Abstract
Social stress exacerbates anxious and depressive behaviors in humans. Similarly, anxiety- and depressive-like behaviors are triggered by social stress in a variety of non-human animals. Here, we tested whether oral administration of the putative anxiolytic probiotic strains Lactobacillus helveticus R0052 and Bifidobacterium longum R0175 reduces the striking increase in anxiety-like behavior and changes in gut microbiota observed following social defeat stress in Syrian hamsters. We administered the probiotic at two different doses for 21 days, and 16S rRNA gene amplicon sequencing revealed a shift in microbial structure following probiotic administration at both doses, independently of stress. Probiotic administration at either dose increased anti-inflammatory cytokines IL-4, IL-5, and IL-10 compared to placebo. Surprisingly, probiotic administration at the low dose, equivalent to the one used in humans, significantly increased social avoidance and decreased social interaction. This behavioral change was associated with a reduction in microbial richness in this group. Together, these results demonstrate that probiotic administration alters gut microbial composition and may promote an anti-inflammatory profile but that these changes may not promote reductions in behavioral responses to social stress.
Similar content being viewed by others
Introduction
The human gastrointestinal tract houses a vastly abundant community of microorganisms, and it has become increasingly clear that the state of this microbial community can meaningfully impact disease states1,2,3. This community of microbes is necessary for general health and vital processes such as digestion4,5, gastrointestinal barrier protection, and immunoregulation6,7. While it is also known that multiple, bidirectional routes of communication exist between the gastrointestinal tract, its microbiota, and the brain8, more research is required to understand the neurological and behavioral consequences of this communication. More recent data demonstrate that the gut microbiota may alter brain and behavior9,10,11,12, but few studies have examined whether this community modifies social behavior in ethologically relevant models of social interaction.
Social stress is the primary form of stress experienced by humans and is a major predictor for the onset of a variety of neuropsychiatric disorders13,14,15, such as mood and anxiety disorders. Animal models of social stress have revealed that socially defeated rodents and monkeys display behavioral responses (i.e., social avoidance, changes in ingestive behavior and sleep, and increases in anxiety- and depressive-like behaviors) that resemble the symptoms of neuropsychiatric disorders in humans15,16,17,18,19,20,21. Extensive research has explored how social stress may increase the likelihood of developing a mood or anxiety disorder15,22,23; however, data on the direct effect of gut microbiota on susceptibility to social stress are limited. This deficiency is important because it has been suggested that the gut microbial community could be a potential target for the development of novel treatments (e.g., “psychobiotics”) for these neuropsychiatric disorders24,25. It is known that the gut is responsive to stress and that stress-induced dysbiosis of the gut microbial community is linked to negative health consequences such as breakdown of the gastrointestinal barrier and a heightened proinflammatory profile26,27,28,29,30,31. Our lab recently demonstrated that even a single social defeat in Syrian hamsters causes dysbiosis of the gut microbial community in both animals that win (e.g., become dominant) and those that lose (e.g., become subordinate), with such alterations being exacerbated following repeated bouts of social stress32. Importantly, specific microbial taxa also appear to predict future dominant or subordinate status following an agonistic encounter, suggesting that the microbial profile may modify future behavioral responses to social conflict32.
In order to better understand how the state of the gut microbial community drives social behavior and to assess whether “psychobiotics” might protect against the deleterious effects of social stress, we asked whether manipulating the gut microbial community with a probiotic containing large quantities of gut-derived microbes that are thought to benefit the host33 would promote resistance to the behavioral consequences of social stress. We used a well-characterized social defeat model in Syrian hamsters16,32,34,35. This species provides an ideal model of social stress because when conspecifics are paired, they readily produce aggressive and territorial behavior that rapidly results in the formation of a stable dominance relationship36. Importantly, agonistic behavior during these brief encounters is highly ritualized and rarely results in tissue damage, allowing us to focus on the psychological, as opposed to physical, aspects of social stress. This advantage also eliminates any confounding effect of physical injury on inflammation and the gut microbial community. Here, we tested whether oral treatment with a probiotic containing Lactobacillus helveticus R0052 and Bifidobacterium longum R0175 is sufficient to reduce stress-induced dysbiosis of the gut microbial community and to decrease defeat-induced social avoidance. Furthermore, we investigated the extent to which probiotic treatment impacts the gut microbial community or has an anti-inflammatory effect.
Results
Probiotic intervention at a low dose increases susceptibility to social stress
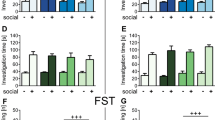
A timeline for the study is shown in Fig. 1a. Following both acute and repeated defeat training, hamsters were tested for social avoidance and social interaction with a novel, caged opponent. Repeated Measures, Two-way ANOVAs with Tukey’s post hoc analyses were run to assess the effect of probiotic intervention (high and low dose or placebo) and number of defeats (acute or repeated) on social behavior. Behavioral data from 3 hamsters in the low dose probiotic group were not scored due to a technical error in saving the videos and thus the number of animals in this group was reduced. Analysis of social avoidance behavior revealed no interaction effect (F(2, 44) = 0.5, p = 0.6, η2 = 0.02) or main effect of defeat number (F(1, 44) = 2.4, p = 0.1, η2 = 0.05) on avoidance behavior; however, there was a significant main effect of probiotic treatment (F(2, 44) = 4.2, p = 0.02, η2 = 0.2). Tukey’s pairwise comparisons indicated that hamsters treated with the low dose of the probiotic exhibited significantly more social avoidance following an acute defeat than did hamsters treated with a high dose of the probiotic (p = 0.04) or the placebo (p = 0.02) (Fig. 1b). Following repeated defeats, no significant differences in avoidance behavior between treatment groups were observed; however, there was a strong trend for hamsters treated with the low dose of the probiotic to spend significantly more time avoiding a caged opponent compared to placebo-treated hamsters (p = 0.055) (Fig. 1b). Analysis of social interaction revealed no interaction effect (F(2, 44) = 1.5, p = 0.2, η2 = 0.06), but there was a significant main effect of defeat number (F(1, 44) = 22.3, p < 0.0001, η2 = 0.3) and a main effect of probiotic treatment on social interaction (F(2, 44) = 4.8, p = 0.01, η2 = 0.2). Tukey’s post hoc analysis revealed that hamsters treated with a low dose of the probiotic spent significantly less time interacting socially with a caged opponent following an acute defeat compared to hamsters treated with a high dose of the probiotic (p = 0.02) or placebo (p = 0.03) (Fig. 1b). Following repeated defeats, hamsters treated with a low dose of the probiotic exhibited less social interaction than did placebo-treated hamsters (p = 0.03), but they did not differ from hamsters receiving the high dose of the probiotic (p = 0.2) (Fig. 1c).
