Abstract
Parasites are widespread in nature. Nevertheless, they have only recently been incorporated into food web studies and community ecology. Earlier studies revealed the large effects of parasites on food web network structures, suggesting that parasites affect food web dynamics and their stability. However, our understanding of the role of parasites in food web dynamics is limited to a few theoretical studies, which only assume parasite-induced mortality or virulence as a typical characteristic of parasites, without any large difference in terms of predation effects. Here, I present a food web model with parasites in which parasites change the mortality and interaction strengths of hosts by affecting host activity. The infected food web shows that virulence and infection rate have virtually no effect on food web stability without any difference in interaction strengths between susceptible and infected individuals. However, if predation rates are weakened through a restriction of the activity of infected individuals, virulence and infection rate can greatly influence stability: diseases with lower virulence and higher transmission rate tend to increase stability. The stabilization is stronger in cascade than random food webs. The present results suggest that parasites can greatly influence food web stability if parasite-induced diseases prevent host foraging activity. Parasite-induced infectious disease, by weaking species interactions, may play a key role in maintaining food webs.
Similar content being viewed by others
Introduction
Parasites are widespread in ecosystems, and can account for a large part of the biomass on earth1,2. Nevertheless, parasites have only recently begun to be considered in community ecology3,4,5,6,7,8,9,10,11,12,13. Earlier studies showed that the effects of including parasites in food webs are so large that they modify network topology by increasing the connectance and food-chain length14,15. Because of the strong relationship between food web topology and population dynamics16,17, parasites may affect community stability14,15. On the other hand, as shown in classical epidemiological theories, parasites can regulate host populations18,19 and, therefore, can affect community and ecosystem dynamics8,20. In spite of the potential large effects of parasites on community dynamics, few theoretical studies have examined how parasites affect the stability of such complex communities, particularly with diverse species and their interactions21,22.
Most earlier efforts to understand the effects of parasites on community dynamics used simple systems with a few species, such as two host-one parasite models23,24. Hence, we know very little about the roles of parasites within large ecological communities with diverse species. Still, a pioneering work, which considered the dynamics of multiple hosts infected by a parasite, demonstrated a role of host diversity in the establishment, persistence, and outbreak of the parasite, but without considering any direct interaction between host species25. A few theoretical studies have introduced parasites into a food web with direct interactions between species21,22. Contrary to the intuitive view that parasites can contribute to community stability through host population regulation, parasites almost never affect food web stability21,22. Rather, parasites may even contribute to food web instability because parasite invasion can sometimes induce population cycles21. In earlier infected food web models, a key role of parasites was to regulate host population growth by increasing host mortality (virulence)21,22. However, this host regulation effect is a similar feature of predators in food webs; thus, the unique features of the parasite itself must be considered to deeply understand the roles of parasites in food web dynamics6,15.
Parasites can affect other important characteristics of hosts, such as species interactions and reproduction15,24. The most intuitive example is inactivity due to an infectious disease, through which infected individuals become less able to capture and/or eat prey resources, and breed and/or lose reproductive ability24. In addition, ailing individuals are expected to be more easily captured by predators24. Thus, parasites, through the infectious disease itself, can change the network structure of food webs in terms of affecting interaction strengths. If so, the parasite-induced infectious disease could largely affect the food web dynamics, because interaction strength plays a key role in food web stability26,27.
