Abstract
The main aim was to assess the short-term effects of active-tDCS (a-tDCS) in the primary motor cortex (anodal stimulation-M1) on sensorimotor variables. These variables included discriminative sensation through the two-point discrimination (2-PD) test, tactile acuity threshold and pressure pain threshold (PPT), and electromyographic (EMG) activity compared with a sham-tDCS (s-tDCS) in healthy individuals. A total of 100 participants were included. Fifty of the participants received the a-tDCS application of 2 mA for 20 min, whereas the remaining fifty received the s-tDCS. The 2-PD and tactile acuity threshold in thenar eminence of the hand and in the dorsal part of the foot and also, PPT and EMG activity during maximal voluntary contraction in the biceps brachii and rectus femoris were assessed before and after the tDCS application. The a-tDCS intervention was not significantly superior to the s-tDCS in any variable. However, significant within-group pre- and post-intervention differences were found in the a-tDCS, such as the tactile acuity threshold in thenar eminence of the hand, with a small effect size (p = .012, d = 0.20) and in the PPT of the rectus femoris, also with a small effect size (p = .001, d = − 0.17). Regarding EMG activity, a trend towards greater activity was observed in participants with a-tDCS compared with s-tDCS, which showed a trend towards decreased EMG activity. In fact, although no differences were found between the groups, within-group differences were statistically significant in the biceps brachii pre- and postintervention (p = .023, d = − 0.16, and p = .002, d = 0.18, respectively), and also in the rectus femoris, only in the a-tDCS, with a small effect size (p = .011, d = − 0.14). This study showed no significant between-group differences in sensorimotor outcomes. A single session of tDCS in isolation appears to produce immediate effects in healthy participants on sensorimotor function; however, these effects were very small.
Similar content being viewed by others
Introduction
Transcranial direct-current stimulation (tDCS) devices have emerged in recent years, providing important benefits in pain management and muscle training that could be used in routine clinical practice1. Moreover, in the past few years, several studies have demonstrated the safety of this technique2,3. Bikson et al.3 had found no risk of brain injury within the following parameters: < 40 min; < 4 milliamperes (mA); and < 7.2 Coulombs. The safety assessment of tDCS has contributed to encouraging further studies.
Currently, the most investigated tDCS effect is cortical excitability modulation. Indeed, tDCS has been shown to be an interesting tool for increasing and also decreasing corticomotor excitability in healthy individuals and in patients with stroke when applied to the primary motor cortex (M1)4.
In addition, most published articles on changes in pain after electric current application on the primary motor cortex in healthy individuals focus on pain tolerance5,6,7. Almost all these studies involved inducing pain by temperature adjustment. Mariano et al.7, for example, had described improvements in pain tolerance with pressure and cold. Angius et al.8 had also observed reduced pain perception during a cold pressure test. However, they did not report any decrease in pain during exercise. Thermoalgesic information and tactile sensitivity travel along distinct afferent pathways to make afference to the central nervous system. The skin contains mechanical pressure receptors such as Ruffini's corpuscles or Merkel's discs, as well as free nerve endings that are activated after exceeding a certain threshold of activation. Both thermoalgesic and tactile information converge in that they are exteroceptive sensitivity. Epicritic tactile sensitivity travels through the dorsal column–medial lemniscus pathway while protopathic tactile sensitivity travels through the anterior spinothalamic cord. Finally, thermoalgesic sensitivity travels through the lateral spinothalamic cord9. In the study of Cobos et al.10 they consider that the modalities of mechanical pain and thermal pain may have different mechanisms due to the different times of onset after neural lesion. This seems to be in agreement with the findings found in the literature due to the difference in effects found depending on the type of pain studied. However, the effects of tDCS on pain modulation are not yet fully understood11.
Given very little has been assessed regarding the effects of tDCS on the pressure pain threshold (PPT) in healthy participants, PPT is an interesting focus of study to enhance our understanding of tDCS effects12,13. tDCS application to the M1 and any resulting changes in sensitivity have not yet been investigated and could provide interesting results.
In addition, tDCS devices have been widely used in recent years in sports-related studies. In the current literature, data have been found both for and against the effect of tDCS on strength. For example, Vargas et al.14 had described an increase in quadriceps strength during maximal voluntary contractions. However, Giboin et al.15 had shown the opposite effect from tDCS. In addition, Hendy & Kidgell16 performed transcranial anodal direct current anodal stimulation in asymptomatic subjects combined with standard strength training with the goal of increasing voluntary dynamic strength in wrist extensors. They found that adding anodal stimulation did not provide any significant improvement in strength enhancement compared to strength training in isolation. However, also Hendy & Kidgell17 found that applying transcranial anodal direct current stimulation during unilateral strength training is able to elicit strength and corticospinal excitability even in the untrained contralateral limb muscle. These contradictions in the literature highlight the need to increase our understanding of the connections between muscle activity and tDCS intervention.