Probiotic intervention at a low dose increases susceptibility to social stress. (a) A timeline for the study is shown in (a). Following the acute defeat, hamsters treated with a low dose of the probiotic (n = 7) (blue dots) avoided a novel opponent more (b) and interacted with the opponent less (c) than did hamsters treated with a high dose of the probiotic (n = 20) (purple dots) or the placebo (n = 20) (orange dots) (*p < 0.05). Following repeated defeats, there were no significant differences in avoidance behavior between treatment groups (b), yet hamsters treated with the lose dose (n = 7) (blue dots) interacted with a novel opponent significantly less than did placebo-treated hamsters (n = 20) (orange dots) (c; *p < 0.05). Data presented as mean +/− standard error of the mean. Note that the videos for three animals in the low dose probiotic group were lost due to a technical problem, so the group n for behavioral analysis is lower than for the other measures analyzed.
Probiotic intervention modestly alters the concentration of probiotic strains in feces
Measurement of the probiotic strains in fecal samples collected from each hamster approximately 1 h before the acute defeat, 24 h after the acute defeat, or 24 h after the final repeated defeat was completed by qPCR. Concentrations were not normally distributed. Therefore, nonparametric Kruskal–Wallis with Dunn’s multiple comparisons test was used to compare the concentrations of probiotic strains across treatment groups. Concentrations of the probiotic strains for each treatment group were collapsed across the three time points for statistical analysis. Hamsters whose concentrations were below the range of the standard curve were given a value of 0 for analysis. Concentrations of unspiked samples were 6.44 log bacteria/g feces for Lactobacillus helveticus R0052 and 6.49 log bacteria/g feces for Bifidocaterium logum R0175. A significant effect of treatment was observed for the concentration of Lactobacillus helveticus R0052 (H(3,150) = 102.9, p < 0.0001). The concentration of Lactobacillus helveticus R0052 was significantly lower in the placebo group compared to the two probiotic groups and the concentration of Lactobacillus helveticus R0052 was higher in hamsters treated with the high dose of the probiotic compared to hamsters treated with the low dose of the probiotic or placebo (Dunn’s multiple comparisons test; high dose v placebo, p < 0.0001, low dose v placebo, p < 0.0001, high dose v low dose, p = 0.028, Fig. 2a). For Bifidobacterium longum R0052, a significant effect of treatment was also observed (H(3,150) = 129.3, p < 0.0001); the concentration of Bifidobacterium longum R0052 was higher in the feces of hamsters treated with the high dose of the probiotic compared to the hamsters treated with the low dose of the probiotic or placebo (Dunn’s multiple comparisons test; high dose v placebo, p < 0.0001, high dose v low dose, p < 0.0001, Fig. 2b).
Probiotic intervention modestly alters the concentration of probiotic strains in feces. Concentrations of Lactobacillus helveticus R0052 (a) and Bifidobacterium longum R0175 (b) detected in fecal samples collected approximately 1 h before the acute defeat (pre defeat, black bar), 24 h following the acute defeat (blue bar), or 24 h following repeated defeats (green bar). Data are presented as mean +/− standard error of the mean; non-shared letters within each panel denote a significant difference (p < 0.03).
Probiotic intervention alters gut microbiota composition
Microbiota composition was analyzed by 16S rRNA Illumina sequencing of fecal DNA samples collected before defeat training (baseline), after the initial (acute) defeat, and after nine (repeated) bouts of social defeat. PERMANOVA analysis of the unweighted UniFrac distance revealed that the microbial composition of all treatment groups significantly differed from one another after probiotic treatment but before any behavioral manipulation (low dose v high dose, p = 0.006; low dose v placebo, p = 0.006; high dose v placebo, p = 0.01) (Fig. 3a). Following acute defeat, the microbial composition of hamsters given the low dose of the probiotic differed significantly from that of hamsters administered the high dose (p = 0.05) (Fig. 3b). After the acute defeat there was also a strong trend for the microbial composition of hamsters given either dose of the probiotic to significantly differ from that of placebo-treated hamsters (low dose v placebo, p = 0.057; high dose v placebo, p = 0.057) (Fig. 3b). Following repeated defeats, the microbial composition of hamsters treated with a low dose of the probiotic differed from that of both hamsters treated with a high dose of the probiotic (p = 0.006) and placebo-treated hamsters (p = 0.04) (Fig. 3c).
Probiotic intervention alters the gut microbial composition before and after social stress. Principle coordinate analysis (PCoA) of the unweighted UniFrac distance at baseline (a), after acute social stress (b), and after repeated social stress (c). Although not immediately obvious based on the PCoA plots, the unweighted UniFrac distance revealed that at “baseline” (after probiotic treatment but before any behavioral manipulations) the microbial composition of each treatment group differed from one another (a) and that, after acute social stress, the microbial composition of hamsters administered the low dose of the probiotic (n = 10) (orange dots) differed from that of hamsters administered the high dose (n = 20) (red dots) (b). Following repeated social stress, unweighted UniFrac revealed that the microbial composition of hamsters treated with a low dose of the probiotic (n = 10) (orange dots) was different than that of both the placebo-treated hamsters (n = 20) (blue dots) and the hamsters treated with a high dose of the probiotic (n = 20) (red dots) (c). P values were determined using PERMANOVA analysis and denoted as significant at p < 0.05 (a–c).
The analysis of alpha diversity of the intestinal microbiota, reflecting the bacterial richness and evenness of the community, revealed a significant effect of probiotic treatment using both phylogeny-based (Faith Phylogenetic Diversity (PD) Whole Tree) (H = 14.54, p = 0.0007) and non-phylogeny-based (Observed Operational Taxonomic Units (OTUs)) (H = 10.33, p = 0.006) measurements. Dunn’s multiple comparisons test revealed a significant decrease in alpha diversity for defeated hamsters treated with a low dose of the probiotic compared to defeated hamsters treated with a high dose of the probiotic (Faith PD Whole Tree, p = 0.001; Observed OTUs, p = 0.01) or with the placebo (Faith PD Whole Tree, p = 0.008; Observed OTUs, p = 0.02) (Fig. 4).
Probiotic administration at a low dose decreases intestinal microbiota diversity. Alpha diversity was determined by Faith PD Whole Tree and Observed OTUs in hamsters treated with a high dose (purple) or low dose (blue) of the probiotic and in placebo-treated hamsters (orange). No differences within groups were observed, thus groups were collapsed across time points. Both Faith PD (a) and Observed OTUs (b) indicated that hamsters given a low dose of the probiotic (blue) had lower diversity than did hamsters given a high dose (purple) or placebo (orange). *denotes p < 0.05; **denotes p ≤ 0.01; ***denotes p ≤ 0.001. Data presented as mean +/− standard error of the mean.
Next, LEfSE analysis (Linear Discriminant Analysis (LDA) Effect Size) was used to identify bacterial taxa that were significantly altered by treatment or defeat training. An LDA threshold of 2 was used to infer significance (LDA > 2, p < 0.05). A relatively small number of taxa were significantly altered by probiotic treatment using this stringent threshold. Notably, genus Bifidobacterium was significantly higher in hamsters treated with a low dose of the probiotic compared to all other treatment groups following acute defeat (LDA > 2, p < 0.05; Fig. 5a). Following repeated defeats, genus Prevotella was significantly higher in hamsters treated with a high dose of the probiotic compared to their pre-defeat baseline and phyla Proteobacteria was significantly higher in placebo-treated animals compared to both probiotic treatment groups (LDA > 2, p < 0.05; Fig. 5b).
