, Asia and North America (see details in Supplementary Table 1).
Reference material: Strain SAG 27.96 and the clonal strain SAG 2645 derived from SAG 27.96 are available at the Culture Collection of Algae (SAG), University of Göttingen, Germany.
Holotype: Two slides (one holotype, one paratype) with protargol-impregnated specimens from the clonal culture SAG 2645, which derived from the reference material SAG 27.96, isolated from the pond of the Old Botanical Garden of the University of Göttingen (Germany), have been deposited in the Oberösterreichisches Landesmuseum at Linz (LI, Austria).
Zoobank Registration LSID: AFD967ED-BC2A-43FD-847E-5DF588BB025C.
Paramecium deuterobursaria sp. nov.
Synonym: Paramecium bibursaria nom. inval.
Description: The strains CCAP 1660/34 and CCAP 1660/36 belong to syngen R2 according to Greczek-Stachura et al.10,11 and differ from other syngens by their SSU and ITS rDNA sequences (OK318487). From morphology, the cells are ellipsoidal to broadly ellipsoidal and dorso-ventrally flattened in vivo. The cells measure 81–167 × 35–83 µm; the single macronucleus is located around mid-cell and measures 24–46 × 10–32 µm; the adjacent single compact micronucleus measures 10–18 × 5–9 µm, no micronucleus seen in live cells of strain CCAP 1660/34; the usually two (rarely one or three) contractile vacuoles, one in the anterior and one in the posterior cell portion have radial collecting channels and 1–3 excretory pores each; the number of ciliary rows/20 µm is 13–22; the length of the caudal cilia is 11–20 µm; the numerous trichocysts located in the cell cortex are 4–6 µm in length. The symbiotic algae belong to M. conductrix; the larger algae measure 5–7 × 4–7 µm; the smaller algal cells measure 3–5 × 2–5 µm.
Geographic distribution: The investigated strains of syngen R2 were found in Europe: Zurich, Switzerland; Lake Piburg, Austria. In addition, this species has been reported from different places in Europe, Asia and Australia (see details in Supplementary Table 1).
Reference material: Strain CCAP 1660/36 is available at the Culture Collection of Algae and Protozoa (CCAP) at the Scottish Association for Marine Science, Oban, Scotland.
Holotype: Two slides (one holotype, one paratype) with protargol-impregnated specimens from the reference material CCAP 1660/36, isolated from Lake Piburg (Tyrol, Austria), have been deposited in the Oberösterreichisches Landesmuseum at Linz (LI, Austria).
Zoobank Registration LSID: D1C20BE6-9A15-4A3D-A7E5-DFC31FF04679.
Paramecium tritobursaria sp. nov.
Synonym: Paramecium tribursaria nom. inval.
Description: The strains CCAP 1660/26, CCAP 1660/28 and CCAP 1660/31 belong to syngen R3 according to Greczek-Stachura et al.10,11 and differ from other syngens by their SSU and ITS rDNA sequences (MT231339). From morphology, the cells are ellipsoidal to broadly ellipsoidal and dorso-ventrally flattened in vivo. The cells measure 80–153 × 49–73 µm; the single macronucleus is located around mid-cell and measures 21–53 × 12–31 µm; the adjacent single compact micronucleus measures 9–17 × 3–6 µm; no micronucleus seen in live cells of strain CCAP 1660/28; the usually two (rarely one or three) contractile vacuoles, one in the anterior and one in the posterior cell portion have radial collecting channels and 1–3 excretory pores each; the number of ciliary rows/20 µm is 12–20; the length of the caudal cilia is 8–19 µm; the numerous trichocysts located in the cell cortex are 4–6 µm in length. The symbiotic algae belong to C. variabilis; the larger algae measure 4–7 × 3–6 µm; the smaller algal cells measure 3–5 × 2–4 µm.
Geographic distribution: The investigated strains of syngen R3 were found in Europe and Asia: Lake Piburg, Austria; Tokyo, Japan; Khabarovsk region, Amur River, Russia. In addition, this species has been reported from different places in Europe, Asia, North and South America as well as in Australia (see details in Supplementary Table 1).
