Abstract
Animal studies demonstrated that paternal alcohol exposure before conception increases the risk of adverse neurodevelopment in offspring, but limited evidence is known in humans. Based on Shanghai-Minhang Birth Cohort Study, we aimed to examine associations between preconceptional paternal alcohol consumption and child behavioral problems. Paternal alcohol consumption during the last 3 months before conception was obtained by maternal report. Children born to fathers who drank alcohol at least once a week were classified as exposed. Child behavioral problems were assessed using the Child Behavior Checklist (CBCL) at age of 2, 4, and 6. Negative binomial regression was used to estimate the rate ratio (RR) of CBCL raw scores in 796 offspring. The risks of rating scores on anxious/depressed were increased by 33% (RR 1.33, 95% CI 1.09, 1.61) and 37% (RR 1.37, 95% CI 1.02, 1.84) among boys in the exposed group at age of 4 and 6, respectively. We also found that risks of somatic complaints were increased by 18% (RR 1.18, 95%CI 1.00, 1.40) and 65% (RR 1.65, 95%CI 1.14, 2.38) among girls in the exposed group at age of 4 and 6. The increased risks of sleep problems (RR 1.25, 95% CI 1.00, 1.55) in girls at age 4, thought problems (RR 1.32, 95% CI 1.01, 1.73) in girls at age 6, rule-breaking behaviors (RR 1.35, 95% CI 1.09, 1.67) in boys at age 6 were also found. The risks of CBCL scores on anxious/depressed and sleep problems in girls at age 4, as well as the risks of somatic complaints and rule-breaking behaviors in boys at age 6 increased with the level of exposure to paternal alcohol consumption. Our findings provided preliminary evidence that preconceptional paternal alcohol consumption may increase risks of child behavioral problems.
Similar content being viewed by others
Introduction
It has been established that heavy maternal alcohol consumption during pregnancy can cause Fetal Alcohol Syndrome Disorders1, a syndrome characterized by physiological and neuropsychological deficits. In particular, emerging research has demonstrated that preconceptional maternal alcohol consumption was associated with lower mean full-scale IQ, overall attention, and sustained attention2, which indicated the possible adverse effects of earlier ethanol exposure on the gamete. As maternal alcohol exposure before conception plays an important role in child development, it is biologically plausible that preconceptional paternal alcohol consumption could also have effects on child neurodevelopment through its impacts on sperm3.
Spermatogenesis is estimated to take approximately 74 days, which is considered to be a process susceptible to exogenous disturbance4,5. The preconceptional period is thus increasingly recognized as a highly sensitive window6. Evidence from animal studies has also shown that preconceptional paternal alcohol consumption can induce genetic and epigenetic alterations in sperm that may increase the risks of adverse neurodevelopment in offspring, including attention deficit hyperactivity disorder-like behaviors, anxiety and depression-like behaviors, and cognitive impairment7,8,9,10,11. In humans, previous studies about the effects of paternal alcohol consumption on adverse neurodevelopment of the offspring were mainly focused on fathers with alcohol dependence, which indicated the crucial role of genetic transmission in their associations12,13. However, we know far less about the effects of alcohol consumption before conception for general fathers, whose alcohol consumption is much lower than those fathers with alcohol dependence.
The present study thus aimed to examine the associations between paternal alcohol consumption during the last 3 months before conception and the risk of child behavioral problems at 2, 4, and 6 years of age.
Methods
Study design and participants
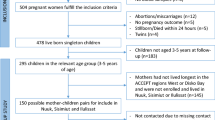
The present study was based on the Shanghai-Minhang Birth Cohort Study (S-MBCS), which was designed to determine the effects of early life environmental exposures on the health of offspring14,15. From April to December 2012, pregnant women who visited Minhang Maternal and Child Health Hospital for their first prenatal care at 12–16 weeks of gestation were consecutively recruited into this prospective cohort study if they: (1) were native Chinese and residents of Shanghai; (2) had no history of major chronic diseases diagnosed by a physician; (3) intended to deliver their babies in the study hospital; and (4) were willing to take part in scheduled interviews during pregnancy and after delivery. A total of 1,225 singleton mother-infant pairs were recruited in the cohort, among whom 1219 women reported their spouses’ alcohol consumption during the last 3 months before conception at recruitment. The closest caregivers (more than 85% were mothers) completed the inventory of Child Behavior Checklist (CBCL/1.5–5) when their children were 2 and 4 years of age and completed the inventory of CBCL/6–18 when their children were 6 years of age. We restricted the study to 796 children who had information of preconceptional paternal alcohol consumption, and who had at least one CBCL assessment at 2, 4, and 6 years of age, as shown in Fig. 1.
