Abstract
Retention is a key element in HIV prevention programs. In Sub-Saharan Africa most data on retention come from HIV clinical trials or people living with HIV attending HIV treatment and control programs. Data from observational cohorts are less frequent. Retention at 6-/12-month follow-up and its predictors were analyzed in OKAPI prospective cohort. From April 2016 to April 2018, 797 participants aged 15–59 years attending HIV Voluntary Counseling and Testing in Kinshasa were interviewed about HIV-related knowledge and behaviors at baseline and at 6- and 12-month follow-ups. Retention rates were 57% and 27% at 6- and 12-month follow up; 22% of participants attended both visits. Retention at 6-month was significantly associated with 12-month retention. Retention was associated with low economic status, being studying, daily/weekly Internet access, previous HIV tests and aiming to share HIV test with partner. Contrarily, perceiving a good health, living far from an antiretroviral center, daily/weekly alcohol consumption and perceiving frequent HIV information were inversely associated with retention. In conclusion, a high attrition was found among people attending HIV testing participating in a prospective cohort in Kinshasa. Considering the low retention rates and the predictors found in this study, more HIV cohort studies in Kinshasa need to be evaluated to identify local factors and strategies that could improve retention if needed.
Similar content being viewed by others
Introduction
Despite a 28% decline in new HIV infections in Eastern and Southern Africa from 2010 to 2018 and of 13% in Western and Central Africa, there are still 25.6 million people living with HIV in Sub-Saharan Africa (SSA), a region where 61% of new infections worldwide take place1. These figures show that there has been an insufficient progress in the prevention, treatment and control of HIV.
Retention at the different stages of the HIV continuum of care is a key element for the success of HIV control programs2,3,4,5,6,7,8,9,10. It can lead to a better medication adherence, enabling dosage or drug modifications, status disclosure to partner, and better social support, thus reducing the risk of antiretroviral therapy (ART) resistance and HIV transmission, morbidity and mortality11,12,13.
Retention is also essential for HIV research studies. High quality longitudinal cohorts and clinical trials within healthcare settings are necessary for the design of adequate HIV programs. Both study designs facilitate data collection, tracing the dynamics of behaviors and identify factors that influence sexual behaviors, testing or treatment adherence.
In SSA, most published research on retention and related factors show data from clinical trials or from people living with HIV within the HIV continuum of care. The problem of attrition among people living with HIV still not under ARV treatment (who have a high risk of HIV transmission) or among participants in prospective observational cohorts has been less frequently evaluated5,14,15,16.
Observational studies can show the real-world context outside the controlled protocols of clinical trials. They can help to properly design preventive interventions, and to improve their feasibility, uptake, acceptability and efficiency17,18,19,20. Longitudinal observational studies can also provide information concerning loss to follow-up (LTFU) and their predictors, which is relevant for the implementation of HIV preventive strategies, and to improve the quality of the data obtained21.
Being LTFU in longitudinal cohorts can be an important source of selection bias22. LTFU participants might have different characteristics from those who complete all study visits19,23,24,25,26. Furthermore, failing to achieve the expected number of participants can reduce the statistical power of a study27 and also prolong the study, modifying available economic resources28. Thus, retention is crucial for the validity of these studies.
Data from observational cohorts evaluating retention in ART programs in SSA have shown attritions at 12 months from 7 to 45%10,23,29. However, attrition rates vary for different regions and studies, being generally high in SSA10,30,31.
Different factors have been shown to be associated with being LTFU in the African context, including some related to the participants, their contact information, the study time, location or staff12,32,33. Frequent changes in cell phone numbers, transportation to study sites or financial constraints contribute largely to these figures28.
Research on retention in longitudinal observational studies in the Democratic Republic of the Congo (RDC) are scarce. Therefore, in this study the retention rates at 6- and 12-month follow-ups and the associated factors were analyzed in a prospective cohort study among people attending HIV testing at a reference hospital in Kinshasa (DRC).
Methods
Study design and participants
The OKAPI (Observational Kinshasa AIDS Prevention Initiative) project is a prospective cohort study designed to evaluate factors associated with changes in HIV knowledge and sexual behaviors after 6- and 12-months of follow-up. From April 2016 to April 2018, people aged 15–59 years attending HIV Voluntary Counseling and Testing (VCT) at a reference hospital in Kinshasa were invited to participate. Those with a previous HIV positive test as well as pregnant women were excluded. Details of the study have been published elsewhere34.
Data collection
Participants were interviewed by local researchers at baseline and after 6- and 12-month follow-up periods. Male and female interviewers were available at all study times. Questionnaires were available in French and Lingala. The baseline questionnaire included 59 questions about their sociodemographics, health-related aspects, HIV knowledge, attitudes, behaviors and exposure to HIV information. Follow-up questionnaires were shorter and included some new questions concerning sociodemographic and health issues.