Although Chhatbar et al.18 showed that electrical current flows through the human brain in vivo with this technique, further investigation is needed to establish and define more precisely the optimal parameters and effects of tDCS stimulation in healthy subjects and in cases of pain and muscle activity. However, the literature already presents clearer parameters for some results. Lefaucheur et al.19 recently developed evidence-based guidelines on the therapeutic use of tDCS which set out some parameters. For example, in neuropathic pain due to spinal cord injury, placing the anode at M1 and the cathode at the supraorbital region, 5 sessions at 2 mA for 20 min have been shown to have an effect on pain modulation. Also, placing the anode and cathode in the same area, at 1 mA, 10 sessions of 20 min have been shown to have an impact on pain reduction in fibromyalgia. However, therapeutic relevance of tDCS needs to be further explored.
The hypothesis is that the application of a-tDCS will cause greater effects in sensorimotor function (higher PPT, greater electromyographic activity and better discriminative sensation) compared to the application of s-tDCS. Due to the lack of literature on the effects of tDCS on sensory-discriminative variables as well as contradictory findings regarding the effects on pain modulation and motor variables, the main aim of the present study was to assess the short-term effects of active-tDCS (a-tDCS) on the M1 regarding sensorimotor variables such as discriminative sensation, PPT and electromyographic activity compared with a sham-tDCS (s-tDCS) in healthy individuals.
Methods
Study design
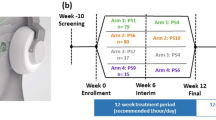
The present study was a quasi-experimental, single-blind, sham-controlled trial. A non-probabilistic sample was used. The participants were assigned to a group depending on their date of registration (Fig. 1). The first 50 were placed in the a-tDCS group and the following 50 were assigned to the s-tDCS group. The study protocol follows the Consolidated Standards of Reporting Trials statement on randomised trials of nonpharmacological treatments20.
All procedures were approved by the Human Research Ethics Committee of the Hospital Infantil Universitario Niño Jesús of Madrid (CI: R-0022/18). This study was registered in the United States Randomized Trials Register on clinicaltrial.gov (trial registry number: NCT04148547 (01/11/2019 Last update).
Participant recruitment
A total of 100 asymptomatic volunteers were recruited from September 2018 to June 2019 from the local community through social media and e-mail. The inclusion criteria were as follows: (a) asymptomatic participants; and (b) men and women aged 18 to 65 years. The exclusion criteria were the following: (a) insomnia; (b) nausea; (c) headache; (d) pregnancy; (e) use of painkillers in the last 24 h; (f) presence of metal inside the head; (g) pacemaker; (h) wound on the area of electrodes’ application; (i) drug consumption; (j) recent application of tDCS; and (k) psychiatric disease impeding understanding of the study. All data were collected at the La Salle University Centre for Advanced Studies. Informed written consent was obtained from all the participants prior to inclusion. All the participants were informed of the study procedures, which were planned under the ethical standards of the Helsinki Declaration.
Blinding
The participants were blinded to their group allocation. Due to the quasi-experimental design of this study, the assessors were not blinded.
Interventions
Active-tDCS
The active tDCS group received 20 min of tDCS application of 2 mA, given no adverse effects had been reported with a 2 mA intensity application3,21. Previous studies have also obtained better results when the tDCS was applied at a 2 mA intensity, increasing the motor evoked potentials amplitude by 40%; this gain had not been reported with a 1 mA stimulation. That outcome suggests a greater motor response of the muscles and axons of the motor descending tracts with a current intensity of 2 mA. The current was gradually increased for 15 s at the beginning and gradually reduced for 15 s at the end.
Sham-tDCS
The control group only received the initial 15 s, during which the current intensity increased and then no current flowed during the rest of the application22. By using this technique and the fact that having had a tDCS application in the past was an exclusion criterion, we ensured that the participant did not know whether they were receiving either the active or sham intervention.
Procedures
After giving their consent to partake in the study, all the participants received a set of questionnaires prior to the intervention. These included a sociodemographic assessment as well as an evaluation of their physical activity.