Reference material: Strain CCAP 1660/26 is available at the Culture Collection of Algae and Protozoa (CCAP) at the Scottish Association for Marine Science, Oban, Scotland.
Holotype: Two slides (one holotype, one paratype) with protargol-impregnated specimens from the reference material CCAP 1660/26, isolated from Japan, have been deposited in the Oberösterreichisches Landesmuseum at Linz (LI, Austria).
Zoobank Registration LSID: CC0FBA7E-9E3A-4C37-B424-C9BFF2018EC0.
Paramecium tetratobursaria sp. nov.
Synonym: Paramecium tetrabursaria nom. inval.
Description: The strains CCAP 1660/25 and CCAP 1660/33 belong to syngen R4 according to Greczek-Stachura et al.10,11 and differ from other syngens by their SSU and ITS rDNA sequences (MT231347). From morphology, the cells are ellipsoidal to broadly ellipsoidal and dorso-ventrally flattened in vivo. The cells measure 65–179 × 37–79 µm; the single macronucleus is located around mid-cell and measures 18–53 × 10–29 µm; the adjacent single compact micronucleus measures 8–18 × 4–10 µm; the usually two (rarely one or three) contractile vacuoles, one in the anterior and one in the posterior cell portion have radial collecting channels and 1–3 excretory pores each; the number of ciliary rows/20 µm is 14–19; the length of the caudal cilia is 12–20 µm; the numerous trichocysts located in the cell cortex are 4–7 µm in length. The symbiotic algae belong to C. variabilis (CCAP 1660/25) and M. conductrix (CCAP 1660/33); the larger algae measure 3–6 × 3–6 µm; the smaller algal cells measure 2–5 × 1–4 µm.
Geographic distribution: The investigated strains of syngen R4 are found in North- and South America: Burlington, North Carolina, USA; San Pedro de la Paz, Laguna Grande, Chile. In addition, this species has been reported from Europe (see details in Supplementary Table 1).
Reference material: Strain CCAP 1660/25 is available at the Culture Collection of Algae and Protozoa (CCAP) at the Scottish Association for Marine Science, Oban, Scotland.
Holotype: Two slides (one holotype, one paratype) with protargol-impregnated specimens from the reference material CCAP 1660/25, isolated from a pond in Burlington (North Carolina, USA), have been deposited in the Oberösterreichisches Landesmuseum at Linz (LI, Austria).
Zoobank Registration LSID: 78BA9923-07A9-4918-AD7C-9E5E15CC9CDB.
Paramecium pentobursaria sp. nov.
Synonym: Paramecium pentabursaria nom. inval.
Description: The strain CCAP 1660/30 belongs to syngen R5 according to Greczek-Stachura et al.10,11 and differs from other syngens by their SSU and ITS rDNA sequences (MT231348). From morphology, the cells are ellipsoidal to broadly ellipsoidal and dorso-ventrally flattened in vivo. The cells measure 161–194 × 76–99 µm; the single macronucleus is located around mid-cell and measures 24–47 × 19–31 µm; the adjacent single compact micronucleus measures 13–20 × 4–9 µm; the usually two (rarely one or three) contractile vacuoles, one in the anterior and one in the posterior cell portion have radial collecting channels and 1–4 excretory pores each; the number of ciliary rows/20 µm is 13–19; the length of the caudal cilia is 14–25 µm; the numerous trichocysts located in the cell cortex are 5–7 µm in length. The symbiotic algae belong to C. variabilis; the larger algae measure 5–6 × 5–6 µm; the smaller algal cells measure 4–5 × 3–4 µm.
Geographic distribution: The investigated strain of Syngen R5 was found in Europe: Astrakhan Nature Reserve, Russia.
Reference material: Strain CCAP 1660/30 is available at the Culture Collection of Algae and Protozoa (CCAP) at the Scottish Association for Marine Science, Oban, Scotland.
Holotype: Two slides (one holotype, one paratype) with protargol-impregnated specimens from the reference material CCAP 1660/30, isolated from Astrakhan Nature Reserve (Russia), have been deposited in the Oberösterreichisches Landesmuseum at Linz (LI, Austria).
Zoobank Registration LSID: 6629FA71-E00F-48C6-83AB-61C0CA4823B6.
