Abstract
The gut microbiota of mosquitoes plays a critical role in the life history of the animal. There is a growing body of research characterising the gut microbiota of a range of mosquito species, but there is still a paucity of information on some members of the Anopheles gambiae complex. In this study, the gut microbiota of four laboratory strains were characterised. SENN (Anopheles arabiensis—insecticide susceptible major vector), SENN DDT (Anopheles arabiensis—insecticide resistant major vector), MAFUS (Anopheles merus—minor vector) and SANGWE (Anopheles quadriannulatus—non-vector) were used in this study. The microbiota of fourth instar larvae, 3-day old, 15-day old non-blood fed and 15-day old blood fed females were characterised by MALDI-TOF mass spectroscopy and 16 s rRNA gene sequencing by next generation sequencing. The four strains differed in species richness but not diversity. The major vectors differ in β-diversity from that of the minor and non-vectors. There was no difference in α- or β-diversity in 15 non-blood fed females and 15-day old females that had 3 blood meals before day 15. These differences may be related to a mixture of the effect of insecticide resistance phenotype as well as a potential relationship to vector competence to a limited extent. Bacterial diversity is affected by species and age. There is also a potential relationship between the differences in gut microbiota and capacity to transmit parasites. This genetic background of the mosquitoes, however, play a major role, and must be considered in this relationship.
Similar content being viewed by others
Introduction
The Anopheles gambiae complex is a group of morphologically identical mosquitoes that differ in their behaviour and ability to transmit the malaria parasite to human hosts1. Three members of the An. gambiae complex are found in South Africa. The major malaria vector An. arabiensis, a minor malaria vector An. merus and a non-malaria vector An. quadriannulatus are vectors that are all found in Africa within the malaria endemic provinces of South Africa2. Currently, the only species implicated in malaria transmission in South Africa is An. arabiensis3.
Anopheles arabiensis is one of the dominant malaria vector species in Sub-Saharan Africa1,4. Anopheles arabiensis exhibits anthropophilic (human biting), zoophilic (animal biting), endophagic (indoor feeding), exophagic (outdoor feeding), endophilic (indoor resting) and exophilic (outdoor resting) behavioural patterns1,5,6.
The members of the An. gambiae complex found in South Africa differ from An. gambiae ss and An. coluzzi which commonly display exophily and exophagy. Yet, they differ markedly in their vector competence1. As such, behavioural differences alone cannot account for these differences. There would also be a molecular basis for these differences, particularly in terms of immunological underpinnings.
To highlight an example of the role of immunity, An. quadriannulatus can be artificially infected with Plasmodium falciparum at low efficiency7, even though this is not usually observed in the wild. The prophenoloxidase system of An. quadriannulatus is initiated at lower levels than the major vector An. gambiae8. This highlights the importance of the immunological response to the parasite. An increased understanding of the immunological differences between vectors and non-vectors would inform on future transmission blocking efforts.
A key factor in determining vector competence is the gut microbiota of the mosquito. The microbiota of the midgut in a mosquito plays crucial roles in development, reproduction, immunity, and vector competence9. The composition of the midgut varies with the life cycle stage and diet10. The larval gut microbiome is crucial for development as gut-sterilised mosquitoes cannot pupate10,11,12. Larvae feed on the contents of the surrounding water as well as drink the surrounding water. This suggests that the diet obtained from the environment is important in the survival and replacement of new bacteria in the midgut13,14.
For malaria to be transmitted by an Anopheles mosquito, an infectious blood meal must be ingested and make it through the developmental bottlenecks in the midgut and salivary glands. The midgut is the first and main bottleneck of parasite development, as this is where the parasite undergoes the sexual stages of reproduction. The success of the escape of the motile ookinetes from the midgut is crucial for successful parasite proliferation15. Therefore, the study of the dynamics of the midgut of the mosquito is a key field of study to develop alternative vector control strategies.
The midgut microbiota of the Anopheles mosquito contributes to the ability of the mosquito to contract and transmit the Plasmodium parasite as it is involved with the immunity of the mosquito16,17. The midgut microbiota protects the mosquito against Plasmodium infection through various pathways. The bacteria in the midgut can act as a physical barrier after a blood meal has been ingested, preventing penetration of the parasite into the midgut wall18. The bacteria in the midgut can directly affect the parasite by producing antimicrobial compounds or by inducing oxidative stress. In addition to these other defence mechanisms, the bacteria can activate the NF-κB dependent Immune-deficiency (imd) pathway to fight Plasmodium infection15.
A search of the term “Anopheles microbiome” on Pubmed has increased from 3 records in 2010 to 23 in 2020. This indicates a slow but steady increase of the information in this field. Although the field of mosquito microbiome studies are advancing, the studies tend to be limited in the species studies, typically focussing on An. stephensi, An. coluzzi and An. gambiae ss, examples of studies includes Sharma et al.; Galeano-Castañeda et al.; Saab et al.17,19,20. Studies of the microbiome of An. arabiensis and other members of the An. gambiae complex are notably lacking. Characterising the gut microbiota of Plasmodium refractory species could provide information on the role of gut microbiota in this phenotype. This study aimed to characterise the dynamic gut microbiota of the minor vector An. merus and the non-vector An. quadriannulatus. Furthermore, this will be compared to the dynamic gut microbiome of insecticide resistant and susceptible An. arabiensis.
Results
Culture-dependent characterisation of the dynamic gut microbiota by MALDI-TOF MS
The species identified by MALDI-TOF MS were dominated by Gram-negative bacteria, specifically γ-proteobacteria of the family Enterobacteriaceae (0.41). The remaining γ-proteobacteria were from the families of Yersiniaceae (0.09), Aeromonadaceae, (0.08). Pseudomonadaceae (0.07), Moraxellaceae (0.05), and Morganellaceae (0.02). The next most abundant classes were the Bacilli (0.02) and Actinobacteria (0.01), which constituted the majority of the Gram-positive bacteria identified. The final significant Gram-negative class was the Weeksellaceae (0.08) family. The results of the MALDI-TOF study are summarised in Supplementary Table S1.