Ethics
This study involved human participants, and all procedures were performed in accordance with the Declaration of Helsinki. The study protocol was approved by the research ethics committees of the Shanghai Institute of Planned Parenthood Research. Both the pregnant women and their husbands gave informed consent before enrollment in the study and at postnatal follow-up visits for their children’s participation.
Paternal alcohol consumption before conception
Considering susceptible period of spermatogenesis4,5, we defined the time window of exposure as the last 3 months before conception. Paternal alcohol consumption was assessed through questionnaires completed by mothers at 12–16 gestational weeks. Children born to fathers who drank alcohol at least once a week during the last 3 months before conception were classified as exposed, and the rest as unexposed16,17. Fathers who drank alcohol were also asked about the type (e.g., beer, liquor, wine, or rice wine) and the average amount of alcohol beverages consumed per week. Alcoholic beverage consumption was recorded in grams as well as in terms of glasses or bottles16,18. In line with our previous study, the average amount of alcohol consumption per week from each type of beverage was calculated according to a specific formula: alcohol dose (g/wk) = beverage volume per week (ml) × alcoholic percentage of the beverage (%) × 0.8 g/ml (density of alcohol)16. The cumulative exposure of alcohol consumption per week was then calculated as the sum of the average amount of alcohol consumption for each type of beverage.
Child behavioral problems assessment
Child behavioral problems were assessed using the CBCL when children were 2, 4, and 6 years of age. The CBCL is a psychometric instrument applied widely in clinical and research settings to assess child behavioral problems. The Chinese version of the CBCL/1.5–5 and CBCL/6–18 have been validated for use among preschool (1.5–5 years) and school-aged (6–18 years) children with high reliability and stability19,20. The instrument instructs the mother to rate her child's behavior using a 3-category scale ranging from not true to often true, with 0 representing not true, 1 representing somewhat or sometimes true, and 2 representing very true or often true. Higher scores indicating greater behavioral problems. The CBCL/1.5–5, consisting of 99 items, provides measurements on 7 subscales obtained by summing the responses for each item within the subscale, including emotionally reactive, anxious/depressed, somatic complaints, withdrawn, sleep problems, attention problems, and aggressive behavior. Two aggregated scales measuring internalizing and externalizing behavioral problems are generated on the basis of the 7 subscales: internalizing behavior scores are derived from the sum of the emotionally reactive, anxious/depressed, somatic complaints, and withdrawn; externalizing behavior scores are derived from the sum of attention problems and aggressive behavior. In the CBCL 6–18, the 113-item scale is also subdivided into several subscales, namely anxious/depressed, withdrawn/depressed, somatic complaints, social problems, thought problems, attention problems, rule-breaking behavior, and aggressive behavior21. Scores on internalizing and externalizing behavior problems of CBCL 6–18, can also be obtained. The Internalizing domain here subsumes three syndrome scales: anxious/depressed, withdrawn/depressed, and somatic complaints. The externalizing domain includes the rule-breaking behavior and aggressive behavior syndrome scales. In the present study, mother–child pairs were followed up by home visits when children were 4, and 6 years of age. At ages 2, the information of CBCL was administered over the mail, as no in- person visits were conducted.
The instrument also provides T scores, which are standardized to a normative population, however, because T scores are truncated, these scores do not allow researchers to differentiate among children with low scores, which are indicative of fewer problems22. Thus, we included the CBCL raw scores in the final analyses.
Other variables
Information was obtained on maternal and paternal demographic characteristics, health condition, medical and reproductive history, and lifestyle factors, such as smoking and drinking habits, at recruitment by a structured questionnaire. Paternal smoking was defined as smoking at least 1 cigarette per week. Passive smoking was defined as indoor inhalation for at least 15 min a day from other smokers16. The depressive symptoms of pregnant women were also assessed at recruitment using the Centre for Epidemiological Studies Depression Scale. Information on children’s birth date, gender, and gestational age at birth was extracted from medical records.