Evaluators
Each of the two physiotherapists who were in charge of the measurements received a specific 10-h training session on how to use the instruments. Training was performed with some individuals whose measurements were not included in the study.
tDCS intervention
The tDCS technique requires two electrodes. The electrodes used in the tDCS intervention of both groups were symmetrical, with a surface of 50 mm per 70 mm. The anode is the active electrode, and the cathode the referent electrode; they were applied in the following way: anode was placed over M1 and the cathode, was placed over the supra-orbital region. Anodal stimulation requires the anode to be placed where it is intended to facilitate the depolarization of the cells. Through this mode of electrical stimulation, the excitability of the motor region is increased, facilitating neuronal activation23.
The Sooma tDCS (Sooma, Helsinki, Finland), was the model we used, given it proposes a placebo protocol, as described previously. The electrodes were saturated with a saline solution to improve current conductivity. Both groups received the exact same measurements, before and after the tDCS application. All variables were measured before and after tDCS application. After the application of tDCS, the measurements were evaluated immediately upon completion.
Outcome measures
Primary outcomes
Discriminative sensation
Tactile acuity threshold Exacta Von Frey monofilaments (North Coast Medical, Morgan Hill, CA, USA) were used, applying a pressure on the skin until the monofilament bends24. The researcher started the assessment with the thinnest monofilament, then gradually increased the diameter until the participant stated that they felt the tactile stimuli. This test was performed three times for each diameter at the thenar eminence of the hand and the back of the foot of the dominant limb. In the hand, the cutaneous pressure threshold has shown an intraclass correlation coefficient (ICC) ranging from 0.96 to 0.9925.
2-PD test
The Aesthesiometer Baseline device (AliMed, Deham, MA, USA) was used for the 2-PD test in the same areas of tactile acuity above26. Dellon et al. and Mielke et al. had found that 2-PD measurements could be determined with acceptable interobserver reliability and reproducibility in the hand26,27.
The participant lay in a supine position. The dominant upper limb remained in external rotation, while the lower limb stayed in a neutral position. The participant’s eyes were closed to ensure they did not know when the examiner brought the monofilaments or the aesthesiometer to his skin. The dominant upper and lower limb were tested.
PPT
The PPT was assessed with a Wagner FDX algometer (Wagner Instruments, Greenwich, CT, USA)28. The PPT has been defined as the minimal amount of pressure at which a sense of pressure first changes to pain or discomfort. The Wagner FDX algometer consisted of a round rubber disk (area: 1 cm2) attached to a pressure (force) gauge. The gauge displayed values in kilograms but given the surface of the rubber tip was 1 cm2, the readings were expressed in kg/cm2. The pressure was applied at a rate of 0.31 kg/s29. Previous studies have reported an intraexaminer reliability of this procedure ranging from 0.6–0.97, whereas the interexaminer reliability ranged from 0.4 to 0.9830.
PPTs were tested in two different locations. These sites included the biceps brachii and the rectus femoris. All the assessments were performed in a quiet room. In order to familiarise the participants with the test procedure, pressure was first applied to an area that would not be tested during the study (nondominant forearm). A total of 3 consecutive measurements of the PPT at the 4 locations at intervals of 30 s, and the mean of these 3 trials, were used for the data analysis30.
Muscle recruitment through electromyographic activity
The activity of surface electromyography (sEMG) during a maximum voluntary isometric contraction was measured31. The long head of the biceps brachii was tested, to determine the impact on the upper limb. The rectus femoris was also chosen to assess the effect on the lower limb. Both tests were performed on the participant’s dominant limb.
Both isometric contractions (upper and lower limb) were performed after a clear explanation by the physical therapist. The participant was informed of the procedure, the position to adopt during the contraction, and the exact way to contract to avoid compensating with other muscles and to optimise biceps brachii and rectus femoris fibre recruitment.
The muscle activity during a maximal voluntary contraction was contrasted with inactivity during the resting phase. The participants performed 3 isometric contractions for 5 s, with an equal resting time after each contraction. Throughout this protocol, we were able to record the amplitude changes in sEMG.
The assessment of the lower limb followed the same protocol in the rectus femoris. The electrodes were placed while in a supine position, and the procedure was the same. A Free-EMG 1000 surface electromyography (EMG) device (BTS Bioengineering, Quincy, MA, USA) was used for muscle contraction capture. The signals were filtered using a band-pass filter, and a rectification was performed for signal normalisation. In order to quantify the maximum voluntary contraction, the root mean square (RMS) was calculated, using a fixed window of 0.02 s. In previous studies, the RMS of the EMG had an excellent reliability (ICC = 0.86–0.93) in the lower extremity muscles during bridging and quadruped exercises32. The reliability of scapular and arm muscles’ sEMG amplitude values, including the biceps brachii, have been found to be excellent (ICC ≥ 0.75)33 (Fig. 2).