The sociodemographic variables considered in the analyses carried out in this paper were: sex, age (quantitative and categorized into 15–19, 20–24 and 25–59 years, for adolescents, young and adults, respectively, following the World Health Organization and other official classifications), education level (no studies/primary studies, secondary studies, university studies), perceived economic level (low, middle, high), working status (unemployed/housewife, studying, working), civil status (single, married, divorced, widowed), church attendance (never/seldom/monthly, daily/weekly), use of the mobile phone and the Internet (daily/weekly). Health-related information included: perceived health status (1 to 5 scale, categorized as 1–3 for lower health status, and 4–5 for better perceived health), HIV risk perceived (no/low risk, medium/high risk), previous HIV testing, attending HIV VCT with couple, intention to share an HIV + result with partner, time to the closest ARV center (< 15, 15–30, 30–60, > 60 min, categorized into > 60 min or not), HIV test result (negative, positive, indeterminate), STI diagnosis in the last 12 months (yes/no), diagnosis of genital ulcer, urinary tract, vaginal or penis infection (yes/no) and alcohol consumption (never/seldom, weekly/daily). With regards to sexual behaviors, participants were asked about ever sex, first sex before 15 years, ≥ 2 sexual partners currently and in the previous 6 months, consistent condom use, oral, anal or paid sex (ever) and forced sex (ever), including physical violence perpetrated by partner, sex being afraid of partner´s reaction and unwanted sexual practices. With regards to HIV misconceptions, believing HIV is caused by witchcraft or God´s punishment was considered. Finally, participants were asked about HIV information in Kinshasa (few/quite enough and much).
HIV diagnosis
A free HIV screening test was carried out at each study visit. A blood sample was collected and rapid diagnostic tests were used, following the routine clinical protocol: Determine HIV-1/2 (Alere) test was performed and, if positive, DoubleCheckGold (Orgenics) and Unigold (Trinity Biotech) rapid immunoassays were used for confirmation. A Dried Blood Spot (DBS) card was collected for HIV molecular analyses among participants getting an HIV positive test.
Follow-up
At baseline, each participant´s telephone number was registered, as well as up to three other contact phone numbers in order to avoid attrition. All participants received a transport fee at each follow-up appointment, as explained at recruitment. One week before each follow-up appointment, the local personnel phoned the participants up to 5 times. Participants were considered lost-to follow up (LTFU) if they did not answer to any phone-calls or they could not or did not want to remain in the study. Reasons for not coming back were recorded. Home visits were offered to LTFU participants that could not come back, in order to facilitate the HIV testing and data collection.
Statistical analyses
Descriptive analyses were first carried out to evaluate participants´ sociodemographics, health, sexual behaviors and HIV knowledge/information at baseline and at 6- and 12-month visits. Paired comparisons between characteristics at baseline and at 6- and 12-month follow-ups, respectively, were carried out using McNemar test. Additionally, at both follow-ups characteristics of the retained and lost-to-follow-up participants were compared by using Chi2 test and Student t test for categorical and quantitative variables, respectively. Crude logistic regression analyses were carried out to evaluate the association between each baseline characteristic and retention in the cohort at 6- and 12-month appointments. A multivariate logistic regression model was fitted including all variables significantly associated with retention in the crude analyses. A different multivariate model was fitted for each follow-up time, considering the 6-, 12- or both 6- and 12-month retention as the dependent variable. An additional multivariate-adjusted model was built including time-varying exposures and a Generalized Estimated Equations (GEE) analysis was carried out for this purpose.
All p-values < 0.05 were considered statistically significant. Analyses were performed using Stata version 15.1 (StataCorp, College Station, TX, USA).
Ethical issues
OKAPI project was approved by the Research Ethics Committees of the two centers involved in the study (University of Navarra, Spain and Monkole Hospital, DRC). A written informed consent was collected from each participant or parent and/or legal guardian if subjects were under 18. All methods were performed in accordance to the principles of the Declaration of Helsinki.
Results
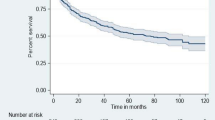
From April 2016 to April 2018, 797 participants were included in the cohort. All people invited to participate agreed to join the study. All of them replied to the baseline interview and were HIV tested. At the 6-month appointment, 456/797 participants were followed up (57% retention rate): 445 (56%) returned to Monkole Hospital and 11 (1%) were interviewed and tested at home. At 12-month follow-up, 219/797 participants returned (27%). Overall, 176/797 (22%) participants attended both the 6-month and the 12-month follow-ups, and 43/797 (5%) participants came back only at the 12-month follow-up (Fig. 1A). Among the 24 participants that received an HIV positive test at baseline, retention rates were 50% at 6-month follow-up, 21% at 12-month appointment and 17% came at both follow-ups (Fig. 1B).
Attending the 6-month appointment was significantly and independently associated with being followed at 12-month (adjusted OR = 4.4; 95% CI = 3.0–6.5).
The mean follow-up time for participant's 6-month and 12 -month visits was 6.6 (1.7) months and 14.8 (5.0) months, respectively. Thus, 50% of participants returned after their expected date for their 6-month follow-up and 83% returned later than expected for their 12-month follow-up.