Statistical analyses
Main analyses on the associations between preconceptional paternal alcohol consumption and CBCL raw scores
The baseline characteristics of children in exposed and unexposed groups were tabulated using counts and percentages for categorical variables and means (standard deviation, SD) for continuous variables. We also described the distributions of the demographic characteristics of the included and excluded children. We considered the CBCL-scores as count data according to its distribution (Supplementary Table S1). To account for greater variability (variance larger than mean) in the data and the better relative fit than would be expected based on a Poisson model (Supplementary Table S1), negative binomial regression was used to estimate the unadjusted and adjusted rate ratios (risks) (RR) and 95% confidence interval (CI) for the association between paternal alcohol consumption and risk of CBCL raw scores in offspring. In preliminary analyses, we included a multiplicative interaction term (exposure × sex) in statistical models. Potential sex-specific effects seemed to be suggested, thus, we also presented the results of stratified analysis by sex of children.
We also explored the dose–response relationship between preconceptional paternal alcohol consumption and CBCL raw scores by sex of children. Fathers who drank alcohol were categorized by tertiles (lowest: 0–30.72 g/week; middle: 30.72–68.48 g/week; and highest tertiles: > 68.48 g/week) according to the amount of cumulative consumption per week, which lead to 4 categories of exposure: non-drinkers (unexposed group), low (0–30.72 g/week), moderate (30.72–68.48 g/week), and highest (> 68.48 g/week) drinking. Trends tests were assessed by entering the 4 categories of the amount of paternal alcohol consumption as ordinal integer values (i.e., 1–4).
Covariates were examined as potential confounders on the basis of prior knowledge on alcohol-mental health relationship23. They were also retained if they caused a ≥ 10% change in the effect estimates for the exposure–outcome associations. Paternal age (years), paternal body-mass index (BMI) (< 18.5, 18.5–24 or > 24.0, kg/m2), paternal education (high school or below, college or university, postgraduate or above), paternal smoking before conception (yes or no), gestational age (weeks), sex of offspring, maternal age at conception (years), maternal parity (primiparous, multiparous), maternal depressive symptoms (yes or no, with the cut-off point at 16)24, maternal folic acid intake before conception (yes or no), and maternal multivitamin intake during pregnancy (yes or no) were included in the negative binomial regression models.
Sensitivity analyses
To further rule out the potential confounding effects of paternal smoking, we restricted the analyses to children whose fathers had never smoked before conception. We also re-ran the analysis among children whose mothers were not exposed to passive smoking during pregnancy to examine the robustness of the results. We did not include maternal smoking and maternal drinking in our analyses because few women were active smokers (2.0%) in Shanghai25, and only nine women (1.4%) reported alcohol consumption during pregnancy (Supplementary Table S2). Finally, we repeated the analyses in parents who received college or university education, or had normal pre-pregnancy BMI (18.5 < BMI < 24 kg/m2) to reduce any concern about the effects of these variables on the results.
All analyses were conducted with SAS 9.4 (SAS Institute Inc, Cary, NC, USA). Two-sided P-values of < 0.05 were regarded as statistically significant.
Results
Participant characteristics
The demographic characteristics of participants are shown in Table 1. In the present study, a total of 253 (31.8%) children were born to fathers who had alcohol consumption during the last 3 months before conception. Compared with the unexposed group, fathers in the exposed group were more likely to receive lower levels of education, and to smoke before conception. Mothers in the exposed group were more likely to report depressive symptoms during pregnancy or to be exposed to passive smoking, and less likely to take folic acid supplements before conception. Compared to the excluded children of the S-MBCS cohort, mothers of included children were older, better educated, and less likely to be depressed during pregnancy. Parental education of included children was a little higher than that of those excluded. No significant differences in other background characteristics were found (Supplementary Table S2).
Associations between paternal alcohol consumption and CBCL raw scores among children at age 2, 4, and 6 years
We found positive patterns (worse neurodevelopment) of paternal alcohol consumption with anxious/depressed, aggressive behavior, and internalizing problem scores in children at 2 years of age, but the associations became attenuated after adjusting for potential confounders.
At age of 4, we found elevated risks of rating scores on emotion reactive, anxious/depressed, sleep problems, and internalizing problems in children born to fathers with alcohol consumption before conception (RR range 1.16–1.23). When we stratified the analyses by sex, the risk of rating scores on anxious/depressed was increased by 33% (RR 1.33, 95% CI 1.09, 1.61) and the risk of rating scores on sleep problems was increased by 25% (RR 1.25, 95% CI 1.00, 1.55) among girls in the exposed group. Boys in the exposed group tend to have higher rating scores on somatic complaints, sleep problems, and internalizing problems. At age 6, paternal alcohol consumption was associated with increased risks of anxious/depressed scores in girls (RR 1.37, 95% CI 1.02, 1.84), and somatic complaints scores in boys (RR 1.65, 95% CI 1.14, 2.38), which was consistent with the findings at age 4. We also found that the risks of thought problem scores were increased by 32% (RR 1.32, 95% CI 1.01, 1.73) among girls in exposed group at age 6. The risks of rule-breaking behaviors and externalizing behaviors scores were increased by 35% (RR 1.35, 95% CI 1.09, 1.67) and 24% (RR 1.24, 95% CI 1.03,1.51), respectively, among boys in the exposed group at age 6, as shown in Table 2.
