Abstract
Metabolic Syndrome is a clustering of obesity, hyperglycemia/insulin resistance, dyslipidemia, and hypertension. We aimed to determine the incidence of metabolic syndrome among Royal Thai Army (RTA) personnel and its risk factors. We conducted a retrospective cohort study using data from 2017 to 2021. Metabolic syndrome was defined by NCEP ATP III (2005 Revision). A total of 98,264 participants were enrolled in the present study. The overall incidence rate of metabolic syndrome was 3.7 per 100 person-year (95% CI 3.7–3.8). The statistically significant risk factors for metabolic syndrome included male sex (aHR 1.40; 95% CI 1.29–1.51), age > 35 years, current alcohol consumption, and no exercise. When stratified by sex, the incidence rate of metabolic syndrome among participants aged ≥ 45 years was higher than those aged < 35 years with aHR 6.34; 95% CI 6.01–6.70 for males and aHR 9.59; 95% CI 7.55–12.19 for females. Our data demonstrated that metabolic syndrome is a common health issue, especially among RTA personnel over 35 years. Alcohol consumption and sedentary behavior played an essential role in facilitating metabolic syndrome in this study population and are potential targets for intervention to enhance primary prevention of the sequelae of metabolic syndrome.
Similar content being viewed by others
Introduction
Metabolic syndrome is a clustering of obesity, hyperglycemia/insulin resistance, dyslipidemia (DLP), and hypertension (HT)1, 2. It identifies patients at high risk of developing type 2 diabetes (T2D) and atherosclerotic cardiovascular diseases (ASCVD)2, 3. Furthermore, metabolic syndrome is associated with a 2.4 times higher mortality rate compared with those without metabolic syndrome3.
The prevalence of metabolic syndrome in each geographic region is diverse. In the Asia–Pacific region, a recent study reported that the prevalence of metabolic syndrome ranged from 11.9 to 37.1%4 while its prevalence in South Asia was 26.1%5. Several studies reported different incidences of metabolic syndrome accounting for 3.1, 21.0, and 22.9 per 1000 person-years in Spain6, Japan7 and Taiwan8, respectively. In Thailand, recent studies have reported that the prevalence of metabolic syndrome among Thai males is 11.7 to 25.8%, while among females, prevalence is 8.2 to 26.3%9,10,11,12,13.
Military personnel constitute a specific population group exhibiting different behavior risk factors from the civilian population. Moreover, the institutional structures of the military convey harmful behaviors among personnel and weaken their desire to embrace healthier habits14. In Thailand, a recent study reported that the prevalence of current alcohol consumption among military personnel was approximately 70%15 higher than that among Thai civilians (68.5%)16. In addition, related evidence among Royal Thai Army (RTA) personnel emphasized that obesity prevalence has continuously risen from 2017 to 202115. A related meta-analysis reported that the overall estimated prevalence of metabolic syndrome among military personnel was 8.0%, according to the National Cholesterol Education Programme Adult Treatment Panel III (NCEP-ATP III) criteria17. However, the evidence on the incidence of metabolic syndrome and risk factors among the military population, including in Thailand, was scarce. Further evidence is needed concerning behavioral risk factors to enhance primary prevention of metabolic syndrome and its sequelae, including noncommunicable diseases (NCDs) and ASCVD.
Nationwide, approximately 130,000 Royal Thai Army (RTA) personnel participate in a yearly health examination provided by the RTA Medical Department18. In the present study, we investigated the incidence of metabolic syndrome among RTA personnel using the physical health examination in the RTA personnel database from 2017 to 2021. Importantly, sex is now evolving as a significant factor in developing metabolic dysregulation19. Furthermore, effect modification among risk factors influencing metabolic syndrome development may hold public health significance20, 21. Therefore, we also evaluated sex- and age-specific associations between behavioral factors and the incidence of metabolic syndrome among RTA personnel.