The main sociodemographic, clinical and behavioral characteristics of participants at baseline and at 6- and 12-month follow-ups are shown in Table 1. At baseline near 60% of the participants were women and the mean age of the study population was 30 years (SD: 9). Around 70% of respondents had university studies, close to 80% reported a middle economic level, 82% were not married, 91% had a high religiosity and the great majority reported a daily/weekly access to the mobile phone (99%) and the Internet (78%). The majority of the participants perceived having a good health status (66%) and a low HIV risk (94%), most of them had been previously HIV tested (64%) and 3% got a new positive HIV diagnosis in our study. At baseline, 10% of the participants reported having had an STI diagnosis in the previous year. Regarding sexual behaviors, most participants were sexually experienced (91%), 11% reported having had sex before 15 years, around 20% had multiple sexual partners and 60, 20 and 10% reported oral, anal or paid sex, respectively. The prevalence of all these risk sexual practices was significantly lower at follow-up when paired comparisons were done at each of the follow-ups. Finally, regarding HIV-related misconceptions, at baseline 38% wrongly believed HIV is caused by witchcraft or God´s punishment. However, the percentage of participants reporting this wrong belief was significantly lower at both follow-ups.
When baseline characteristics among retained and lost-to-follow-up participants were compared, at 6-month follow-up retained participants were significantly more likely to have reported being studying/working at baseline (48 vs. 30%, p < 0.001), to have a more frequent baseline use of the Internet (82 vs. 74%, p = 0.016), having a lower perception of good health (61 vs. 74%, p < 0.001), reporting genital ulcers (10 vs. 6%, p = 0.038) or urinary tract infections (14 vs. 9%, p = 0.05), having had more repeated HIV testing (68 vs. 59%, p = 0.008), having the intention to share a positive HIV test with their partner (34 vs. 20%, p < 0.001), having suffered from forced sex (37 vs. 22%, p < 0.001), living closer to ARV distribution centers (85 vs. 77%, p = 0.003), less frequently reporting common alcohol consumption (14 vs.21%, p = 0.007) and having received much information on HIV (16 vs. 23%, p = 0.0.12). At 12-month follow-up significant differences among retained and lost-to-follow-up participants were found for the frequent Internet use (88 vs. 75%, p < 0.001), repeated HIV testing (71 vs. 61%, p = 0.012), intention to share a positive result with partner (36 vs. 25%, p = 0.002) and less reporting of forced sex (11 vs. 18%, p = 0.028).
Variables significantly associated with retention in crude analyses were included in multivariate models (Table 2). Retention at 6- and 12-month follow-ups was independently associated with reporting a low economic status, being studying or working, having a daily/weekly access to the Internet, having been previously HIV tested, having the intention to share an HIV positive test with their partner and reporting forced sex. On the contrary, at 6-month follow-up, participants living far from an ARV centre, were significantly less likely to come back. Sex, age and sexual risk behaviors were not associated with retention at any follow-up.
Discussion
Research on retention in HIV studies in SSA has been focused on people living with HIV within the HIV continuum of care. Fewer studies have analyzed the retention rates and associated factors in observational cohorts. In this prospective HIV cohort including people attending HIV Voluntary Counseling and Testing in Kinshasa, and having both positive and negative tests, low retention rates were found: 57% at the 6-month follow-up appointment and 27% at the 12-month follow-up, with only 22% of the participants completing the two scheduled visits. Among participants who got an HIV positive test result, retention rates were 50% and 21% at 6- and 12-month visits, respectively.
Data on retention rates in the DRC come from retrospective cohorts including patients living with HIV within HIV care and ARV programs29,32,35,36,37. They all show higher retention rates than ours, varying from 67 to 96%. Recent studies from different countries in SSA show retention rates between 17 and 83% after 12 months10,15,23,31,38,39,40. Most of these studies were conducted among people HIV positive involved in HIV care programs, which makes them more motivated to continue in the study. On the contrary, most of our participants were HIV negative, with no need of coming back to healthcare. In an Ugandan cohort that evaluated factors associated with dropout among high-risk participants invited for HIV testing and having a negative result, researchers found that 92% were followed up after 6 months, 85% at 12 months and 76% at 18 months19, thus, retention rates much higher than those observed in our study in Kinshasa were found. This was probably because they were invited for HIV screening (compared to the client-initiated VCT in our cohort), they were high-risk individuals which may have moved them to attend follow-up visits, they included more home visits and also because in the study condoms and medical care including STIs diagnosis and treatment were offered to them. In another study published by the same research group they analyzed retention among high-risk women with an HIV negative test and they found that over the 18-month follow-up period, 93% came back at least once41. The higher retention observed may have been influenced by the fact that previous to the screening participants attended community meetings in which information about the study was provided. Also, participants compliant with study follow-ups were trained as leaders that reminded other participants to attend their visits and reported about participants who had migrated. Finally, the same research team published another study that similarly evaluated retention among high-risk HIV negative people in the same Ugandan fishing communities. It was an HIV vaccine preparedness study in which door-to-door HIV counseling and testing was offered. Over the 24-month follow-up period, retention was 70%42. Another HIV vaccine trial preparedness cohort in South Africa found a 85% retention among participants who were HIV negative. Factors that probably contributed were the group workshops on HIV and vaccines hold along the study period and the HIV, syphilis and pregnancy tests performed at each 3-month follow-up visits43. Mutagoma et al. found a 92% retention rate after 12-month follow-up among participants in the Rwandan AIDS indicator and HIV incidence longitudinal household survey. The authors discussed factors that helped to have good retention rates such as the small size of Rwanda which facilitates transportation and the knowledge about people relocations and migrations among community members and the support of the national media23. Conley and cols. evaluated retention in a prospective cohort study among HIV-1 serodiscordant heterosexual couples and found a retention around 80%. All couples received at least one home visit, support groups were available, trimethoprim-sulfamethoxazole prophylaxis was supplied for partners HIV-infected, free drinks were offered during clinic visits, and transportation fees to and from the clinic were covered44.