Syngen Affiliation related to Ciliate Morphology, Endosymbionts and Geographic Distribution
Pearson-correlations of morphometric, syngen-specific and endosymbiont datasets of the P. bursaria strains revealed four significant positive correlations (p < 0.05 and − 0.75 > r > 0.75) between ciliate cell length (BLEN) and width (BWID), BWID and macronucleus width (MACWID), as well as length and width of large symbiotic algae (LSALEN and LSAWID; Fig. 7).
Pearson-correlations of morphometric, symbiont and syngen data of Paramecium strains under study. Colored dots indicate the strength of correlation, and the size of dots represent p-values. Bold squares highlight significant correlations, with − 0.75 > r > 0.75 and p < 0.05. Abbreviations: ANVAC number of excretory pores in anterior contractile vacuole, ALSPEC algal species, BLEN body/cell length, BWID body/cell width, CAUCIL caudal cilia length, CILROW number of ciliary rows, EXTLEN extrusome/trichocyst length, GEO geographical region, LSALEN large symbiotic algae length, LSAWID large symbiotic algae width, MACLEN macronucleus length, MACWID macronucleus width, MICLEN micronucleus length, MICWID micronucleus width, POVAC number of excretory pores in posterior contractile vacuole, SSALEN small symbiotic algae length, SSAWID small symbiotic algae width, SYN syngen affiliation.
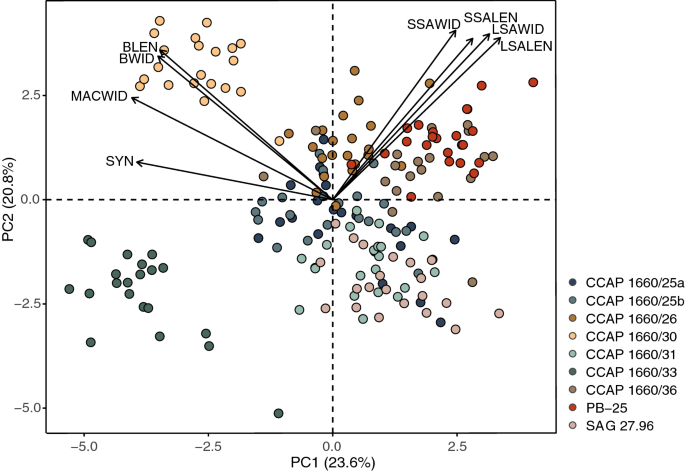
The results of the principal component analysis (PCA) are summarized in the ordination diagram in Fig. 8. The first two axes explain 44.4% of the total variation in the investigated features. Only the first five components (out of 18) had eigenvalues > 1, accounting for 73.1% variation in total (Supplementary Table 3). Principal component axis 1 (PC1) appears to be most negatively weighted by syngen (SYN) and width of the macronucleus (MACWID), separating CCAP 1660/30 and CCAP 1660/33 from the other strains. Principal component axis 2 (PC2) is primarily positively influenced by symbiotic algae characteristics (LSALEN, LSAWID, small symbiotic algal length (SSALEN) and width (SSAWID)) and, ciliate cell length (BLEN) and width (BWID; Supplementary Table 4), partitioning strain PB-25, CCAP 1660/26 and CCAP 1660/36 from CCAP 1660/31 and SAG 27.96 (Fig. 8).
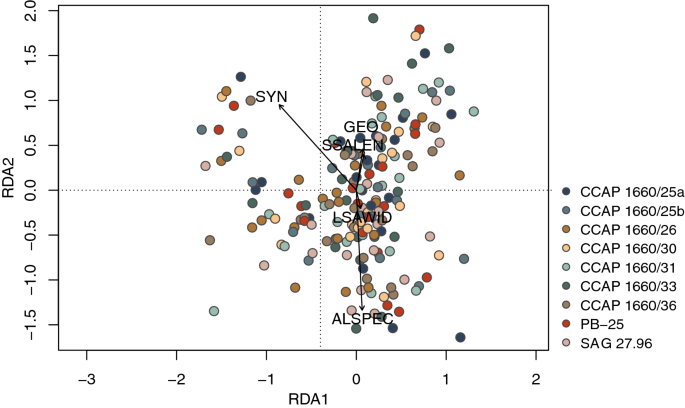
The redundancy analysis (RDA; Fig. 9) revealed a large difference between morphometric features and the tested set of explanatory variables (i.e., algal species (ALSPEC), LSAWID, SSALEN, SYN and GEO) as only 26.9% of the total variation could be explained.