Baseline outcome
Level of physical activity
The level of physical activity was evaluated using the international physical activity questionnaire (IPAQ), which divides the participants into 3 groups according to their level of activity: high, moderate or low/inactive34. This questionnaire has shown acceptable validity for measuring total physical activity; its reliability is reported to be approximately 0.65 (r = 0.76; 95% CI 0.73–0.77)35.
Data analysis
The data analysis was performed using the Statistical Package for the Social Sciences (SPSS 25.00, IBM, Chicago, IL, USA). For the data analysis, we used a confidence interval of 95%, considering all those variables with a p value of less than 0.05 to be statistically significant. Descriptive statistics used to summarise data for continuous variables are presented as mean ± standard deviation with a 95% confidence interval, and the categorical variables are presented as an absolute number or relative frequency percentage. A repeated measures analysis of variance (ANOVA) was conducted to study the effect on the dependent variables of the between-participant factor ‘intervention group’ with 2 levels (a-tDCS and s-tDCS) and the within-participant factor called ‘time’ with 2 levels (pre- and postintervention). Partial eta squared (ƞp2) was calculated as a measure of effect size (strength of association) for each main effect and interaction in the ANOVAs, with 0.01–0.059 representing a small effect, 0.06–0.139 a medium effect and > 0.14 a large effect36. The effect size (Cohen’s d) was calculated for the main variables. According to Cohen’s method, the effect was considered as small (0.20–0.49), medium (0.50–0.79) or large (> 0.80)37.
Results
A total of 100 healthy participants were included and were allocated into 2 groups of 50 participants per group. No adverse events were reported in either group. No statistically significant preintervention between-group differences in demographic data were found (Table 1).
Discriminative sensation
Regarding the tactile acuity threshold assessment in the upper limb, the ANOVA revealed significant changes over time (F = 6.28, p = 0.014, ƞp2 = 0.06) but not for the group*time interaction (F = 1.272, p = 0.262, ƞp2 = 0.013). The post hoc analysis revealed no significant postintervention between-group differences (p = 0.832). However, significant within-group pre- and postintervention differences were found in the a-tDCS group, with a small effect size (p = 0.012, d = 0.20). There were no significant within-group pre- and postintervention differences in the s-tDCS group (p = 0.332) (Fig. 3).
Regarding the tactile acuity threshold assessment in the lower limb, the ANOVA revealed no significant differences in group*time (F = 3.33, p = 0.071, ƞp2 = 0.033) nor over time (F = 0.229, p = 0.633, ƞp2 = 0.002).
Regarding the 2-PD assessment in the upper limb, the ANOVA revealed no significant differences in group*time (F = 1.037, p = 0.311, ƞp2 = 0.01) nor over time (F = 0.225, p = 0.637, ƞp2 = 0.002).
With regard to the 2-PD assessment in the lower limb, the ANOVA revealed no significant differences in group*time (F = 1.978, p = 0.163, ƞp2 = 0.02) nor over time (F = 2.78, p = 0.098, ƞp2 = 0.028).
Pressure pain threshold
Regarding the PPT assessment in the upper limb, the ANOVA revealed no significant differences in group*time (F = 0.014, p = 0.906, ƞp2 = 0.0) nor over time (F = 0.977, p = 0.325, ƞp2 = 0.01).
However, with regard to the PPT assessment in the lower limb, the ANOVA revealed significant differences in group*time (F = 4.51, p = 0.036, ƞp2 = 0.044) and over time (F = 6.82, p = 0.01, ƞp2 = 0.065). The post hoc analysis revealed no significant postintervention between-group differences (p = 0.413). However, significant within-group pre- and postintervention differences were found in the a-tDCS group, with a small effect size (p = 0.001, d = − 0.17). There were no significant within-group pre- or postintervention differences in the s-tDCS group (p = 0.732) (Fig. 4).
EMG activity
Regarding the sEMG assessment in the long head of the biceps brachii, the ANOVA revealed significant differences in group*time (F = 15.30, p < 0.001, ƞp2 = 0.135) but not over time (F = 0.413, p = 0.522, ƞp2 = 0.004). The post hoc analysis revealed significant preintervention between-group differences, with a moderate effect size (p = 0.004, d = − 0.56) but no significant postintervention between-group differences (p = 0.259). In addition, significant pre- and postintervention within-group differences were found both in the a-tDCS group (p = 0.023, d = − 0.16) and in the s-tDCS group (p = 0.002, d = 0.18), with a small effect size (Fig. 5).