Methods
Study design and participants
We conducted a retrospective cohort study using data from 2017 to 2021. We retrieved data from the annual health examination database of RTA personnel after obtaining approval from the RTA Medical Department in Bangkok, Thailand. The missions of the RTA Medical Department include providing healthcare services, promoting health, and preventing disease for both military personnel and Thai civilians. The RTA Medical Department provides annual health examinations for RTA personnel through the Army Institute of Pathology, Armed Forces Research Institute of Medical Sciences and 37 RTA hospitals nationwide. The records are then reported to the RTA Medical Department in Bangkok. Eligible participants consisted of RTA personnel aged 18 to 60 years nationwide who had health examination records from 2017 to 2020. Of the approximately 130,000 RTA personnel, 125,211 (96.3%) participated in physical health examinations. Participants with a history of metabolic syndrome (n = 11,455; 8.8%) and those undergoing physical health examination at baseline only (n = 10,534; 8.1%) were excluded. A final target of 103,222 (79.4%) participants without a diagnosis of metabolic syndrome participated in the baseline review. We excluded 4958 (2.9%) participants because they had missing data concerning the diagnosis of metabolic syndrome. Therefore, a total of 98,264 (75.6%) participants without a diagnosis of metabolic syndrome at baseline were enrolled in the present study and followed up until 2021. The flow of the study is presented in Fig. 1.
Data collection
Annually, the Army Institute of Pathology, Armed Forces Research Institute of Medical Sciences and 37 RTA hospitals provide health examinations for RTA personnel. A self-report guide was created using a standardized case report form to obtain demographic characteristics, comorbidities and behavioral risk factors, including age, sex, health scheme, smoking status, history of alcohol consumption and exercise. The comorbidities included a history of HT, DLP and T2D.
The region of residence consisted of Bangkok, the north, northeast, central and south. The health scheme of the participants included Civil Servant Medical Benefits, Social Security and Universal Coverage. Behavioral risk factors were obtained from the self-reported questionnaire. Smoking status was divided in four groups including (1) never smoked, (2) ex-smoker defined as being smoke-free for 12 months, (3) irregular current smoker and (4) regular current smoker. Alcohol consumption was categorized in four groups including (1) never consumed, (2) exdrinker defined as alcohol-free for 12 months, (3) irregular current drinker and (4) regular current drinker. A history of T2D, HT and DLP comorbidity of study participants was defined using the information from the responses to the questionnaire, (1) “Have you ever been diagnosed with T2D or taken antihyperglycemic drugs?”, (2) “Have you ever been diagnosed with HT or taken antihypertensive drugs?” and (3) “Have you ever been diagnosed with DLP or taken lipid-lowering drugs?”.
The annual health examination dataset at baseline and follow-up also included anthropometric measurements of waist circumference, weight, height, systolic blood pressure and diastolic blood pressure. The information on laboratory testing and anthropometric measurements came from several RTA hospitals nationwide providing the measurements, which trained technicians performed, and the standard and quality of services of all RTA hospitals were certified by Healthcare Accreditation Institute, Thailand. Waist circumference was measured at midline level (between the inferior margin of the ribs and the superior border of the iliac crest), with a plastic tape22. The participant’s body weight was measured in kilograms and the height in centimeters. Blood pressure (BP) was measured using an automatic blood pressure monitor by an operator trained in standardized technique following the Thai guidelines on HT treatment. The participants were advised to avoid caffeine and smoking for at least 30 min before measuring. Two measurements were taken, and the average was recorded23. Laboratory data included fasting plasma glucose, triglycerides, high-density lipoprotein cholesterol.
We defined metabolic syndrome according to NCEP ATP III (2005 Revision)24, requiring at least three of the following components: (1) abdominal obesity (waist ≥ 102 cm (≥ 40 inches) for men ≥ 88 cm (≥ 35 inches) for women), (2) triglycerides ≥ 150 mg/dL or pharmacologic treatment, (3) HDL cholesterol ≤ 40 mg/dL for men or 50 mg/dL for women or pharmacologic treatment, (4) systolic blood pressure ≥ 130 mmHg or diastolic blood pressure ≥ 85 mmHg or taking antihypertensive medication, and (5) fasting plasma glucose ≥ 100 mg/dL or pharmacologic treatment.
Statistical analysis
We performed statistical analyses using Stata, Version 17 (StataCorp. 2021, College Station, TX) and (2) SPSS, Version 27.0 (IBM Corp. 2020. Armonk, NY, USA). We calculated the frequency distribution of baseline characteristics, behavioral data and comorbidities to describe the study sample. Due to the nature of an observational study, the information on some variables was missing, including smoking status (5.0%), alcohol consumption (4.5%) and exercise (5.7%). However, the study consisted of a large sample size; therefore, the existing data would be included in the analysis. Categorical variables including sex, age groups, regions, health scheme, smoking status, history of alcohol consumption and exercise history were presented as percentages. We calculated the person-time of observation for each participant as the duration between the participant's baseline data and the year at which metabolic syndrome occurred or the end of the study (2021), whichever occurred first. We calculated incidence rates of metabolic syndrome with 95% confidence intervals (CI) per 100 person-years of observation, and used the log-rank test to compare the incidence rates of metabolic syndrome across characteristics. Cox proportional hazard regression analysis was used to investigate behavioral and demographic risk factors for metabolic syndrome and presented the magnitude of associations as unadjusted and adjusted hazard ratios (HR) with 95% confidence intervals. A two-sided p-value less than 0.05 was considered statistically significant. We also conducted a sensitivity analysis to assess the incidence rates of metabolic syndrome according to the International Diabetes Federation (IDF) 2005 and explored the association between incidence rates of metabolic syndrome and behavioral factors.