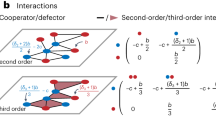
In this paper, using a food web model with a disease, I show that disease-induced changes in the strengths of species interactions play a key role in stabilizing complex communities. I consider a cascade food web model28 comprising N species, any pair of which is connected with a probability of C (connectance). For each pair of species, i, j = 1,…, N with i < j, species i never consumes species j, whereas species j may consume species i. The infectious disease is caused by a specialist parasite to each host species (coinfection and cross-species infection are not considered). Once a disease spreads, each population has susceptible or uninfected (S) and infected (I) individuals. The infection occurs through contact with infected individuals, where the infection rate of each species is notated by βi, and infected individuals recover at a constant rate γi into susceptible individuals. The parasites do not increase through vertical transmission. The infected individuals have a higher death rate than susceptible individuals; the additional death rate is defined as virulence, notated by vi. Population dynamics are driven by interspecific predator–prey interactions and host-parasite interactions via the infectious disease (see Methods). The interaction strength is a key factor affecting the food web dynamics. Here, I consider that the interaction strengths are changed by the disease; to do this, I control the relative interaction strength to a basal predator–prey interaction in a food web model without diseases. Each interaction strength is normalized by the interaction strength among S (predator) vs. S (prey). The relative interaction strength among S (predator) vs. I (prey) is defined as A1, and that of I is defined as A2 (Fig. 1a). A typical expectation of the changes in interaction strength due to diseases would be weak interactions of infected individuals and/or strong interactions of susceptible individuals to infected individuals (Fig. 1b). In addition to an increase in mortality due to a disease, reproduction rate can be decreased. The reduction of the reproduction is controlled by a parameter, R, which is a constant that controls the degree of growth rate reduction from the susceptible to infected individuals. In this study, I mainly control the relative interaction strengths (A1 and A2) to examine how diseases affect the stability of food webs, as evaluated via community persistence, with the probability that all species persist for a given time (see Methods for details).
Schematic diagram of the simplest infected food web. (a) Model structure. Si and Ii represent a susceptible and infected individual, respectively (i = 1; predator, i = 2; prey). A0 is the normalized interaction strength among susceptible predators and prey. A1 and A2 are the relative strength of interaction among predators and prey, indicated by each arrow to A0. Each species has a disease with transmission rate βi, virulence vi and recovery rate γi. (b) Effects of the disease to interaction strengths between predator and prey. The strength of interactions are represented by the arrow thickness in (a). (c) Effects of disease to birth and death rates. In the main text, birth rate is same among susceptible and infected individuals, although the effect of birth reduction due to disease is shown in Supplemental Information.
Results
Consider a healthy food web without diseases (βi = 0). In such a situation, it is difficult for complex communities with diverse species to persist, as well known from previous food web models29,30. This suggests that a healthy world is unstable. However, in nature, there are no healthy ecosystems without parasite-induced infectious diseases.
Here, consider the introduction of parasites or pathogens into the food web (βi > 0), which induces infectious diseases in each species. This infection can increase host mortality (virulence effect). When the infectious disease only has the virulence effect, the virulence level has not a significant effect to stability (Fig. 2a), although higher virulence can slightly increase stability when infection rate is high (Fig. S1); this suggests that virulence due to an infectious disease does not play a significant role in food web stability.
Effects of virulence and infection rate on community stability with varying interaction strengths. Each figure (a–h) has different interaction strength values, as indicated in the upper side of each panel. Color represents the mean virulence values. N = 30 and C = 0.3. Parameters are R = 1, \(\overline{r}\) = 1, \(\overline{d}\) = 0.005, \(\overline{a}\) = 0.05, and \(\overline{\gamma }\) = 0.005.
However, this is not true if predator–prey interactions are altered by infection; this can change predation rates. First, when the predation rates of infected individuals are weaker than those of susceptible individuals (A1 > A2), infection rate can largely affect community stability, with stability increasing as infection rates increase (Fig. 2b). In addition, as the predation rates of infected individuals become weaker (A1 > > A2), virulence beings to affect stability, wherein a higher virulence tends to decrease stability (Fig. 2c,d). These patterns are qualitatively held, even when the predation rates of susceptible individuals to infected individuals are stronger than those of susceptible individuals (A1 > 1; Fig. 2e–h). On the other hand, the effects of the predation rates of susceptible individuals to infected individuals (A1) on stability depend on the infection rate. When infection rates are low, higher values of A1 tend to decrease stability (Fig. 3a,b); however, when infection rates are high, the values of A1 almost never affect stability (Fig. 3c). These results are not affected by the reproduction rates of infected individuals. Infertility due to infection does not change the results, although infertility can have a somewhat stabilizing effect when the infection rate is high and the disease has a lower effect on interaction strengths (Fig. S2). The recovery rate tends to decrease stability (Fig. S3), suggesting that the continuous emergence of infected individuals with a loss of appetite plays a key role in food web stability. The stability of infected food webs is so strong that it is almost never reduced by an increase in food web complexity, species richness, and connectance, contrary to a food web without infection (Fig. S4).
