Abstract
Nailfold capillaroscopy (NFC) is a safe and non-invasive imaging tool for evaluating microvascular abnormalities. This retrospective cross-sectional study aimed to analyze the NFC outcomes and clinical characteristics in patients and an asymptomatic carrier with transthyretin (TTR) gene mutation. The participants consist of eight patients with genetically and clinically confirmed hereditary amyloidogenic transthyretin (ATTRv) amyloidosis and one asymptomatic carrier. The TTR gene mutant forms of six male and three female participants from six families were Asp38Ala (five patients), Lys35Asn (three patients), and Ala36Pro (one patient). All participants showed decreased capillary density, dilatated capillaries, and destructed architecture in NFC. Early progression identification of a carrier to patients with symptoms is a major concern from a therapeutic viewpoint in ATTRv amyloidosis. Therefore, further studies with a larger number of subjects will be needed to determine the use of NFC as an early detection tool.
Similar content being viewed by others
Introduction
Nailfold capillaroscopy (NFC) is a safe and non-invasive imaging tool for evaluating the microvascular abnormalities and capillary structure in the nailbed area. So far, NFC application mainly included rheumatic disease assessment and their clinical presentation, such as systemic sclerosis and Raynaud’s phenomenon1. Recently, NFC has been attempted on various diseases that are expected to have microcirculation pathology beyond rheumatic diseases. The most representative example related to these new diagnostic attempts is a diabetic complication, wherein patients with diabetes show reduced capillary length, irregular distribution, abnormal morphology, and reduced density compared with normal subjects in NFC2,3. Particularly, a positive correlation was found in the NFC score in diabetic retinopathy and polyneuropathy3,4.
Hereditary amyloidogenic transthyretin (ATTRv) amyloidosis is a rare, autosomal dominant, adult-onset systemic disease caused by a point mutation in the gene encodes transthyretin (TTR). TTR is a 55-kDa homotetramer protein that is largely synthesized by the liver and accounts for approximately 90% of TTR production. Additionally, TTR is also synthesized by retinal pigment epithelium cells and choroid plexuses of the brain5. Systemic and localized amyloid accumulation is related to the disease. Among them, the ocular involvement after liver transplant is observed although plasma TTR cannot cross the blood-retina barrier. Thus, ocular manifestations are largely due to the local TTR production6. Until 1990, ATTRv amyloidosis was considered an endemic rare disease that was observed in Portugal, Sweden, and Japan7,8,9. Since then, several endemic areas were additionally reported in a recent study that estimated the cumulative number of people with ATTRv amyloidosis in 10,186 persons, with a range of 5526–38,46810. The epidemiologic study of endemic areas and the incidence is likely to be updated with increasing disease awareness among clinicians and wider use of the genetic test.
One of the suggested pathomechanisms is that axon loss occurs due to microangiopathy as ATTRv progresses although the pathophysiology of neuropathy in ATTRv is unclear5,11. This argument is based on the pathological finding that endoneurial blood vessels are invaded and destroyed by amyloid deposition12. We evaluated whether NFC can support this view as an easy and non-invasive way to observe microcirculation abnormalities in patients with ATTRv. Additionally, this study aimed to identify the disease onset in carriers promptly through NFC and determine whether NFC can reflect disease progression in patients with ATTRv amyloidosis. This study reports the NFC outcomes in eight patients with ATTRv amyloidosis and one asymptomatic carrier. The NFC patterns of the participants were investigated, which reported an association between these patterns and the clinical features of ATTRv amyloidosis.
Methods
Participants and ethical statements
This retrospective cross-sectional study included eight patients who were genetically and clinically confirmed with ATTRv amyloidosis and one asymptomatic carrier. The medical records of participants at a single tertiary medical center from April 2017 to December 2020 were investigated. Exclusion criteria were the presence of preexisting diseases, such as systemic connective tissue diseases or diabetes that affect the vascular architecture, or a history of trauma to the periungual fold of the fingers. This study was approved by the Institutional Review Board of Konkuk University Medical Center (KUMC-IRB), Seoul, Korea (No. 2021-04-046), and informed consent was exempted under the supervision of KUMC-IRB in consideration of the retrospective characteristics of this study, which is limited to medical record reviews that do not contain personal identification information. This study conformed to the ethical norms and standards of the Declaration of Helsinki.