Identification of microbial taxa that were differentially altered across groups following acute or repeated social defeat. LEfSE analysis (Version 1, available at: https://huttenhower.sph.harvard.edu/galaxy/))82 was used to identify microbial taxa that differed among or between groups in hamsters given either a low dose of the probiotic (green), high dose of the probiotic (red), or placebo (blue) after an acute defeat (a) or repeated defeats (b). Blue represents taxa higher in placebo-treated hamsters compared to both doses of the probiotic; green, taxa higher in hamsters treated with a low dose of the probiotic compared to the high dose or placebo; red, taxa higher in hamsters treated with a high dose of the probiotic compared to the low dose or placebo. Only taxa meeting an LDA significant threshold of > 2.0 are represented.
Probiotic intervention increases circulating anti-inflammatory cytokines
A Bio-Plex Pro™ Rat Cytokine 23-Plex Assay was used to analyze circulating levels of cytokines. A rat assay was chosen because no equivalent multiplex assay exists for hamster. Only serum concentrations (pg/mL) of IL-7, IL-4, IL-10, GRO/KC, IL-5, and MIP-3α were detected in the majority of hamsters and were thus able to be reliably analyzed. Hamsters whose concentrations were out of range were given a value of 0 for analysis. It should be noted that the majority of the values were on the low end of the standard curve, which can increase variability and decrease reproducibility of results. No effect of treatment was found for IL-7, GRO/KC, and MIP-3α. One-way ANOVA revealed an effect of treatment for the anti-inflammatory cytokine IL-4 (F(2, 48) = 12.12, p < 0.0001) and Tukey’s post hoc analysis revealed that hamsters treated with both the low and high dose of the probiotic had elevated IL-4 compared to placebo-treated hamsters (high dose vs. placebo, p = 0.0004; low dose v placebo, p = 0.0004; Fig. 6a). The concentrations of IL-10 and IL-5 were not normally distributed, thus the nonparametric Kruskal–Wallis with Dunn’s multiple comparisons test was used and revealed a significant effect of treatment for IL-10 (H(3,51) = 19.29, p < 0.0001) and IL-5 (H(3,51) = 19.66, p < 0.0001). Hamsters given either dose of the probiotic had higher concentrations of both the anti-inflammatory cytokine IL-10 and the anti-inflammatory chemokine IL-5 compared to hamsters treated with placebo (Dunn’s multiple comparisons test; IL-10, high dose v placebo, p = 0.002, low dose v placebo, p = 0.0021, Fig. 6b; IL-5, high dose v placebo, p = 0.003, low dose v placebo, p = 0.0002, Fig. 6c).
Probiotic intervention alters cytokines following social stress. Following repeated social defeat, circulating concentrations (pg/mL) of IL-4 (a), IL-10 (b), and IL-5 (c) were significantly increased in hamsters treated with both doses of the probiotic (low dose, n = 7, blue dots) (high dose, n = 20, purple dots) compared with placebo (n = 20, orange dots) (*significantly higher than placebo, p < 0.01). Data presented as mean +/− standard error of the mean.
Discussion
Our results demonstrate that a probiotic intervention at a dose that is equivalent to that used in humans can induce increases in social avoidance, decreases in social interaction, alterations in the gut microbial community, along with modest changes in serum anti-inflammatory cytokines in hamsters. We selected the commercially available probiotic CEREBIOME® (formerly called Probio’stick®) containing the organisms Lactobacillus helveticus R0052 and Bifidobacterium longum R0175 based on previous published findings that this probiotic at similar doses can reduce stress responses following water submersion or maternal separation37,38 as well as anxiety- and depressive-like behavior in humans and other animals39,40,41,42. Thus, our behavioral findings contrast with the previous literature and were opposite of what was expected33,43,44,45,46,47,48,49,50. One possible explanation for our unexpected finding may be that our behavioral endpoint measures something different than do the standard tests of anxiety-like behavior such as the open field or light–dark box tests that are commonly used in mice and other rodents45,48. It is possible that the response to an ethologically relevant stressor, such as social stress, is very different than that observed after exposure to a more artificial stressor. It may also be the case that the effects of probiotics are specific and do not necessarily translate across strains or species. This possibility is supported by evidence that different mouse strains exhibit strain-specific probiotic effects45,48. Ultimately, it is unclear why our data differ from the majority of the previous literature indicating that probiotics are anxiolytic. At the very least our data serve as a caution that the behavioral effect of putative anxiolytic probiotic treatments may not always be as predicted. A careful examination of differences in probiotic-induced changes to the gut microbial community and/or cytokine signaling, however, is needed to help elucidate why probiotic intervention at varying doses drives different responses in different models.
The unweighted UniFrac metric revealed that the microbial composition of all treatment groups differed from one another prior to stress (but after 14 days of probiotic treatment). Differences in microbiota based on treatment but before behavioral manipulation suggest that probiotic intervention in the absence of stress is sufficient to alter the microbial composition. Further, in addition to the anxiogenic behavioral profile observed in hamsters treated with a low dose of the probiotic, an altered microbial profile was observed in this group compared to the high dose-treated and placebo-treated hamsters after repeated social defeat. This indicates that the social stress effects on both microbiota and behavior seem to be altered differently by the same probiotic given at different doses. This finding underscores the potential importance of dosage in the ultimate outcome of probiotic treatment.
Alpha diversity, a measure of microbial richness and abundance, was assessed using a phylogeny-based (Faith PD Whole Tree) and non-phylogeny-based (Observed OTUs) measurement. The measurements complimented one another, with both demonstrating a reduction in richness in hamsters given the low dose versus the high dose or the placebo. It is possible that a reduction in alpha diversity following social defeat in hamsters given the low dose of the probiotic drives, in part, the anxiogenic behavioral profile observed in this group51,52. This is an interesting possibility that should be examined further in future studies. Alternatively, it is also possible that the excipients present in a higher dose in the placebo and high dose probiotic groups drove greater microbiobial richness. This possibility is discussed further below (see paragraph on limitations).
LEfSe analysis identifies particular microbial taxa that drive differences in beta and alpha diversity across groups. Although several microbial taxa were altered between treatment groups or within groups following defeat, there were fewer changes observed compared to our previous study assessing the gut microbial community of hamsters following acute and repeated social defeat with no probiotic intervention32. One possibility is that the excipients (xylitol, maize-derived maltrodextrin, plum flavor, and malic acid) present in both the placebo and probiotic solutions, themselves, had some effect on the gut microbiota and that this, in turn, buffered or masked the effect of social stress on the gut microbiota. The latter possibility is supported by the recent demonstration that maltodextrin is a stressor for the gut53. In the present study, genus Bifidobacterium was significantly higher in hamsters treated with a low dose of the probiotic after the acute defeat. Bifidobacterium longum is present in the probiotic formulation; thus, ingestion of the probiotic at a low dose may have allowed for heightened colonization of Bifidobacterium in the gut and/or increased proliferation of Bifidobacterium species. It is not clear why this did not also occur following administration of the high dose probiotic. The phyla Proteobacteria was significantly higher in placebo-treated hamsters following repeated social stress and genus Prevotella was higher in hamsters given the high dose of the probiotic compared to their pre-defeat baseline. In our previous study, both Proteobacteria and Prevotella predicted dominance in an agonistic encounter32, and other research suggests these taxa may be beneficial or stress-protective for the host50,54. Therefore, the greater abundance of Proteobacteria or Prevotella in hamsters treated with the placebo or high dose probiotic may, in part, have driven the reduced behavioral response to social stress observed in these two groups.