Ethics considerations
The study was reviewed and approved by the Institutional Review Board, RTA Medical Department, Bangkok, Thailand in compliance with international guidelines such as the Declaration of Helsinki, the Belmont Report, CIOMS Guidelines and the International Conference on Harmonization of Technical Requirements for Registration of Pharmaceuticals for Human Use—Good Clinical Practice (ICH—GCP) (Approval number S067h/64). Because we used secondary data, the Institutional Review Board, RTA Medical Department granted a waiver of informed consent.
Results
Baseline characteristics
Table 1 shows the baseline demographic, behavioral, and clinical characteristics of the 98,264 RTA personnel included in the study population. The majority (91.1%) were males. The average age of the study participants was 35.6 ± 11.0 years. Approximately one-third (32.1%) of participants were current smokers. The baseline prevalence of regular alcohol use was 7.4%, while the prevalence of irregular alcohol intake was 63.3%. Regular exercise at baseline was reported by 63.3% of study participants.
Incidence of metabolic syndrome among RTA personnel
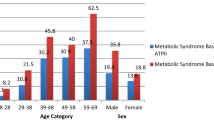
A total of 11,178 (11.4%) RTA personnel developed metabolic syndrome during the follow-up period, representing an incidence rate of 3.7 per 100 person-years (95% CI 3.7–3.8). The incidence rates among males and females were 3.8 per 100 person-years (95% CI 3.7–3.8) and 3.2 per 100 person-years (95% CI 2.9–3.4), respectively. Table 2 shows incidence of metabolic syndrome among RTA personnel by characterirtics.
Risk factors of metabolic syndrome among RTA personnel
Multivariable adjusted hazard ratios from the Cox model are shown in Table 3. After mutually adjusting for demographic, behavioral, and clinical characteristics, the incidence rate of metabolic syndrome was higher for males than females (adjusted HR 1.40; 95% CI 1.29–1.51), RTA personnel over age 35 years, those residing in Cental and Northeast, and those who reported current alcohol consumption. On the other hand, former smokers had a lower incidence rate of metabolic syndrome than lifelong non-smokers. Finally, those who reported regular exercise had a lower incidence rate of metabolic syndrome than sedentary participants (adjusted HR 0.79; 95% CI 0.74–0.86). There was a significant effect modification (p-value = 0.0001) between sex and age on the incidence rate of metabolic syndrome.
With study participants stratified by sex (Table 4), the higher age, the higher the incidence rate of metabolic syndrome in both sex. In males, the incidence rate of metabolic syndrome was significantly higher among those with a current alcohol intake, while there is no significant association in females. In addition, males who had a history of regular exercise had a lower incidence rate of metabolic syndrome than sedentary participants; however, it was not significantly different among females.
Table 5 shows factors associated with incidence rate of metabolic syndrome stratified by age. The incidence rate of metabolic syndrome was higher in males than females (adjusted HR 1.81, 1.66, and 1.18) among participants aged \(\le\) 35, 35–44, and ≥ 45 years, respectively. Regular alcohol intake was associated with the higher incidence rate of metabolic syndrome among RTA personnel aged 35–44 and ≥ 45 years but was not associated with the younger RTA personnel aged 35 years. Participants aged \(\le\) 35 and ≥ 45 years who reported regular exercise had a significantly lower incidence rate of metabolic syndrome than those with a sedentary lifestyle; however, there was no association among RTA aged 35–44 years.
Sensitivity analysis
In the sensitivity analysis, the incidence rate of metabolic syndrome, according to IDF 2005, was 4.3 per 100 person-years (95% CI 4.3–4.4). The incidence rates among males and females were 4.4 per 100 person-years (95% CI 4.3–4.4) and 4.1 per 100 person-years (95% CI 3.9–4.3) (supplement Table 1). We found a consistent association between demographic and behavioral factors. RTA personnel over 35 years had a higher incidence of metabolic syndrome than younger participants with a dose–response relationship. Regarding behavioral factors, the RTA personnel who reported regular exercise had a lower incidence rate of metabolic syndrome than sedentary participants. In contrast, alcohol intake was not associated with the incidence rate of metabolic syndrome according to IDF 2005 (supplement Table 2).