Investigation of clinical manifestations
Clinical data were collected from available medical records, consisting of age at onset, medical history, family history, the genotype of TTR mutation, clinical manifestations, and adjusted treatment. The main clinical presentation of each patient with ATTRv amyloidosis was classified as neuropathy, cardiomyopathy, or mixed types, and all participants were subjected to a series of related tests during initial diagnosis13. They underwent echocardiography and Holter monitoring to confirm amyloidogenic cardiomyopathy. Additionally, a nerve conduction study and autonomic nervous function screening test (heart rate variability, Valsalva maneuver, and tilt-table test) were performed to evaluate amyloidogenic peripheral neuropathy. Carpal tunnel syndrome (CTS), among neuropathy, was determined by clinical criteria and supplementary diagnostic methods14. Clinical criteria consist of compatible neuropathic pain, Tinel’s sign, or Phalen’s maneuver before the occurrence of lower extremity abnormalities. Disproportional solitary damage of the median nerve in the supplementary methods was confirmed by nerve conduction study or ultrasonography15,16. Neuropathy severity of patients with ATTRv amyloidosis was classified by Polyneuropathy disability (PND) scoring system17. This system includes the following five stages: stage 0, no nerve impairment and visible symptoms; stage 1, a sensory disturbance in the lower extremities but the walking capacity of the patient is not affected; stage 2, the patient might have some difficulty in walking but usually does not require walking aids, such as sticks or crutches; stage 3a, the patient might require support from a single walking stick or crutch; stage 3b, the patient might require support from two sticks or crutches, and stage 4, the patient is wheelchair bounded or bedridden. All participants had valid medical records that confirm the retinal capillary and vitreous structure status to assess ocular symptoms of amyloidosis.
Nailfold capillaroscopy
The NFC test was performed by a skilled examiner who was blinded to the patients’ clinical amyloidosis status. Each patient was acclimatized for 20 min at a room temperature of 24 °C before the NFC examination. The examiner used a drop of immersion oil to increase the visibility of the microvessels in the nailfolds. The capillaries on the left and the right four fingers, except the thumb, were observed with a microscope (× 100 and × 400 magnifications; Olympus SZ-PT, Olympus, Tokyo, Japan), and photographs were taken of the last distal row of capillaries using a digital camera (Polaroid, Minnetonka, MN, USA). Four images were taken from eight fingers (2 of each finger at each magnification), and the evaluation was performed in a total of 32 fields.
The recently described semiquantitative rating scale (0: no changes; 1: < 33% of capillary alterations/reduction; 2: 33%–66% of capillary alterations/reduction; and 3: > 66% of capillary alterations/reduction) was adopted1,18. The following six capillaroscopic parameters were obtained: capillary density, dilated capillaries, giant capillaries, hemorrhage, ramification, and architectures. Normal nailfold capillaries consist of one loop with a diameter of < 20 μm. Meanwhile, ramified capillaries show patterns of bushes by additional several coils that originate from normal capillaries. The nailfold capillary architecture indicates the vessel arrangement consistency. The cut-off value for decreased nailfold capillary density was an average of nine uniform vascular loops per 1 mm in fingers. The alignment is lost, and the shape of the loops cannot be distinguished under pathological conditions. Each parameter was scored by a semiquantitative rating system and measured on each finger, and an average score of the eight fingers was recorded. The total score is calculated by adding up their averages. The lowest score means close to the normal condition, and a higher score indicates a distorted microvascular limb status.