There are some important limitations of this study that should be recognized in the context of the above discussion, however. The first, mentioned above, is that the low dose probiotic group also ingested lower doses of the excipients than did the placebo and high dose probiotic groups, which received equal concentrations of the excipients. Thus, it is possible that the lower levels of these additives in the low-dose group underlies the differential effects on the microbiome and behavior. We were not initially concerned about this because there are ample data indicating that these excipients do not have behavioral or physiological effects, including a recent finding in humans that ingestion of 75 g of sugar but not 75 g of maltodextrin increases the stress response to the Trier Social Stress Test compared to placebo55. In fact, these additives are often used as control treatments and are classified as inert by the United States Food and Drug Administration. As noted above, however, there are some recent data suggesting that maltrodextrin, at least, can alter the gut microbiome56. Future studies should certainly take into account the possibility that excipients might have independent effects, and it is critical for the field that this possibility be examined directly. A related limitation of this study is that the effect of treatment was only examined in socially stressed animals. Future research should determine whether probiotic or excipient treatment alters social avoidance or social investigation in unstressed individuals. Additionally, the concentrations of the probiotic bacterial strains were only examined in fecal samples and not in the gut, itself, which would provide a better measure of the actual colonization by these bacteria. Given that we used a repeated defeat stressor and collected samples before, during, and after social stress, obtaining gut samples, such as from caecum, was not possible. Finally, there is a recent report that treatment with CEREBIOME® decreases food intake and body weight gain in male but not in female rats57. This suggests that, going forward, measures of food intake and body mass should be included in studies such as this one.
It has been suggested that gut microbiota may drive differences in social behavior by altering cytokine signaling, a well-characterized route of communication between the gut and brain3,29,58. Thus, we examined whether an increase in the expression of pro-inflammatory cytokines was correlated with changes to the gut microbial community and increased behavioral susceptibility to social defeat following administration of the low dose of the probiotic. This prediction was based on previous evidence indicating that increases in proinflammatory cytokines exacerbate depressive- and anxiety-like behavior in animal models59,60,61,62,63,64,65. Unfortunately, many of the cytokines and chemokines targeted in the Luminex assay were out of detectable range in one or more groups. Thus, we were unable to analyze several of the cytokines in which we had the most interest such as IL-6 and TNFα. Of the six cytokines or chemokines that were detectable (IL-7, IL-4, IL-10, GRO/KC, IL-5, MIP-3α), three were significantly altered by treatment. Although we observed an effect of probiotic dose on behavior and on the gut microbial community, this dose-dependency was not apparent when analyzing circulating cytokine signaling. Three anti-inflammatory cytokines, IL-4, IL-10 and IL-5, were significantly elevated in both probiotic (low and high dose) groups compared to placebo. The proinflammatory cytokines IL-7 and MIP-3α and proinflammatory chemokine GRO/KC were not altered across groups. These results support previous work showing an anti-inflammatory effect of Lactobacillus helveticus R0052 and Bifidobacterium longum R0033 and R017566,67. Therefore, a therapeutic benefit of probiotic treatment may be to bias the immune system toward an anti-inflammatory profile following stress. Given the finding that we observed so few changes in cytokine concentrations, however, it could be the case that the use of the rat multiplex assay to assess hamster cytokines/chemokines limited our findings, that our model of social stress is too mild, that the current probiotic is ineffective in causing a robust increase in pro- or anti-inflammatory signaling and/or that it is not via alterations in immune signaling that the probiotics altered behavior in this study.
Collectively, the results of the current study demonstrate that the effect of probiotics on behavior and on gut microbial composition are likely species- and dose-dependent. This highlights the importance of testing the effect of probiotics in multiple animal models and in various environmental and behavioral contexts. Further, the dose-dependent differences in behavior and alterations to gut microbiota following probiotic treatment clearly illustrate that a higher probiotic dose does not necessarily predict a greater behavioral response. We have demonstrated that probiotic intervention can alter both behavioral responses to social stress and gut microbiota, but future work is necessary to establish whether the changes to the gut microbiota are necessary for probiotic-induced behavioral alterations and by what mechanism these changes occur.
Methods
Animals
Adult male Syrian hamsters (Mesocricetus auratus), weighing between 120 and 130 g, were obtained from Charles River Laboratory (Kingston, NY) at approximately 3 months of age. Hamsters were group-housed until the beginning of the experiment when they were individually housed in polycarbonate cages (24 × 33 × 20 cm). Animals were housed on corncob bedding, given cotton nesting material, and maintained in a temperature-controlled colony room under a 14:10 h light/dark cycle, which is standard to maintain reproductive gonadal status in hamsters. It is important to note that individual housing is not stressful for Syrian hamsters68. Food and water were available ad libitum. All hamsters were handled daily for 7 days to acclimate them to handling stress before the beginning of the experiment. All protocols and procedures were approved by the Georgia State University Institutional Animal Care and Use Committee prior to experimentation, and all methods align with the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
Probiotic intervention
At the beginning of the study, group-housed Syrian hamsters (n = 50) were matched by weight within cages and randomly assigned across treatment groups from each cage and individually housed. Hamsters were then treated with either the commercial probiotic formulation CEREBIOME® (Lallemand Health Solutions Inc., Montreal, QC, Canada) containing freeze-dried lactic acid bacteria strains, Lactobacillus helveticus R0052 and Bifidobacterium longum R0175 mixed with excipients (xylitol, maize derived matrodextrin, plum flavor, and malic acid) or the placebo formulation containing the excipients. Hamsters were assigned to one of three treatment groups: placebo, probiotic at a low dose of 109 colony forming units per day that is thought to be comparable to a dose normally consumed by humans, or probiotic high dose (tenfold higher than low dose) of 1010 colony forming units per day. Placebo and probiotic solutions were prepared prior to administration as per manufacturer instructions following careful procedures to prevent cross-contamination of treatments (for a full description, see Myles et al.42). Each hamster received a daily dose of 0.2 g in distilled water. Hamsters received either the probiotic intervention or placebo for 14 days prior to the behavior experiment and throughout testing for a total treatment length of 21 days. Each day, hamsters were given 0.3 mL of the appropriate solution by syringe feeding69, which all animals readily ingested, at the start of the active phase of the daily activity cycle.
Behavioral procedures
All behavioral manipulations were conducted during the dark phase of the daily light:dark cycle to control for circadian variation in behavior and because this is when hamsters are active and exhibit the majority of their agonistic behavior. All hamsters were moved into the behavior suite 30 min prior to any manipulation to allow time to acclimate. Behavior trials were run under dim red light and were recorded with a CCD camera.
