Abstract
The nutritional integrity of wheat is jeopardized by rapidly rising atmospheric carbon dioxide (CO2) and the associated emergence and enhanced virulence of plant pathogens. To evaluate how disease resistance traits may impact wheat climate resilience, 15 wheat cultivars with varying levels of resistance to Fusarium Head Blight (FHB) were grown at ambient and elevated CO2. Although all wheat cultivars had increased yield when grown at elevated CO2, the nutritional contents of FHB moderately resistant (MR) cultivars were impacted more than susceptible cultivars. At elevated CO2, the MR cultivars had more significant differences in plant growth, grain protein, starch, fructan, and macro and micro-nutrient content compared with susceptible wheat. Furthermore, changes in protein, starch, phosphorus, and magnesium content were correlated with the cultivar FHB resistance rating, with more FHB resistant cultivars having greater changes in nutrient content. This is the first report of a correlation between the degree of plant pathogen resistance and grain nutritional content loss in response to elevated CO2. Our results demonstrate the importance of identifying wheat cultivars that can maintain nutritional integrity and FHB resistance in future atmospheric CO2 conditions.
Similar content being viewed by others
Introduction
Wheat is the most cultivated crop world-wide and accounts for nearly one fifth of all human dietary protein1,2. The nutritional integrity of wheat is endangered by rapidly rising atmospheric carbon dioxide (CO2) and the associated emergence and enhanced virulence of plant pathogens3,4,5. Grain protein and mineral content decline at elevated CO2 in major C3 photosynthetic cereal crops such as wheat, negatively impacting end-use quality and ultimately food security5,6,7,8. Furthermore, climate change is predicted to increase the risk of mycotoxin contamination associated with fungal diseases such as Fusarium Head Blight (FHB)9, and elevated CO2 may provide a strain-specific pathogenic advantage on hosts with greater losses in nutritional content10. FHB outbreaks can substantially diminish grain yield and end-use quality due to sterile florets and withered, mycotoxin contaminated grain kernels11. Cultivating wheat germplasm that are climate resilient and possess some resistance to FHB is a key control strategy for maintaining food safety and security.
Previous studies have shown that wheat grown at elevated CO2, with sufficient water and fertilization, will have substantially increased yields but the grain will contain higher carbohydrate content and lower relative amounts of proteins, minerals, and lipids12,13,14. This effect, often referred to as dilution, is generally caused by an excess accumulation of carbohydrates from enhanced photosynthetic carbon metabolism; however, disproportionate losses in specific nutrients indicate a more complex biological partitioning mechanism may be partially responsible6,13,15,16. Carbohydrate dilution of grain protein results in wheat flour with reduced baking quality, limiting the end-use utility and producing less nutritious, lower value food goods7,17,18.
The impact of elevated CO2 on grain quality is cultivar dependent, and even known adaptive traits, such as high nitrogen use efficiency and improved root vigor, are not sufficient to counteract the effects on grain protein and recover end-use quality19,20. The effects of rising CO2 not only impacts wheat nutritional content but can also increase plant disease susceptibility, particularly to FHB3,21,22,23. The combined adverse effects of elevated CO2 on wheat nutritional content and disease susceptibility is a significant threat to wheat producers and consumers. Currently, plant differential response to elevated CO2 has been studied in a very limited number of cultivars and crops; greater screening efforts could uncover meaningful differences in elevated CO2 response, which could be exploited by plant breeders12. It is vitally important to determine whether FHB resistance is associated with greater grain nutritional losses at elevated CO2 and to identify climate and FHB resilient cultivars.
In spring wheat, moderate resistance to FHB is predominantly derived from the genetic background of the Sumai 3 cultivar, largely due to the Fhb1 Quantitative Trait Locus (QTL). Although, there are other sources of resistance (Fhb2, Fhb4, Fhb5, Fhb7), there is no combination of these, or other genes, that provide complete resistance to FHB24. While individual QTLs provide a degree of resistance, the introgression of Fhb1, Fhb2, and Fhb5, as well as the stacking of two or more FHB QTLs, has been associated with reduced grain protein content25,26. We recently observed that the difference in grain protein content between wheat grown at 400 ppm CO2 versus 800 ppm was significantly greater in the FHB moderately resistant hard red spring wheat cultivar Alsen, which contains the Fhb1 and Fhb5 loci, in comparison to the susceptible cultivar Norm, which does not10. Consequently, we investigated whether FHB resistance factors from the Sumai 3 background could impact grain nutritional content in future atmospheric conditions.
Based on our results with the wheat cultivars Alsen and Norm, we hypothesized that the direct effects of elevated CO2 more severely impact the grain composition of FHB moderately resistant wheat cultivars than susceptible cultivars. To test this hypothesis, six FHB susceptible cultivars and nine moderately resistant cultivars (Table 1) were grown in triplicate experiments at ambient 400 ppm (a[CO2]) or elevated 1000 ppm (e[CO2]), and then evaluated for differences in plant development, physiology, yield, and nutritional composition. Differences between wheat cultivars and FHB resistance scores (Table 1) were identified to evaluate whether the presence of FHB resistance genes will be a nutritional liability for wheat cultivars in the future.