Culture-independent characterisation of the dynamic gut microbiota by 16S rRNA gene sequencing
Alpha diversity determined by 16S sequencing
When comparing the alpha diversity of the complete groups, the species richness indices (a measure of the total number of species in the relevant strain and treatment) Chao1 and ACE (summarised in Table 2) indicate a significant difference between the strains. Species evenness indicators (a measure of relative abundance of species in the relevant strain and treatment), Shannon and Simpson (summarised in Table 3), do not indicate a significant difference. Chao 1 indices indicated a significant difference between strains (Kruskal–Wallis One-way ANOVA: p < 0.01). SENN (An. arabiensis-insecticide susceptible) had a significantly higher index than SENN DDT (An. arabiensis-insecticide resistant) (p = 0.01), as well as SANGWE (An. quadriannulatus) (p = 0.01) and MAFUS (An. merus) (p < 0.01) (Fig. 1A). Similarly, with ACE indicators, there was a significant difference between the strains (Kruskal–Wallis One-way ANOVA: p = 0.03). SENN had a significantly higher ACE index than SENN-DDT (p = 0.02). Although SENN did not have a significantly different ACE index than MAFUS (p = 0.05), it did have a significantly higher ACE index than SANGWE (p = 0.02). MAFUS and SANGWE did not differ in ACE indices (p = 0.13) (Fig. 1B).
Overall, the Shannon diversity indices of the strains did not differ (Kruskal–Wallis One-way ANOVA: p = 0.14). SANGWE had a significantly higher Shannon diversity index than SENN (p = 0.03) as did MAFUS (p = 0.02). SENN DDT did not differ significantly from SENN (p = 0.44) (Fig. 1C). The Simpson’s diversity index was also not significantly different (Kruskal–Wallis One-way ANOVA: p = 0.14). The only significant difference in Simpson’s index was significantly higher indices in MAFUS than SENN DDT (p < 0.01) (Fig. 1D).
When comparing alpha diversity between the life stages of the strains, the Chao 1 index of the fourth instar larvae was significantly higher than that of 15-day old sugar fed adults (p < 0.01) and 15-day old blood fed adults (p = 0.02). The ACE index of the fourth instar larvae was significantly higher than that of 3-day old adults (p = 0.02), 15-day old non-blood fed adults (p < 0.01), and 15-day old blood fed adults (p < 0.01). The Shannon index of the larvae was significantly higher than that of 3-day old adults (p < 0.01), 15-day old non-blood fed adults (p < 0.01) and 15-day old blood fed adults (p < 0.01). The Simpson diversity index of the larvae was significantly higher than that of 3-day old adults (p < 0.01), 15-day old non-blood fed adults (p < 0.01) and 15-day old blood fed adults (p < 0.01). There was no difference in species richness between older blood fed and non-blood fed adults as Chao 1 indices did not differ significant (p = 0.71) neither did ACE indices differ (p = 0.63). This was also true for species evenness, as there were no significant differences in Shannon diversity indices (p = 0.41) or Simpson diversity indices (p = 0.93) (Supplementary Table S2).
Within the strains, only MAFUS had a significant difference within Chao1 index between the life stages (Kruskal–Wallis One-way ANOVA: p = 0.03). There was no difference in ACE indices between the life stages of SENN (p = 0.31), SENN DDT (p = 0.44), MAFUS (p = 0.05) and SANGWE (p = 0.07). When examining indices examining evenness rather than the richness, there was a significant difference in Shannon diversity indices between life stages in SANGWE (p = 0.03) as well as MAFUS (p = 0.03), but not in SENN (p = 0.09) or SENN DDT (p = 0.08). When examining Simpson diversity indices, only SENN had a significant difference in indices between life stages (p = 0.04) (Supplementary Table S2). The variation in the findings is confirmed by rarefaction analysis, with larval diversity being the greatest in all samples. This analysis also confirms the depth of sequencing (Fig. 2A). Alpha diversities indices are summarised in Tables 1 and 2.
Rarefaction curves and beta diversity plots of the An. gambiae complex. (A) rarefaction curves of MAFUS (An. merus), SANGWE (An. quadriannulatus), SENN (An. arabiensis-insecticide susceptible) and SENN-DDT (An. arabiensis-insecticide resistant). (B) Principle Coordinates Analysis plot of beta-diversity highlighting the differences between strains. (C) Principal Component Analysis plot of beta-diversity highlighting differences between life stages.
Beta diversity analysis determined by 16S sequencing
Two β-diversity analyses were performed, and both confirmed the same patterns. NMDS analysis demonstrated that MAFUS (An. merus) and SANGWE (An. quadriannulatus) clustered together and were significantly different from SENN (An. arabiensis-insecticide susceptible) and SENN DDT (An. arabiensis-insecticide resistant) (PERMANOVA: p = 0.01, F = 6.59) (Fig. 2B). Principle Co-ordinates analysis (PCoA) also confirmed this finding (PERMANOVA: p = 0.01, F = 6.59). There was also a significant difference in β-diversity of larvae and 15-day old adults, with 3-day old females overlapping the different age groups (PERMANOVA: p = 0.01, F = 4.89) (Fig. 2C).
Comparison of bacterial species overlaps and differential abundance in mosquito strains
When examining the overlap in bacterial specimens identified, all four strains had 17 families in common. SENN (An. arabiensis-insecticide susceptible) and SENN DDT (An. arabiensis-insecticide resistant) shared 6 families, while SANGWE (An. quadriannulatus) and MAFUS (An. merus) shared only one family (Fig. 3A). When examining overlapping genera, all four strains had 33 genera in common. MAFUS and SENN DDT had the most unique genera with four each. There were 14 shared genera between SENN and SENN DDT, but only 4 shared between MAFUS and SANGWE (Fig. 3B). When examining species composition, MAFUS had 10 unique species, the most of any of the strains. SANGWE had 4 unique species, and SENN DDT had 3 unique species. SENN had no unique species present (Fig. 3C).