For acute defeat training, hamsters were placed in the home cage of a novel, same-sex aggressor for 15 min34. For repeated defeat training, hamsters were placed in a novel, same-sex resident aggressor’s home cage for 5 min twice a day for 4 days. The first pairing occurred at the start of the dark phase and the second occurred 4 h later. A clear plastic lid was placed over the resident’s cage during each pairing to prevent escape. The resident aggressor reliably attacked the experimental subject and the latter exhibited submissive and defensive behaviors such as upright defense, flee, and tail lift36.
Social behavior testing (duration 5 min) took place approximately 24 h after acute and repeated defeat training, as described previously70. Hamsters were placed in a novel polycarbonate cage with a novel aggressor. These aggressors were confined to a small box on one side of the polycarbonate cage, allowing the subject to see, hear, and smell the aggressor, but preventing any direct contact. Testing sessions were recorded in the same manner as the defeat training and were later analyzed by observers blinded to condition to determine the time spent “far” (in the opposite half of the polycarbonate cage from the caged resident aggressor), which we operationally define as social avoidance as described previously70,71, and time spent in social interaction (defined as nose to caged aggressor). Social behavior comparisons were analyzed by Repeated Measures, Two-way ANOVA with Tukey’s post hoc analysis on GraphPad Prism 8.2.0 (GraphPad Software, La Jolla, CA) and effect sizes were calculated. Differences in post hoc analyses were denoted as significant at *p < 0.05. While published reports indicate that the incidence of adverse events associated with these treatments are extremely unlikely72, hamsters were monitored daily throughout treatment and testing by the experimenters and the Department of Animal Resources staff, as well as veterinary technicians or veterinarians to assess coat quality and to make sure that food and water intake remained constant. Additionally, experimenters carefully observed all hamsters during each agonistic encounter for coprophagia and for any injury. No coprophagia or tissue damage occurred during training or testing.
Fecal collection and microbiota composition analysis by 16S rRNA gene sequencing
Fresh fecal samples were collected just before the beginning of the active (dark) phase of the daily light:dark cycle at three time points: 1) prior to the initial defeat (baseline samples), 2) 24 h after the acute defeat (acute defeat samples), and 3) 24 h after the final defeat (repeated defeat sample) to assess the microbial community before any stress, after one bout of social defeat, and after repeated bouts of social defeat. To avoid additional stress to the animal, hamsters were transferred into a clean cage and fecal samples were collected from the bedding approximately 1 h later. Samples were collected in RNase-free microcentrifuge tubes and were immediately frozen and stored at − 80 °C until further processing.
Characterization of microbial communities was performed by 16S rRNA gene sequencing as previously described32,73. Briefly, extracted DNA was used to construct sequencing libraries according to Illumina’s “16 S Metagenomic Sequencing Library Preparation” guide (Part # 15044223 Rev. B), with the exception of using Qiagen HotStar MasterMix for the first PCR (“amplicon PCR”) and halving reagent volumes for the second PCR (“index PCR”). The template specific primers were (without the overhang adapter sequence) the following: forward (5′-CCTACGGGNGGCWGCAG-3′) and reverse (5′-GACTACHVGGGTATCTAATCC-3′), targeting the V3–V4 hypervariable region74 specific to bacterial organisms and generating a fragment of around 460 bp. The first PCR (“amplicon PCR”) was carried out for 25 cycles with annealing temperatures of 55 °C. Diluted pooled samples were loaded on an Illumina MiSeq and sequenced using a 500-cycle (paired-end sequencing configuration of 2 × 250 bp) MiSeq Reagent Kit v3.
16S rRNA gene sequencing analysis
The sequences were demultiplexed and quality filtered using the Quantitative Insights Into Microbial Ecology 2 (QIIME 2, version 3.5.5) software package75. Forward and reverse Illumina reads were joined using the fastq-join method. We used the QIIME 2 default parameters for quality filtering75. Sequences were clustered using amplicon sequencing variant tables with Deblur76. Clusters were then classified taxonomically using the Greengenes reference database, Version 13.577. Clusters that did not match any Greengenes Operational taxonomic units (OTUs) were kept. A single representative sequence for each OTU was aligned and a phylogenetic tree was built using FastTree78.
The phylogenetic tree described above was used to assess beta and alpha diversity79,80. Unweighted UniFrac81 was used to compute distances and to measure beta diversity between groups using rarefied OTU table count. Principal coordinates analysis (PCoA) plots were used to further assess and visualize beta diversity. Groups were compared for distinct clustering using PERMANOVA method using vegan R-package through QIIME 2. The phylogeny-based metric, phylogenetic diversity whole tree (PD whole tree) measurement and the non phylogeny-based metric, Observed OTUs, were determined with QIIME 2. Kruskal–Wallis with Dunn’s multiple comparison test was used to determine differences among groups on GraphPad Prism 8.2.0 (GraphPad Software, La Jolla, CA). Lastly, LEfSE (Linear Discriminate Analysis Effect Size) was used to compare abundance of specific taxa between groups82.
Analysis of fecal samples for probiotic strains via quantitative PCR
The concentrations of the specific bacterial strains fed to the hamsters was measured from fecal samples using qPCR methodology and primers as described in detail in Myles et al.42.
Multiplex assay procedure
24 h after the final defeat and immediately following behavior testing, hamsters were briefly anesthetized with isoflurane and euthanized by cervical dislocation. Trunk blood was collected and allowed to clot at room temperature for 2 h. After 2 h, blood was centrifuged for 20 min at 2000×g to obtain serum, which was immediately frozen and stored at − 80 °C.
A Bio-Plex Pro™ Rat Cytokine 23-Plex Assay was conducted on hamster serum using a fluorescent bead-based instrument Bio-Plex 200 (Bio-Rad, Hercules, CA). Bio-Plex instruments were validated using a Bio-Plex Validation kit 24 h prior to conducting the assay, and instruments were calibrated immediately prior to performing the assay using a Bio-Plex Calibration Kit. The assay was conducted per the manufacturer’s protocol using the recommended sample dilution (4×) and standard curve concentrations. All samples and standards were assayed in duplicates. This multiplex technology uses “xMAP”-based microspheres to detect protein concentrations of target analytes. The concentration of each analyte was determined by Bio-Plex Manager, and the analyte concentrations were compared across groups by ANOVA with Tukey’s post hoc comparisons or Kruskal–Wallis with Dunn’s multiple comparisons test on GraphPad Prism 8.2.0 (GraphPad Software, La Jolla, CA). A rat assay was chosen because no equivalent multiplex assay exists for hamster. Notably, most of the sequences for the analytes in the multi-plex assay share a 77–90% homology with hamster sequences, suggesting a high likelihood of cross-reactivity with hamster proteins.
Data availability
All raw sequence data resulting from the 16S rRNA gene sequencing are available on NCBI under accession number PRJNA669011.
References
Eckburg, P. B. et al. Diversity of the human intestinal microbial flora. Science 308, 1635–1638. https://doi.org/10.1126/science.1110591 (2005).
Cryan, J. F. et al. The microbiota-gut-brain axis. Physiol. Rev. 99, 1877–2013. https://doi.org/10.1152/physrev.00018.2018 (2019).
