Abstract
Preserved ratio impaired spirometry (PRISm) is associated with an increased mortality rate; however, its characteristics have not been clearly identified in Japan. This cross-sectional study of community-dwelling older adults compared physical function between people with PRISm and those with no respiratory issues, from 2014 to 2019. We collected demographic data through interviews and measured respiratory and physical functions. We included 668 older adults (male, 23.5%; mean age, 72.8 ± 5.6 years); the prevalence of PRISm was 12%, while the prevalence of obstruction was 6.9%. Propensity score matching was used to identify control subjects with normal spirometry (n = 80) while minimizing the effects of confounders during comparisons with the PRISm population (n = 80). Compared with community-dwelling older adults with normal lung capacity, older adults with PRISm had a lower forced vital capacity (%FVC; 68.7 ± 9.1% vs. 92.5 ± 12.7%, p < 0.001), lower core muscle endurance (sit-up test: 6.7 ± 5.8 vs. 8.7 ± 6.0, p = 0.032), and a longer one-leg stance duration (52.4 ± 41.1 s vs. 36.4 ± 34.1 s, p = 0.008). In multivariable logistic regression, %FVC and increased one-leg stance were independent predictors of PRISm status. The prevalence of PRISm among community-dwelling elderly Japanese exceeds that of obstructive lung disease and is associated with reduced %FVC and better performance on balance testing.
Similar content being viewed by others
Introduction
Preserved ratio impaired spirometry (PRISm), generally known as the ‘restrictive ventilatory defect’, refers to a condition in which spirometry shows a ratio of forced expiratory volume in the first second (FEV1) to forced vital capacity (FVC) of ≥ 70% and a predicted FEV1 (%FEV1) of < 80%1. The prevalence of PRISm has been reported to be between 12 and 24%1,2,3,4. Its association with respiratory symptoms3,5,6,7,8,9 and increased mortality2,9,10,11,12 means that PRISm cannot be ignored in clinical settings13,14. Risk factors include age, female sex, a history of smoking, diabetes mellitus, and obesity5,7,15,16. Moreover, increased dyspnoea, reduced 6-min walk distance1, and reduced health-related quality of life17,18 have been reported in individuals with PRISm.
The Genetic Epidemiology of COPD (COPDGene) study and other reports discussing PRISm1,2,3,8,18 have explained the condition using medical data4,6,9. However, few studies have focused on community-dwelling individuals7. In Japan, awareness of PRISm remains low, and its prevalence is unknown. To date, no community-based studies have been conducted.
Past research has reported the prevalence of chronic obstructive pulmonary disease (COPD) in community-dwelling persons19,20, as well as physical function and cognitive function characteristics of community-dwelling individuals suspected of having COPD21,22,23,24,25. Physical characteristics of PRISm, such as reduced lung capacity and being overweight5,7,16, have also been elucidated but, aside from a reduced 6-min walk distance1, physical function characteristics remain unexplored. Previous studies have found that individuals with PRISm have a shorter 6-min walk distance; therefore, we hypothesised that PRISm would also be associated with a decline in physical function, such as reduced limb skeletal muscle strength and flexibility. If patients with PRISm had poorer physical function than individuals with normal respiratory function, it would be possible to use physical function parameters and physical characteristics as early warning signs of PRISm.
Therefore, in this study, we aimed to clarify the prevalence of hidden PRISm in Japan and to investigate differences in terms of attributes and physical function between community-dwelling older adults with normal respiratory function and those with PRISm.
Results
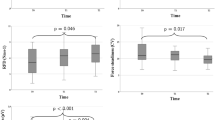
A total of 668 community-dwelling older adults were included in this study (Fig. 1). The mean age of the participants was 72.8 ± 5.6 years. There were 157 males (23.5%) and 511 females (76.5%). The PRISm group comprised 80 participants (12.0%), the normal respiratory function group comprised 542 participants (81.1%), and there were 46 participants (6.9%) with obstructive ventilation. The study participants’ attributes are shown in Tables 1 and 2. Propensity score matching was used to eliminate confounding bias in relation to community-dwelling older adults with PRISm and those with normal respiratory function, and the two groups were compared. The results of the comparison of attributes between the two groups are shown in Table 3, and physical function measurements are shown in Table 4. Significant between-group differences were found for FVC (p < 0.001), %FVC (p < 0.001), FEV (p < 0.001), %FEV (p < 0.001), FEV/FVC (p = 0.041), the sit-up test (p = 0.032), and the one-leg stance test (p = 0.008). No significant differences were found in terms of diabetes mellitus, respiratory disease, and any of the other items.