Discussion
Among strains of P. bursaria, six syngens have been discovered so far by mating experiments5,9,10. Our phylogenetic analyses using a concatenated dataset of SSU and ITS sequences revealed five highly supported lineages among the investigated P. bursaria strains, which clearly corresponded to the cryptic species assigned to syngens R1-R5 according to Greczek-Stachura et al.10,11. All syngens could be individually distinguished by their molecular signatures (Fig. 3), however, isolates belonging to syngens R1 and R2 could not be recognized by sequencing their SSU rDNA only.
Paramecium bursaria are distributed worldwide (Fig. 4). Only the syngens R1 and R5 have been found in Europe, whereas the other syngens have been recorded from Europe, Asia, North and South America and Australia. However, very little is known from other regions of the world such as South America, Australia or Africa.
The available strains of P. bursaria were mostly isolated for studying their green algal endosymbionts. Originally, these endosymbionts were differentiated into two groups: an American (or Southern) and a European (or Northern) type15,16,17,18,19,20. Pröschold et al.1 taxonomically revised both groups and emended the description of the two species C. variabilis and M. conductrix based on the authentic strains (SAG 211–6 and SAG 241.80). Considering both of these strains, Spanner et al.21 developed an easy diagnostic PCR approach for the isolation and identification of the zoochlorellae living in P. bursaria revealing that both endosymbiotic species were found in almost all syngens (Fig. 2). In syngen R3, only C. variabilis was detected. Interestingly, in the strain CCAP 1660/10, belonging to syngen R4, C. vulgaris has been reported19. The assignment to syngen R4 of this strain is surprising because this is the only record from Europe. This has to be taken with caution because the assignment of another strain CCAP 1660/11 to syngen R5 by Hoshina & Imamura19 is incorrect as demonstrated in our study. This strain belongs to syngen R1 (Fig. 2; Table S1). Unfortunately, this strain is lost and the syngen assignment cannot be proven. Chlorella vulgaris occurred either free-living or as endosymbiont of ciliates such as Euplotes daidaleos, Coleps hirtus, Climacostomum virens and P. bursaria1. Unfortunately, those ciliates are neither available in public culture collections nor their molecular datasets in public databases. Consequently, the ciliate host/syngen from which C. vulgaris had originally been isolated remains unknown. Recently, Greczek-Stachura et al.22 reported that Chlorella sorokiniana, a free-living species from warm-temperate habitats, also occurred in three isolates of P. bursaria collected in Lake Baikal and the Kamchatka region (Asian part of Russia). The investigations were based on the partial nuclear large subunit (LSU) rDNA and chloroplast genes encoding the ribosomal protein L36 (rpl36) and translation initiation factor IF-1 (infA). Unfortunately, no ITS of these isolates has been sequenced and, accordingly, the assignment to C. sorokiniana is questionable. For example, the Chinese P. bursaria strain Cs2 (R3) bears the “American” type of endosymbiont as demonstrated by Hoshina et al.16, which is C. variabilis and not C. sorokiniana1. Moreover, two other reports of Greczek-Stachura et al.22 were probably incorrect: the strains AZ20-1 (according to CCAP 1660/30 in our study; R5) and Yad1-g (R3), both have C. variabilis, and not C. vulgaris as endosymbiont21,23 (this study; see also Supplementary Table 1). Both molecular markers (partial LSU, rpl36-infA) do not have the diagnostic power for a discrimination of green algal endosymbionts at the species level.