Finally, regarding the sEMG assessment in the rectus femoris, the ANOVA revealed significant differences in group*time (F = 7.12, p = 0.009, ƞp2 = 0.068) but not over time (F = 0.99, p = 0.322, ƞp2 = 0.01). The post hoc analysis revealed no significant postintervention between-group differences (p = 0.795). However, significant pre- and postintervention within-group differences were found in the a-tDCS group, with a small effect size (p = 0.011, d = − 0.14). There were no significant pre- and postintervention within-group differences in the s-tDCS group (p = 0.239) (Fig. 6).
Discussion
The main aim of the present study was to assess the short-term effects of a-tDCS in the M1 on sensorimotor variables including discriminative sensation, PPT and EMG activity compared with a sham-tDCS in healthy individuals. Although the results showed some significant within-group differences in the a-tDCS group between the pre- and postintervention interval, none of the results showed that the a-tDCS group had significant between-group differences compared with the sham group.
Regarding EMG activity, anodal stimulation usually increases the excitability of neurons in the area underneath the tDCS scalp electrodes38. In our study, a significant increase in the number of muscle fibres recruited during maximal isometric contractions was expected but was not observed, which coincides with Flood et al.39 However, a trend towards greater activity was observed in participants with a-tDCS compared with the sham group, which showed a trend towards decreased EMG activity. In fact, although no differences were found between the groups, the within-group differences were statistically significant in the upper limb. Krishnan et al.40 showed that anodal tDCS increased the magnitude of biceps brachii activation at 37.5% and 50% of maximum and also resulted in an increase in the peak force and EMG values during maximal muscle contractions in healthy subjects. However, Flood et al.39 had found that tDCS did not significantly raise maximal strength production in healthy volunteers. Suzuki et al.41 had reported an increase in motor evoked potentials in healthy participants; however, this increase was higher in stroke patients, suggesting that the potential effects of tDCS were different in a clinical population compared with healthy individuals.
Cycling exercises have been studied with the application of tDCS in other brain regions, such as the dorsolateral prefrontal cortex. Holgado et al.42 had found that tDCS over the left dorsolateral prefrontal cortex did not affect self-paced exercise performance in trained asymptomatic cyclists. However, Angius et al.43 had observed a reduced perception of effort and increased endurance in asymptomatic cyclists following anodal stimulation of the M1 when the cathode was placed on the contralateral shoulder, but not when placed over the prefrontal region. In addition, Vargas et al.14 had shown an increase in quadriceps strength during a maximal voluntary contraction after tDCS, yet Giboin et al.15 had found the opposite effect. In addition, Hendy et al.44 had found that the use of a-tDCS in combination with strength training of the wrist extensor muscles in healthy individuals did not result in improvements to the training itself when combined with a sham tDCS intervention. However, strength training with a-tDCS appears to differentially modulate cortical plasticity, which did not occur following strength training in isolation.
These data suggest that the application of tDCS produces immediate changes in healthy participants although these effects seem small and the data may also suggest that by itself, tDCS is not powerful enough to lead to changes in key variables such as strength or electromyographic activity. However, it appears that it can cause changes in regional neuronal activity during use45. Given the findings are somewhat contradictory and unclear, more research is needed.
Regarding the PPT, Borckardt et al.46 had reported similar findings as those observed in the present study where no significant effects were found in mechanical PPTs. However, Reidler et al.47 had shown that a-tDCS in the M1 significantly increased the PPT in healthy participants. In chronic pain and also in neuropathic pain due to spinal cord injury, the a-tDCS has showed good results in the increase of PPT in combination with other techniques such as TENS and visual illusion respectively48,49. In addition, the systematic review and meta-analysis conducted by Vaseghi et al.50 found that anodal tDCS stimulation was effective in the increase of PPT in healthy population and also, it showed a decrease of pain level in patients with chronic pain.
Pinto et al.51 had reviewed several tDCS studies on chronic pain, and concluded that despite evidence in favour of tDCS in combination with other treatments for chronic pain, it is still not possible to conclude whether tDCS is associated with a useful clinical effect for the treatment of maintained pain. Again, it appears that the results obtained in the present study are controversial, as are those found in the current scientific literature.
Regarding the tactile acuity threshold, the results showed statistically significant within-group differences in the a-tDCS group only in the upper limb. However, these differences were not superior to the s-tDCS group. There is a lack of literature regarding the influence of tDCS on somatosensory variables, especially when the stimulation is in the primary motor cortex. Falcone et al.52 had found that the application of tDCS in the prefrontal cortex led to significant improvements in perceptual sensitivity using signal detection theory by distinguishing between the perceptual sensitivity itself and the response bias. These improvements were retained for at least 24 h. Hilgenstock et al.53 had studied the effects of tDCS on somatosensory performance and concluded that, compared with a placebo application, tDCS promoted tactile learning by reducing the 2-PD threshold. The main difference with respect to the present study is that they carried out the training over 5 days and not in a single session. Thus, a single session of tDCS might be insufficient to bring about changes in an asymptomatic population.