Results
Study population and clinical characteristics
The study examined eight patients and one asymptomatic carrier in six families with pathogenic TTR gene mutation (Table 1). Of them, six were males and three were females with a mean age of 54 years (range 44–64 years). All participants were Koreans of East Asian descent. The mean disease duration of ATTRv amyloidosis was 7.5 years (range 5–11 years) upon NFC testing in 8 patients. Three mutation types of TTR gene were included, namely Asp38Ala (five patients), Lys35Asn (three patients), and Ala36Pro (one patient). Patient No. 3 had a neuropathy type amyloidosis, and the remaining seven patients were mixed type. Patients No. 1 and No. 6 showed vitreous opacity due to amyloid deposits without retinal vessel change. All eight patients had peripheral neuropathy, and five of them had additional neuropathology limited to the hand called CTS. Among the autonomic disorders, orthostatic hypotension occurred in all eight patients, and intractable diarrhea was repeated in six patients. Additionally, two patients and one asymptomatic carrier were smokers during the NFC testing. Except for the asymptomatic carrier, all eight enrolled patients were treated with transthyretin stabilizer (tafamidis) or small interfering RNAs (siRNAs) targeting the TTR gene (patisiran).
NFC measurement
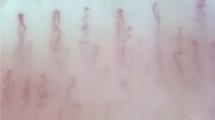
All eight patients and one asymptomatic carrier showed abnormalities in the components of capillary density, dilatated capillaries, and architecture (Table 1). The mean of the 9 participants’ total score was 3.403 (range 2.000–6.125). Patient No. 1 with the longest duration of illness and severe neuropathic symptoms (PND score of 3b) among all participants had the lowest, near-normal result in the total score (Fig. 1A). Two patients with ocular amyloid pathology due to localized TTR production (Patients No. 1 and 4) had relatively low scores among the subjects with total scores of 2.000 and 2.500. The highest score was 3.500 in the five patients with CTS. The asymptomatic carrier (Patient No. 6) showed the lowest capillary density or highest avascular areas in NFC and had the highest score of 6.125 (Fig. 1B). Two (Patients No. 6 and 8) of the three smokers had high scores (6.125 and 5.250, respectively), wherein one was an asymptomatic carrier and the other had a severe status with a PND score of 3a. The NFC result for each type of TTR gene mutation, excluding carriers, revealed two patients (Patient No. 1 and 4) with the longest disease duration (Lys35Asn and Asp38Ala, respectively) with the lowest scores (2.000 and 2.500, respectively) in each group.
Patient No. 1 showed slightly decreased density and dilatation (arrowhead) of capillaries (A), and Patient No. 6 had few capillaries because of extensive architecture damage, and hemorrhage (arrow) in the nailbed area (B). These were taken from the bilateral second and fourth fingers. (× 400 magnifications; Olympus SZ-PT, Olympus, Tokyo, Japan; R: right, L: left, 2: second finger, 4: fourth finger.
Discussion
Microangiopathy caused by blood-nerve-barrier disruption and extracellular amyloid is considered one of the major pathophysiologies in ATTRv amyloidosis5. Retinal and choroidal microvasculature destruction due to localized amyloid deposit has also been reported with ATTRv amyloidosis, which has been claimed as the main evidence that suggests clinical microangiopathy19. Another key feature of ATTRv is autonomic neuropathy, in which small fiber damage is prominent. Selective and preferential A-delta and C fiber damage before extensive systemic abnormalities appear responsible for these features20. In a previous case report, Raynaud's phenomenon is one of the autonomic dysfunctions of the microvasculature and has been reported as one of the major clinical phenotypes of ATTRv21. NFC results can be expected to be abnormal in patients with ATTRv because of the presence of microvascular pathology and autonomic nervous system dysfunction in ATTRv. However, the pattern or findings of NFC reflecting microvasculature and autonomic nervous system in patients with ATTRv amyloidosis has never been described yet.