Scriven, M., Dinan, T. G., Cryan, J. F. & Wall, M. Neuropsychiatric disorders: Influence of gut microbe to brain signalling. Diseases 6, 77. https://doi.org/10.3390/diseases6030078 (2018).
Sommer, F. & Backhed, F. The gut microbiota–masters of host development and physiology. Nat. Rev. Microbiol. 11, 227–238. https://doi.org/10.1038/nrmicro2974 (2013).
Hill, M. J. Intestinal flora and endogenous vitamin synthesis. Eur. J. Cancer Prev. 6(Suppl 1), S43-45 (1997).
Hrncir, T., Stepankova, R., Kozakova, H., Hudcovic, T. & Tlaskalova-Hogenova, H. Gut microbiota and lipopolysaccharide content of the diet influence development of regulatory T cells: Studies in germ-free mice. BMC Immunol. 9, 65. https://doi.org/10.1186/1471-2172-9-65 (2008).
Tlaskalova-Hogenova, H. et al. Commensal bacteria (normal microflora), mucosal immunity and chronic inflammatory and autoimmune diseases. Immunol. Lett. 93, 97–108. https://doi.org/10.1016/j.imlet.2004.02.005 (2004).
Dinan, T. G. & Cryan, J. F. Regulation of the stress response by the gut microbiota: Implications for psychoneuroendocrinology. Psychoneuroendocrinology 37, 1369–1378. https://doi.org/10.1016/j.psyneuen.2012.03.007 (2012).
Cryan, J. F. & Omahony, S. The microbiome-gut-brain axis: From bowel to behavior. Neurogastroenterol. Motil. 23, 187–192 (2011).
Bercik, P. & Collins, S. M. The effects of inflammation, infection and antibiotics on the microbiota-gut-brain axis. Adv. Exp. Med. Biol. 817, 279–289. https://doi.org/10.1007/978-1-4939-0897-4_13 (2014).
Martin, C. R. & Mayer, E. A. Gut-brain axis and behavior. Nestle Nutr. Inst. Worksh. Ser. 88, 45–53. https://doi.org/10.1159/000461732 (2017).
Mao, J. H. et al. Genetic and metabolic links between the murine microbiome and memory. Microbiome 8, 53. https://doi.org/10.1186/s40168-020-00817-w (2020).
Brown, G. W. & Prudo, R. Psychiatric disorder in a rural and an urban population: 1. Aetiology of depression. Psychol. Med 11, 581–599 (1981).
Kessler, R. C. The effects of stressful life events on depression. Annu. Rev. Psychol. 48, 191–214. https://doi.org/10.1146/annurev.psych.48.1.191 (1997).
Bjorkqvist, K. Social defeat as a stressor in humans. Physiol. Behav. 73, 435–442 (2001).
Huhman, K. L. Social conflict models: Can they inform us about human psychopathology?. Horm. Behav. 50, 640–646. https://doi.org/10.1016/j.yhbeh.2006.06.022 (2006).
Foster, M. T., Solomon, M. B., Huhman, K. L. & Bartness, T. J. Social defeat increases food intake, body mass, and adiposity in Syrian hamsters. Am. J. Physiol. Regul. Integr. Comp. Physiol. 290, R1284-1293. https://doi.org/10.1152/ajpregu.00437.2005 (2006).
Solomon, M. B., Foster, M. T., Bartness, T. J. & Huhman, K. L. Social defeat and footshock increase body mass and adiposity in male Syrian hamsters. Am. J. Physiol. Regul. Integr. Comp. Physiol. 292, R283-290. https://doi.org/10.1152/ajpregu.00330.2006 (2007).
Kinn, A. M. et al. A double exposure to social defeat induces sub-chronic effects on sleep and open field behaviour in rats. Physiol. Behav. 95, 553–561 (2008).
Shively, C. A. Social subordination stress, behavior, and central monoaminergic function in female cynomolgus monkeys. Biol. Psychiatry 44, 882–891 (1998).
Hammels, C. et al. Differential susceptibility to chronic social defeat stress relates to the number of Dnmt3a-immunoreactive neurons in the hippocampal dentate gyrus. Psychoneuroendocrinology 51, 547–556. https://doi.org/10.1016/j.psyneuen.2014.09.021 (2015).
Agid, O., Kohn, Y. & Lerer, B. Environmental stress and psychiatric illness. Biomed. Pharmacother. 54, 135–141. https://doi.org/10.1016/s0753-3322(00)89046-0 (2000).
Lederbogen, F. et al. City living and urban upbringing affect neural social stress processing in humans. Nature 474, 498–501. https://doi.org/10.1038/nature10190 (2011).
Long-Smith, C. et al. Microbiota-gut-brain axis: New therapeutic opportunities. Annu. Rev. Pharmacol. Toxicol. 60, 477–502. https://doi.org/10.1146/annurev-pharmtox-010919-023628 (2020).
Dinan, T. G. & Cryan, J. F. Brain-gut-microbiota axis and mental health. Psychosom. Med. 79, 920–926. https://doi.org/10.1097/psy.0000000000000519 (2017).
Maltz, R. M. et al. Prolonged restraint stressor exposure in outbred CD-1 mice impacts microbiota, colonic inflammation, and short chain fatty acids. PLoS ONE 13, e0196961. https://doi.org/10.1371/journal.pone.0196961 (2018).
Lyte, M., Vulchanova, L. & Brown, D. R. Stress at the intestinal surface: Catecholamines and mucosa-bacteria interactions. Cell Tissue Res. 343, 23–32. https://doi.org/10.1007/s00441-010-1050-0 (2011).
Holder, M. K. et al. Dietary emulsifiers consumption alters anxiety-like and social-related behaviors in mice in a sex-dependent manner. Sci. Rep. 9, 172. https://doi.org/10.1038/s41598-018-36890-3 (2019).
Dinan, T. G., Stilling, R. M., Stanton, C. & Cryan, J. F. Collective unconscious: hoW gut microbes shape human behavior. J. Psychiatr. Res. 63, 1–9. https://doi.org/10.1016/j.jpsychires.2015.02.021 (2015).
Parashar, A. & Udayabanu, M. Gut microbiota regulates key modulators of social behavior. Eur. Neuropsychopharmacol. 26, 78–91. https://doi.org/10.1016/j.euroneuro.2015.11.002 (2016).
Chassaing, B. et al. Dietary emulsifiers impact the mouse gut microbiota promoting colitis and metabolic syndrome. Nature 519, 92–96. https://doi.org/10.1038/nature14232 (2015).
Partrick, K. A. et al. Acute and repeated exposure to social stress reduces gut microbiota diversity in Syrian hamsters. Behav. Brain Res. 345, 39–48. https://doi.org/10.1016/j.bbr.2018.02.005 (2018).
Dinan, T. G., Stanton, C. & Cryan, J. F. Psychobiotics: A novel class of psychotropic. Biol. Psychiatry 74, 720–726. https://doi.org/10.1016/j.biopsych.2013.05.001 (2013).
Huhman, K. L. et al. Conditioned defeat in male and female Syrian hamsters. Horm. Behav. 44, 293–299 (2003).