Subsequently, a logistic regression analysis was performed with the PRISm group as the dependent variable and with hypertension, respiratory disease, other diseases, %FVC, knee extension strength, sit-up test, and one-leg stance test as independent variables (φ, > 0.1; d, > 0.2). The results showed significant relationships for %FVC (p < 0.001) and the one-leg stance test (p = 0.004; Table 5). The receiver operating characteristic (ROC) analysis, which was performed to determine the %FVC cut-off value, showed an area under the curve (AUC) of 0.961. The %FVC cut-off value based on the Youden index was 83.6% (sensitivity, 0.988; specificity, 0.800; Table 6).
Discussion
This study aimed to clarify the prevalence of hidden PRISm in Japan and to explore differences in attributes and physical function between community-dwelling older adults with normal respiratory function and those with PRISm. The key findings, which we consider novel and important, are as follows: the prevalence of PRISm among community-dwelling older adults in Japan was 12.0%, older adults with PRISm had significantly decreased sit-up test times (used to determine physical function), and the %FVC cut-off value for predicting PRISm was 83.6%.
Previous studies have reported prevalence rates for PRISm as between 12 and 24%1,2,3,4. In this study, the prevalence rate was 12.0%, which was similar to that reported in some other countries. However, the community-dwelling older adults who participated in this study were participants in a health-support project sponsored by the local municipality; therefore, they were likely to have had a relatively high level of health awareness. Smoking rates and, therefore, PRISm prevalence, in Japan, may well have been higher if a larger sample (including those with low health awareness) had been surveyed, which needs to be considered in further studies in Japan. In this study, the percentage of patients with spirometric obstruction was 6.9%, with a higher percentage found in the PRISm group. We found a greater prevalence of PRISm compared with spirometric obstruction among community-dwelling older adults, which was a novel finding. Additionally, 13 participants had respiratory disease, none of whom were in the PRISm group. Based on these results, it would appear that the presence of respiratory disease is not a risk factor for PRISm.
After comparing the PRISm group with the normal respiratory function group, significant between-group differences for each indicator of respiratory function were found and, in terms of physical function, the PRISm group showed a significant decrease in sit-up test times. Wan et al.1 found that the 6-min walk distance drops significantly for individuals with PRISm. As such, we hypothesised that if individuals with PRISm had a shorter 6-min walk distance, a decline in physical function in relation to limb skeletal muscle strength and core muscle strength would also likely be observed. We identified no significant differences in indicators of limb skeletal muscle power, such as grip strength, or in performance, such as walking speed; however, as noted, the PRISm group showed a significant decrease in sit-up test times. The sit-up test is also considered useful for assessing core muscle endurance26. In this study, we were unable to undertake a 6-min walk test due to setting issues. However, this significant decrease in sit-up test times (an assessment of core muscle endurance) suggested that those with PRISm had reduced endurance. The PRISm group had significantly better one-leg stance test results. Participants in Bohannon et al.’s study27 undertook a one-leg stance test five times and the measurements were recorded. They found that the mean stance time was 14.2 ± 9.3 s for participants in their 70 s and 22.5 ± 8.6 s for those in their 60 s. Although the one-leg stance test results obtained in our study were higher than those reported by Bohannon et al., we do not consider the difference to be clinically important. These findings suggest that individuals with PRISm have reduced overall stamina (6-min walk distance) and core stamina but are unlikely to show reduced limb muscle strength or balance. Wan et al.2 found that PRISm was associated with an increased mortality rate and noted that 22.2% of the participants in their study transitioned to a diagnosis of COPD (GOLD 0), and that 25.1% had progressed to COPD (GOLD 1–4) after 5 years. Horie et al.21 found that community-dwelling older adults who exhibited obstructive ventilatory defects but who had not been diagnosed with COPD did not have reduced physical function compared with a group of older adults with normal respiratory function. Therefore, similar to COPD, PRISm in community-dwelling older adults is difficult to predict using physical function tests alone, and the active use of spirometry is essential28,29.