Considering the retention observed in other African cohorts and that rates of follow-up around 80% have been recommended in cohort studies45, more work is needed to monitor and improve retention in Kinshasa, working on different factors that assure better follow-ups in cohort studies or clinical trials.
In our study significant associations were not found for participants´ age and sex, although most studies show that retention rates increase with age and among females12,19,46,47,48,49. In our cohort, education level was neither a predictive factor of better retention. Most of the evidence coming from SSA studies show that having no education or lower levels of education is associated with higher attrition rates5,12,15,23,40,47,50, with the exception of one study in which patients with secondary or higher education were more frequently LTFU32. In our cohort being a student (89% of them university students) was associated with higher retention at 6-month follow-up, probably due to being more responsible or sensitive with research. Also, participants working at baseline were more likely to be retained at 6-month follow-up compared to unemployed participants, although no significant associations were found for 12-month follow-up and for those attending both follow-up appointments. Other studies have found that working is associated with higher attrition rates, probably as a result of inflexible work schedules19,28,50,51. However, some authors have found opposite results, with lack of formal employment being a barrier to retention33,52. Within HIV care, higher lost-to-care rates have been found among patients with a paid profession maybe as a result of being more able to transfer to a private clinic5.
Regarding economic level, literature shows that reporting a lower socioeconomic status, family financial constraints or perceiving social class as poor are associated with being LTFU15,28,30,52,53. In our study participants with a low economic level were significantly more likely to return at 6- and 12-month follow-ups. This may be due to the fact that all participants had a monetary incentive for transportation at follow-up, which probably motivated them to come back to the appointments. The same effect of economic incentives for transportation has been shown by other authors54,55. In fact, a key factor affecting retention rates are transportation costs or lack of transportation15,24,28,30,33,46,50,52,56. Teague and colleagues carried out a systematic review in 2018 including 143 longitudinal cohort studies to assess the effectiveness of different retention strategies and found that `barrier-reduction´ strategies were associated with higher retention27. Related to transportation barriers, reporting a longer distance to the closest center for ARV distribution was inversely associated with retention at 6-month follow-up, as previously reported by other authors4,15,25,30,52,57. As recommended by other researchers27,46, home visits were offered to improve retention in the cohort. Although some LTFU participants were retained, home visits were not an easy nor efficient strategy in our study.
With respect to the perceived health status, as shown by other authors25, participants perceiving they had a good health were less likely to attend the 6-month follow-up visit (significance was lost for the 12-month follow-up). It makes sense that people with good health do not perceive the need to be followed up.
Participants previously HIV tested were significantly more likely to attend follow-ups. As shown by other authors23,58, the wish to know one´s serostatus encourages people to participate in studies and to continuously want to be tested. However, other authors have shown that perceiving that an HIV negative test will not change, may be a barrier for repeating HIV testing59, which could explain why many of the participants in our cohort, who were mostly HIV negative, did not return for the follow-up visit. In this sense, some authors suggest HIV pre-exposure prophylaxis (PrEP) availability and provision could be a potential incentive to improve retention among people attending HIV testing60,61. In the DRC the knowledge and access to the PrEP are still low today61,62, adherence to antiretrovirals is not high63 and ARV resistance is present64. Therefore, it does not seem to be an option today in the country although it might be an option to be considered in the future for high risk population attending HIV testing.
Regarding disclosure of HIV status, higher retention rates were found among participants that had the intention to share a positive HIV test result with their partner; this was consistent with other studies33,46,50,52. The intention of non-disclosure is more frequent in patients perceiving or fearing stigma, which may cause participants to miss their appointments12,30,46,52,65. In fact, among women, fear of repercussions from their partners has been associated with attrition15,65.
No significant associations were found between sexual risk behaviors and retention. Some studies have shown that having multiple sexual partners or using condoms were associated with participant dropouts12,19, however, there is few evidence on this.