Discussion
To our knowledge, the present study is the largest recent epidemiologic study of the incidence of metabolic syndrome among RTA personnel in Thailand. These data provide essential evidence on behavioral risk factors for metabolic syndrome in this population.
Several studies have reported data on the prevalence of metabolic syndrome in diverse populations in different regions of the world. However, the results of these studies are difficult to compare because of variation in the criteria used to define metabolic syndrome, with some relying on the NCEP ATP III, the IDF, and the World Health Organization (WHO)2 criteria.
We found that the overall incidence rate of metabolic syndrome among RTA personnel was 3.7 per 100 person-years. This rate is relatively high compared with results from a nine-year follow-up study in a rural community in Thailand, reporting an incidence rate of metabolic syndrome of 3.5 per 100 person-years25. Furthermore, this incidence rate was higher than that reported in a Japanese7 and a Taiwanese8 cohort indicating a rate of 2.1 and 2.3 per 100 person-years, repectively. This finding may be explained by the institutional structures of the military that convey detrimental behaviors and weaken personnel’s efforts to embrace healthier habits14; for example, in the present study a higher prevalence was found of regular alcohol consumption compared with the study participants in a Japanese7 and a Taiwanese8 cohort.
When considering the association between sex and the incidence of metabolic syndrome, our finding of a higher rate among males than females was consistent with results from a Japanese cohort7, but other studies, including a cohort of US military personnel26, and cohorts from Korea27 and Thailand25 have reported higher rates among females than males. Beliefs about masculinity embedded in the culture of military personnel may establish the behavioral patterns of men in styles affecting their health awareness28. Compared with females, on average, males have higher risk for cardiovascular diseases29, 30 and premature death31. The majority of the RTA population is male; therefore, targeting behaviors, that may mitigate the disparity in metabolic syndrome incidence between males and females has the potential to lower the incidence of sequelae of metabolic syndrome, including NCDs and ASCVD in particular.
In the present study, we intended to determine associations between the incidence rate of metabolic syndrome and behavioral factors at baseline. According to NCEP ATP III (2005), metabolic syndrome consisted of laboratory testing, history of pharmacologic treatment and anthropometric measurements. The RTA personnel, after participating in the annual physical health examination, received their health examination results and may have been advised by healthcare workers to modify their lifestyle. If the RTA personnel improved their health behaviors, it may have positively affected their laboratory testing results and anthropometric measurements during the follow-up period. Therefore, the incidence rates of metabolic syndrome in this population may have been underestimated.
We found that participants older than 35 years exhibited a higher incidence of metabolic syndrome than younger participants with a dose–response relationship in both sexes. Although our findings were consistent with a report from Japan reporting a higher incidence of metabolic syndrome among older than younger adults, a few studies did not find an increase of metabolic syndrome with increasing age25, 27, suggesting that the association with age was not inevitable. Several mechanisms can explain a higher incidence of metabolic syndrome with increasing age. For example, central adiposity, accumulation of fatty infiltration of the liver and sedentary lifestyle have a higher prevalence among older than younger individuals32. Furthermore, an age-related increase in oxidative stress plays a significant role in contributing vascular alterations by triggering the biochemical processes concomitant with metabolic syndrome33,34,35. When stratified by sex, the incidence rate of metabolic syndrome among participants aged ≥ 45 years was higher than those aged < 35 years with adjusted HR 6.34 (95%CI 6.01 to 6.70) for males, and 9.59 (95% CI 7.55 to 12.19) for females. Alterations in circulating female sex hormone levels, such as lower estrogen levels, were associated with distinct changes in adipose distribution patterns, reverting to visceral accumulation and raising the likelihood of increases in WC19, 36. Therefore, our study suggested that preventing the development of metabolic syndrome with increasing age is an essential target for the primary prevention of NCDs and ASCVD later in life.