Previous research has identified significant differences between participants with PRISm and those with normal respiratory function regarding respiratory function and the presence or absence of a smoking history1,5,7,15,16. Our study findings showed that there were many older adults who smoked among those with PRISm (Table 1). Our results are consistent with those of previous studies and show that Japanese individuals follow a similar trend in this regard. Furthermore, cumulative exposure to tobacco smoke has been associated with an increased risk of death2. Although previous studies have found associations between PRISm and older age, female sex, body mass index (BMI), and the presence or absence of diabetes mellitus, we did not find any significant differences among these risk factors. Jones et al.’s study of the effects of BMI on lung volume30 reported that obesity alone did not necessarily reduce lung capacity. Our study results showed no significant difference in relation to BMI, possibly because only a small number of study participants were obese.
In this study, we investigated factors likely to be associated with PRISm, using logistic regression analysis. We found that %FVC and the one-leg stance test results were associated with PRISm. Based on these results, we calculated the %FVC cut-off value using ROC analysis. The %FVC cut-off value for predicting PRISm was 83.2%, with an AUC of 0.961, indicating very high accuracy. A sensitivity of 98.8% and a specificity of 80.0% indicated high predictive power. Although we focussed on airway obstruction, we consider that the effect of reduced FVC was also an important risk factor. These findings support the importance of spirometry in PRISm detection.
Our study findings, similar to those of previous studies, indicated that older adults with PRISm had reduced endurance. However, a lack of significant between-group differences in physical function, except for trunk endurance, is an important novel finding. Regan et al.31 reported that chronic smokers may exhibit clinical and physiological impairments due to smoking, even if they do not meet the diagnostic criteria for COPD. They reported that spirometry alone is likely to underestimate respiratory disease. As such, surveying physical function in addition to respiratory function and comparing it between individuals with PRISm and those with normal respiratory function is likely to be significant both clinically and in terms of social welfare generally. Moreover, conditions such as COPD and lower back pain have been identified as primary contributors to years spent living with disability, due to disease and injury sequelae32. Thus, it may be possible to extend healthy lifespans through early detection of PRISm, which may otherwise advance to COPD in some people, followed with appropriate medical management and health-related support.
Limitations
This study was limited in terms of its small sample size for the logistic regression analysis, the uneven distribution of males and females, and its cross-sectional research design. Furthermore, as we recruited community-dwelling older adults, many participants were those involved in a community socialising program for older adults who did not need support. Therefore, we cannot exclude the possibility that our study participants were individuals with relatively high health awareness. Future studies are needed involving a sample of community-dwelling individuals who either do not or cannot participate in such programs. We did not use the 6-min walk test due to limited space in the study setting; therefore, a reliable endurance assessment could not be undertaken. Endurance assessments should be conducted in future studies, in the same way as in previous studies. However, it was useful to include patient-reported outcomes such as running or jogging durations.
Conclusions
This study is the first to report the prevalence of PRISm in community-dwelling individuals in Japan (12.0%). A significant difference in core muscle endurance was found between participants with PRISm and those with normal respiratory function; however, we found no significant between-group differences in terms of other physical functions. As expected, our findings confirmed that restrictive ventilatory defect is a major issue. Our findings may facilitate health promotion planning and clinical goal setting, and serve to reinforce the importance of spirometry.
Methods
Participants
This study involved a cross-sectional survey of community-dwelling older adults, with annual measurements taken every September from 2014 to September 2019. Participants were recruited using flyers distributed in Yasu City, Shiga Prefecture, from June 2014 to August 2019. The flyers specified that no form of compensation would be offered for participation. Inclusion criteria comprised the following: (i) age, ≥ 60 years old; (ii) a mini-mental state examination (MMSE) score of < 24, with no apparent warning signs of cognitive impairment; and (iii) no spirometry performed after registration. Exclusion criteria comprised participants who were unable to understand the instructions for physical or cognitive tests or who were unable to complete all of the tests.
Participants ranged in age from 47 to 90 years at enrolment and included individuals with and without a history of smoking. This study was undertaken in accordance with the Declaration of Helsinki and was approved by the Ethics Committee of Kyoto Tachibana University (approval no. 19–5). Written informed consent was obtained from all participants prior to participation in the study.
After collecting data concerning age, height, and weight at a group physical assessment, the study participants completed the MMSE. Data concerning prevalence rates for hypertension, dyslipidemia, osteoporosis, orthopaedic diseases, diabetes mellitus, cardiovascular diseases, respiratory diseases, kidney diseases, and cancer were also collected from the participants. Smoking history was self-reported and included whether the participant was a current or former smoker. Finally, data from 614 study participants were analysed (Fig. 1).