Table 3 presents the dose–response relationship between preconceptional paternal alcohol consumption and the CBCL raw scores in the children. The risks of CBCL scores on anxious/depressed and sleep problems in girls at age 4, as well as the risks of somatic complaints and rule-breaking behaviors in boys at age 6 increased with the level of exposure to paternal alcohol consumption (Supplementary Table S3): compared with girls in unexposed group, the risks of anxious/depressed and sleep problems were increased by 47% (RR 1.47, 95% CI 1.07, 2.02) and 46% (RR 1.46, 95% CI 1.02, 2.08), respectively, for girls in the highest drinking group; we also found elevated risks of somatic complaints in boys born to fathers with low drinking (RR 1.85, 95% CI 1.07, 3.19) and highest drinking (RR 2.18, 95% CI 1.31, 3.63), and elevated risks of rule-breaking behaviors (RR 1.42, 95% CI 1.04, 1.92) in boys born to fathers with moderate drinking. Some low-dose effects of paternal alcohol consumption on CBCL raw scores on CBCL scores were also indicated, like the increased risk of somatic complaints, sleep problems in boys at 4 years of age and anxious/depressed and thought problems in girls at 6 years of age. We did not observe patterns of dose–response relationship between preconceptional paternal alcohol consumption and the CBCL raw scores in the children at 2 years of age (Supplementary Table S4).
Sensitivity analyses
When we repeated the analyses in children without paternal smoking, nearly all the effect sizes were more pronounced, especially for sleep problems of boys and anxious/depressed of girls at 2 years of age (Table 4). Similar patterns were also observed in the children whose mothers were not exposed to passive smoking during pregnancy and received college or university education, whose fathers had normal BMI and received college or university education, although some associations became non-significant mostly due to the reduced sample size (Supplementary Table S5-S8). We also found that the associations of paternal alcohol consumption with anxious/depressed, sleep problems, and thought problems in girls whose mothers had normal pre-pregnancy BMI were attenuated (Supplementary Table S9).
Discussion
This prospective cohort study provided the first epidemiological evidence implying that children born to fathers who drank alcohol before conception may have higher risks of experiencing behavioral problems. Paternal alcohol consumption before conception was associated with higher rating scores for anxious/depressed in girls and somatic complaints in boys at 4 and 6 years of age. A dose–response relationship was observed on anxious/depressed and sleep problems among girls at 4 years of age, and on somatic complaints and rule-breaking behaviors among boys at 6 years of age.
Animal studies have provided clear evidence that offspring sired by ethanol-exposed males in the absence of maternal ethanol exposure showed more adverse behavioral development11, including increased risk of anxiety- and depression-like behaviors7,10, increased in activity and sensorimotor integration deficits, as well as decreased balance, coordination, and short-term motor learning26. Previous human studies mostly focused on the effects of paternal alcoholism on psychiatric disorders, including increased risk of externalizing symptoms and attention-deficit hyperactivity disorder12,13,27,28. However, none of these studies collected information on paternal alcohol consumption before conception. Although epidemiological studies on the potential effects of paternal preconceptional alcohol consumption on neurodevelopment are still limited, the hypothesis of increased risks of development problems of the offspring associated with paternal preconceptional alcohol consumption is not novel. Several human studies have shown that preconceptional paternal alcohol consumption was associated with lower birthweight29, microcephaly30, fetal birth defects31, increased risk of acute lymphoblastic leukemia32, and poor reproductive development16. Combined with our findings in the present study, these results suggested that preconception period may be a critical window for offspring development.