Ciliate descriptions and taxonomic assignments basically require the detailed study of species-specific diagnostic features, relevant literature, and biogeographical aspects24. It is consequently necessary that molecular and microscopic approaches are closely linked for a certain population or strain, especially when the ciliate’s ecology is in the focus of a study25,26. Nevertheless, as molecular approaches are becoming major tools in ciliate ecology, the morphological identification of a ciliate still remains hidden in many cases27. Since the first description by Ehrenberg28, P. bursaria was often identified only by the presence of green algal endosymbionts despite reported findings of free-living and naturally algal-free individuals29. Moreover, the symbiotic algae can be artificially ‘removed’ from P. bursaria for experimental approaches30,31. Detailed morphological investigations of this species were lacking for a long time under the assumption that all ‘green’ paramecia were assignable to P. bursaria. Kreutz et al.12 provided a detailed description on a population of P. bursaria and another green congener, P. chlorelligerum, a rare species that was originally established by Kahl32. Despite Kalmus33 already mentioned a high variability of especially the cell shape among Paramecium species, very little is known about their phenotypic plasticity. However, from our detailed morphometric analyses of the studied strains, we can confirm that the morphological features unequivocally revealed P. bursaria and showed that the characteristics tended to be highly variable (Supplementary Table 2) as already reported by Foissner et al.34 in their identification key.
Conclusions
The P. bursaria species complex is widely distributed around the world. As shown, sequencing and analyzing of the SSU and ITS rDNA of isolated samples and strains can clearly assign them to the syngen level. The five lineages revealed by our phylogenetic analyses clearly corresponded to the syngen affiliations. Unfortunately, the syngens could not be identified by morphology only. Further studies are needed to get more insights about the geographical distribution of the P. bursaria species complex and its endosymbionts, which both can be clearly determined using our molecular tools presented here. The usage of diagnostic PCR approach provided an easy method for identification of the green algal endosymbionts.
Methods
Origin of the investigated strains and cultivation of ciliates and their endosymbionts
The origin of the investigated P. bursaria strains is summarized in Table 2. As the respective strains preferred different media, we used modified Bold Basal Medium (3N-BBM + V; medium 26a in Schlösser35) with the addition of 30 ml of soil extract per liter (S/BBM; see Spanner et al.21), modified Woods Hole MBL (WC) medium36 mixed with Volvic® (V) mineral water, in various concentrations, V/WC 1:1, and V/WC 5:1 v/v. All cultures were maintained at 15–21 °C under a light:dark cycle of 12:12 h (photon flux rate up to 50 μmol m−2 s−1). The isolated green algal endosymbionts were cultivated under the same culture conditions in Basal Medium with beef extract (ESFl; medium 1a in Schlösser37).
DNA extraction, PCR and sequencing
Genomic DNA of the P. bursaria strains was extracted using the DNeasy Plant Mini Kit (Qiagen GmbH, Hilden, Germany). The SSU and ITS rDNA were amplified using the Taq PCR Mastermix Kit (Qiagen GmbH, Hilden, Germany) with the primers EAF3 and ITS055R as described in Spanner et al.21. The datasets generated and analyzed during the current study are available in GenBank (https://www.ncbi.nlm.nih.gov). The GenBank accession numbers are given in Table 2.
Identification of the green algal endosymbionts
The green algal endosymbionts were identified using three different approaches: (i) the diagnostic PCR approach21, (ii) direct sequencing using the green algal specific primers G500F and G800R as described by Darienko et al.38, and (iii) isolation using the method introduced by Spanner et al.21 and sequencing of the SSU and ITS rDNA with the green algal specific primers. The respective identification method used is given in Table 2.
Phylogenetic and network analyses
All sequences were aligned to their secondary structures as demonstrated for strain SAG 27.96 (Fig. 1; Supplementary Fig. 1). The secondary structures were folded using the software mfold39, which uses the thermodynamic model (minimal energy) for RNA folding. The visualization of the structures was manually done using the program Illustrator CS5.1 (Adobe Inc.). For the phylogenetic analyses, we calculated the log-likelihood values of 56 models using the automated selection tool implemented in PAUP version 4.0b16940 to test which evolutionary model fit best for the dataset. The best model according to the Akaike criterion by PAUP was chosen. The settings of the best model were given in the figure legends. The following methods were used for the phylogenetic analyses: distance, maximum parsimony, and maximum likelihood, all included in PAUP version 4.0b16940.