Overlapping midgut bacteria identified between the different mosquito strains. The blue dots represent the number of overlaps per strain, with single dots indicating the number of unique families/genera/species per strain. (A) Comparison of common families. There are 17 common families between all four strains. (B) Comparison of common genera. There are 33 common genera between all four strains. (C) Comparison of common species. There are 34 common species between all strains.
Differential abundance was assessed between strains. For each pairwise comparison, genera fold changes with adjusted p-values (q-value) < 0.01 were considered differentially abundant. When comparing SENN to SANGWE Aeromonas and Aquitalea had the greatest fold increased abundance in SENN, and Mesorhizobium and Sphingopyxis having the smallest fold change. Notable abundance differences were increased Elizabethkingia and Rhanella in SENN and increased Pseudomonas, Klebsiella and Serratia in SANGWE. Similarly, Aquitalea and Aeromonas had the greatest fold increased abundance in SENN DDT compared to SANGWE, and Mesorhizobium and Microbacterium having the smallest fold change. Notable abundance differences were higher abundances of Rhanella and Elizabethkingia in SENN-DDT, and increased abundances of Pseudomonas and Klebsiella in SANGWE.
When examining differential abundance between MAFUS and the two An. arabiensis strains, SENN and SENN DDT, Serratia was the genus with the greatest differential abundance in both SENN and SENN DDT compared to MAFUS. The smallest differential abundance in both SENN and SENN DDT compared to MAFUS are Sphingopyxis and Mesorhizobium. Pseudomonas and Enterobacter were more differentially abundant in MAFUS than either SENN or SENN DDT. When comparing the differential abundance of MAFUS and SANGWE, Acromobacter, Sphingobacterium and Elizabethkingia had the greatest differential abundance in MAFUS. Mycobacterium, Leucobacterium and Polynucleobacter had the least differential abundance in MAFUS. There were no genera differentially abundant between SENN and SENN DDT at q-value < 0.01 significance level. When using a less stringent significance level of q-value < 0.05, differentially abundant species were largely observed when comparing blood-fed females. Klebsiella were more abundant in SENN, while Rhanella, Aeromonas and Serratia were more abundant in SENN DDT (Fig. 4). With the same adjustment, Cedecea, Klebsiella, Enterobacter and Rhanella had a greater differential abundance in 3-day old SENN and Zooglea had the lowest differential abundance. The fold changes associated with the differential abundance are summarised in Tables 3 and 4.
Comparison of differential abundance of genera between strains. (A) Differential abundance of genera in SENN vs SANGWE at an 99% Confidence interval. (B) Differential abundance of genera in SENN-DDT vs SANGWE at a 99% Confidence interval. (C) Differential abundance of genera in MAFUS vs SANGWE at a 99% Confidence interval. (D) Differential abundance of genera in SENN vs MAFUS at a 99% Confidence interval. (E) Differential abundance of genera in SENN vs SANGWE at an 99% Confidence interval. (F) Differential abundance of genera in 15-day bloodfed SENN vs 15 -day bloodfed SENN-DDT at a 95% confidence interval. No genus was found to be significantly abundantly expressed between these two strains overall at the 99% confidence interval. The dotted line at 0 represents the change from over representation to underrepresentation of bacterial genera. The species name on either side of the dotted line where the relevant bacterial species are abundant.. Each dot represents a single OTU.
Discussion
The amount of mosquito species where the gut microbiota is being characterised is increasing12,21,22,23 and as such the catalogue of microbiome information available for comparison is increasing. The use of MALDI-TOF MS resulted in a markedly less species identified than NGS. These findings were congruent with a recent finding that analysed the microbiota of laboratory and wild An. arabiensis by MALDI-TOF MS where similar numbers and species were identified21. For the MS technique to be used for larger scale microbiome studies, culture protocols will have to be optimised. This will include optimising the choice of culture media as well as growing plates under anaerobic conditions to maximise the diversity of samples cultured. The use of an alternate database could also potentially improve the quality of analysis. Therefore, although less data was obtained by MALDI-TOF analysis, it is still a viable technique for identifying mosquito gut microbiota.
NGS was the more sensitive technique. This technique, however, are more prone to contamination which can result in misidentification. A common source of contamination is often the DNA extraction kit. In support of this, a study by Mancini et al.24 demonstrated that some of the most common species associated with kit-borne contamination found in mosquito microbiome study included Mesorhizobium, Phyllobacterium, Rhizobium, Comamonas, Delftia, Variovorax and Escherichia-Shigella24. In the current study Phyllobacterium and Rhizobium were not identified, while Mesorhizobium, Comamonas, Delftia, Variovorax and Escherichia-Shigella were detected at low abundance (< 0.01% prevalence). The human skin contaminants Corynebacterium (12 OTUs) and Streptococcus (119 OTUs) were also identified at low abundance (< 0.01% prevalence), while Propionibacterium, another skin-associated contaminant was not identified at all.
There have been several studies associating gut microbiota with insecticide resistance22,23,25. These studies compared the gut microbiota of fenitrothion resistant and susceptible An. albimanus populations23 as well as observing distinct gut microbiota in pyrethroid resistance in An. gambiae22. The insecticide susceptible SENN strain had a greater species richness than the insecticide resistant SENN-DDT strain, but this did not translate into a difference in species diversity. Both strains also had overlapping β-diversity. There were also no bacterial species differentially abundant in the strains at the 99% confidence, unlike when compared to other strains. The increased number of species in SENN suggests a reduction in bacterial species due to selection for resistance23. However, the presence of species and genera associated with insecticide degradation in both species suggests that this is not a significant contributor to insecticide resistance in the SENN DDT strain. As the SENN DDT strain is fixed for the L1014F mutation as well as having elevated detoxification enzymes26, it is likely that the contribution of insecticide degrading bacteria to the resistant phenotype is minimal. It is, however, worth noting that Sphingobacterium, an insecticide degrading genus22, was significantly reduced in the insecticide susceptible SANGWE. A previous study found Microbacterium was unique to insecticide susceptible An. albimanus23. In this study, Microbacterium had a greater relative abundance in the insecticide susceptible SANGWE and MAFUS, which has low level insecticide tolerance.