McCann, K. E., Sinkiewicz, D. M., Rosenhauer, A. M., Beach, L. Q. & Huhman, K. L. Transcriptomic analysis reveals sex-dependent expression patterns in the basolateral amygdala of dominant and subordinate animals after acute social conflict. Mol. Neurobiol. 56, 3768–3779. https://doi.org/10.1007/s12035-018-1339-7 (2019).
36Albers, H. E., Huhman, K. L. & Meisel, R. L. in Hormones, Brain and Behavior (eds Arthur P. Arnold, Susan E. Fahrbach, Anne M. Etgen, & Robert T. Rubin) 393–433 (Academic Press, London, 2002).
Ait-Belgnaoui, A. et al. Bifidobacterium longum and Lactobacillus helveticus synergistically suppress stress-related visceral hypersensitivity through hypothalamic–pituitary–adrenal axis modulation. J. Neurogastroenterol. Motil. 24, 138–146. https://doi.org/10.5056/jnm16167 (2018).
Ait-Belgnaoui, A. et al. Probiotic gut effect prevents the chronic psychological stress-induced brain activity abnormality in mice. Neurogastroenterol. Motil. 26, 510–520. https://doi.org/10.1111/nmo.12295 (2014).
Messaoudi, M. et al. Assessment of psychotropic-like properties of a probiotic formulation (Lactobacillus helveticus R0052 and Bifidobacterium longum R0175) in rats and human subjects. Br. J. Nutr. 105, 755–764. https://doi.org/10.1017/s0007114510004319 (2011).
Arseneault-Breard, J. et al. Combination of Lactobacillus helveticus R0052 and Bifidobacterium longum R0175 reduces post-myocardial infarction depression symptoms and restores intestinal permeability in a rat model. Br. J. Nutr. 107, 1793–1799. https://doi.org/10.1017/s0007114511005137 (2012).
Kazemi, A., Noorbala, A. A., Azam, K., Eskandari, M. H. & Djafarian, K. Effect of probiotic and prebiotic vs placebo on psychological outcomes in patients with major depressive disorder: A randomized clinical trial. Clin. Nutr. 38, 522–528. https://doi.org/10.1016/j.clnu.2018.04.010 (2019).
Myles, E. M. et al. Guidelines for best practice in placebo-controlled experimental studies on probiotics in rodent animal models. Benef. Microbes 11, 245–254. https://doi.org/10.3920/BM2019.0144 (2020).
Liu, W.-H. et al. Alteration of behavior and monoamine levels attributable to Lactobacillus plantarum PS128 in germ-free mice. Behav. Brain Res. 298, 202–209 (2016).
Pirbaglou, M. et al. Probiotic supplementation can positively affect anxiety and depressive symptoms: A systematic review of randomized controlled trials. Nutr. Res. 36, 889–898. https://doi.org/10.1016/j.nutres.2016.06.009 (2016).
Bravo, J. A. et al. Ingestion of Lactobacillus strain regulates emotional behavior and central GABA receptor expression in a mouse via the vagus nerve. Proc. Natl. Acad. Sci. U A 108, 16050–16055. https://doi.org/10.1073/pnas.1102999108 (2011).
O’Mahony, S. M., Hyland, N. P., Dinan, T. G. & Cryan, J. F. Maternal separation as a model of brain-gut axis dysfunction. Psychopharmacology 214, 71–88. https://doi.org/10.1007/s00213-010-2010-9 (2011).
Maehata, H. et al. Heat-killed Lactobacillus helveticus strain MCC1848 confers resilience to anxiety or depression-like symptoms caused by subchronic social defeat stress in mice. Biosci. Biotechnol. Biochem. 83, 1239–1247. https://doi.org/10.1080/09168451.2019.1591263 (2019).
Bharwani, A., Mian, M. F., Surette, M. G., Bienenstock, J. & Forsythe, P. Oral treatment with Lactobacillus rhamnosus attenuates behavioural deficits and immune changes in chronic social stress. BMC Med. 15, 7 (2017).
Reber, S. O. et al. Immunization with a heat-killed preparation of the environmental bacterium Mycobacterium vaccae promotes stress resilience in mice. Proc. Natl. Acad. Sci. USA 113, E3130-3139. https://doi.org/10.1073/pnas.1600324113 (2016).
Warda, A. K. et al. Heat-killed lactobacilli alter both microbiota composition and behaviour. Behav. Brain Res. 362, 213–223. https://doi.org/10.1016/j.bbr.2018.12.047 (2019).
Simpson, C. A., Mu, A., Haslam, N., Schwartz, O. S. & Simmons, J. G. Feeling down? A systematic review of the gut microbiota in anxiety/depression and irritable bowel syndrome. J. Affect Disord. 266, 429–446. https://doi.org/10.1016/j.jad.2020.01.124 (2020).
Pascale, A., Marchesi, N., Govoni, S. & Barbieri, A. Targeting the microbiota in pharmacology of psychiatric disorders. Pharmacol Res. 157, 104856. https://doi.org/10.1016/j.phrs.2020.104856 (2020).
Laudisi, F. et al. The food additive maltodextrin promotes endoplasmic reticulum stress-driven mucus depletion and exacerbates intestinal inflammation. Cell Mol. Gastroenterol. Hepatol. 7, 457–473. https://doi.org/10.1016/j.jcmgh.2018.09.002 (2019).
Thompson, R. S. et al. Dietary prebiotics and bioactive milk fractions improve NREM sleep, enhance REM sleep rebound and attenuate the stress-induced decrease in diurnal temperature and gut microbial alpha diversity. Front. Behav. Neurosci. 10, 240–240. https://doi.org/10.3389/fnbeh.2016.00240 (2017).
Zänkert, S., Kudielka, B. M. & Wüst, S. Effect of sugar administration on cortisol responses to acute psychosocial stress. Psychoneuroendocrinology 115, 104607. https://doi.org/10.1016/j.psyneuen.2020.104607 (2020).
Arnold, A. R. & Chassaing, B. Maltodextrin, modern stressor of the intestinal environment. Cell. Mol. Gastroenterol. Hepatol. 7, 475–476. https://doi.org/10.1016/j.jcmgh.2018.09.014 (2019).
Myles, E. M. et al. Supplementation with combined Lactobacillus helveticus R0052 and Bifidobacterium longum R0175 across development reveals sex differences in physiological and behavioural effects of western diet in Long-Evans rats. Microorganisms https://doi.org/10.3390/microorganisms8101527 (2020).
Ramakrishna, B. S. Role of the gut microbiota in human nutrition and metabolism. J. Gastroenterol. Hepatol. 28(Suppl 4), 9–17. https://doi.org/10.1111/jgh.12294 (2013).
Yirmiya, R. et al. Effects of antidepressant drugs on the behavioral and physiological responses to lipopolysaccharide (LPS) in rodents. Neuropsychopharmacology 24, 531. https://doi.org/10.1016/S0893-133X(00)00226-8 (2001).
Hannestad, J., DellaGioia, N. & Bloch, M. The effect of antidepressant medication treatment on serum levels of inflammatory cytokines: A meta-analysis. Neuropsychopharmacology 36, 2452–2459. https://doi.org/10.1038/npp.2011.132 (2011).