Effects of interaction strengths on community stability with varying infection rates. (a) \(\overline{\beta }\) = 0.25. (b) \(\overline{\beta }\) = 0.5. (c) \(\overline{\beta }\) = 2.5. Color represents values of A1. Other information is identical to that in Fig. 2.
I also tested other food web models: an interspecific infection model, a random network model, a free-living parasite model and vertical transmission model (Supplemental Online text). Interspecific infection causes stability in two ways, depending on the intraspecific infection rate. The stability tends to increase with an increase in interspecific infection rate when intraspecific infection rate is low, whereas the tendency is reversed when intraspecific infection rate is high (Fig. S5). Contrary to the cascade model, random food webs show lower stability (Fig. S6). In addition, with a large A1, an increase in infection rate can decrease stability, particularly when A2 or virulence is large. This instability is so large that high values of A1 decrease stability even when infection rates are high (Fig. S6). On the other hand, a difference in the assumption on an infection process does not change the qualitative results. I consider a model in which infection occurs only through contact with a free-living parasite (Supplemental Online text); even in this case, the main results are qualitatively held (Fig. S7). Further, vertical transmission can contribute to stability, although the contribution is not significant (Fig. S8).
Discussion
The present study showed that a parasite-induced infectious disease can play a key role in stabilizing food web dynamics via a change in interaction strengths caused by the disease. If parasites do not cause any difference in traits, other than mortality to the infected host individuals, parasites almost do not affect food web stability. However, once the parasite-induced infectious disease can influence the interaction strengths, the difference in interaction strengths among susceptible and infected individuals allow the key characteristics of infectious diseases (infection rate and virulence) to have an effect on food web stability, wherein a higher infection rate and lower virulence can contribute to food web stability. The stabilizing effect of an infectious disease becomes large when infection causes a loss of appetite or decreases predation rates. This stabilizing effect does not depend on whether parasites cause infertility nor have a free-living period, but it does depend on the food web network structure. Infectious disease is likely to have a more stabilizing role in a cascade food web than in a random food web. These results suggest that the suppression of activity caused by infectious diseases can play a key role in stabilizing natural food webs.
Why can infected food webs be stable? Infectious diseases inherently have two stabilizing effects. First, less active, infected predators can prevent a large reduction in the number of prey. If infection rates are high, abundant predators can be suppressed by the infectious disease due to disease-induced mortality and appetite loss24,31. The predator suppression due to disease can prevent a large reduction in the number of prey. In addition, predation of infected prey can also suppress the infectious disease in the prey, resulting in the prevention of a reduction of the prey. Because the number of prey is not reduced as normal, infected prey can also act as an alternative resource for susceptible predators32, preventing a large reduction in the number of predators. However, susceptible predator is not likely to be more abundant as explained above, the predation effect is maintained at a low level (because the consumption rare itself does not change but predator abundance is low). As a consequence, both predator and prey are unlikely to be less and more abundant, resulting in a stabilization of population dynamics. This suggests a disease induced self-maintenance mechanism in food webs. However, the mechanism is not likely to stabilize when the food web structure is not a cascade (i.e., random). In a cascade network, top predators and basal prey resources always exist. Since a top predator is never eaten by another species, an abundant predator is likely to be regulated by the disease. On the other hand, in a random network, top predators and/or basal prey species may not exist; if so, susceptible predators become less abundant because of predation from other species. Altogether, this results in the prevention of predator regulation and leads to destabilization of the food web.
Parasites or infectious disease-induced interaction change are critical in leading a stabilizing role in food web dynamics; this has been supported by a simple predator–prey system with a parasite31,33, but it has never been demonstrated in more complex systems with diverse species. The present study suggests that revealing a network structure of infected food webs with respect to interaction strengths is important for understanding the role of infectious diseases in ecological communities. Conversely, from a community stability standpoint, we need to focus on the parasites that alter interaction structures, among all diverse parasites. On the other hand, the present model is not applicable to all types of parasites; however, it is at least applicable to a disease occurring through intra- and interspecific infection or contact with a free-living parasite. Interspecific infection can promote the stabilization of an infectious disease when intraspecific infection is less frequent since it can merely contribute to increasing the number of infected individuals. However, when both intra- and interspecific infection rates are high, the extinction of less abundant susceptible individuals would prevent a high stabilization effect. Nevertheless, examining the types of parasites that largely affect ecological community stability will be an important future research area.