Definitions
PRISm was defined as a post-bronchodilator FEV1 < 80% of the predicted FEV1 and a ratio of FEV1/FVC ≥ 70%.
Spirometry
For spirometry, the Autospiro AS-507 (Minato Medical Science, Osaka, Japan) was used to measure the flow-volume curve twice, according to the spirometry guidelines of the American Thoracic Society/European Respiratory Society33. Measurements included FVC, %FVC, FEV1, %FEV1, and FEV1/FVC.
Body composition
To assess body composition, the InBody430 (InBody Japan Inc., Tokyo, Japan) was used to measure BMI, skeletal muscle mass, skeletal muscle mass index (SMI), body fat, and body fat percentage using the impedance method. The SMI was calculated through dividing skeletal muscle mass by height squared (kg/m2)34.
Physical functions
Skeletal muscle strength
Grip strength was assessed using a digital hand dynamometer (T.K.K. 5401 Grip-D, Takei Scientific Instruments Co., Ltd., Niigata, Japan). This assessment tool has been shown to be both valid and reliable35. Participants were instructed to let their arms hang freely beside their bodies and stand upright so that the dynamometer did not touch them. Both sides were measured twice, and the maximum value (kg) was used.
Knee extension strength was evaluated using a handheld dynamometer (μTasF-1, Anima Corporation, Tokyo, Japan), as described in detail by Bohannon36. This assessment tool has been shown to be valid and reliable37,38. The participants were seated in a chair with the knee bent at a 90° angle, and the sensory pad was fixed to the distal end of the leg with a band. Two measurements were performed for each leg. The maximum measured value (kgf) was divided by body weight (kg) to calculate the body weight-normalised percentage (%), which was then used to represent knee extension strength.
Toe grip strength was assessed using a toe grip dynamometer (T.K.K. 3362; Takei Scientific Instruments Co., Ltd., Niigata, Japan). This assessment tool has been shown to be valid and reliable39,40. Measurements were taken with the knee bent at a 90º angle while the participant was seated in a chair in a neutral ankle position, neither dorsi- nor plantarflexed, nor internally or externally rotated41. Both feet were measured twice, and the maximum value (kg) was used.
A sit-up performance test was conducted using the method described by Abe et al.42. This assessment tool has been used to assess core muscle strength and endurance26. The participants were positioned on a mat in a supine position with the knees bent at an angle of approximately 90º. The participants performed a complete sit-up, reaching an upright position with the elbows touching the thighs and then returned to the supine position. The number of repetitions completed in 30 s was recorded.
30-s stand test (CS-30)
The CS-30 was performed using the method described by Jones et al.43. The participants were seated in a chair (height, 40 cm) with their arms crossed over the chest. Rising to a standing position with the knees completely extended, then returning to a seated position was considered as one cycle. Participants were instructed to complete as many cycles as possible within 30 s after the starting signal had been given. The number of sit-stand-sit cycles completed in 30 s was counted.
Sit-and-reach test
The sit-and-reach test was performed using a digital sit-and-reach test box (T.K.K. 5412, Takei Scientific Instruments Co., Ltd., Niigata, Japan). This assessment tool has been shown to be valid44. The distance covered by the fingertips while reaching was measured. Measurements were conducted twice, and the maximum observed distance (cm) was used.
One-leg stance test
The one-leg stance test time was assessed using a digital stopwatch. The length of time the participant could maintain a one-leg stance with their eyes open was measured39. Two measurements were taken for each leg, and the maximum length of time was used for further analyses. The maximum time for the one-leg stance test was restricted to 120 s.
Fastest gait speed
The fastest gait speed was assessed using a digital stopwatch on an 11-m walkway divided into two 3-m practice sections flanking a 5-m measurement section. At the start of the measurement, the participants were verbally instructed to walk ‘as quickly as possible’, and their walking time was measured. The gait speed (m/s) was calculated using the recorded time (s).
Timed up-and-go test
The timed up-and-go (TUG) test was performed while walking ‘as quickly as possible’, as explained by Shumway-Cook et al.45. At the start signal, the participants rose from a seated position in an armless chair (height, 40 cm), walked around a pole 3 m in front of them, and returned to sit in their original chair. This process was timed using a digital stopwatch. Measurements were taken twice, and the shortest time was used.
Cognitive function
Cognitive function was assessed using the MMSE. The MMSE is a widely used general test for assessing cognitive function. The examination comprises 11 items with a maximum possible score of 30 points46. A score of ≤ 23 points indicates cognitive impairment47,48.