Regardless of strains, larvae had the greatest species richness and diversity of all the life stages. This is congruent with previous findings on the dynamic gut microbiota of An. gambiae12, where species richness decreased with age and blood feeding status. The shift in microbiota between the aquatic and aerial stages has been observed previously10,27. Furthermore, in an examination of the dynamic gut microbiota of An. gambiae, diversity decreased with age and blood feeding status12,28. This was confirmed by comparing the differential abundance of the combined groups, where larvae had nineteen genera with a higher differential abundance compared to six in 15-day old females which had multiple blood meals. Asaia, Elizabethkingia, Serratia and Rhanella were more abundant in blood fed females, while Aquitalea, Sphingobacterium and Sphiyngopyxis were the most abundantly expressed in larvae. Consistent with previous findings, this represents a shift from aerobic to facultatively anaerobic29. Unlike other studies, this study did not examine the direct effect of blood on gut microbiota. Rather, it examined the longer-term effects of multiple blood meals. This is because multiple blood meals are a common feeding strategy (e.g., Norris et al.)30 which is known to increase the Plasmodium infectivity31. The lack of species richness and diversity in the adult stages examined in this study suggests that specific bacterial species, rather than population changes contribute to the microbial effect on life history.
A range of factors affect the composition of the gut microbiota. This includes the rearing water, food and sugar source20. Like with previous studies, several common species constituted the majority of gut microflora. These include Enterobacter, Rhanella, Klebsiella, Serratia, Pseudomonas, Elizabethkingia, Asaia and Raoultella. This supports the suggestion of a core gut microbiota. A core microbiome has been suggested to exist for An. gambiae and An. coluzzi32. As the gut microbiome is markedly less diverse than that of the salivary glands and reproductive organs24,33, it is plausible that a set of bacteria could constitute the core gut microbiota of anophelines. It has also been suggested that the core microbiota persists over time34, which is also supported by this study.
There have not been many studies that have characterised members of the An. gambiae complex. The majority of studies have focused on the major vector An. gambiae (e.g., Gimonneau et al.; Boissière et al.)33,35. There are fewer examples of An. arabiensis to compare to (E’Silva et al.; Barnard et al.)21,36 and even less for An. quadriannulatus and An. merus24. As such, there are not many databases for to compare the data generated in this study. A notable conservation is that in An. arabiensis from the E’Silva study, which constitutes young, unfed females are dominated by Serratia as are both An. arabiensis strains in this study. Interestingly, this congruency does not hold true for the dominance of Pseudomonas in An. merus and An. quadriannulatus when compared to Mancini et al.24.
The amount of microbiome data available for members of the An. gambiae complex outside that of the nominal member33 is relatively limited. A single previous study examined the microbiota of several tissues in nine mosquito species, including An. gambiae, An. arabiensis, An. merus and An. quadriannulatus24. The present study is more similar to Wang et al.12 which examined the dynamic gut microbiota of An. arabiensis. Although this took place in laboratory strains, the uniformity in larval feeding, sugar feeding and provision of blood meals should limit the rate of false discovery, this cannot be fully guaranteed20.
Indices that estimate richness (Chao1 and ACE) indicate a significant difference between the two An. arabiensis strains with the insecticide susceptible strain having a significantly higher species richness (number of bacteria). This difference did not extend to species diversity, as determined by Shannon and Simpson indices. Anopheles merus and An. quadriannulatus did not differ in α-diversity but had higher Shannon indices than the insecticide susceptible An. arabiensis. Despite the lack of difference in alpha diversity, there is a marked difference in beta diversity between the strains, with the major vector species SENN and SENN DDT clustering separately from the minor vector MAFUS and the non-vector species SANGWE. This is markedly different to the lack of beta diversity between wild An. gambiae and An. coluzzi33. Laboratory reared mosquitoes have been described as having lower gut microbial diversity than their wild counterparts (as reviewed in Dada et al.)37. Despite this, a significant difference in beta diversity was observed in the present study.
Anopheles gambiae and An. coluzzi are both efficient major vectors of malaria that do not differ in microbial beta diversity. By contrast, An. arabiensis, An. merus and An. quadriannulatus differ in vector competence. This difference in vector competence is unlikely to be explained fully by behaviour. Anopheles arabiensis, An. merus and An. quadriannulatus are all primarily zoophilic and outdoor biting. Therefore, the differences in vector competence may have further molecular underpinnings, such as variable immune responses (e.g., Habtewold et al.)38. The differences in gut microbiota between the major, minor, and non-vectors may therefore contribute vector competence, although this needs to be confirmed by Plasmodium-infection studies.
Due to their anti-Plasmodium activities, Asaia, Pantoea, Pseudomonas, Enterobacter, Serratia are gaining considerable attention as promising candidates for paratransgenic modifications for vector control strategies24. Pseudomonas putida, Pantoea sp and S. marcescens were capable of blocking Plasmodium development in vivo when introduced as a sugar meal. Comamonas sp., Acinetobacter sp., P. putida and P. rhodesiae, Pantoea sp, S. marcescens and E. anophelis demonstrated Plasmodium blocking activity in vitro39. The Enterobacter isolate Esp_Z inhibits Plasmodium development from gametocyte to ookinete by in vivo and in vitro production of reactive oxygen species40. When comparing differential abundance of An. quadriannulatus to both An. arabiensis strains, Pseudomonas was more abundant in the former. Additionally, Serratia and Enterobacter was more abundant in An. quadriannulatus compared to the insecticide susceptible An. arabiensis. Pseudomonas, Pantoea, and Enterobacter had a greater differential abundance in An. meus. This further suggests that differential gut microbiota may contribute to differences in vector competence between the strains Serratia had a higher differential abundance in An. quadriannulatus. Serratia, however, had a greater differential abundance in the An. arabiensis strains. It is worth noting, however, that despite the association of Serratia with anti-Plasmodium defense, this is strain dependent41. Serratia marcescens, however, is known to degrade several insecticides23. The higher expression in the An. arabiensis strains may be related more to insecticide resistance than anti-parasite defense.