The secondary structures of the SSU and ITS rRNA sequences were compared to find genetic synapomorphies, which were used for the construction of haplotype networks. To establish an overview on the distribution of each syngen, the SSU and ITS haplotypes were used for a BLASTn search (100% coverage, > 97% identity; Altschul et al.13). To construct the haplotype networks, we used the Templeton-Crandall-Sing (TCS) network tool41,42 implemented in PopART43. The COI sequences presented in Greczek-Stachura et al.10,11 were analyzed to find synapomorphies at the amino acid level.
Morphological investigations of ciliates and endosymbionts
The morphology of the P. bursaria strains and their endosymbionts was studied mainly from living individuals, which were cloned using the isolation method (steps 1 and 2) described in Spanner et al.21. After 24 h of starvation, the single ciliate cells were cultivated in 24-well plates (Biomedica) each in the cultivation media mentioned above. To reveal their ciliary pattern, additionally, a dry silver nitrate impregnation was applied44. All protists were studied under bright field and differential interference contrast optics with an Olympus BX51 and an Olympus BX60 microscope (Olympus, Vienna, Austria) with 40–1000 × magnifications. For documentation and measurements, two digital image analysis systems were used (ProgRes SpeedXT core 5 2.9.0.1. and ProgRes Capture Pro imaging system version 2.9.0.1, Jenoptik, Jena, Germany). The ciliates were identified by means of the key of Foissner et al.34 and Kreutz et al.12 and standard morphometric calculations were done. The green algae were identified by comparison with the descriptions presented in Pröschold et al.1. Type slides (holotypes, paratypes) were stained with protargol (Skibbe method)45.
Multivariate analyses of morphometric, symbiont and syngen data of Paramecium strains
All correlation and multivariate analyses were conducted in R version 4.1.1 using the stats and vegan packages. Statistical analyses included all morphometric, syngen and geographic origin information, as well as algal symbiont features of the Paramecium strains under study (Figs. 7, 8, 9). Strains CCAP 1660/28 and CCAP 1660/34 were excluded from downstream analyses as no micronucleus data (= no micronucleus could be seen in the ciliates) were available.
All data were first checked for normality with a Shapiro–Wilk test and then used to run standard Pearson correlations between each other to rule out any correlations. Correlations were considered significant if p < 0.05 and − 0.75 > r > 0.75. The overall variation in the dataset was summarized with a PCA (unconstrained ordination). The relationship between morphometric features (response variables) and explanatory variables, representing syngen and symbiont features, was summarized using an RDA (constrained ordination) with centered data. Features GEO, LSALEN and SSAWID were removed from analysis due to multicollinearity with SYN, LSAWID and SSALEN, respectively (Supplementary Tables 3–4). The significance of the observed relationship was tested with a Monte Carlo permutation test using 999 permutations.
References
Pröschold, T., Darienko, T., Silva, P. C., Reisser, W. & Krienitz, L. The systematics of “Zoochlorella" revisited employing an integrative approach. Environ. Microbiol. 13, 350–364 (2011).
Reisser, W. & Häder, D. P. Role of endosymbiotic algae in photokinesis and photophobic responses of ciliates. Photochem. Photobiol. 39, 673–678 (1984).
Sommaruga, R. & Sonntag, B. Photobiological aspects of the mutualistic association between Paramecium bursaria and Chlorella. In: Endosymbionts in Paramecium (ed. Fujishima, M.) Microbiol. Monogr. 12, 111–130 (2009).
Fujishima, M. Endosymbionts in Paramecium. Microbiol. Monogr. 12, 1–252 (2009).
Sonneborn, T.M. Breeding systems, reproductive methods, and species problems in Protozoa. In: The Species Problem. American Association for the Advancement of Science 155–324 (1957).
Jennings, H. S. Paramecium bursaria: Mating types and groups, mating behavior, self-sterility. Am. Nat. 73, 414–431 (1939).
Jennings, H. S. & Opitz, P. Genetics of Paramecium bursaria. IV. A fourth variety from Russia. Lethal crosses with an American variety. Genetics 29, 576–583 (1944).
Chen, T. T. Varieties and mating types in Paramecium bursaria. I. New variety and types from England, Ireland and Czechoslovakia. Proc. Natl. Acad. Sci. USA 32, 173–181 (1946).
Bomford, R. The syngens of Paramecium bursaria: New mating types and inter-syngenic mating reactions. J. Protozool. 13, 501–504 (1966).