May mathematically demonstrated that complex communities comprised of diverse species with many interaction links are inherently unstable29,30. As this prediction was in contrast to persisting, real ecological communities, ecologists have searched for the key unknown factors of natural ecosystems, and May’s model has not worked. Weak interactions have been suggested to be a major stabilizing factor26,27. Real communities appear to be characterized by many weak interactions34,35,36. I propose that infected food webs may be one of mechanisms that cause weak interactions, which can play a key role in maintaining complex communities. A healthy world may be unstable, because in nature, there are no healthy ecosystems without parasite-induced infectious diseases.
Methods
Consider a cascade food web28 in which pairs of species i and j (i, j = 1,…, N) are connected by a trophic interaction with a probability of C, which is defined as the proportion of realized interaction links L in the possible maximum interaction links Lmax of a given network model (L = CLmax). For each pair of species, i, j = 1,…, N with i < j, species i never consumes species j, whereas species j may consume species i. To examine the generalization of the main result, random food webs37 was also be tested (Fig. S6). The maximum link number Lmax is calculated from N(N − 1)/2 in both the cascade and random models.
Consider that disease transmission occurs only within each population. Then, each species can have susceptible and infected individuals, denoted by S and I, respectively. Infection occurs horizontally through contact with infected individuals within the same species; the vertical transmission of parasites does not occur. Infected individuals have a higher mortality rate than susceptible individuals. Infected individuals have the same species interaction links with susceptible individuals, whereas interaction strengths and/or reproduction rate will be different from those of susceptible individuals. The food web model with diseases is defined by an ordinary differential equation:
where Xki is the abundance of species i and Xi = XSi + XIi. The subscript k represents the states of individuals, where S and I represent a susceptible and infected host, respectively. rki is the intrinsic rate of change in a species i, di is the basal death rate of species i, vi is the additional death rate due to disease (virulence), βi is the transmission rate of disease, and γi is the recovery rate of disease. M is the terms on predator–prey interactions. MS = ΣjMSSijXSj + ΣjMSIijXIj represents the population change rate through species interactions of susceptible hosts in species i, MI+ = ΣjMISijXSj + ΣjMIIijXIj (> 0) represents the population increase rate through prey consumption by infected host species i, and MI− = ΣjMSIjiXSj + ΣjMIIjiXIj (< 0) represents the population decrease rate through consumption of infected host prey by predators. Mlij is the interaction coefficient between species i and j. The subscript l represents a particular interaction structure. “SS” equates to predator–prey interactions among susceptible host species i and j, “SI” to predator–prey interactions between susceptible host species i and infected host species j, “IS” to predator–prey interactions between infected host species i and susceptible host species j, and “II” to predator–prey interactions among infected host species i and j. Interaction coefficients are defined as Mlij = elijalij and Mlji = − alij, where alij is the consumption rate and elij (< 1) is the conversion efficiency.
Generally, disease can decrease the activity of a host, such that the consumption rate and/or reproduction rate of infected individuals will decrease. On the other hand, infected individuals can be easily consumed by non-infected predators. To reflect this, for simplicity, I assume that (1) eIij = Reij (R ≤ 1), where eSSij = eSIij = eij and eISij = eIIij = eIij; (2) rIi = Rri (R ≤ 1), where rSi = ri; and (3) aSIij = A1aij (A1 ≥ 1) and aIij = A2aij (A2 ≤ 1), where aSSij = aij and aISij = aIIij = aIij. R, A1, and A2 are constant, which control the relative magnitude of each parameter of infected individuals to that of susceptible individuals.