Several mechanisms could account for the effects of preconceptional paternal alcohol exposure on neurodevelopment in offspring. First, preconceptional alcohol exposure could increase the risk of child behavior problems via epigenetic modifications of sperm. Environmental conditions during the preconception period have been demonstrated to shape sperm epigenetics that can be heritable and influence offspring phenotypes33,34. The association between paternal alcohol exposure and behavioral alterations in offspring may be explained by altered genomic imprinting mediated by changes in DNA methylation of specific genes10. Additionally, small noncoding RNAs have also been confirmed to play a key role in paternal alcohol consumption34,35. Second, another potential mechanism underlying the adverse effects of paternal alcohol consumption on child behavioral problem might be genetic factors. Fathers with alcohol dependence may transmit genetic vulnerability to offspring not only for alcohol exposure but also for behavioral problems13,27. Genetic transmission was also supported by many family, twin and adoption studies, which found that strong genetic components are in the link between parental drinking and behavioral control in their offspring12,13,27. Additionally, that alcohol might produce alterations in the germ cell line which lead to chromosomal abnormalities or gene mutations in sperm and then transferred to offspring was also suggested36.
Studies that have examined the associations between alcohol consumption and adverse health outcomes in adults point towards different dose–response curves: a U-shape association37, a J-shape association38, inverted-U shape39 between consumption patterns and health outcomes were all reported. Although we tried to demonstrate the dose–response relationship in our present study, our ability is limited due to the relatively small sample size in each dose category and lack of subjects with paternal heavy alcohol consumption before conception (1.6%). Nevertheless, statistically significant dose–response relationship of some domains (e.g., anxious/depressed) was found in total population at different ages, although they did not reach significance after sex-stratification largely due to limited statistical power. Future studies with larger sample size are warranted to confirm the dose–response relationship.
The present study has several strengths. One major strength of the present study was the prospective design, which could provide potential causality between preconceptional paternal alcohol consumption and the risk of child behavioral problem and reduce recall bias. Additionally, we collected detailed information on covariates, which ensured careful consideration on a number of variables related to paternal alcohol consumption or outcome of interest in the analyses.
Our findings should also be considered in the context of some limitations. First, it has been reported that hyperactivity and diminished cognitive abilities in children are related to biological fathers with alcohol dependence rather than to adoptive fathers36,40,41, which indicates that the observed associations are less likely to be explained by social and child-raising factors. However, it cannot be ruled out that social and child-raising factors specific to fathers who drink alcohol may contribute to the observed associations. Second, information on preconceptional paternal alcohol consumption was reported by the children’s mothers during pregnancy. Although there was a very good inter-rater agreement between the parental reports of spouses’ drinking practices23, alcohol consumption by fathers was not evaluated by objective measurements of biomarkers such as gamma-glutamyl transferase and thus the misclassification of exposure may have been introduced. In addition, we only collected information on paternal alcohol consumption during the last 3 months before conception on the basis that spermatogenesis is a susceptible period. However, there may have been fathers who ceased drinking when they planned to conceive and thus whose children were classified as unexposed. Earlier paternal alcohol consumption can also affect the epigenetic milieu of sperm that could be passed on to offspring11. Such misclassification would lead to under-estimation of the true association. Third, quantitative measurement of exposure in this study relied on average alcohol consumption per week. Averaging alcohol exposure could obscure the fact that there were important subgroups of fathers who were binge drinkers or who drank more heavily in one period than in the rest42. The effects of the pattern of alcohol consumption may need to be evaluated in future studies. Fourth, family-based designs have shown that parental history of alcohol dependence/ abuse has adverse effects on neurodevelopment of the offspring12,13. If heavy alcohol use was defined as drinking at least 21 units (one unit corresponds to 8 g of alcohol) per week according to the UK alcohol guidelines23, only 13 (1.6%) were categorized as heavy drinking in the present study. Although we did not collect the information about the maternal preconceptional alcohol consumption, it was reported that only a few women drink (4.5%) in China25,43. Thus, the results were less likely be affected by the parental history of alcohol dependence/abuse in this study. Additionally, we lack information about paternal psychiatric disorders prior to conception, which has been reported to be associated with the offspring development44. Fifth, our study population may under-represent the sample of lower socio-economic status because more than 70% of parents have college degree or above, which may restrict the generalizability of our findings. Lastly, CBCL at ages of 2 years was assessed by mail in our study, and the samples were relatively small, which may contribute to the nonsignificant results in children at 2 years of age. Future studies with larger samples are needed to confirm our findings.
The present study added to the epidemiological evidence that children born to fathers with preconceptional alcohol consumption may have a higher risk of experiencing neurobehavioral problems. Life course epidemiology and Mendelian inheritance have long been grounded in assumptions on the causal inference of maternal exposures occurring during pregnancy and early life45. While maternal antenatal risk factors for adverse offspring development remain an important focus for intervention, findings of the present study combined with converging evidence highlight the effects of paternal exposure prior to conception on offspring development. Determining if the paternal preconceptional period represent a window of heightened vulnerability would improve our understanding of modifiable risk factors for children’s health and wellbeing33. Besides, our findings calls for greater attention to this issue given that exposure to paternal alcohol consumption is widespread (nearly one-third of fathers consumed alcohol before conception in China31, and many pregnancies are unplanned46. Importantly, there are no guidelines on safe drinking levels for men in couples trying for a pregnancy30. While the present study need to be confirmed by further studies, our findings highlight the risk of paternal alcohol consumption prior to conception as well as the necessity for future research in this field.