Greczek-Stachura, M. et al. Identification of Paramecium bursaria syngens through molecular markers—Comparative analysis of three loci in the nuclear and mitochondrial DNA. Protist 163, 671–685 (2012).
Greczek-Stachura, M., Rautian, M. & Tarcz, S. Paramecium bursaria—A complex of five cryptic species: Mitochondrial DNA COI haplotype variation and biogeographic distribution. Diversity 13, 589 (2021).
Kreutz, M., Stoeck, T. & Foissner, W. Morphological and molecular characterization of Paramecium (Viridoparamecium nov. subgen.) chlorelligerum Kahl 1935 (Ciliophora). J. Eukaryot. Microbiol. 59, 548–563 (2012).
Altschul, S. F., Gish, W., Miller, W., Myers, E. W. & Lipman, D. J. Basic local alignment search tool. J. Mol. Biol. 215, 403–410 (1990).
Mayr, E. Systematics and the Origin of Species (Columbia University Press, 1942).
Hoshina, R., Kamako, S. & Imamura, N. Phylogenetic position of endosymbiotic green algae in Paramecium bursaria Ehrenberg from Japan. Plant Biol. 6, 447–453 (2004).
Hoshina, R., Kato, Y., Kamako, S. & Imamura, N. Genetic evidence of “American” and “European” type symbiotic algae of Paramecium bursaria Ehrenberg. Plant Biol. 7, 526–532 (2005).
Hoshina, R., Iwataki, M. & Imamura, N. Chlorella variabilis and Micractinium reisseri sp. nov. (Chlorellaceae, Trebouxiophyceae): Redescription of the endosymbiotic green algae of Paramecium bursaria (Peniculia, Oligohymenophorea) in the 120th year. Phycol. Res. 58, 188–201 (2010).
Gaponova, I.N., Andronov, E.E., Migunova, A.V., Vorobyev, K.P., Chizhevskaja, E.P. & Kvitko, K.V. Genomic dactyloscopy of Chlorella sp., symbionts of Paramecium bursaria. Protistology 4, 311–317 (2006/2007).
Hoshina, R. & Imamura, N. Multiple origins of the symbioses in Paramecium bursaria. Protist 159, 53–63 (2008).
Hoshina, R. & Imamura, N. Origins of algal symbionts of Paramecium bursaria. Microbiol. Monogr. 12, 1–29 (2009).
Spanner, C., Darienko, T., Biehler, T., Sonntag, B. & Pröschold, T. Endosymbiotic green algae in Paramecium bursaria: A new isolation method and a simple diagnostic PCR approach for the identification. Diversity 12, 240 (2020).
Greczek-Stachura, M., Zagata Lesnicka, P., Tarcz, S., Rautian, M. & Mozdzern, K. Genetic diversity of symbiotic green algae of Paramecium bursaria syngens originating from distant geographical locations. Plants 10, 609 (2021).
Kodama, Y. et al. Comparison of gene expression of Paramecium bursaria with and without Chlorella variabilis symbionts. BMC Genomics 15, 183 (2014).
Agatha, S., Ganser, M. H. & Santoferrara, L. F. The importance of type species and their correct identification: A key example from tintinnid ciliates (Alveolata, Ciliophora, Spirotricha). J. Eukaryot. Microbiol. 68, e12865 (2021).
Pröschold, T. et al. An integrative approach sheds new light onto the systematics and ecology of the widespread ciliate genus Coleps (Ciliophora, Prostomatea). Sci. Rep. 11, 5916 (2021).
Frantal, D. et al. Molecular data reveal a cryptic diversity in the genus Urotricha (Alveolata, Ciliophora, Prostomatida), a key player in freshwater lakes, with remarks on their morphology, food preferences, and distribution. Front. Microbiol. 12, 787290 (2022).
Warren, A. et al. Beyond the “Code”: A guide to the description and documentation of biodiversity in ciliated protists (Alveolata, Ciliophora). J. Eukaryot. Microbiol. 64, 539–554 (2017).
Ehrenberg, C.G. Über die Entwickelung und Lebensdauer der Infusionsthiere; nebst ferneren Beiträgen zu einer Vergleichung ihrer organischen Systeme. Abh. Preuss. Akad. Wiss. Phys. Math. Kl. 1831, 1–154 (1831).