Limitations
This study presents several limitations. First, there was no randomisation of the participants, for technical reasons. Second, there was only one session of t-DCS, and in an asymptomatic population. Perhaps with a greater number of sessions different results would have been obtained. Third, due to the design of this study, the assessors were not blinded, and this also should consider as a limitation.
In addition, because tDCS appears to be more powerful in combination with other intervention tools, future studies should be directed towards increasing the number of sessions and combining it with other interventions. A questionnaire to evaluate the effectivity of the sham protocol was not applied and this should be considered as a limitation. Finally, it should be stressed that, although we place the electrode over the M1 as precisely as possible, tDCS is usually placed in a general way over the cerebral cortex.
Conclusions
Although the results of the present study showed several significant within-group differences in the a-tDCS group, none of the results showed that the a-tDCS group had significant between-group differences compared with the s-tDCS group. However, a single session of anodal stimulation of tDCS in isolation over M1 appears to produce slight changes in the excitability of the primary motor cortex region in healthy individuals although these effects seem very small. These results should be taken with caution due to the small effect sizes obtained.
Abbreviations
- tDCS:
-
Transcranial direct current stimulation
- 2-PD:
-
Two-point discrimination test
- PPT:
-
Pressure pain threshold
- EMG:
-
Electromyography
- sEMG:
-
Surface electromyography
- ANOVA:
-
Analysis of variance
- a-tDCS:
-
Active transcranial direct current stimulation
- s-tDCS:
-
Sham transcranial direct current stimulation
- mA:
-
Milliamperes
References
Brunoni, A. R. et al. Clinical research with transcranial direct current stimulation (tDCS): Challenges and future directions. Brain Stimul. 5, 175–195 (2012).
Iannone, A. et al. Safety of transcranial direct current stimulation in a patient with deep brain stimulation electrodes. Arq. Neuropsiquiatr. 77, 174–178 (2019).
Bikson, M. et al. Safety of transcranial direct current stimulation: Evidence based update 2016. Brain Stimul. 9, 641–661 (2016).
Bastani, A. & Jaberzadeh, S. Does anodal transcranial direct current stimulation enhance excitability of the motor cortex and motor function in healthy individuals and subjects with stroke: A systematic review and meta-analysis. Clin. Neurophysiol. 123, 644–657 (2012).
Powers, A. et al. Effects of combining a brief cognitive intervention with transcranial direct current stimulation on pain tolerance: A randomized controlled pilot study. Pain Med. 19, 677–685 (2018).
Saldanha, J. S. et al. Impact of age on tDCS effects on pain threshold and working memory: Results of a proof of concept cross-over randomized controlled study. Front. Aging Neurosci. 12, 189 (2020).
Mariano, T. Y. et al. Effects of transcranial direct current stimulation (tDCS) on pain distress tolerance: A preliminary study. Pain Med. 16, 1580–1588 (2015).
Angius, L., Hopker, J. G., Marcora, S. M. & Mauger, A. R. The effect of transcranial direct current stimulation of the motor cortex on exercise-induced pain. Eur. J. Appl. Physiol. 115, 2311–2319 (2015).
Nathan, P. W., Smith, M. & Deacon, P. The crossing of the spinothalamic tract. Brain 124, 793–803 (2001).
Cobos, E. J. et al. Mechanistic differences in neuropathic pain modalities revealed by correlating behavior with global expression profiling. Cell Rep. 22, 1301–1312 (2018).
Fregni, F. & Pascual-Leone, A. Technology insight: Noninvasive brain stimulation in neurology—Perspectives on the therapeutic potential of rTMS and tDCS. Nat. Clin. Pract. Neurol. 3, 383–393 (2007).
Aslaksen, P. M., Vasylenko, O. & Fagerlund, A. J. The effect of transcranial direct current stimulation on experimentally induced heat pain. Exp. Brain Res. 232, 1865–1873 (2014).
Mordillo-Mateos, L. et al. Effects of transcranial direct current stimulation on temperature and pain perception. Sci. Rep. 7, 2946 (2017).
Vargas, V. Z. et al. Modulation of isometric quadriceps strength in soccer players with transcranial direct current stimulation. J. Strength Cond. Res. 32, 1336–1341 (2018).
Giboin, L.-S. & Gruber, M. Anodal and cathodal transcranial direct current stimulation can decrease force output of knee extensors during an intermittent MVC fatiguing task in young healthy male participants. J. Neurosci. Res. 96, 1600–1609 (2018).