The microbiota of An. merus and An. quadriannulatus is poorly examined. Although this study does contribute information to this small pool, there are several limitations to this study. The first limitation is that this study requires replication on field specimens (as described in Romoli and Gendrin)15 Secondly, experiments need to be performed examining the effect of Plasmodium infection on gut microbial diversity to confirm the species-specific role of microbiota in vector competence. It would also be worth examining changes in the microbiota of the salivary glands after infection. Furthermore, it is worth considering the effect of saltwater breeding on the microbiota of An. merus. As an example, Rhanella is a freshwater bacterial species42, potentially explaining the low expression of this species in An. merus.
The An. merus strain was reared in salt water, but as an osmotolerant species, considerations must be made of the effect of salt concentration on the gut microbiota of this species. The variation in beta diversity between the two An. arabiensis strains and the An. merus and An. quadriannulatus may be due to the evolutionary distance between the species, but the lack of beta diversity between An. quadriannulatus and An. merus should be taken into account as motivation for the validity of these findings. Although the findings could suggest a role for gut microbiota in differences in Plasmodium transmission, these findings must be replicated with wild mosquitoes, and preferably with experiments that examine the changes in gut microbiota before and after Plasmodium infection. These experiments are, however, beyond the scope of this study.
In conclusion, this study characterised the dynamic gut microbiota of An. merus, An. quadriannulatus and An. arabiensis by culture-dependent and culture independent techniques. Although α-diversity did not differ greatly, there was a significant difference in β-diversity of the major vector An. arabiensis and minor vector An. merus and the non-vector An. quadriannulatus. This diversity, in conjunction with the decreased relative abundance of genera associated with anti-Plasmodium effects in An. arabiensis suggests that this difference may be associated with vector competence. Although both the insecticide susceptible and resistant An. arabiensis harboured insecticide degrading bacteria, genera associated with insecticide susceptibility was associated with the insecticide susceptible An. quadriannulatus strain. Therefore, this study suggests a role for differential microbial diversity in the life history of zoophilic members of the An. gambiae complex.
Materials and methods
Mosquito collection and preparation for characterisation
Laboratory reared Anopheles mosquitoes used were housed and collected from the Botha de Meillon insectary of the National Institute of Communicable Disease (NICD), Johannesburg, South Africa. Four strains, representing three species were used in this study. The An. arabiensis strain SENN is an insecticide susceptible strain originated in Sennar, Sudan. SENN has been in colony since 1980. The An. arabiensis strain SENN DDT is an insecticide resistant strain that has continuously been selected for DDT resistance from the SENN strain since 1995. This strain is still being selected for DDT resistance. SENN DDT is fixed for the kdr L1014F mutation. It also has elevated cytochrome P450, general esterase as well as Glutathione S-transferase activity. This accounts for the strain’s resistance to DDT, permethrin, deltamethrin, λ-cyhalothrin and malathion26,43,44. The An. quadriannulatus strain (SANGWE) is insecticide susceptible and originated from Zimbabwe. MAFUS is an An. merus strain originating from Malahlapanga, South Africa and is tolerant to insecticides. It has been in colony since 2012 and was reared in 50% seawater.
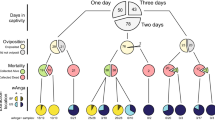
Larvae were reared in reverse osmosis water at 25 °C (± 2 °C), 80% relative humidity (± 5%) with a 12-h light/dark cycle and a 30-min dusk/dawn cycle45. Larvae were fed a mixture of powder Beano™ dog biscuits and yeast. Several life stages of An. arabiensis (SENN and SENN DDT), An. merus and An. quadriannulatus were collected: fourth instar larvae, 3-day old female adults, 15-day old non-blood fed female adults and 15-day old blood fed female adults. Blood feeding took place at the age of 3, 7 and 11 days. Blood was supplied by a single consenting human volunteer, the author Shüné V. Oliver, as per the ethics waiver provided by the University of the Witwatersrand (Ethics waiver number: 09-06-2020-O). Blood was supplied as a live feeding volunteer, and no other individuals besides the author were involved in providing the blood meal. For MALDI-TOF mass spectrometric analysis of the midgut microbiota, 180 female mosquitoes and 60 larvae were used. For 16S rRNA gene sequencing analysis of the midgut microbiota, 180 female mosquitoes and 60 larvae were used.
Matrix assisted laser desorption ionisation-time of flight (MALDI-TOF) mass spectrometric analysis of the midgut microbiota
Midgut resection and culturing procedure
Larvae were chilled at 4 °C to minimise their activity and adult female mosquitoes were cold killed at − 70 °C for 5 min prior to midgut resection. Strict sterile conditions were implemented, the forceps, microscope, microscope slides and mosquitoes were sterilised with 70% ethanol. The midguts from the respective samples were dissected using a dissection microscope (Olympus, SZ2-ILST) with a magnification of 40×. Five replicates consisting of three midguts each were suspended in 200 µl of 0.1 M Phosphate Buffered Saline (PBS) pH 7.2.
The extracted guts were aseptically added to 2 ml of Lysogeny Broth (LB broth). The culturing tubes were incubated at 37 °C overnight with constant shaking at approximately 120 rpm. The negative control consisted of PBS in uninoculated LB broth. Each of the samples were plated onto three types of media, namely, Brain Heart Infusion BHI agar (for the culture of fastidious organisms), a 10% blood agar (for the culture of fastidious organisms) and a MacConkey agar (selective for lactose fermenters). All culture plates were obtained from Diagnostic Media Products (South Africa). Single colonies were obtained by streak plating with subsequent incubation at 37 °C for 18 h.
Bacterial identification using MALDI-TOF spectroscopy
The mass spectrometry system used in this study is housed in the Centre for Healthcare-Associated Infections, Antimicrobial Resistance and Mycoses (CHARM) at the National Institute of Communicable Diseases in Johannesburg, South Africa.