Retention at 6-month follow-up was lower for those participants thinking there was frequent HIV information in Kinshasa. They probably did not find it necessary to contact the healthcare center again, once they were HIV tested and counseled. Other studies have shown an association between poor knowledge on STI or HIV and being LTFU12,52, which can be a result of not perceiving the infections negative consequences.
Despite the repeated reminder phone calls in our study a low retention was found. People in Kinshasa, as well as in other SSA, frequently lose or change their phone numbers28,50. Likewise, Teague and colleagues showed that retention was significantly higher in studies not including phone call reminders compared to those with phone call reminders. In our study, reporting a weekly/daily access to the Internet at baseline was significantly associated with a better retention at 6-month, 12-month as well as 6- and 12-month follow-ups. Considering that the access to the Internet is increasing in Kinshasa (as observed among our participants most of whom reported a frequent access), other innovative approaches could be a good option for maintaining regular contacts and participants´ motivation and improve retention, as proposed by different authors11,28,66,67. There is few evidence on new strategies based on modern technology that need to be evaluated in future studies to improve participants´ retention.
Based on the factors that have been found to be associated with better retention in our cohort, and taking into account Teague and cols´ findings for their four proposed groups of retention strategies (barrier-reduction; community-building; follow-up strategies (eg. incentives)/reminders; and tracing), it can be highlighted that a better design of home visits and of alternative methods of data collection for barrier-reduction is needed. According to Teague and cols, barrier-reduction strategies, such as home visits, and community-building strategies (i.e. emphasizing the benefits of the study, giving feedback and sending thank you and birthday messages to the participants) were the most efficient in lowering attrition27. This could similarly help improve retention in cohorts in the DRC. Regarding follow-up/reminders strategies, the different options they analyzed did not show to improve retention, neither did tracing strategies, such as the tracing via alternative contacts that was considered in our cohort. They did not improve retention. Finally, the different Internet options within all different retention approaches need to be evaluated.
Our study has some limitations that need to be considered. First, the reasons for being LTFU were not well documented and information regarding death, migration or other physical or psychological diseases, described in other SSA studies3,29 was lacking. Considering that the vast majority of our participants tested HIV negative, a high mortality rate is quite unlikely in our cohort. Other reasons such as going to other health facilities for new HIV/STI testing, the distance to our hospital or the presence of other comorbidities do need to be taken into account in future studies. Secondly, considering the 100% response rate observed in our project, there seems to be a possibility that agreeing to participate was a polite and socially desirable answer, possibly explaining the low retention rates in our cohort.
Despite these limitations, our study has some strengths that need to be highlighted. This is the first study in the DRC evaluating the retention rates and associated factors in an HIV research cohort including participants attending HIV Voluntary Counseling and Testing and not in the continuum of HIV care and treatment. Secondly, 797 participants were included in the cohort, a sample size that allowed evaluating multiple factors using multivariate analyses and that can help the local policymakers to improve retention in different HIV research studies in the country.
In conclusion, our results show a high attrition among people attending HIV VCT and participating in a prospective cohort in Kinshasa. Considering the factors associated with a better retention, such as the economic incentive at follow-up, the repeated HIV test or the use of the Internet, this study underscores the need of designing and implementing new strategies to improve retention in HIV research studies in Kinshasa.
References
UNAIDS. UNAIDS Data 2019. Geneva, 2019. https://www.unaids.org/en/resources/documents/2019/2019-UNAIDS-data (last accessed Feb 14, 2020].
van der Kop, M. L. et al. Retention in clinic versus retention in care during the first year of HIV care in Nairobi, Kenya: A prospective cohort study. J. Int. AIDS Soc. 21, e25196. https://doi.org/10.1002/jia2.25196 (2018).
Seifu, W., Ali, W. & Meresa, B. Predictors of loss to follow up among adult clients attending antiretroviral treatment at Karamara general hospital, Jigjiga town, Eastern Ethiopia, 2015: A retrospective cohort study. BMC Infect. Dis. 18, 280. https://doi.org/10.1186/s12879-018-3188-4 (2018).
Ameyan, W. et al. A participatory approach to improving retention in HIV treatment and care for newly diagnosed patients in a secondary health facility in Sierra Leone. J. AIDS Clin. Res. 8, 4. https://doi.org/10.4172/2155-6113.1000686 (2017).
Stolka, K. et al. Characteristics of antiretroviral therapy-naïve patients lost-to-care in HIV clinics in Democratic Republic of Congo, Cameroon, and Burundi. AIDS Care 28, 913–918 (2016).
Breger, T. L. et al. Self-disclosure of HIV status, disclosure counseling, and retention in HIV care in Cameroon. AIDS Care 29, 838–845. https://doi.org/10.1080/09540121.2016.1271390 (2017).
Carlucci, J. G. et al. Attrition of HIV-exposed infants from early infant diagnosis services in low- and middle-income countries: A systematic review and meta-analysis. J. Int. AIDS Soc. 21, e25209. https://doi.org/10.1002/jia2.25209 (2018).
Chammartin, F. et al. Outcomes of patients lost to follow-up in african antiretroviral therapy programs: Individual patient data meta-analysis. Clin. Infect. Dis. 67, 1643–1652. https://doi.org/10.1093/cid/ciy347 (2018).