Materials and methods
Wheat cultivars and growth conditions
FHB susceptible hard red spring wheat cultivars with FHB scores ranging from 5 to 9, Linkert27, MN00269, MN10281-1-98, MN11492-6, Ulen28, Wheaton29, and FHB moderately resistant (MR) cultivars with FHB scores ranging from 2 to 4, (Bolles30, Glenn31, Lang-MN32, MN08172-3-10, MN11394-6, RB0733, Rollag34, Sabin35, Shelly36) (Table 1), were grown in controlled environment growth chambers according to the methods outlined in Hay, et al.10. The relative FHB resistance of wheat cultivars is based on evaluations in 6 or more inoculated, misted field environments for FHB traits: incidence, severity, Visual Scabby Kernels, and DON contamination. New cultivars are compared against cultivars with an established 1–9 FHB rating for classification32. For the cultivation and harvest of wheat, all local and national regulations were followed, and all relevant permissions were acquired. No genetically modified plant cultivars were examined in the study.
The growth chambers were blocked into pairs, with each block containing a chamber set to ambient [CO2] (420 ± 20 ppm, a[CO2]) and a chamber set to 1000 ± 20 ppm [CO2] (e[CO2]). Experiments were performed in triplicate between 2018 and 2019. The wheat cultivars were grown in a completely random block design. For each cultivar, five plants were grown in a 20 × 15-cm plastic pot, filled with approximately 4 L of SunGrow Horticulture potting mix (Agawam, MA, U.S.A.). Growth chambers were programmed at 25/23 °C day/night, respectively, a 14 h photoperiod at 550 μmol m−2 s−1 photosynthetic photon flux density, and 50–60% relative humidity. Plants were watered daily and were fertilized biweekly with soluble Peters 20–20–20 nutrient supplement (The Scotts Company, Marysville, OH, U.S.A.) until anthesis. Plant positions were randomized after each watering. The developmental timings of heading (Feekes 10.2) and flowering (Feekes 10.5.2) were monitored, and tiller number and height were measured once plants reached physiological maturity (Feekes 11.3). After ripening (Feekes 11.4), grain was harvested for yield and compositional evaluations. Wheat grain moisture and protein were determined by near-infrared spectroscopy (NIR), using a DA 7250 NIR analyzer (Perten Instruments, Springfield, IL). Samples were then milled into whole wheat flour using a Retsch ZM200 ultra centrifugal mill (Retsch, Haan, Germany), equipped with a 12-tooth stainless steel rotator spinning at 10,000 rpm with a 0.75 mm stainless steel screen.
Carbohydrate analysis
For each growth experiment, each sample was prepared in triplicate (i.e. 3 replicates × 3 growth experiments × 15 cultivars × 2 [CO2] = 270 samples). Water soluble carbohydrate content (WSC) for each sample was determined in triplicate by high-performance anion-exchange chromatography-pulsed amperometric detection (HPAEC-PAD), following the protocol outlined in Hay, et al.10. Grain starch and fructan content was determined in triplicate by using a sequential procedure as outlined by Dien, et al.37. Grain soluble carbohydrates were extracted from 100 mg of the whole wheat flour with 5 mL of 80% v/v ethanol at 60 °C for 1 h. Samples were then transferred to a 4 °C refrigerator and stored for 16–22 h before centrifugation at 1925× g for 15 min. The fructan concentration of the supernatant was determined by hydrolyzing samples to fructose for colorimetric measurement of monomeric fructose38. To hydrolyze starch to glucose, the alcohol-insoluble residue was treated with 6 µg, (3.5 Units where one unit will liberate 1.0 mg of maltose from starch in 3 min at pH 6.9 at 20 °C) of heat-stable α-amylase (from Bacillus licheniformis, Sigma-Aldrich, Saint Louis, MO) for 1 h at 90 °C in 5 mL of 0.1 M acetate buffer, pH 5. The sample was subsequently treated with 600 µg (8.6 Units where one unit will liberate 1.0 mg of glucose from starch in 3 min at pH 4.5 at 55 °C.) amyloglucosidase (from Aspergillus niger, Roche, Mannheim, Germany) for 3 h at 60 °C. Finally, 20 mL of each sample was analyzed in by a high-performance liquid chromatography (Ultimate 3000 (Thermo Scientific, Waltham, MA)) for glucose using a refractive index detector through an Aminex-87H column running 5 mM sulfuric acid isocratically at 0.6 mL/min flow rate at 60 °C. Sample temperature was maintained at 4 °C in the autosampler prior to injection.