Parameters in each simulation are randomly determined from a uniform distribution with a lower limit of zero. The default mean values of each parameter in the main text were: \(\overline{r}\) = 1, \(\overline{d}\) = 0.005, \(\overline{a}\) = 0.05 and \(\overline{\gamma }\) = 0.005 (hereafter, the bar represents the mean parameter values of all species). For simplicity, I assume eij = 0.2. I systematically changed the values \(\overline{\beta }\), \(\overline{v}\), R, A1, and A2 to examine the effects of disease on food web dynamics stability. When \(\overline{\beta }\) = 0, the system approximated the original food web model without disease. Once \(\overline{\beta }\) > 0, the food web was infected by disease. Then, if R = A1 = A2 = 1, we can see the effects of virulence on food web dynamics. If R = 1, but Ai is varied, we can see the effects of disease-induced interaction changes on food web dynamics. If R < 1, we can see the effects of infertility due to disease on food web dynamics.
In each iterated simulation, the initial species abundance was randomly selected from a uniform distribution (XSi = 0–1.0, XIi = 0–0.01). Community persistence38, used as a representative of stability, was then calculated by measuring the fraction of simulation runs in which the entire community persisted (ΣkXki > 10−13 for all i) after a sufficiently long time (t = 2 × 103, which corresponded to the time taken for community persistence to reach an asymptote) in 500 runs. In this model, local stability analysis does not capture the model behavior, because even if the coexistence equilibrium (all variables have positive equilibrium) is locally stable, some species can lose the infected population depending on the initial conditions.
Data availability
All data generated and analyzed during this study are included in this published article.
References
Dobson, A., Lafferty, K. D., Kuris, A. M., Hechinger, R. F. & Jetz, W. Homage to Linnaeus: How many parasites? How many hosts?. Proc. Natl. Acad. Sci. 105, 11482–11489 (2008).
Kuris, A. M. et al. Ecosystem energetic implications of parasite and free-living biomass in three estuaries. Nature 454, 515–518 (2008).
Seabloom, E. W. et al. The community ecology of pathogens: Coinfection, coexistence and community composition. Ecol. Lett. 18, 401–415 (2015).
French, R. K. & Holmes, E. C. An ecosystems perspective on virus evolution and emergence. Trends Microbiol. 28, 165–175 (2020).
Hudson, P. J., Dobson, A. P. & Lafferty, K. D. Is a healthy ecosystem one that is rich in parasites?. Trends Ecol. Evol. 21, 381–385 (2006).
Raffel, T. R., Martin, L. B. & Rohr, J. R. Parasites as predators: Unifying natural enemy ecology. Trends Ecol. Evol. 23, 610–618 (2008).
Johnson, P. T. J. et al. When parasites become prey: Ecological and epidemiological significance of eating parasites. Trends Ecol. Evol. 25, 362–371 (2010).
Frainer, A., McKie, B. G., Amundsen, P. A., Knudsen, R. & Lafferty, K. D. parasitism and the biodiversity-functioning relationship. Trends Ecol. Evol. 33, 260–268 (2018).
Jephcott, T. G., Sime-Ngando, T., Gleason, F. H. & Macarthur, D. J. Host-parasite interactions in food webs: Diversity, stability, and coevolution. Food Webs 6, 1–8 (2016).
Rohr, J. R. et al. Towards common ground in the biodiversity–disease debate. Nat. Ecol. Evol. 4, 24–33 (2020).
Johnson, P. T. J., De Roode, J. C. & Fenton, A. Why infectious disease research needs community ecology. Science 349, 1259504 (2015).
Marcogliese, D. J. & Cone, D. K. Food webs: A plea for parasites. Trends Ecol. Evol. 12, 320–325 (1997).
Chen, H.-W. et al. Network position of hosts in food webs and their parasite diversity. Oikos 117, 1847–1855 (2008).
Lafferty, K. D., Dobson, A. P. & Kuris, A. M. Parasites dominate food web links. Proc. Natl. Acad. Sci. USA 103, 11211–11216 (2006).
Lafferty, K. D. et al. Parasites in food webs: The ultimate missing links. Ecol. Lett. 11, 533–546 (2008).
Dunne, J. A. The network structure of food webs. In Ecological Networks: Linking Structure to Dynamics (eds Pascual, M. & Dunne, J. A.) 27–28 (Oxford University Press, 2005).