Murray, K. R. et al. Improving retention in HIV care among adolescents and adults in low- and middle-income countries: A systematic review of the literature. PLoS ONE 12, e0184879. https://doi.org/10.1371/journal.pone.0184879 (2017).
Haas, A. D. et al. Retention and mortality on antiretroviral therapy in sub-Saharan Africa: Collaborative analyses of HIV treatment programmes. J. Int. AIDS Soc. 21, e25084. https://doi.org/10.1002/jia2.25084 (2018).
Fernández-Luis, S. et al. Reengagement of HIV-infected children lost to follow-up after active mobile phone tracing in a rural area of Mozambique. J. Trop. Pediatr. 65, 240–248. https://doi.org/10.1093/tropej/fmy041 (2019).
Tsadik, M., Berhane, Y., Worku, A. & Terefe, W. The magnitude of, and factors associated with, loss to follow-up among patients treated for sexually transmitted infections: A multilevel analysis. BMJ Open 7, e016864. https://doi.org/10.1136/bmjopen-2017-016864 (2017).
Whembolua, G. L., Conserve, D. F., Thomas, K., Tshiswaka, D. I. & Handler, L. HIV serostatus disclosure in the Democratic Republic of the Congo: A systematic review. AIDS Care 31(4), 489–493. https://doi.org/10.1080/09540121.2018.1510103 (2019).
Zachariah, R. et al. Retention and attrition during the preparation phase and after start of antiretroviral treatment in Thyolo, Malawi, and Kibera, Kenya: Implications for programmes?. Trans. R. Soc. Trop. Med. Hyg. 105, 421–430 (2011).
da Silva, M. et al. Patient loss to follow-up before antiretroviral therapy initiation in rural Mozambique. AIDS Behav. 19, 666–678 (2015).
WHO. Retention in HIV Programmes: Defining the Challenges and Identifying Solutions: Meeting Report (2011).
Sohn, A. H. et al. Using observational data to Inform HIV policy change for children and youth. J. Acquir. Immune Defic. Syndr. 78(Suppl 1), S22–S26 (2018).
Ford, N. et al. The contribution of observational studies in supporting the WHO “treat all” recommendation for HIV/AIDS. J. Virus Erad. 4, 5–8 (2018).
Abaasa, A. et al. Factors associated with dropout in a long term observational cohort of fishing communities around Lake Victoria, Uganada. BMC Res. Notes 8, 815 (2015).
Lodi, S. et al. Effect estimates in randomized trials and observational studies: Comparing apples with apples. Am. J. Epidemiol. 188, 1569–1577. https://doi.org/10.1093/aje/kwz100 (2019).
Hønge, B. L. et al. Loss to follow-up occurs at all stages in the diagnostic and follow-up period among HIV-infected patients in Guinea-Bissau: A 7-year retrospective cohort study. BMJ Open 3, e003499. https://doi.org/10.1136/bmjopen-2013-003499 (2013).
Alonso, A. et al. Predictors of follow-up and assessment of selection bias from dropouts using inverse probability weighting in a cohort of university graduates. Eur. J. Epidemiol. 21, 351–358 (2006).
Mutagoma, M. et al. The role of community health workers and local leaders in reducing attrition among participant in the AIDS indicator survey and HIV incidence in a national cohort study in Rwanda. BMC Public Health 18, 338. https://doi.org/10.1186/s12889-018-5243-x (2018).
Marete, I. et al. Lost to follow-up among pregnant women in a multi-site community based maternal and newborn health registry: a prospective study. Reprod. Health 12, S4. https://doi.org/10.1186/1742-4755-12-S2-S4 (2015).
Badenhorst, D. H. S., Van der Westhuizen, C. A., Britz, E., Burger, M. C. & Ferreira, N. Lost to follow-up: Challenges to conducting orthopaedic research in South Africa. S. Afr. Med. J. 108, 917–921. https://doi.org/10.7196/SAMJ.2018.v108i11.13252 (2018).
Elul, B., Saito, S., Chung, H., Hoos, D. & El-Sadr, W. Attrition from human immunodeficiency virus treatment programs in Africa: A longitudinal ecological analysis using data from 307 144 patients initiating antiretroviral therapy between 2005 and 2010. Clin. Infect. Dis. 64, 1309–1316. https://doi.org/10.1093/cid/cix162 (2017).
Teague, S. et al. Retention strategies in longitudinal cohort studies: A systematic review and meta-analysis. BMC Med. Res. Methodol. 18, 151. https://doi.org/10.1186/s12874-018-0586-7 (2018).
Icard, L. D. et al. Retention of South African adolescents in a 54-month longitudinal HIV risk reduction trial. Prev. Sci. 18, 534–540. https://doi.org/10.1007/s11121-017-0793-1 (2017).
Fox, M. P. & Rosen, S. Patient retention in antiretroviral therapy programs up to three years on treatment in sub-Saharan Africa, 2007–2009: systematic review. Trop. Med. Int. Health 15(Suppl 1), 1–15 (2010).