All eight patients with ATTRv amyloidosis and one asymptomatic carrier showed decreased capillary density, dilatated capillaries, and destructed architecture although differences in severity were seen in this study. Nevertheless, interestingly, a participant with asymptomatic carrier status had the most severe form of impairment, and patients with a long disease duration showed relatively good NFC outcomes. Hence, it can be inferred that risk factors, other than TTR gene mutation, affect NFC results, and smoking is likely considered a risk factor in this study. Smoking is one of the main risk factors for endothelial damage, and asymptomatic smokers have been reported to be more commonly accompanied by NFC abnormalities than nonsmokers22. However, how smoking affects ATTRv amyloidosis or how smoking negatively affects NFC in the presence of comorbid underlying medical conditions has not been established. Therefore, additional studies comparing the NFC results by classifying carriers with TTR gene abnormalities as smokers and nonsmokers would be recommended. Another factor in this outcome may be the effect of adjusted treatment. Two patients with the longest disease duration also received siRNA treatment for the longest duration among patients (Table 1). The microvascular damage is uncertain to have a reversible course in this study, assuming that ATTRv and microangiopathy are related. Additionally, confirming the treatment effect on their good NFC test results is impossible. However, this suggests that subsequent studies should consider the treatment effects, as well as the association of ATTRv and NFC results.
This is the first report to apply NFC as an evaluation tool for ATTRv amyloidosis. NFC abnormalities may have been presented to accompany the group with TTR gene mutation, but significant limitations need to be considered. First, the number of evaluated subjects was very small because ATTRv amyloidosis is a very rare disease. A previous study that analyzed NFC results with systemic diseases other than rheumatic diseases demonstrated the association between the longer period and severity of diabetes mellitus status and more severe capillary damage measured by NFC23. Additionally, NFC abnormalities were confirmed to be more prominent in patients with glaucoma and essential hypertension than in the non-disease group23. Whereas, the disease duration, clinical severity, and NFC results did not show proportional results in this study, which is likely due to the small number of participants. Second, from a viewpoint related to the ATTRv research, surveying the relationship between genotype and phenotype in TTR gene mutation is important, but such an analysis was not possible in this study. Third, as a cross-sectional study, the time course of the disease and co-risk factors were not adequately adjusted. Particularly, the peripheral neuropathy status of the upper extremity of the participants would all vary depending on the disease duration, the patient’s underlying condition, and treatment experience. All patients’ severity of polyneuropathy and carpal tunnel syndrome was not measured in detail. A follow-up study is needed that considering the specific NFC results obtained in the group in which the disease does not start in the upper extremity even with polyneuropathy. Additionally, a separate evaluation is estimated to be necessary for patients with only CTS among patients with ATTRv. Fourth, the NFC assessment method should be applied differently depending on the disease although we applied the standard method in rheumatoid disease and diabetes. However, this limitation is expected to be improved as further studies are conducted.
From the therapeutic perspective of ATTRv amyloidosis, the current greatest interest is finding a suitable group for early treatment because early treatment is effective in preventing irreversible damage and poor quality of life5. However, early ATTRv diagnosis is difficult due to disease rarity. Additionally, the criteria for determining the onset and treatment initiation may be ambiguous even in a group that monitors the disease onset due to family history. Therefore, the authors wanted to investigate the effectiveness of NFC as an applicable and simple test method considering the pathophysiology of ATTRv. Hence, this pilot study suggests conducting additional larger and well-controlled studies to verify the possibility of NFC as an early diagnostic tool in asymptomatic carriers and patients with ATTRv amyloidosis.
References
Cutolo, M. et al. Nailfold videocapillaroscopic patterns and serum autoantibodies in systemic sclerosis. Rheumatology 43, 719–726 (2004).
Barchetta, I. et al. High prevalence of capillary abnormalities in patients with diabetes and association with retinopathy. Diabet. Med. 28, 1039–1044 (2011).
Hsu, P.-C. et al. Nailfold capillary abnormalities are associated with type 2 diabetes progression and correlated with peripheral neuropathy. Medicine 95, e5714 (2016).
Rajaei, A., Dehghan, P. & Farahani, Z. Nailfold capillaroscopy findings in diabetic patients (a pilot cross-sectional study). J. Pathol. 05, 65–72 (2015).
Adams, D., Koike, H., Slama, M. & Coelho, T. Hereditary transthyretin amyloidosis: A model of medical progress for a fatal disease. Nat. Rev. Neurol. 15, 387–404 (2019).
Minnella, A. M. et al. Ocular involvement in hereditary amyloidosis. Genes (Basel) 12, 955 (2021).