Conclusions
Our study provided very preliminary evidence that paternal alcohol consumption during the last 3 months before conception may increase the risk of child behavioral problems. Although consistent with converging evidence from animal models, replication in birth cohort studies with larger samples is needed.
Data availability
The datasets generated and/or analyzed during the current study are not publicly available due to subject confidentiality but are available from the corresponding author on reasonable request.
References
Jones, K. L., Smith, D. W., Ulleland, C. N. & Streissguth, P. Pattern of malformation in offspring of chronic alcoholic mothers. Lancet 1, 1267–1271. https://doi.org/10.1016/s0140-6736(73)91291-9 (1973).
Kesmodel, U. S. et al. The association of pre-pregnancy alcohol drinking with child neuropsychological functioning. BJOG 122, 1728–1738. https://doi.org/10.1111/1471-0528.13172 (2015).
Finegersh, A., Rompala, G. R., Martin, D. I. & Homanics, G. E. Drinking beyond a lifetime: New and emerging insights into paternal alcohol exposure on subsequent generations. Alcohol 49, 461–470. https://doi.org/10.1016/j.alcohol.2015.02.008 (2015).
Schmidt, C. W. Chips off the old block: How a father’s preconception exposures might affect the health of his children. Environ. Health Perspect. 126, 022001. https://doi.org/10.1289/ehp2348 (2018).
Cordier, S. Evidence for a role of paternal exposures in developmental toxicity. Basic Clin. Pharmacol. Toxicol. 102, 176–181. https://doi.org/10.1111/j.1742-7843.2007.00162.x (2008).
Toivonen, K. I., Oinonen, K. A. & Duchene, K. M. Preconception health behaviours: A scoping review. Prev. Med. 96, 1–15. https://doi.org/10.1016/j.ypmed.2016.11.022 (2017).
Kim, P. et al. Chronic exposure to ethanol of male mice before mating produces attention deficit hyperactivity disorder-like phenotype along with epigenetic dysregulation of dopamine transporter expression in mouse offspring. J. Neurosci. Res. 92, 658–670. https://doi.org/10.1002/jnr.23275 (2014).
Beeler, E., Nobile, Z. L. & Homanics, G. E. Paternal preconception every-other-day ethanol drinking alters behavior and ethanol consumption in offspring. Brain Sci. 9, 56. https://doi.org/10.3390/brainsci9030056 (2019).
Meek, L. R., Myren, K., Sturm, J. & Burau, D. Acute paternal alcohol use affects offspring development and adult behavior. Physiol. Behav. 91, 154–160. https://doi.org/10.1016/j.physbeh.2007.02.004 (2007).
Liang, F. et al. Paternal ethanol exposure and behavioral abnormities in offspring: Associated alterations in imprinted gene methylation. Neuropharmacology 81, 126–133. https://doi.org/10.1016/j.neuropharm.2014.01.025 (2014).
Rathod, R. S. et al. Effects of paternal preconception vapor alcohol exposure paradigms on behavioral responses in offspring. Brain Sci. 10, 658. https://doi.org/10.3390/brainsci10090658 (2020).
Knopik, V. S. et al. Contributions of parental alcoholism, prenatal substance exposure, and genetic transmission to child ADHD risk: A female twin study. Psychol. Med. 35, 625–635. https://doi.org/10.1017/s0033291704004155 (2005).
Knopik, V. S., Jacob, T., Haber, J. R., Swenson, L. P. & Howell, D. N. Paternal alcoholism and offspring ADHD problems: A children of twins design. Twin Res. Hum. Genet. 12, 53–62. https://doi.org/10.1375/twin.12.1.53 (2009).
Luan, M. et al. Prenatal polybrominated diphenyl ethers exposure and anogenital distance in boys from a Shanghai birth cohort. Int. J. Hyg. Environ. Health 222, 513–523. https://doi.org/10.1016/j.ijheh.2019.01.008 (2019).