Mesic, A. et al. Facilitators and barriers to adherence to antiretroviral therapy and retention in care among adolescents living with HIV/AIDS in Zambia: A mixed methods study. AIDS Behav. 23, 2618–2628. https://doi.org/10.1007/s10461-019-02533-5 (2019).
Kariminia, A. et al. Mortality and losses to follow-up among adolescents living with HIV in the IeDEA global cohort collaboration. J. Int. AIDS Soc. 21, e25215. https://doi.org/10.1002/jia2.25215 (2018).
Akilimali, P. Z. et al. Disclosure of HIV status and its impact on the loss in the follow-up of HIV-infected patients on potent anti-retroviral therapy programs in a (post-) conflict setting: A retrospective cohort study from Goma Democratic Republic of Congo. PLoS ONE 12, e0171407. https://doi.org/10.1371/journal.pone.0171407 (2017).
Gill, M. M. et al. Addressing early retention in antenatal care among HIV-positive women through a simple intervention in Kinshasa, DRC: The Elombe “Champion” Standard Operating Procedure. AIDS Behav. 22, 860–866. https://doi.org/10.1007/s10461-017-1770-1 (2018).
Carlos, S. et al. Heterosexual oral and anal sex in Kinshasa (D.R. Congo): Data from OKAPI prospective cohort. PLoS ONE 14, e0210398. https://doi.org/10.1371/journal.pone.0210398 (2019).
Ditekemena, J. et al. Antiretroviral treatment program retention among HIV-infected children in the Democratic Republic of Congo. PLoS ONE 9, e113877 (2014).
Koole, O. et al. Retention in a NGO supported antiretroviral program in the Democratic Republic of Congo. PLoS ONE 7, e40971. https://doi.org/10.1371/journal.pone.0040971 (2012).
Ross, J. et al. Association between pregnancy at enrollment into HIV care and loss to care among women in the Democratic Republic of Congo, 2006–2013. PLoS ONE 13, e0195231. https://doi.org/10.1371/journal.pone.0195231 (2018).
Clouse, K. et al. Mobility and clinic switching among postpartum women considered lost to HIV care in South Africa. J. Acquir. Immune Defic. Syndr. 74, 383–389. https://doi.org/10.1097/QAI.0000000000001284 (2017).
Kiplagat, J. et al. Retention in care among older adults living with HIV in western Kenya: A retrospective observational cohort study. PLoS ONE 13, e0194047. https://doi.org/10.1371/journal.pone.0194047 (2018).
Namuwenge, P. M. et al. Loss to follow up from isoniazid preventive therapy among adults attending HIV voluntary counseling and testing sites in Uganda. Trans. R. Soc. Trop. Med. Hyg 106, 84–89 (2012).
Ssetaala, A. et al. Recruitment and retention of women in fishing communities in HIV prevention research. Pan. Afr. Med. J. 21, 104 (2015).
Bahemuka, U. M. et al. Retention of adults from fishing communities in an HIV vaccine preparedness study in Masaka, Uganda. PLoS ONE 14, e0198460 (2019).
Middelkoop, K. et al. Adolescent and adult participation in an HIV vaccine trial preparedness cohort in South Africa. J. Adolesc. Health 43, 8–14 (2008).
Conley, N. J. et al. Distance from home to study clinic and risk of follow-up interruption in a cohort of HIV-1-discordant couples in Nairobi, Kenya. PLoS ONE 7, e43138 (2012).
Aschengrau, A. & Seage, G. R. Essentials of Epidemiology in Public Health (Jones and Bartlett Publishers, Inc., 2003).
Nabaggala, M. S. et al. Re-engagement in HIV care following a missed visit in rural Uganda. BMC Res. Notes 11, 762 (2018).
Bulsara, S. M., Wainberg, M. L. & Newton-John, T. R. O. Predictors of adult retention in HIV care: A systematic review. AIDS Behav. 22, 752–764. https://doi.org/10.1007/s10461-016-1644-y (2018).
Mee, P. et al. Changes in patterns of retention in HIV care and antiretroviral treatment in Tanzania between 2008 and 2016: An analysis of routinely collected national programme data. J. Glob. Health 9, 010424. https://doi.org/10.7189/jogh.09.010424 (2019).
Sikazwe, I. et al. Retention and viral suppression in a cohort of HIV patients on antiretroviral therapy in Zambia: Regionally representative estimates using a multistage-sampling-based approach. PLoS Med. 16, e1002811 (2019).
Kiwanuka, N. et al. A randomized trial to assess retention rates using mobile phone reminders versus physical contact tracing in a potential HIV vaccine efficacy population of fishing communities around Lake Victoria, Uganda. BMC Infect. Dis 18, 591. https://doi.org/10.1186/s12879-018-3475-0 (2018).
Lopez-Varela, E. et al. Continuum of HIV care in rural Mozambique: The implications of HIV testing modality on linkage and retention. J. Acquir. Immune Defic. Syndr. 78, 527–535 (2018).