To perform the analysis individual bacterial colonies were selected manually from each plate based on the size, colour, growth pattern or shape and transparency. The individual colony was selected and applied to a 96-well MALDI target plate (Bruker Daltonics, Wissembourg, France; cat no. 8280800) in duplicate. Brucker Matrix HCCA (α-Cyano-4-hydroxycinnamic acid), portioned at 2.5 ± 0.3 mg matrix (Hain Life Sciences) was dissolved in 250 µl Standard solvent (acetonitrile 50%, water 47.5% and trifluoroacetic acid 2.5%) (Sigma-Aldrich). One microlitre of Bruker HCCA Bruker (10 mg Bruker HCCA/mL) was added to each occupied target spot position and left to dry at room temperature.
The target plate was placed into the Brucker Matrix-assisted Laser Desorption Ionising Biotyper System with a benchtop Microflex LT/SH mass spectrometer (Bruker Daltonics, Wissembourg, France). The bacteria were identified on 16 s rRNA spectra as per manufacturer’s instructions. For each spectrum obtained a maximum of 100 peaks were used and compared with other peaks in the MBT 7854 MSP Library (Bruker Daltonics, Wissembourg, France; ref no. 182903). The bacteria were accepted as correctly identified if the identification score was ≥ 1.946.
For all downstream analysis α-diversity indices were divide into species richness and species evenness indicators. The Chao1 and ACE indices were used to measure species richness, a direct count of the Operational Taxonomic Units (OTUs). The Shannon and Simpson diversity indices were used as measures of species evenness. The Shannon and Simpson diversity index is a measure of diversity which considers both richness as well as their relative abundance or evenness.
16S rRNA gene sequencing analysis of the midgut microbiota
Midgut dissection and DNA extraction
The midguts of the female adult mosquitoes and larvae were dissected as previously mentioned however, five replicates consisting of five midguts each were suspended in 200 µl of 0.1 M PBS pH 7.2. To ensure sterile conditions, all equipment and surfaces used were sterilised using 70% ethanol and 3% hydrogen peroxide. An extraction negative control of 0.1 M PBS pH 7.2 only was used during the course of the DNA extraction.
Total genomic DNA from the midgut of the mosquito was extracted using the DNeasy PowerSoil® Kit (QIAGEN: 12888-50). The tubes containing the midguts were centrifuged at 5000×g and approximately 150 µl of PBS was discarded, the remaining PBS and midguts were vortexed briefly and added to the PowerBead™ tubes. The protocol was then followed as per manufacturer’s instructions.
Amplification of the V3-V4 hypervariable region of the 16S rRNA gene
The V3-V4 hypervariable region of the bacterial 16S rRNA gene was amplified using universal bacterial primers. Forward Primer (5′ GTC TCG TGG GCT CGG AGA TGT GTA TAA GAG ACA GGA CTA CHV GGG TAT CTA ATC C 3′)(Integrated DNA Technologies) and Reverse Primer (5′ TCG TCG GCA GCG TCA GAT GTG TAT AAG AGA CAG CCT ACG GGN GGC WGC AG 3′) (4 nmol Ultramer® DNA Oligo 55 bases : Integrated DNA Technologies)47. Library preparation was performed according to the standard instructions of the 16S Metagenomic Sequencing Library Preparation protocol (IlluminaTM, Inc., San Diego, CA, United States). The size of the amplicons were then visualised using the 4200 TapeStation (Agilent Technologies). Two negative controls were used in the amplicon preparation step. The extraction negative control as well as no template control were used in the initial amplification step. No amplification was observed in either of the negative controls.
Libraries were then sequenced on the MiSeq platform using MiSeq Reagent kit v3 (Illumina) and paired-end 2 × 300 bp sequencing was performed at the Sequencing Core Facility, National Institute for Communicable Diseases, South Africa.
Bioinformatic analysis
QIAGEN CLC Genomics Workbench 20 (CLC Bio Qiagen) was used to identify the operational taxonomic units (OTU’s) of each of the specimens analysed. Amplicon-based analysis workflows were used for data quality control and OTU clustering.
Sequences were filtered, trimmed and subjected to quality control to ensure clean data was used for clustering. The raw reads were trimmed with a 0.05 quality limit. Clustering of the data was done at a 99% similarity threshold with the SILVA 16S v132 database. The OTU’s were assigned to sequences based on 97% identity. The OTU table was then used to obtain a visual representation of the midgut bacteria of each respective sample in the form of a bar plot.
Bacterial diversity analysis was done using raw reads, which were quality controlled and filtered (Q > 20 and length > 50 bp) using fastqc (v0.11.8) and trimGalore (v0.6.4_dev; https://github.com/FelixKrueger/TrimGalore), respectively48,49. In addition, trimGalore was used for adapter removal.
All the downstream analyses were performed in R (v3.6.1), including classification, abundance estimations, statistical analysis, and visualisation. Clean reads were pre-processed using dada2 package (v1.12.1)50, including quality inspection, trimming, dereplication, merging paired-end reads and removal of chimeric sequences.
Taxonomy was assigned to the obtained amplicon sequence variants (ASVs) and the ASV abundance estimates determined using training sequence sets based on the SILVA reference database (v138; https://zenodo.org/record/1172783#.XvCmtkUzY2w).
Ordinations for beta diversity, abundance bar plots, alpha diversity and richness estimates, and heatmaps were generated using the phyloseq package (v1.28.0)51, ggplot2 (v3.2.1) and AmpVis2 package (v2.6.4)52. Data clustering in NMDS were assessed using PERMANOVA (permutation test with pseudo-F ratios) as implemented in the adonis function in the vegan package (https://github.com/vegandevs/vegan).
Kruskal–Wallis rank-sum tests were used to compare alpha diversity between groups. Venn diagrams were generated using Venn Diagram (v1.6.20) and UpsetR (v1.4.0)53. Differential abundance analysis between sample groups was performed using DESeq2 (v1.24.0)54.
References
Sinka, M. E. et al. The dominant Anopheles vectors of human malaria in Africa, Europe and the Middle East: Occurrence data, distribution maps and bionomic précis. Parasites Vectors 2010(3), 1–34 (2010).
Dahan-Moss, Y. et al. Member species of the Anopheles gambiae complex can be misidentified as Anopheles leesoni. Malar. J. 2020(19), 1–9 (2020).