Tian, Y. et al. Determinants of plasma concentrations of perfluoroalkyl and polyfluoroalkyl substances in pregnant women from a birth cohort in Shanghai, China. Environ. Int. 119, 165–173. https://doi.org/10.1016/j.envint.2018.06.015 (2018).
Xia, R. et al. Association Between Paternal Alcohol Consumption Before Conception and Anogenital Distance of Offspring. Alcohol Clin Exp Res 42, 735–742. https://doi.org/10.1111/acer.13595 (2018).
Kim, S. Y., Min, C., Oh, D. J. & Choi, H. G. Tobacco smoking and alcohol consumption are related to benign parotid tumor: A nested case-control study using a national health screening cohort. Clin. Exp. Otorhinolaryngol. 12, 412–419. https://doi.org/10.21053/ceo.2018.01774 (2019).
Klonoff-Cohen, H., Lam-Kruglick, P. & Gonzalez, C. Effects of maternal and paternal alcohol consumption on the success rates of in vitro fertilization and gamete intrafallopian transfer. Fertil. Steril. 79, 330–339. https://doi.org/10.1016/s0015-0282(02)04582-x (2003).
Liu, J., Cheng, H. & Leung, P. W. The application of the preschool Child Behavior Checklist and the caregiver-teacher report form to Mainland Chinese children: Syndrome structure, gender differences, country effects, and inter-informant agreement. J. Abnorm. Child Psychol. 39, 251–264. https://doi.org/10.1007/s10802-010-9452-8 (2011).
Achenbach, T. M., Dumenci, L. & Rescorla, L. A. DSM-oriented and empirically based approaches to constructing scales from the same item pools. J. Clin. Child Adolesc. Psychol. 32, 328–340. https://doi.org/10.1207/s15374424jccp3203_02 (2003).
Guerrera, S. et al. Assessment of psychopathological comorbidities in children and adolescents with autism spectrum disorder using the child behavior checklist. Front. Psychiatry 10, 535. https://doi.org/10.3389/fpsyt.2019.00535 (2019).
Cowell, W. J. et al. Prenatal exposure to polybrominated diphenyl ethers and child attention problems at 3–7 years. Neurotoxicol. Teratol. 52, 143–150. https://doi.org/10.1016/j.ntt.2015.08.009 (2015).
Mahedy, L. et al. Parental alcohol use and risk of behavioral and emotional problems in offspring. PLoS ONE 12, e0178862. https://doi.org/10.1371/journal.pone.0178862 (2017).
Weissman, M. M., Sholomskas, D., Pottenger, M., Prusoff, B. A. & Locke, B. Z. Assessing depressive symptoms in five psychiatric populations: A validation study. Am. J. Epidemiol. 106, 203–214. https://doi.org/10.1093/oxfordjournals.aje.a112455 (1977).
Chinese Association On Tobacco Control. http://www.catcprc.org.cn/index.aspx?menuid=4&type=articleinfo&lanmuid=8&infoid=8289&language=cn (2017).
Conner, K. E., Bottom, R. T. & Huffman, K. J. The impact of paternal alcohol consumption on offspring brain and behavioral development. Alcohol Clin. Exp. Res. 44, 125–140. https://doi.org/10.1111/acer.14245 (2020).
Haber, J. R., Jacob, T. & Heath, A. C. Paternal alcoholism and offspring conduct disorder: Evidence for the “common genes” hypothesis. Twin Res. Hum. Genet. 8, 120–131. https://doi.org/10.1375/1832427053738782 (2005).
Furtado, E. F., Laucht, M. & Schmidt, M. H. Gender-related pathways for behavior problems in the offspring of alcoholic fathers. Braz. J. Med. Biol. Res. 39, 659–669. https://doi.org/10.1590/s0100-879x2006000500013 (2006).
Little, R. E. & Sing, C. F. Father’s drinking and infant birth weight: Report of an association. Teratology 36, 59–65. https://doi.org/10.1002/tera.1420360109 (1987).
Zuccolo, L. et al. Pre-conception and prenatal alcohol exposure from mothers and fathers drinking and head circumference: Results from the Norwegian Mother-Child Study (MoBa). Sci. Rep. 7, 39535. https://doi.org/10.1038/srep39535 (2016).
Zhou, Q. et al. Association of preconception paternal alcohol consumption with increased fetal birth defect risk. Jama Pediatr. 175, 742–743. https://doi.org/10.1001/jamapediatrics.2021.0291 (2021).
Milne, E. et al. Parental alcohol consumption and risk of childhood acute lymphoblastic leukemia and brain tumors. Cancer Causes Control 24, 391–402. https://doi.org/10.1007/s10552-012-0125-5 (2013).