Tonooka, Y. & Watanabe, T. A natural strain of Paramecium bursaria lacking symbiotic algae. Eur. J. Protistol. 38, 55–58 (2002).
Summerer, M., Sonntag, B. & Sommaruga, R. An experimental test of the symbiosis specificity between the ciliate Paramecium bursaria and strains of the unicellular green alga Chlorella. Environ. Microbiol. 9, 2117–2122 (2007).
Summerer, M., Sonntag, B., Hörtnagl, P. & Sommaruga, R. Symbiotic ciliates receive protection against UV damage from their algae: A test with Paramecium bursaria and Chlorella. Protist 160, 233–243 (2009).
Kahl, A. Urtiere oder Protozoa I: Wimpertiere oder Ciliata (Infusoria) 4. Peritricha und Chonotricha. Tierwelt Dtl. 30, 651–886 (1935).
Kalmus, H. Paramecium—Das Pantoffeltierchen. Eine monographische Zusammenfassung der wichtigsten Kenntnisse. Gustav Fischer Jena, 188 p (1931).
Foissner, W., Berger, H. & Kohmann, F. Taxonomische und ökologische Revision der Ciliaten des Saprobiensystems. Band III: Hymenostomata, Prostomatida, Nassulida. Informationsber. Bayer. Landesamt für Wasserwirtschaft: Munich, Germany 1/94, 1–548 (1994).
Schlösser, U. G. Additions to the culture collection of algae since 1994. Bot. Acta. 110, 424–429 (1997).
Guillard, R. R. L. & Lorenzen, C. J. Yellow-green algae with chlorophyllide C 2. J. Phycol. 8, 10–14 (1972).
Schlösser, U. G. SAG-Sammlung von Algenkulturen at the University of Göttingen, Catalogue of Strains 1994. Bot. Acta 107, 113–186 (1994).
Darienko, T., Rad-Menéndez, C., Campbell, C. & Pröschold, T. Are there any true marine Chlorella species? Molecular phylogenetic assessment and ecology of marine Chlorella-like organisms, including a description of Droopiella gen. nov. Syst. Biodivers. 17, 811–829 (2019).
Zuker, M. Mfold web server for nucleic acid folding and hybridization prediction. Nucleic Acid Res. 31, 3406–3615 (2003).
Swofford, D.L. PAUP* Phylogenetic Analysis Using Parsimony (*and Other Methods), Version 4.0b10. Sinauer Associates (2002).
Clement, M., Posada, D. & Crandall, K. A. TCS: a computer program to estimate gene genealogies. Mol. Ecol. 9, 1657–1659 (2000).
Clement, M., Snell, Q., Walker, P., Posada, D. & Crandall, K. TCS: Estimating gene genealogies. Parallel Distrib. Process. Symp. Int. Proc. 2, 184 (2002).
Leigh, J. W. & Bryant, D. POPART: Full-feature software for haplotype network construction. Methods Ecol. Evol. 6, 1110–1116 (2015).
Foissner, W. An update of basic light and scanning electron microscopic methods for taxonomic studies of ciliated protozoa. Int. J. Syst. Evol. Microbiol. 64, 271–292 (2014).
Skibbe, O. An improved quantitative protargol stain for ciliates and other planktonic protists. Arch. Hydrobiol. 130, 339–347 (1994).
Acknowledgements
The study was funded by the Austrian Science Fund (FWF): P28333-B25. We thank Hans-Dieter Görtz, Renu Gupta, Thomas Posch, Ulrike Scheffel, Ulrike Koll and Monika Summerer for the collection of strains and help in the laboratory. We thank two anonymous reviewers and Alexey Potekhin for constructive comments on an earlier version of our manuscript.
Author information
Authors and Affiliations
Contributions
All authors conceived and designed the study and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Spanner, C., Darienko, T., Filker, S. et al. Morphological diversity and molecular phylogeny of five Paramecium bursaria (Alveolata, Ciliophora, Oligohymenophorea) syngens and the identification of their green algal endosymbionts. Sci Rep 12, 18089 (2022). https://doi.org/10.1038/s41598-022-22284-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-22284-z
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.