Kouassi Kan V. et al. Factors Associated with Loss to Follow-up Status among ART Patients in Cote d’Ivoire. Research Report Summary. Published by the USAID Health Care Improvement Project. Bethesda, MD: University Research Co., LLC (URC) (2014).
Yotebieng, M. et al. Conditional cash transfers improve retention in PMTCT services by mitigating the negative effect of not having money to come to the clinic. J. Acquir. Immune Defic. Syndr. 74, 150–157 (2017).
Bonsu, J. M., Frasso, R. & Curry, A. E. Lessons from the field: The conduct of randomized controlled trials in Botswana. Trials 18, 503. https://doi.org/10.1186/s13063-017-2237-4 (2017).
Booker, C. L., Harding, S. & Benzeval, M. A systematic review of the effect of retention methods in population-based cohort studies. BMC Public Health 11, 249. https://doi.org/10.1186/1471-2458-11-249 (2011).
Yotebieng, K. A., Fokong, K. & Yotebieng, M. Depression, retention in care, and uptake of PMTCT service in Kinshasa, the Democratic Republic of Congo: A prospective cohort. AIDS Care 29, 285–289. https://doi.org/10.1080/09540121.2016.1255708 (2017).
Freeman, A. et al. Patient outcomes in Lubumbashi, Democratic Republic of Congo after a disruption in HIV care due to decreased global fund appropriations. AIDS Behav. 18, 2135–2143. https://doi.org/10.1007/s10461-014-0761-8 (2014).
Regan, S. et al. Factors associated with self-reported repeat HIV testing after a negative result in Durban, South Africa. PLoS ONE 8, e62362. https://doi.org/10.1371/journal.pone.0062362 (2013).
Harichund, C., Kunene, P., Simelane, S., Abdool, K. Q. & Moshabela, M. Repeat HIV testing practices in the era of HIV self-testing among adults in KwaZulu-Natal, South Africa. PLoS ONE 14, e0212343. https://doi.org/10.1371/journal.pone.0212343 (2019).
World Health Organization. WHO Implementation tool for pre-exposure prophylaxis (PrEP) of HIV infection. Module 5: Monitoring and evaluation. Geneva, 2018 (WHO/CDS/HIV/18.10). Licence: CC BY-NC-SA 3.0 IGO.
Irungu, E. M. & Baeten, J. M. PrEP rollout in Africa: status and opportunity. Nat. Med. 26(5), 655–664. https://doi.org/10.1038/s41591-020-0872-x (2020).
Bepouka, B. I. et al. Care providers’ knowledge and willingness to prescribe pre-exposure prophylaxis (PrEP) in Kinshasa, Democratic Republic of Congo (DRC). Pan. Afr. Med. J. 34, 166. https://doi.org/10.11604/pamj.2019.34.166.18025 (2019).
Izizag, B. B. et al. Determinants of non-compliance with antiretroviral therapy in adult patients in Kinshasa. Pan. Afr. Med. J. 37, 157. https://doi.org/10.11604/pamj.2020.37.157.13261 (2020).
Kwon, E. H. et al. HIV-1 subtypes and drug resistance mutations among female sex workers varied in different cities and regions of the Democratic Republic of Congo. PLoS ONE 15(2), e0228670. https://doi.org/10.1371/journal.pone.0228670 (2020).
Gichane, M. W. et al. Intimate partner violence and prevention of mother to child transmission of HIV: Evidence from Kinshasa Democratic Republic of Congo. PLoS ONE 13, e0203471. https://doi.org/10.1371/journal.pone.0203471 (2018).
Daher, J. et al. Do digital innovations for HIV and sexually transmitted infections work? Results from a systematic review (1996–2017). BMJ Open 7, e017604. https://doi.org/10.1136/bmjopen-2017-017604 (2017).
Rhyne, J. M. et al. The South African Rea Phela Health Study: A randomized controlled trial of communication retention strategies. PLoS ONE 13, e0196900. https://doi.org/10.1371/journal.pone.0196900 (2018).
Acknowledgements
The authors want to thank Monkole Hospital, the interviewers for their essential work and the generous participants who make the research possible. The authors also thank the Government of Spain (Fondo de Investigación en Salud-PI16/01908), the Government of Navarra (045-2015) and the Institute for Culture and Society of the University of Navarra for the funding provided.
Author information
Authors and Affiliations
Contributions
S.C. conducted the literature review, statistical analyses and wrote the first draft of the manuscript. E.B., A.N. and G.R. have contributed to study design and writing the paper. CL-B and A.O. have provided advice on methodological aspects and helped write this article. B.M. has critically revised this manuscript. J.I. has supervised all phases of the study design and has contributed in writing all versions of the paper. All authors have read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Carlos, S., Burgueño, E., Ndarabu, A. et al. Predictors of retention in the prospective HIV prevention OKAPI cohort in Kinshasa. Sci Rep 11, 5431 (2021). https://doi.org/10.1038/s41598-021-84839-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-84839-w
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.