Sousa, A., Coelho, T., Barros, J. & Sequeiros, J. Genetic epidemiology of familial amyloidotic polyneuropathy (FAP)-type I in Póvoa do Varzim and Vila do Conde (north of Portugal). Am. J. Med. Genet. 60, 512–521 (1995).
Sousa, A., Anderson, R., Drugge, U., Holmgren, G. & Sandgren, O. Familial amyloidotic polyneuropathy in Sweden: Geographical distribution, age of onset, and prevalence. Hum. Hered. 43, 288–294 (1993).
Kato-Motozaki, Y. et al. Epidemiology of familial amyloid polyneuropathy in Japan: Identification of a novel endemic focus. J. Neurol. Sci. 270, 133–140 (2008).
Schmidt, H. H. et al. Estimating the global prevalence of transthyretin familial amyloid polyneuropathy. Muscle Nerve. 57, 829–837 (2018).
Tozza, S. et al. The neuropathy in hereditary transthyretin amyloidosis: A narrative review. J. Peripher. Nerv. Syst. 26, 155–159 (2021).
Planté-Bordeneuve, V. & Said, G. Familial amyloid polyneuropathy. Lancet Neurol. 10, 1086–1097 (2011).
Coelho, T., Maurer, M. S. & Suhr, O. B. THAOS–the transthyretin amyloidosis outcomes survey: Initial report on clinical manifestations in patients with hereditary and wild-type transthyretin amyloidosis. Curr. Med. Res. Opin. 29, 63–76 (2013).
Padua, L. et al. Carpal tunnel syndrome: Clinical features, diagnosis, and management. Lancet Neurol. 15, 1273–1284 (2016).
AAEM Quality Assurance Committee et al. Literature review of the usefulness of nerve conduction studies and electromyography for the evaluation of patients with carpal tunnel syndrome. Muscle Nerve 16, 1392–1414 (1993).
Aseem, F., Williams, J. W., Walker, F. O. & Cartwright, M. S. Neuromuscular ultrasound in patients with carpal tunnel syndrome and normal nerve conduction studies. Muscle Nerve 55, 913–915 (2017).
Adams, D. Recent advances in the treatment of familial amyloid polyneuropathy. Ther. Adv. Neurol. Diso. 6, 129–139 (2013).
Sulli, A., Secchi, M. E., Pizzorni, C. & Cutolo, M. Scoring the nailfold microvascular changes during the capillaroscopic analysis in systemic sclerosis patients. Ann. Rheum. Dis. 67, 885–887 (2008).
Rousseau, A. et al. Angiographic signatures of the predominant form of familial transthyretin amyloidosis (Val30Met Mutation). Am. J. Ophthalmol. 192, 169–177 (2018).
Conceição, I., Costa, J., Castro, J. & de Carvalho, M. Neurophysiological techniques to detect early small-fiber dysfunction in transthyretin amyloid polyneuropathy. Muscle Nerve 49, 181–186 (2014).
Magy, N. et al. Transthyretin mutation (TTRGly47Ala) associated with familial amyloid polyneuropathy in a French family. Amyloid 9, 272–275 (2002).
Yuksel, E. P., Yuksel, S., Soylu, K. & Aydin, F. Microvascular abnormalities in asymptomatic chronic smokers: A videocapillaroscopic study. Microvasc. Res. 124, 51–53 (2019).
Ciaffi, J. et al. Nailfold capillaroscopy in common non-rheumatic conditions: A systematic review and applications for clinical practice. Microvasc. Res. 131, 104036 (2020).
Author information
Authors and Affiliations
Contributions
Conceptualization and design: K.C., J.O.; Data acquisition and analysis: D.K., H.K.M., H.R.K.; Data interpretation: D.K., H.K.M., H.R.K.; Drafting of the manuscript: D.K.; Critical revisions to manuscript: K.C.; Overall supervision: K.C., J.O.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kim, D., Oh, J., Min, H.K. et al. A pilot study of nailfold capillaroscopy in hereditary transthyretin amyloidosis. Sci Rep 12, 11715 (2022). https://doi.org/10.1038/s41598-022-15779-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-15779-2
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.