Dandalo, L. C. et al. Population Dynamics and Plasmodium falciparum (Haemosporida: Plasmodiidae) Infectivity Rates for the Malaria Vector Anopheles arabiensis (Diptera: Culicidae) at Mamfene, KwaZulu-Natal, South Africa. J. Med. Entomol. 54, 1758–1766 (2017).
Gillies, M. T. & De Meillon, B. The Anophelinae of Africa south of the Sahara (Ethiopian Zoogeographical Region). Anophelinae Africa south Sahara (Ethiopian Zoogeographical Reg. (1968).
Killeen, G. F., Govella, N. J., Lwetoijera, D. W. & Okumu, F. O. Most outdoor malaria transmission by behaviourally-resistant Anopheles arabiensis is mediated by mosquitoes that have previously been inside houses. Malar. J. 2016(15), 1–10 (2016).
Mahande, A., Mosha, F., Mahande, J. & Kweka, E. Feeding and resting behaviour of malaria vector, Anopheles arabiensis with reference to zooprophylaxis. Malar. J. 2007(6), 1–6 (2007).
Takken, W. et al. Susceptibility of Anopheles quadriannulatus theobald (Diptera: Culicidae) to Plasmodium falciparum. Trans. R. Soc. Trop. Med. Hyg. 93, 578–580 (1999).
Habtewold, T., Povelones, M., Blagborough, A. M. & Christophides, G. K. Transmission blocking immunity in the malaria non-vector mosquito Anopheles quadriannulatus species A. PLoS Pathog. 4, e1000070 (2008).
Jayakrishnan, L., Sudhikumar, A. V. & Aneesh, E. M. Role of gut inhabitants on vectorial capacity of mosquitoes. J. Vector Borne Dis. 55, 69 (2018).
Coon, K. L., Vogel, K. J., Brown, M. R. & Strand, M. R. Mosquitoes rely on their gut microbiota for development. Mol. Ecol. 23, 2727–2739 (2014).
Akorli, J., Namaali, P. A., Ametsi, G. W., Egyirifa, R. K. & Pels, N. A. P. Generational conservation of composition and diversity of field-acquired midgut microbiota in Anopheles gambiae (sensu lato) during colonization in the laboratory. Parasites Vectors 2019(12), 1–9 (2019).
Wang, Y., Gilbreath, T. M., Kukutla, P., Yan, G. & Xu, J. Dynamic gut microbiome across life history of the malaria mosquito Anopheles gambiae in Kenya. PLoS ONE 6, 1–9 (2011).
Ranasinghe, H. A. K. & Amarasinghe, L. D. Naturally occurring microbiota in dengue vector mosquito breeding habitats and their use as diet organisms by developing larvae in the Kandy district, Sri Lanka. Biomed Res. Int. 2020, 5830604 (2020).
Chen, S. et al. Multiple factors determine the structure of bacterial communities associated with Aedes albopictus under artificial rearing conditions. Front. Microbiol. 5, 605 (2020).
Romoli, O. & Gendrin, M. The tripartite interactions between the mosquito, its microbiota and Plasmodium. Parasit. Vectors 11, 1–8 (2018).
Grogan, C., Bennett, M., Moore, S. & Lampe, D. Novel Asaia bogorensis signal sequences for Plasmodium inhibition in Anopheles stephensi. Front. Microbiol. 12, 1–14 (2021).
Sharma, P. et al. Altered gut microbiota and immunity defines Plasmodium vivax survival in Anopheles stephensi. Front. Immunol. 11, 1–13 (2020).
Kumar, S., Molina-Cruz, A., Gupta, L., Rodrigues, J. & Barillas-Mury, C. A Peroxidase/dual oxidase system modulates midgut epithelial immunity in Anopheles gambiae. Science 327, 1644–1648 (2010).
Galeano-Castañeda, Y., Bascuñán, P., Serre, D. & Correa, M. M. Trans-stadial fate of the gut bacterial microbiota in Anopheles albimanus. Acta Trop. 201, 105204 (2020).
Saab, S. A. et al. The environment and species affect gut bacteria composition in laboratory co-cultured Anopheles gambiae and Aedes albopictus mosquitoes. Sci. Rep. 10, 1–13 (2020).
Silva, B. E., Matsena Zingoni, Z., Koekemoer, L. L. & Dahan-Moss, Y. L. Microbiota identified from preserved Anopheles. Malar. J. 20, 1–18 (2021).
Omoke, D. et al. Western Kenyan Anopheles gambiae showing intense permethrin resistance harbour distinct microbiota. Malar. J. 2021(20), 1–14 (2021).
Dada, N., Sheth, M., Liebman, K., Pinto, J. & Lenhart, A. Whole metagenome sequencing reveals links between mosquito microbiota and insecticide resistance in malaria vectors. Sci. Rep. 2018(8), 1–13 (2018).
Mancini, M. V. et al. Estimating bacteria diversity in different organs of nine species of mosquito by next generation sequencing. BMC Microbiol. 18, 1–10 (2018).
Dada, N. et al. Pyrethroid exposure alters internal and cuticle surface bacterial communities in Anopheles albimanus. ISME J. 2019(13), 2447–2464 (2019).
Oliver, S. V. & Brooke, B. D. The effect of larval nutritional deprivation on the life history and DDT resistance phenotype in laboratory strains of the malaria vector Anopheles arabiensis. Malar. J. 2013(12), 1–9 (2013).
Minard, G., Mavingui, P. & Moro, C. V. Diversity and function of bacterial microbiota in the mosquito holobiont. Parasit. Vectors 6, 1–12 (2013).
Muturi, E. J., Dunlap, C., Ramirez, J. L., Rooney, A. P. & Kim, C. H. Host blood-meal source has a strong impact on gut microbiota of Aedes aegypti. FEMS Microbiol. Ecol. 95, fiy213 (2018).
Coon, K. L. et al. Bacteria-mediated hypoxia functions as a signal for mosquito development. Proc. Natl. Acad. Sci. USA 114, E5362–E5369 (2017).