Hodes, G. E. et al. Individual differences in the peripheral immune system promote resilience versus susceptibility to social stress. Proc. Natl. Acad. Sci. USA 111, 16136–16141. https://doi.org/10.1073/pnas.1415191111 (2014).
Zhang, J. C. et al. Blockade of interleukin-6 receptor in the periphery promotes rapid and sustained antidepressant actions: A possible role of gut-microbiota-brain axis. Transl. Psychiatry 7, e1138. https://doi.org/10.1038/tp.2017.112 (2017).
Monje, F. J. et al. Constant darkness induces IL-6-dependent depression-like behavior through the NF-κB signaling pathway. J. Neurosci. 31, 9075–9083 (2011).
Simen, B. B., Duman, C. H., Simen, A. A. & Duman, R. S. TNFα signaling in depression and anxiety: Behavioral consequences of individual receptor targeting. Biol. Psychiatry 59, 775–785 (2006).
Goshen, I. et al. Brain interleukin-1 mediates chronic stress-induced depression in mice via adrenocortical activation and hippocampal neurogenesis suppression. Mol. Psychiatry 13, 717 (2008).
Audy, J., Mathieu, O., Belvis, J. & Tompkins, T. A. Transcriptomic response of immune signalling pathways in intestinal epithelial cells exposed to lipopolysaccharides, Gram-negative bacteria or potentially probiotic microbes. Benef. Microbes 3, 273–286. https://doi.org/10.3920/bm2012.0027 (2012).
MacPherson, C. W., Shastri, P., Mathieu, O., Tompkins, T. A. & Burguière, P. Genome-wide immune modulation of TLR3-mediated inflammation in intestinal epithelial cells differs between single and multi-strain probiotic combination. PLoS ONE 12, e0169847. https://doi.org/10.1371/journal.pone.0169847 (2017).
Ross, A. P. et al. Social housing and social isolation: Impact on stress indices and energy balance in male and female Syrian hamsters (Mesocricetus auratus). Physiol. Behav. 177, 264–269. https://doi.org/10.1016/j.physbeh.2017.05.015 (2017).
Tillmann, S. & Wegener, G. Syringe-feeding as a novel delivery method for accurate individual dosing of probiotics in rats. Benef. Microbes 9, 311–315. https://doi.org/10.3920/bm2017.0127 (2018).
McCann, K. E. & Huhman, K. L. The effect of escapable versus inescapable social defeat on conditioned defeat and social recognition in Syrian hamsters. Physiol. Behav. 105, 493–497. https://doi.org/10.1016/j.physbeh.2011.09.009 (2012).
McCann, K. E., Bicknese, C. N., Norvelle, A. & Huhman, K. L. Effects of inescapable versus escapable social stress in Syrian hamsters: The importance of stressor duration versus escapability. Physiol. Behav. 129, 25–29 (2014).
Foster, L. M., Tompkins, T. A. & Dahl, W. J. A comprehensive post-market review of studies on a probiotic product containing Lactobacillus helveticus R0052 and Lactobacillus rhamnosus R0011. Benef. Microbes 2, 319–334. https://doi.org/10.3920/BM2011.0032 (2011).
Macpherson, A., Khoo, U., Forgacs, I., Philpott-Howard, J. & Bjarnason, I. Mucosal antibodies in inflammatory bowel disease are directed against intestinal bacteria. Gut 38, 365–375 (1996).
Klindworth, A. et al. Evaluation of general 16S ribosomal RNA gene PCR primers for classical and next-generation sequencing-based diversity studies. Nucleic Acids Res. 41, e1–e1. https://doi.org/10.1093/nar/gks808 (2013).
Bolyen, E. et al. Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nat. Biotechnol. 37, 852–857. https://doi.org/10.1038/s41587-019-0209-9 (2019).
Amir, A. et al. Deblur rapidly resolves single-nucleotide community sequence patterns. mSystems https://doi.org/10.1128/mSystems.00191-16 (2017).
McDonald, D. et al. An improved Greengenes taxonomy with explicit ranks for ecological and evolutionary analyses of bacteria and archaea. ISME J. 6, 610–618. https://doi.org/10.1038/ismej.2011.139 (2012).
Price, M. N., Dehal, P. S. & Arkin, A. P. FastTree: Computing large minimum evolution trees with profiles instead of a distance matrix. Mol. Biol. Evol. 26, 1641–1650. https://doi.org/10.1093/molbev/msp077 (2009).
Mackos, A. R., Maltz, R. & Bailey, M. T. The role of the commensal microbiota in adaptive and maladaptive stressor-induced immunomodulation. Horm. Behav. 88, 70–78. https://doi.org/10.1016/j.yhbeh.2016.10.006 (2017).
Sekirov, I., Russell, S. L., Antunes, L. C. & Finlay, B. B. Gut microbiota in health and disease. Physiol. Rev. 90, 859–904. https://doi.org/10.1152/physrev.00045.2009 (2010).
Lozupone, C. & Knight, R. UniFrac: A new phylogenetic method for comparing microbial communities. Appl. Environ. Microbiol. 71, 8228–8235. https://doi.org/10.1128/aem.71.12.8228-8235.2005 (2005).
Segata, N. et al. Metagenomic biomarker discovery and explanation. Genome Biol. 12, R60. https://doi.org/10.1186/gb-2011-12-6-r60 (2011).
Acknowledgements
The authors acknowledge the helpful contribution of Thomas Tompkins, Chad MacPherson, and Amanda Piano at Rosell Institute for Probiotics and Microbiome. This work was supported by Lallemand Health Solutions, the National Institutes of Health R01MH062044 awarded to K.L.H., and a GSU Brains and Behavior Fellowship awarded to K.A.P. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health, the Brains and Behavior Program, the Center for Behavioral Neuroscience, Lallemand Health Solutions, or Georgia State University.
Author information
Authors and Affiliations
Contributions
K.P. ran all aspects of the study, analyzed data, and wrote the manuscript. A.R., A.A,. N.R., L.J., M.L., S.A. substantively contributed to data collection. J.A. oversaw the 16s rRNA gene sequencing and analysis and contributed to the manuscript. B.C. and K.L. H. oversaw the study and analysis and wrote the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Partrick, K.A., Rosenhauer, A.M., Auger, J. et al. Ingestion of probiotic (Lactobacillus helveticus and Bifidobacterium longum) alters intestinal microbial structure and behavioral expression following social defeat stress. Sci Rep 11, 3763 (2021). https://doi.org/10.1038/s41598-021-83284-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-83284-z
This article is cited by
-
Effect of dietary supplementation with Lactobacillus helveticus R0052 on seizure thresholds and antiseizure potency of sodium valproate in mice
Psychopharmacology (2024)
-
Metronidazole and ciprofloxacin differentially affect chronic unpredictable mild stress-induced changes in the colon, cecum and ileum microbiota
Applied Microbiology and Biotechnology (2022)
-
Gut–Brain Axis, Neurodegeneration and Mental Health: A Personalized Medicine Perspective
Indian Journal of Microbiology (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.