Excessive alcohol consumption is a known behavioral risk factor for the incidence of metabolic syndrome and NCDs37, 38. This study found that participants reporting alcohol consumption (regular or irregular) were more likely to develop metabolic syndrome than abstainers. A recent report from a contemporary Japanese cohort reported that compared with abstainers, consumption of more than 60 g of alcohol daily was associated with a higher rate of metabolic syndrome. In contrast, less than 20 g of alcohol daily was associated with a lower risk16. In 2019, The 6th National Health Examination Survey (NHES VI) in Thailand reported that the prevalence of alcohol consumption among Thai adults was 44.6, 59.0 and 31.0% in total, and among males and females, respectively16. Compared the NHES VI, our findings indicated that the prevalence of current alcohol consumption was higher among RTA personnel (70.7%) than that of the general Thai population16. Therefore, alcohol consumption may be an important contributor to the incidence of metabolic syndrome, especially among male RTA personnel. According to tradition among males RTA personnel, reduced harmful use of alcohol may constitute a priority39. Therefore, additional pattern and intensity of alcohol consumption should be assessed in the annual physical health examination section; then motivational interventions such as a Brief Negotiated Interview should be offered to support consumers to modify their risky behaviors40.
We found that compared with lifelong nonsmokers, former smokers had a lower incidence of metabolic syndrome. This finding may result from the lifestyle modification among former smokers who may have relatively higher health awareness than lifelong nonsmokers. For example, a related study in China reported that former smokers had significantly more knowledge of all health effects than those who had never smoked41. Stratified by age, younger RTA personnel, aged < 35 years, compared with lifelong non-smokers, current smokers exhibited a lower incidence of metabolic syndrome. This observation was likely due to the well-documented negative relationship between smoking and obesity42, 43 and the finding that current smokers have less subcutaneous and visceral adipose tissue than those who never have smoked38, 44. However, not all studies have found a negative association between smoking and metabolic syndrome. For example, a report from the CARDIA study in the US did not find a significant association between smoking and risk of metabolic syndrome (RR 1.16; 95% CI 0.96 to 1.40)45. Furthermore, a meta-analysis of prospective studies found an overall significantly positive association between active smoking and the incidence of metabolic syndrome46.
In terms of regular exercise, RTA personnel may have more physical activity than the general civilian population16. However, RTA personnel serve in various departments with different characteristics of duty, for instance, military training units, healthcare workers in military hospitals and office workers in headquarters. Hence, the level of physical activity varied across our study population. We found that regular exercise was associated with a lower incidence of metabolic syndrome among RTA personnel. Regular exercise was negatively associated with obesity, a component of metabolic syndrome47. Therefore, our study suggested that regular exercise should be encouraged to lower the incidence of metabolic syndrome and ultimately NCDs, including ASCVD48, 49. Because vigorous physical activity can rarely trigger acute cardiovascular events or heat-related illness50,51,52, physical activity and structured exercise should be performed based on relevant guidelines53.
This study encountered several limitations. First, the present study was conducted among RTA personnel comprising a greater proportion of male participants than that of the general population. However, the results reported the real-world situation in the RTA population.
Because this constituted a retrospective cohort study using previously collected data, some variables were collected very broadly. For example, we did not have detailed data on how many days per week participants consumed alcohol or the number of alcoholic beverages consumed daily. Likewise, we did not have details of the smoking history, such as the current number of cigarettes smoked daily or pack-years of past exposure. We also did not have detailed data on the frequency, type or intensity of physical activity. Because we used collected data, unmeasured confounders such as family history, socioeconomic status, total calorie intake and nutritional status were excluded in the analysis. Due to the nature of an observational study, the information on some variables was unobtainable, including smoking status (5.0%), alcohol consumption (4.5%) and exercise (5.7%). Nonetheless, the available data provided valuable evidence regarding the associations between these health behaviors and the incidence of metabolic syndrome. The information on laboratory testing and anthropometric measurements came from several RTA hospitals nationwide providing the measurements; however, the measurements were performed by trained technicians, and the standard and quality of services of all RTA hospitals were certified by Healthcare Accreditation Institute, Thailand. In the present study, we aimed to determine associations between the incidence rate of metabolic syndrome and behavioral factors at baseline. However, the RTA personnel received their health examination results and may have been advised by healthcare workers to modify their lifestyle; thus, their behaviors may be changed over time. Thus, time-varying covariates may be considered to approach in future research.
Our study also exhibited significant strengths; of the approximately 130,000 RTA personnel, 98,264 (75.6%) participants, without a diagnosis of metabolic syndrome at baseline, were enrolled in the present study, representing a large sample of RTA personnel. Thus, our findings provide valuable insights into the demographics and behavioral and clinical risk factors, for the incidence of metabolic syndrome in this population. These data may contribute to strategies for the primary prevention of NCDs, ASCVD and premature death in Thai populations.