Hendy, A. M. & Kidgell, D. J. Anodal tDCS applied during strength training enhances motor cortical plasticity. Med. Sci. Sport. Exerc. 45, 1721–1729 (2013).
Hendy, A. & Kidgell, D. Anodal-tDCS applied during unilateral strength training increases strength and corticospinal excitability in the untrained homologous muscle STEPS Study View project Effects of vitamin D supplementation on neuroplasticity in older adults View project. Exp. Brain Res. https://doi.org/10.1007/s00221-014-4016-8 (2014).
Chhatbar, P. Y. et al. Evidence of transcranial direct current stimulation-generated electric fields at subthalamic level in human brain in vivo. Brain Stimul. 11, 727–733 (2018).
Lefaucheur, J. P. et al. Evidence-based guidelines on the therapeutic use of transcranial direct current stimulation (tDCS). Clin. Neurophysiol. 128, 56–92 (2017).
Schulz, K. F., Altman, D. G., Moher, D. & Group, C. CONSORT 2010 Statement : Updated guidelines for reporting parallel group randomised trials. Ann. Intern. Med. 152, 726–732 (2010).
Murray, L. M. et al. Intensity dependent effects of transcranial direct current stimulation on corticospinal excitability in chronic spinal cord injury. Arch. Phys. Med. Rehabil. 96, S114–S121 (2015).
Neri, F. et al. A novel tDCS sham approach based on model-driven controlled shunting. Brain Stimul. 13, 507–516 (2020).
Sanches, C. et al. Testing the therapeutic effects of transcranial direct current stimulation (tDCS) in semantic dementia: A double blind, sham controlled, randomized clinical trial. Trials 20, 632 (2019).
Bell-Krotoski, J. A., Fess, E. E., Figarola, J. H. & Hiltz, D. Threshold detection and Semmes-Weinstein monofilaments. J. Hand Ther. 8, 155–62
Novak, C. B., Mackinnon, S. E., Williams, J. I. & Kelly, L. Establishment of reliability in the evaluation of hand sensibility. Plast. Reconstr. Surg. 92, 311–322 (1993).
Dellon, A. L., Mackinnon, S. E. & Crosby, P. M. Reliability of two-point discrimination measurements. J. Hand Surg. Am. 12, 693–696 (1987).
Mielke, K., Novak, C. B., Mackinnon, S. E. & Feely, C. A. Hand sensibility measures used by therapists. Ann. Plast. Surg. 36, 292–296 (1996).
Kinser, A. M., Sands, W. A. & Stone, M. H. Reliability and validity of a pressure algometer. J. Strength Cond. Res. 23, 312–314 (2009).
Chesterton, L. S., Sim, J., Wright, C. C. & Foster, N. E. Interrater reliability of algometry in measuring pressure pain thresholds in healthy humans, using multiple raters. Clin. J. Pain 23, 760–766 (2007).
Takala, E. P. Pressure pain threshold on upper trapezius and levator scapulae muscles. Repeatability and relation to subjective symptoms in a working population. Scand. J. Rehabil. Med. 22, 63–8 (1990).
Jang, M. H. et al. Validity and reliability of the newly developed surface electromyography device for measuring muscle activity during voluntary isometric contraction. Comput. Math. Methods Med. 2018, 1–9 (2018).
Ekstrom, R. A., Donatelli, R. A. & Carp, K. C. Electromyographic analysis of core trunk, hip, and thigh muscles during 9 rehabilitation exercises. J. Orthop. Sport. Phys. Ther. 37, 754–762 (2007).
de Araújo, R. C. et al. Reliability of electromyographic amplitude values of the upper limb muscles during closed kinetic chain exercises with stable and unstable surfaces. J. Electromyogr. Kinesiol. 19, 685–694 (2009).
Roman-Viñas, B. et al. International physical activity questionnaire: Reliability and validity in a Spanish population. Eur. J. Sport Sci. 10, 297–304 (2010).
Mantilla Toloza, S. C. & Gómez-Conesa, A. El Cuestionario Internacional de Actividad Física. Un instrumento adecuado en el seguimiento de la actividad física poblacional. Rev. Iberoam. Fisioter. Kinesiol. 10, 48–52 (2007).
Cohen, J. Eta-squared and partial eta-squared in fixed factor Anova designs. Educ. Psychol. Meas. 33, 107–112 (1973).
Cohen, J. Statistical Power Analysis for the Behavioral Sciences. 2nd edn. (Lawrence Earlbaum Associates, 1988).
Woods, A. J. et al. A technical guide to tDCS, and related non-invasive brain stimulation tools. Clin. Neurophysiol. 127, 1031–1048 (2016).