Statistical analysis
Participants with FEV1/FVC ≥ 70% and a %FEV1 < 80% were assigned to the PRISm group, whereas those with FEV1/FVC ≥ 70% and %FEV1 ≥ 80% were classified into the normal respiratory function group. Propensity score matching was used to identify control subjects with normal spirometry (n = 80) while minimizing the effects of confounders during comparisons with the PRISm population (n = 80).
Covariates for propensity score matching were determined to be age, sex, smoking history, and SMI. Participant attributes and physical functions were then compared between the groups. Student’s t-tests were performed for continuous variables, and chi-square tests were performed for nominal variables. The φ and d values were calculated as measures of the effect size. Logistic regression analysis was performed using the PRISm group as the dependent variable. Previous studies have found that PRISm was associated with age, sex, BMI, diabetes mellitus, smoking history, and %FVC5,7,15,16. Therefore, we created a logistic regression model that incorporated indicators with an φ value of ≥ 0.1 and a ‘d’ value of ≥ 0.2 as independent variables, after excluding the covariates in the propensity score matching. Lastly, a ROC curve was drawn using the results of the logistic regression analysis and indicator cut-off values determined using the Youden Index. All tests were two-tailed and used a statistical significance level of 5%. Statistical analyses were conducted using SPSS version 24 software.
References
Wan, E. S. et al. Epidemiology, genetics, and subtyping of preserved ratio impaired spirometry (PRISm) in COPDGene. Respir. Res. 15, 89. https://doi.org/10.1186/s12931-014-0089-y (2014).
Wan, E. S. et al. Longitudinal phenotypes and mortality in preserved ratio impaired spirometry in the COPDGene study. Am. J. Respir. Crit. Care Med. 198, 1397–1405. https://doi.org/10.1164/rccm.201804-0663OC (2018).
Fortis, S. et al. Low FVC/TLC in preserved ratio impaired spirometry (PRISm) is associated with features of and progression to obstructive lung disease. Sci. Rep. 10, 5169. https://doi.org/10.1038/s41598-020-61932-0 (2020).
Schwartz, A. et al. Preserved ratio impaired spirometry in a spirometry database. Respir. Care 66, 58–65. https://doi.org/10.4187/respcare.07712 (2021).
Guerra, S. et al. Health-related quality of life and risk factors associated with spirometric restriction. Eur. Respir. J. https://doi.org/10.1183/13993003.02096-2016 (2017).
Wei, X. et al. Imaging features of chronic bronchitis with preserved ratio and impaired spirometry (PRISm). Lung 196, 649–658. https://doi.org/10.1007/s00408-018-0162-2 (2018).
Park, H. J. et al. Significant predictors of medically diagnosed chronic obstructive pulmonary disease in patients with preserved ratio impaired spirometry: A 3-year cohort study. Respir. Res. 19, 185. https://doi.org/10.1186/s12931-018-0896-7 (2018).
Diaz, A. A. et al. Disease severity dependence of the longitudinal association between CT lung density and lung function in smokers. Chest 153, 638–645. https://doi.org/10.1016/j.chest.2017.10.012 (2018).
Wijnant, S. R. A. et al. Trajectory and mortality of preserved ratio impaired spirometry: The Rotterdam Study. Eur. Respir. J. https://doi.org/10.1183/13993003.01217-2019 (2020).
Mannino, D. M., Buist, A. S., Petty, T. L., Enright, P. L. & Redd, S. C. Lung function and mortality in the United States: Data from the First National Health and Nutrition Examination Survey follow up study. Thorax 58, 388–393 (2003).
Mannino, D. M., Ford, E. S. & Redd, S. C. Obstructive and restrictive lung disease and functional limitation: Data from the Third National Health and Nutrition Examination. J. Intern. Med. 254, 540–547 (2003).
Guerra, S. et al. Morbidity and mortality associated with the restrictive spirometric pattern: A longitudinal study. Thorax 65, 499–504. https://doi.org/10.1136/thx.2009.126052 (2010).
Washko, G. R. et al. Lung volumes and emphysema in smokers with interstitial lung abnormalities. N. Engl. J. Med. 364, 897–906. https://doi.org/10.1056/NEJMoa1007285 (2011).
Fabbri, L. M. et al. COPD guidelines: The important thing is not to stop questioning. Am. J. Respir. Crit. Care Med. 176, 527–528. https://doi.org/10.1164/rccm.200706-854ED (2007).