Conclusion
Our data demonstrated that metabolic syndrome is a common health issue, especially among males and RTA personnel over age 35. Alcohol consumption and sedentary behavior appear to play an important role in the incidence of metabolic syndrome in this population and are potential targets for interventions to enhance primary prevention of the sequelae of metabolic syndrome, including NCDs and ASCVD.
Data availability
Data cannot be shared publicly because the data set contains identifying information; additionally, the data belongs to the Royal Thai Army Medical Department. Thus, ethics and confidentiality restrictions exist on the distribution of the data set. Data are available from the Royal Thai Army Medical Department, Bangkok, Thailand, for researchers who meet the criteria to access confidential data.
Abbreviations
- NCDs:
-
Noncommunicable diseases
- ASCVD:
-
Atherosclerotic cardiovascular diseases
- RTA:
-
The Royal Thai Army
- H.R.:
-
Hazard ratio
- CI:
-
Confidence interval
- SD:
-
Standard deviation
References
Eckel, R. H., Grundy, S. M. & Zimmet, P. Z. The metabolic syndrome. Lancet 365, 1415–1428 (2005).
Huang, P. L. A comprehensive definition for metabolic syndrome. Dis. Model. Mech. 2, 231–237 (2009).
Mottillo, S. et al. The metabolic syndrome and cardiovascular risk: A systematic review and meta-analysis. J. Am. Coll. Cardiol. 56, 1113–1132 (2010).
Ranasinghe, P., Mathangasinghe, Y., Jayawardena, R., Hills, A. P. & Misra, A. Prevalence and trends of metabolic syndrome among adults in the Asia-pacific region: A systematic review. BMC Public Health 17, 1–9 (2017).
Grundy, S. M. Metabolic syndrome pandemic. Arteriosclerosis Thrombosis Vasc. Biol. 28 Preprint at https://doi.org/10.1161/ATVBAHA.107.151092 (2008).
Pimenta, A. M. et al. Dietary indexes, food patterns and incidence of metabolic syndrome in a Mediterranean cohort: The SUN project. Clin. Nutr. 34, 508–514 (2015).
Haruyama, Y. et al. Incidence of metabolic syndrome in young japanese adults in a 6-year cohort study: The uguisudani preventive health large-scale cohort study (uphls). J. Epidemiol. 30, 219–226 (2020).
Shang, P. et al. Veganism does not reduce the risk of the metabolic syndrome in a taiwanese cohort. Asia Pacific J. Clin. Nutr. 20, 404–410 (2011).
Nillakupt, K. & Viravathana, N. A survey of metabolic syndrome and its components in Thai medical cadets. J. Med. Assoc. Thail. Chotmaihet Thangphaet 93, S179–S185 (2010).
Satirapoj, B., Supasyndh, O., Mayteedol, N., Chaiprasert, A. & Choovichian, P. Metabolic syndrome and its relation to chronic kidney disease in a Southeast Asian population. Southeast Asian J. Trop. Med. Public Health 42, 176 (2011).
Podang, J., Sritara, P. & Narksawat, K. Prevalence and factors associated with metabolic syndrome among a group of Thai working population: a cross sectional study. J Med Assoc Thai 96(Suppl. 5), 1–9 (2013).
Limpijankit, T. et al. Cardio-ankle vascular index as a predictor of major adverse cardiovascular events in metabolic syndrome patients. Clin. Cardiol. 44, 1628 (2021).
Lohsoonthorn, V., Dhanamun, B. & Williams, M. A. Prevalence of metabolic syndrome and its relationship to white blood cell count in a population of Thai men and women receiving routine health examinations. Am. J. Hypertens. 19, 339 (2006).
Courtenay, W. H. Constructions of masculinity and their influence on men’s well-being: A theory of gender and health. Soc. Sci. Med. 50, 1385–1401 (2000).
Sakboonyarat, B. et al. Rising trends in obesity prevalence among Royal Thai Army personnel from 2017 to 2021. Sci. Rep. 12, 7726 (2022).
Aekplakorn, W.. Thai National Health Examination VI (NHES VI). https://online.fliphtml5.com/bcbgj/znee/#p=187 (2019).
Rostami, H., Tavakoli, H. R., Rahimi, M. H. & Mohammadi, M. Metabolic syndrome prevalence among Armed Forces Personnel (Military Personnel and Police Officers): A systematic review and meta-analysis. Military Med. 184, e417 (2019).
The International Institute for St. The Military Balance 2021. The Military Balance 2021 (2021). https://doi.org/10.4324/9781003177777.