Flood, A., Waddington, G., Keegan, R. J., Thompson, K. G. & Cathcart, S. The effects of elevated pain inhibition on endurance exercise performance. PeerJ 5, e3028 (2017).
Krishnan, C., Ranganathan, R., Kantak, S. S., Dhaher, Y. Y. & Rymer, W. Z. Anodal transcranial direct current stimulation alters elbow flexor muscle recruitment strategies. Brain Stimul. 7, 443–450 (2014).
Suzuki, K. et al. Comparison of the after-effects of transcranial direct current stimulation over the motor cortex in patients with stroke and healthy volunteers. Int. J. Neurosci. 122, 675–681 (2012).
Holgado, D. et al. Transcranial direct current stimulation (tDCS) over the left prefrontal cortex does not affect time-trial self-paced cycling performance: Evidence from oscillatory brain activity and power output. PLoS One 14 (2019).
Angius, L., Pageaux, B., Hopker, J., Marcora, S. M. & Mauger, A. R. Transcranial direct current stimulation improves isometric time to exhaustion of the knee extensors. Neuroscience 339, 363–375 (2016).
Hendy, A. M. & Kidgell, D. J. Anodal tDCS applied during strength training enhances motor cortical plasticity. Med. Sci. Sports Exerc. 45, 1721–1729 (2013).
Lang, N. et al. How does transcranial DC stimulation of the primary motor cortex alter regional neuronal activity in the human brain?. Eur. J. Neurosci. 22, 495–504 (2005).
Borckardt, J. J. et al. A pilot study of the tolerability and effects of high-definition transcranial direct current stimulation (HD-tDCS) on pain perception. J. Pain 13, 112–120 (2012).
Reidler, J. S. et al. Effects of motor cortex modulation and descending inhibitory systems on pain thresholds in healthy subjects. J. Pain 13, 450–458 (2012).
Soler, M. D. et al. Effectiveness of transcranial direct current stimulation and visual illusion on neuropathic pain in spinal cord injury. Brain 133, 2565–2577 (2010).
Boggio, P. S. et al. Transcranial DC stimulation coupled with TENS for the treatment of chronic pain: A preliminary study. Clin. J. Pain 25, 691–695 (2009).
Vaseghi, B., Zoghi, M. & Jaberzadeh, S. Does anodal transcranial direct current stimulation modulate sensory perception and pain? A meta-analysis study. Clin. Neurophysiol. 125, 1847–1858 (2014).
Pinto, C. B., Teixeira Costa, B., Duarte, D. & Fregni, F. Transcranial direct current stimulation as a therapeutic tool for chronic pain. J. ECT 34, e36–e50 (2018).
Falcone, B., Coffman, B. A., Clark, V. P. & Parasuraman, R. Transcranial direct current stimulation augments perceptual sensitivity and 24-hour retention in a complex threat detection task. PLoS One 7 (2012).
Hilgenstock, R., Weiss, T., Huonker, R. & Witte, O. W. Behavioural and neurofunctional impact of transcranial direct current stimulation on somatosensory learning. Hum. Brain Mapp. 37, 1277–1295 (2016).
Acknowledgements
We would like to thank el Centro Superior de Estudios Universitarios CSEU La Salle for making this study possible.
Author information
Authors and Affiliations
Contributions
S.L.L.: research project execution; statistical analysis design, execution; writing of the first draft; manuscript review and critique; M.M. and M.G.: research project execution, writing of the first draft and organisation; R.L.T. and F.C.M.: manuscript review and critique. All authors have read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lerma-Lara, S., De Cherade Montbron, M., Guérin, M. et al. Transcranial direct-current stimulation (tDCS) in the primary motor cortex and its effects on sensorimotor function: a quasi-experimental single-blind sham-controlled trial. Sci Rep 11, 6566 (2021). https://doi.org/10.1038/s41598-021-85989-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-85989-7
This article is cited by
-
Effect of tDCS targeting the M1 or left DLPFC on physical performance, psychophysiological responses, and cognitive function in repeated all-out cycling: a randomized controlled trial
Journal of NeuroEngineering and Rehabilitation (2023)
-
Anodal tDCS over the left DLPFC but not M1 increases muscle activity and improves psychophysiological responses, cognitive function, and endurance performance in normobaric hypoxia: a randomized controlled trial
BMC Neuroscience (2023)
-
The impact of bilateral anodal transcranial direct current stimulation of the premotor and cerebellar cortices on physiological and performance parameters of gymnastic athletes: a randomized, cross-over, sham-controlled study
Scientific Reports (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.