Mannino, D. M. et al. Restricted spirometry in the Burden of Lung Disease Study. Int. J. Tuberc. Lung Dis. 16, 1405–1411. https://doi.org/10.5588/ijtld.12.0054 (2012).
Kurth, L. & Hnizdo, E. Change in prevalence of restrictive lung impairment in the U.S. population and associated risk factors: The National Health and Nutrition Examination Survey (NHANES) 1988–1994 and 2007–2010. Multidiscip. Respir. Med. 10, 7. https://doi.org/10.1186/s40248-015-0003-6 (2015).
Charbonnier, J. P. et al. Airway wall thickening on CT: Relation to smoking status and severity of COPD. Respir. Med. 146, 36–41. https://doi.org/10.1016/j.rmed.2018.11.014 (2019).
Parekh, T. M. et al. Factors influencing decline in quality of life in smokers without airflow obstruction: The COPDGene Study. Respir. Med. 161, 105820. https://doi.org/10.1016/j.rmed.2019.105820 (2020).
Buist, A. S. et al. International variation in the prevalence of COPD (the BOLD Study): A population-based prevalence study. Lancet 370, 741–750. https://doi.org/10.1016/S0140-6736(07)61377-4 (2007).
Fukuchi, Y. et al. COPD in Japan: The Nippon COPD Epidemiology study. Respirology 9, 458–465. https://doi.org/10.1111/j.1440-1843.2004.00637.x (2004).
Horie, J. et al. Factors that delay COPD detection in the general elderly population. Respir. Care 56, 1143–1150. https://doi.org/10.4187/respcare.01109 (2011).
Gosker, H. R. et al. Skeletal muscle fibre-type shifting and metabolic profile in patients with chronic obstructive pulmonary disease. Eur. Respir. J. 19, 617–625. https://doi.org/10.1183/09031936.02.00762001 (2002).
Hernandes, N. A. et al. Reproducibility of 6-minute walking test in patients with COPD. Eur. Respir. J. 38, 261–267. https://doi.org/10.1183/09031936.00142010 (2011).
Seymour, J. M. et al. The prevalence of quadriceps weakness in COPD and the relationship with disease severity. Eur. Respir. J. 36, 81–88. https://doi.org/10.1183/09031936.00104909 (2010).
Spruit, M. A. et al. An official American Thoracic Society/European Respiratory Society statement: Key concepts and advances in pulmonary rehabilitation. Am. J. Respir. Crit. Care Med. 188, e13–e64. https://doi.org/10.1164/rccm.201309-1634ST (2013).
Bianco, A. et al. The sit up test to exhaustion as a test for muscular endurance evaluation. Springerplus 4, 1–8. https://doi.org/10.1186/s40064-015-1023-6 (2015).
Bohannon, R. W., Larkin, P. A., Cook, A. C., Gear, J. & Singer, J. Decrease in timed balance test scores with aging. Phys. Ther. 64, 1067–1070. https://doi.org/10.1093/ptj/64.7.1067 (1984).
Heo, I. R., Kim, H. C. & Kim, T. H. Health-related quality of life and related factors in persons with preserved ratio impaired spirometry: Data from the Korea National Health and Nutrition Examination Survey. Medicina (Kaunas) 57, 4. https://doi.org/10.3390/medicina57010004 (2020).
Young, K. A. et al. Pulmonary subtypes exhibit differential global initiative for chronic obstructive lung disease spirometry stage progression: The COPDGene(R) Study. Chronic Obstr. Pulm. Dis. 6, 414–429. https://doi.org/10.15326/jcopdf.6.5.2019.0155 (2019).
Jones, R. L. & Nzekwu, M. M. The effects of body mass index on lung volumes. Chest 130, 827–833. https://doi.org/10.1378/chest.130.3.827 (2006).
Regan, E. A. et al. Clinical and radiologic disease in smokers with normal spirometry. JAMA Intern. Med. 175, 1539–1549. https://doi.org/10.1001/jamainternmed.2015.2735 (2015).
Vos, T. et al. Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990–2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet 380, 2163–2196 (2012).
Miller, M. R. et al. Standardisation of spirometry. Eur. Respir. J. 26, 319–338. https://doi.org/10.1183/09031936.05.00034805 (2005).