Faulkner, J. L. & Belin De Chantemèle, E. J. Sex hormones, aging and cardiometabolic syndrome. Biol. Sex Diff. 10 Preprint at https://doi.org/10.1186/s13293-019-0246-6 (2019).
Lee, Y. & An, J. Sex differences in risk factors for metabolic syndrome in the Korean population. Int. J. Environ. Res. Public Health 17, 9513 (2020).
Liu, P. L. et al. Association of age and sex with metabolic syndrome in taiwanese adults. Int. J. General Med. 14, 1403 (2021).
Alberti, G., Zimmet, P., Shaw, J. & Grundy, S. M. The IDF consensus worldwide definition of the metabolic syndrome. Brussels Int. Diabetes Federation 23, 469–480 (2006).
Thai Hypertension Society. 2019 Thai Guidelines on The Treatment of Hypertension. (2019).
Grundy, S. M. et al. Diagnosis and management of the metabolic syndrome: An American Heart Association/National Heart, Lung, and Blood Institute scientific statement. Circulation 112 Preprint at https://doi.org/10.1161/CIRCULATIONAHA.105.169404 (2005).
Sangbangmud, J. et al. Incidence and risk factors of metabolic syndrome and 9-year follow-up in Na Yao Community, Sanam Chai Khet District, Chacheongsao, Thailand. J. Southeast Asian Med. Res. 2, 7–15 (2018).
Williams, V. F., Oh, G.-T. & Stahlman, S. Incidence and prevalence of the metabolic syndrome using ICD-9 and ICD-10 diagnostic codes, active component, U.S. Armed Forces, 2002–2017. MSMR 25, (2018).
Pietroiusti, A. et al. Incidence of metabolic syndrome among night-shift healthcare workers. Occup. Environ. Med. 67, 54 (2010).
Williams, D. R. The health of men: Structured inequalities and opportunities. Am. J. Public Health 93 Preprint at https://doi.org/10.2105/AJPH.93.5.724 (2003).
Sakboonyarat, B. & Rangsin, R. Prevalence and associated factors of ischemic heart disease (IHD) among patients with diabetes mellitus: A nation-wide, cross-sectional survey. BMC Cardiovasc. Disord. 18, 151 (2018).
Chantkran, W., Chaisakul, J., Rangsin, R., Mungthin, M. & Sakboonyarat, B. Prevalence of and factors associated with stroke in hypertensive patients in Thailand from 2014 to 2018: A nationwide cross-sectional study. Sci. Rep. 11, 17614 (2021).
Organization, W. H. Noncommunicable diseases country profiles 2018. (2018).
Dominguez, L. J. & Barbagallo, M. The biology of the metabolic syndrome and aging. Curr. Opin. Clin. Nutr. Metabolic Care. 19 Preprint at https://doi.org/10.1097/MCO.0000000000000243 (2016).
Beckman, K. B. & Ames, B. N. The free radical theory of aging matures. Physiol. Rev. 78 Preprint at https://doi.org/10.1152/physrev.1998.78.2.547 (1998).
Cortopassi, G. A., Shibata, D., Soong, N. W. & Arnheim, N. A pattern of accumulation of a somatic deletion of mitochondrial DNA in aging human tissues. Proc. Natl. Acad. Sci. U S A 89, 7370 (1992).
Bonomini, F., Rodella, L. F. & Rezzani, R. Metabolic syndrome, aging and involvement of oxidative stress. Aging Dis. 6 Preprint at https://doi.org/10.14336/AD.2014.0305 (2015).
Janssen, I., Powell, L. H., Jasielec, M. S. & Kazlauskaite, R. Covariation of change in bioavailable testosterone and adiposity in midlife women. Obesity 23, 488–494 (2015).
World Health Organization. Noncommunicable diseases. https://www.who.int/news-room/fact-sheets/detail/noncommunicable-diseases (2021).
Vieira, B. A. et al. Timing and type of alcohol consumption and the metabolic syndrome - ELSA-Brasil. PLoS One 11, e0163044 (2016).
Organization, W. H. Regional technical consultation with member states on the working document for development of the action plan (2022–2030) to effectively implement the global strategy to reduce the harmful use of alcohol as a public health priority: virtual meeting, 25–26 March 2021: meeting report. (2021).
Fernandez, W. G., Hartman, R. & Olshaker, J. Brief interventions to reduce harmful alcohol use among military personnel: Lessons learned from the civilian experience. Military Med. 171, 538–543 (2006).