Dunne, J. A., Williams, R. J. & Martinez, N. D. Network structure and biodiversity loss in food webs: Robustness increases with connectance. Ecol. Lett. 5, 558–567 (2002).
Hudson, P. J., Rizzoli, A., Grenfell, B. T., Heesterbeek, H. & Dobson, A. P. The Ecology of Wildlife Diseases. (Oxford University Press, Oxford, 2002).
Anderson, R. M. & May, R. M. Infectious Diseases of Humans: Dynamics and Control (Oxford University Press, 1992).
McCallum, H. & Dobson, A. Detecting disease and parasite threats to endangered species and ecosystems. Trends Ecol. Evol. 10, 190–194 (1995).
De Castro, F. & Bolker, B. M. Parasite establishment and host extinction in model communities. Oikos 111, 501–513 (2005).
McQuaid, C. F. & Britton, N. F. Parasite species richness and its effect on persistence in food webs. J. Theor. Biol. 364, 377–382 (2015).
Holt, R. D., Dobson, A. P., Begon, M., Bowers, R. G. & Schauber, E. M. Parasite establishment in host communities. Ecol. Lett. 6, 837–842 (2003).
Hatcher, M. J. & Dunn, A. M. Parasites in Ecological Communities: From Interactions to Ecosystems (Cambridge University Press, 2011).
Dobson, A. Population dynamics of pathogens with multiple host species. Am. Nat. 164, S64–S78 (2004).
McCann, K., Hastings, A. & Huxel, G. R. Weak trophic interactions and the balance of nature. Nature 395, 794–798 (1998).
Neutel, A. M., Heesterbeek, J. A. P. & de Ruiter, P. C. Stability in real food webs: Weak links in long loops. Science 296, 1120–1123 (2002).
Chen, X. & Cohen, J. E. Transient dynamics and food–web complexity in the Lotka–Volterra cascade model. Proc. R. Soc. Lond. Ser. B Biol. Sci. 268, 869–877 (2001).
May, R. M. Stability in multispecies community models. Math. Biosci. 12, 59–79 (1971).
May, R. M. Will a large complex system be stable?. Nature 238, 413–414 (1972).
Hilker, F. M. & Schmitz, K. Disease-induced stabilization of predator-prey oscillations. J. Theor. Biol. 255, 299–306 (2008).
Hethcote, H. W., Wang, W., Han, L. & Ma, Z. A predator–prey model with infected prey. Theor. Popul. Biol. 66, 259–268 (2004).
Kooi, B. W., van Voorn, G. A. K. & Das, K. P. Stabilization and complex dynamics in a predator-prey model with predator suffering from an infectious disease. Ecol. Complex. 8, 113–122 (2011).
Winemiller, K. O. Spatial and temporal variation in tropical fish trophic networks. Ecol. Monogr. 60, 331–367 (1990).
Paine, R. T. Food-web analysis through field measurement of per capita interaction strength. Nature 355, 73–75 (1992).
Wootton, J. T. Estimates and tests of per capita interaction strength: Diet, abundance, and impact of intertidally foraging birds. Ecol. Monogr. 67, 45–64 (1997).
Cohen, J. E., Briand, F. & Newman, C. M. Community Food Webs: Data and Theory (Springer, 1990).
Mougi, A. Diversity of biological rhythm and food web stability. Biol. Lett. 17, 20200673 (2021).
Acknowledgements
This study was supported by a Grant-in-Aid for Scientific Research (C) (#20K06826) from the Japan Society for the Promotion of Science. The author appreciates the support from the Faculty of Life and Environmental Sciences at Shimane University for providing financial aid to publish this article.
Funding
This work was supported by a Grant-in-Aid for Scientific Research (C) (Grant Number: #20K06826) from the Japan Society for the Promotion of Science.
Author information
Authors and Affiliations
Contributions
A.M. performed all research and wrote the entire manuscript.
Corresponding author
Ethics declarations
Competing interests
The author declares no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mougi, A. Infected food web and ecological stability. Sci Rep 12, 8139 (2022). https://doi.org/10.1038/s41598-022-11968-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-11968-1
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.