Norris, L. C., Fornadel, C. M., Hung, W. C., Pineda, F. J. & Norris, D. E. Frequency of multiple blood meals taken in a single gonotrophic cycle by Anopheles arabiensis mosquitoes in Macha, Zambia. Am. J. Trop. Med. Hyg. 83, 33–37 (2010).
Shaw, W. R. et al. Multiple blood feeding in mosquitoes shortens the Plasmodium falciparum incubation period and increases malaria transmission potential. PLOS Pathog. 16, e1009131 (2020).
Segata, N. et al. The reproductive tracts of two malaria vectors are populated by a core microbiome and by gender-and swarm-enriched microbial biomarkers OPEN. Nat. Publ. Gr. https://doi.org/10.1038/srep24207 (2016).
Gimonneau, G. et al. Composition of Anopheles coluzzii and Anopheles gambiae microbiota from larval to adult stages. Infect. Genet. Evol. 28, 715–724 (2014).
Tchioffo, M. T. et al. Dynamics of Bacterial community composition in the malaria mosquito’s epithelia. Front. Microbiol. 6, 1500 (2016).
Boissière, A. et al. Midgut microbiota of the malaria mosquito vector Anopheles gambiae and interactions with Plasmodium falciparum infection. PLoS Pathog. 8, 1–12 (2012).
Barnard, K., Jeanrenaud, A. C. S. N., Brooke, B. D. & Oliver, S. V. The contribution of gut bacteria to insecticide resistance and the life histories of the major malaria vector Anopheles arabiensis (Diptera: Culicidae). Sci. Rep. 2019(9), 1–11 (2019).
Dada, N. et al. Considerations for mosquito microbiome research from the Mosquito Microbiome Consortium. Microbiome 9, 1–16 (2021).
Habtewold, T., Groom, Z. & Christophides, G. K. Immune resistance and tolerance strategies in malaria vector and non-vector mosquitoes. Parasites Vectors 2017(10), 1–12 (2017).
Bahia, A. C. et al. Exploring Anopheles gut bacteria for Plasmodium blocking activity. Environ. Microbiol. 16, 2980–2994 (2014).
Cirimotich, C. M. et al. Natural microbe-mediated refractoriness to Plasmodium infection in Anopheles gambiae. Science 332, 855–858 (2011).
Bai, L. et al. A gut symbiotic bacterium Serratia marcescens renders mosquito resistance to Plasmodium infection through activation of mosquito immune responses. Front Microbiol. 10, 1580 (2019).
Koczura, R., Mokracka, J. & Makowska, N. Environmental isolate of Rahnella aquatilis harbors class 1 integron. Curr. Microbiol. 72, 64–67 (2016).
Oliver, S. V. & Brooke, B. D. The effect of elevated temperatures on the life history and insecticide resistance phenotype of the major malaria vector Anopheles arabiensis (Diptera: Culicidae). Malar. J. 2017(16), 1–13 (2017).
Oliver, S. V. & Brooke, B. D. The role of oxidative stress in the longevity and insecticide resistance phenotype of the major malaria vectors Anopheles arabiensis and Anopheles funestus. PLoS ONE 11, e0151049 (2016).
Hunt, R. H., Brooke, B. D., Pillay, C., Koekemoer, L. L. & Coetzee, M. Laboratory selection for and characteristics of pyrethroid resistance in the malaria vector Anopheles funestus. Med. Vet. Entomol. 19, 271–275 (2005).
Seng, P. et al. Ongoing revolution in bacteriology: Routine identification of bacteria by matrix-assisted laser desorption ionization time-of-flight mass spectrometry. Clin. Infect. Dis. 49, 543–551 (2009).
Klindworth, A. et al. Evaluation of general 16S ribosomal RNA gene PCR primers for classical and next-generation sequencing-based diversity studies. Nucleic Acids Res. 41, e1–e1 (2013).
Wood, D. E., Lu, J. & Langmead, B. Improved metagenomic analysis with Kraken 2. Genome Biol. 2019(20), 1–13 (2019).
Ondov, B. D., Bergman, N. H. & Phillippy, A. M. Interactive metagenomic visualization in a Web browser. BMC Bioinform. 12, 1–10 (2011).
Callahan, B. J. et al. DADA2: High-resolution sample inference from Illumina amplicon data. Nat. Methods 2016(13), 581–583 (2016).
McMurdie, P. J. & Holmes, S. phyloseq: An R package for reproducible interactive analysis and graphics of microbiome census data. PLoS ONE 8, e61217 (2013).
Andersen, K. S., Kirkegaard, R. H., Karst, S. M. & Albertsen, M. ampvis2: An R package to analyse and visualise 16S rRNA amplicon data. BioRxiv https://doi.org/10.1101/299537 (2018).
Conway, J. R., Lex, A. & Gehlenborg, N. UpSetR: An R Package for the Visualization of Intersecting Sets and Their Properties https://doi.org/10.1093/bioinformatics/btx364.
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014(15), 1–21 (2014).
Acknowledgements
The authors acknowledge the assistance of the staff of the Centre for Hospital and Acquired infections at the National Institute for Communicable Diseases for their assistance with the MALDI-TOF MS. Dr. Rodney Hull is thanked for his helpful comments of the manuscript.
Funding
This work was supported by the National Research Foundation of South Africa Competitive Support for Unrated Researchers [Grant Number: SRUG190313423259]; and the National Health Laboratory Services Research Trust Development Grant [Grant Number: 2017-2DEV-08-SOL2] to SVO.
Author information
Authors and Affiliations
Contributions
A.S.: performed experiments, analysed data, produced initial draft of manuscript. Z.T.H.K.: performed NGS sequencing runs, manuscript editing. S.K.: bioinformatics analysis, manuscript editing. M.A.: bioinformatics analysis, project administration, manuscript editing. A.I.: manuscript editing. S.V.O.: conceived project, obtained funding, project administration, produced final draft of the manuscript. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Singh, A., Allam, M., Kwenda, S. et al. The dynamic gut microbiota of zoophilic members of the Anopheles gambiae complex (Diptera: Culicidae). Sci Rep 12, 1495 (2022). https://doi.org/10.1038/s41598-022-05437-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-05437-y
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.