Yang, J., Hammond, D., Driezen, P., Fong, G. T. & Jiang, Y. Health knowledge and perception of risks among Chinese smokers and non-smokers: Findings from the Wave 1 ITC China Survey. Tob Control 19(Suppl 2), i18 (2010).
Clair, C. et al. Dose-dependent positive association between cigarette smoking, abdominal obesity and body fat: Cross-sectional data from a population-based survey. BMC Public Health 11, 1–10 (2011).
Matsushita, Y. et al. Associations of smoking cessation with visceral fat area and prevalence of metabolic syndrome in men: The Hitachi health study. Obesity 19, 647–651 (2011).
Terry, J. G. et al. Association of smoking with abdominal adipose deposition and muscle composition in coronary artery risk development in young adults (CARDIA) participants at mid-life: A population-based cohort study. PLoS Med. 17, 1003223 (2020).
Carnethon, M. R. et al. Risk factors for metabolic syndrome: The coronary artery risk development in young adults (CARDIA) study, 1985–2001. Diabetes Care 27, 2707–2715 (2004).
Sun, K., Liu, J. & Ning, G. Active smoking and risk of metabolic syndrome: A meta-analysis of prospective studies. PLoS ONE 7, e47791 (2012).
Hatthachote, P., Rangsin, R., Mungthin, M. & Sakboonyarat, B. Trends in the prevalence of obesity among young Thai men and associated factors: From 2009 to 2016. Mil. Med. Res. 6, 13 (2019).
Ko, I.-G. & Choi, P.-B. Regular exercise modulates obesity factors and body composition in sturdy men. J. Exerc. Rehabilitation 9, 256 (2013).
Tanasescu, M. et al. Exercise type and intensity in relation to coronary heart disease in men. J. Am. Med. Assoc. 288, 1994 (2002).
Nutong, R. et al. Personal risk factors associated with heat-related illness among new conscripts undergoing basic training in Thailand. PLoS ONE 13, e0203428–e0203428 (2018).
World Health Organization, t. Global recommendations on physical activity for health. (World Health Organization, 2010).
Mittleman, M. A. et al. Triggering of acute myocardial infarction by heavy physical exertion – protection against triggering by regular exertion. New England J. Med. 329, 1677 (1993).
Piercy, K. L. et al. The physical activity guidelines for Americans. JAMA J. Am. Med. Assoc. 320, 2020 (2018).
Acknowledgements
The authors are exceedingly grateful to all those involved for successfully conducting the study, including staff of the Professional Division, and the Health Promotion and Preventive Medicine Division, the Royal Thai Army Medical Department. The authors thank all staff members of the Army Institute of Pathology, Armed Forces Research Institute of Medical Sciences and 37 RTA hospitals nationwide for their support in completing this study. This study was supported by the Research Unit for Military Medicine, Phramongkutklao College of Medicine in Bangkok, Thailand.
Funding
This research was supported by (1) the FETP-NCD, Division of Epidemiology, Department of Disease Control, Ministry of Public Health, Thailand, and (2) the Reserch Unit for Military Medicine, Phramongkutklao College of Medicine, Bangkok, Thailand.
Author information
Authors and Affiliations
Contributions
The concept for the study was developed by B.S., R.R., and M.M. B.S., and R.R. collected the data. B.S. and MM analyzed the data. B.S. wrote the first draft. All authors contributed and approved the final version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sakboonyarat, B., Rangsin, R. & Mittleman, M.A. Incidence and risk factors of metabolic syndrome among Royal Thai Army personnel. Sci Rep 12, 15692 (2022). https://doi.org/10.1038/s41598-022-19024-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-19024-8
This article is cited by
-
Associations of triglyceride-glucose index with hyperuricemia among Royal Thai Army personnel
BMC Endocrine Disorders (2024)
-
Association between raised blood pressure and elevated serum liver enzymes among active-duty Royal Thai Army personnel in Thailand
BMC Cardiovascular Disorders (2023)
-
Relationship between triglyceride-glucose index and aminotransferase among Royal Thai Army personnel 2017–2021: a serial cross-sectional study
Lipids in Health and Disease (2023)
-
Rising trends in the predicted 10-year risk of cardiovascular diseases among Royal Thai Army personnel from 2017 to 2021
Scientific Reports (2023)
-
Trends in Serum Total Cholesterol and High Total Cholesterol Prevalence Among Royal Thai Army Personnel in Thailand, 2017–2022
High Blood Pressure & Cardiovascular Prevention (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.