Braun, J. M., Messerlian, C. & Hauser, R. Fathers matter: Why it’s time to consider the impact of paternal environmental exposures on children’s health. Curr. Epidemiol. Rep. 4, 46–55. https://doi.org/10.1007/s40471-017-0098-8 (2017).
Krawetz, S. A. et al. A survey of small RNAs in human sperm. Hum. Reprod. 26, 3401–3412. https://doi.org/10.1093/humrep/der329 (2011).
Rompala, G. R. et al. Heavy chronic intermittent ethanol exposure alters small noncoding RNAs in mouse sperm and epididymosomes. Front. Genet. 9, 32. https://doi.org/10.3389/fgene.2018.00032 (2018).
Abel, E. Paternal contribution to fetal alcohol syndrome. Addict. Biol. 9, 127–133. https://doi.org/10.1080/13556210410001716980 (2004) (discussion 135-126).
Gemes, K., Moeller, J., Engstrom, K. & Sidorchuk, A. Alcohol consumption trajectories and self-rated health: Findings from the Stockholm Public Health Cohort. BMJ Open 9, e028878. https://doi.org/10.1136/bmjopen-2018-028878 (2019).
Kimura, Y. et al. Alcohol consumption and incidence of proteinuria: A retrospective cohort study. Clin. Exp. Nephrol. 22, 1133–1142. https://doi.org/10.1007/s10157-018-1568-0 (2018).
Young, C. K. & McNaughton, N. Mixed effects of low-dose ethanol on cortical and hippocampal theta oscillations. Neuroscience 429, 213–224. https://doi.org/10.1016/j.neuroscience.2020.01.007 (2020).
Tarter, R. E., Hegedus, A. M., Goldstein, G., Shelly, C. & Alterman, A. I. Adolescent sons of alcoholics: Neuropsychological and personality characteristics. Alcohol Clin. Exp. Res. 8, 216–222. https://doi.org/10.1111/j.1530-0277.1984.tb05842.x (1984).
Hegedus, A. M., Alterman, A. I. & Tarter, R. E. Learning achievement in sons of alcoholics. Alcohol Clin. Exp. Res. 8, 330–333. https://doi.org/10.1111/j.1530-0277.1984.tb05522.x (1984).
Sood, B. et al. Prenatal alcohol exposure and childhood behavior at age 6 to 7 years: I. Dose-response effect. Pediatrics 108, E34. https://doi.org/10.1542/peds.108.2.e34 (2001).
Liang, H. et al. Estimation of cancer incidence and mortality attributable to alcohol drinking in China. BMC Public Health 10, 730. https://doi.org/10.1186/1471-2458-10-730 (2010).
Spry, E. A. et al. Parental mental health before and during pregnancy and offspring birth outcomes: A 20-year preconception cohort of maternal and paternal exposure. EClinicalMedicine 27, 100564. https://doi.org/10.1016/j.eclinm.2020.100564 (2020).
Sharp, G. C., Lawlor, D. A. & Richardson, S. S. It’s the mother!: How assumptions about the causal primacy of maternal effects influence research on the developmental origins of health and disease. Soc. Sci. Med. 213, 20–27. https://doi.org/10.1016/j.socscimed.2018.07.035 (2018).
Moreira, L. R. et al. Trends and inequalities in unplanned pregnancy in three population-based birth cohorts in Pelotas, Brazil. Int. J. Public Health 65, 1635–1645. https://doi.org/10.1007/s00038-020-01505-0 (2020).
Acknowledgements
We are greatly indebted to all the pregnant women and their families who voluntarily participated in the present study. Thank all staffs of the Shanghai-Minhang Birth Cohort Study team for their invaluable contribution during data collection. This work was supported by the National key research and development program (2016YFC1000505; 2018YFC1002801).
Author information
Authors and Affiliations
Contributions
M.L. and X.H.Z. collected data, carried out the initial analyses, drafted the initial manuscript, and revised the manuscript. H.L., F.Y., Y.C., and X.X.S. designed the data collection instruments, collected data, and reviewed and revised the manuscript. M.H.M. and W.Y. conceptualized and designed the study, coordinated and supervised data collection, and critically reviewed the manuscript for important intellectual content. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Luan, M., Zhang, X., Fang, G. et al. Preconceptional paternal alcohol consumption and the risk of child behavioral problems: a prospective cohort study. Sci Rep 12, 1508 (2022). https://doi.org/10.1038/s41598-022-05611-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-05611-2
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.