Nonaka, K. et al. Physical characteristics vary according to body mass index in Japanese community-dwelling elderly women. Geriatrics (Basel) https://doi.org/10.3390/geriatrics3040087 (2018).
Yu, R., Ong, S., Cheung, O., Leung, J. & Woo, J. Reference values of grip strength, prevalence of low grip strength, and factors affecting grip strength values in Chinese adults. J. Am. Med. Dir. Assoc. 18, 551. https://doi.org/10.1016/j.jamda.2017.03.006 (2017).
Bohannon, R. W. Test-retest reliability of hand-held dynamometry during a single session of strength assessment. Phys. Ther. 66, 206–209. https://doi.org/10.1093/ptj/66.2.206 (1986).
Martin, H. J. et al. Is hand-held dynamometry useful for the measurement of quadriceps strength in older people? A comparison with the gold standard Bodex dynamometry. Gerontology 52, 154–159. https://doi.org/10.1159/000091824 (2006).
Katoh, M., Isozaki, K., Sakanoue, N. & Miyahara, T. Reliability of isometric knee extension muscle strength measurement using a hand-held dynamometer with a belt: A study of test-retest reliability in healthy elderly subjects. J. Phys. Ther. Sci. 22, 359–363 (2010).
Uritani, D., Fukumoto, T. & Matsumoto, D. Intrarater and interrater reliabilitiesfor a toe grip dynamometer. J. Phys. Ther. Sci. 24, 639–643 (2012).
Mickle, K. J., Munro, B. J., Lord, S. R., Menz, H. B. & Steele, J. R. ISB Clinical Biomechanics Award 2009: Toe weakness and deformity increase the risk of falls in older people. Clin. Biomech. (Bristol, Avon) 24, 787–791. https://doi.org/10.1016/j.clinbiomech.2009.08.011 (2009).
Soma, M. et al. Examinations of factors influencing toe grip strength. J. Phys. Ther. Sci. 28, 3131–3135. https://doi.org/10.1589/jpts.28.3131 (2016).
Abe, T. et al. Associations of sit-up ability with sarcopenia classification measures in Japanese older women. Interv. Med. Appl. Sci. 8, 152–157. https://doi.org/10.1556/1646.8.2016.4.7 (2016).
Jones, C. J., Rikli, R. E. & Beam, W. C. A 30-s chair-stand test as a measure of lower body strength in community-residing older adults. Res. Q. Exerc. Sport 70, 113–119. https://doi.org/10.1080/02701367.1999.10608028 (1999).
Macrae, P. G., Lacourse, M. & Moldavon, R. Physical performance measures that predict faller status in community-dwelling older adults. J. Orthop. Sports Phys. Ther. 16, 123–128. https://doi.org/10.2519/jospt.1992.16.3.123 (1992).
Shumway-Cook, A., Brauer, S. & Woollacott, M. Predicting the probability for falls in community-dwelling older adults using the timed up & go test. Phys. Ther. 80, 896–903 (2000).
Folstein, M. F., Folstein, S. E. & McHugh, P. R. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr. Res. 12, 189–198. https://doi.org/10.1016/0022-3956(75)90026-6 (1975).
Cullen, B. et al. Screening for dementia in an Irish community sample using MMSE: A comparison of norm-adjusted versus fixed cut-points. Int. J. Geriatr. Psychiatry 20, 371–376. https://doi.org/10.1002/gps.1291 (2005).
Tsoi, K. K., Chan, J. Y., Hirai, H. W., Wong, S. Y. & Kwok, T. C. Cognitive tests to detect dementia: A systematic review and meta-analysis. JAMA Intern. Med. 175, 1450–1458. https://doi.org/10.1001/jamainternmed.2015.2152 (2015).
Acknowledgements
We would like to thank all the volunteers who participated in this study. We would also like to thank Editage (www.editage.com) for English language editing.
Funding
This research was funded by the Japanese Physical Therapy Association. Hideki Nakano received funding for this research.
Author information
Authors and Affiliations
Contributions
K.A. and S.M. were responsible for the conception of the study. All authors were involved in investigation. K.A. and S.M. analysed and interpreted the data and revised the manuscript. All authors reviewed and edited the manuscript and approved the submitted final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Anami, K., Murata, S., Nakano, H. et al. Physical performance in relation to preserved ratio impaired spirometry: a cross-sectional study of community-dwelling older Japanese adults. Sci Rep 11, 17411 (2021). https://doi.org/10.1038/s41598-021-96830-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-96830-6
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.