Fatty acid analysis
The ratios of grain fatty acids were determined by hydrolysis and fatty acid methyl ester formation modified from the procedure of Hartman and Lago39, and analysis by gas chromatography mass spectrometry (GC–MS) in triplicate. Briefly, 60 mg of whole wheat flour was treated with 1 mL of 0.25 M sodium methoxide at 60 °C for 30 min. The sample was cooled to room temperature before 1 mL of hexane and 1 mL of saturated NaCl solution were added and allowed to phase separate without disturbance for 10 min. A 0.5 mL aliquot of the organic layer of each sample was analyzed for fatty acid methyl esters by a Shimadzu GC-2010 Plus GC–MS (Tokyo, Japan), using a capillary column (Supelco SP-2380; 30 m × 0.25 mm). Operating conditions were as follows: Initial oven temperature was set to 130 °C, with a programmed ramp of 20 °C/min to 265 °C which was held for 2.25 min. The split ratio was 100:1 with a column flow rate of 1.2 mL/min and a septum purge rate of 4 mL/min. Injector and detector were set to 250 °C, and hexane was used for control blanks and rinse vials.
Grain mineral content
Select grain mineral content was determined by inductively coupled plasma-optical emission spectroscopy with a Perkin-Elmer 7000DV ICP (Shelton, CT), according to the protocols outlined in Hay, et al.10. All samples were tested in triplicate to determine phosphorous, potassium, magnesium, calcium, manganese, zinc, iron, and copper content.
Wheat stem physical characteristics
Evaluations of wheat stem physical characteristics were performed following the modified protocols of Miller, et al.40. Straw samples from mature wheat plants were equilibrated at 23 °C and 50% RH for one week and cut to lengths of 76 mm from the middle of the 2nd internode region. A 3-Point bend test was conducted on an Instron (Norwood, MA) Model 3300 Controller with a 3365 Frame equipped with a 1 kN load cell using Bluehill Universal software. Samples were centered over supports having a 50.8 mm support span. A circular diameter confirmation was used for the tests. The rate of movement was 10 mm/min with a data sampling rate of 20 ms. Testing was stopped with an event displacement of 25 mm. Internal and external diameters, to determine cross-sectional area, were determined approximately 13 mm from each side of the bend site using a micrometer. The bending stress at material failure was calculated using the Equations41:
where Di is the inner diameter, Do is the outer diameter, Fmax is the absolute resistance of the stem sample to break under-load, Ro is the outer radius, Ri is the inner radius, the span of two Instron supports in cm.
Statistical analyses
Results were evaluated by a generalized linear mixed model analysis of variance, with experimental replicate as a random effect (JMP V15.0), to determine significant differences between cultivars and FHB resistance groups due to the effects of elevated CO2 (α = 0.05). Information on pairwise comparisons are detailed within the table and figure legends. Significant correlations between independent variables (α = 0.05) were evaluated by multivariate analysis and a Pearson correlation (r) was reported, which ranges from − 1 to 1; where 0 represent no correlation, − 1 represents a perfectly negative correlation, and 1 a perfectly positive correlation. Discriminant analysis was performed using a stepwise variable selection (Smallest P to enter ≤ 0.05) and a linear fitting method (JMP V15.0).
Results
Effects of elevated CO2 on plant development, height, and yield
To determine whether changes in grain composition due to growth at elevated CO2 were more severe in FHB MR wheat compared with susceptible, 15 cultivars with varying FHB disease resistance (Table 1) were grown to maturity at a[CO2] and e[CO2]. Wheat height, stem strength, tiller number, and yield were all significantly impacted by growth at e[CO2]. Although plant development was not impacted by elevated CO2, wheat cultivars had distinctly different heading and flowering times (supplemental Table 1). The average plant height increased by 9 cm under e[CO2] (supplemental Fig. 1); however, the height increases were only statistically significant for the MR group (P = 0.043; Fig. 1a).
(a) Differences in plant height by Fusarium head blight (FHB) resistance at ambient CO2 (a[CO2]) and elevated CO2 (e[CO2]). (b) Growth response of moderately resistant (MR) and susceptible wheat cultivars at e[CO2], as determined by differences in plant height at a[CO2] compared with the same cultivars grown at e[CO2]. Different letters represent statistically significant differences determined by a Tukey HSD analysis of variance (P < 0.05); MR (n = 27), and susceptible (n = 18), for each CO2 growth condition. Asterisks (*) denote statistically significant differences (P < 0.05).
There was a significant cultivar × [CO2] interaction (P = 0.016) (supplemental Table 2), and the tallest cultivar, MN08173-3-10, had an average increase in height of 15 cm at e[CO2]. The MR cultivars were taller than susceptible cultivars (Fig. 1a) and showed more vigorous vegetative growth at e[CO2] (Fig. 1b); the % change in plant height for the MR cultivars was significantly greater than susceptible cultivars (P = 0.0002).
Because increases in plant height can negatively impact structural integrity (e.g., lodging), stem strength was evaluated. Stem strength, as determined by bending stress at material failure, was moderately less (6%) in wheat grown at e[CO2] (P = 0.009), but this was mostly driven by a significant cultivar × [CO2] interaction (P = 0.0007) and did not correlate with FHB resistance level or difference in plant height at e[CO2]. While the stem strength of most cultivars was not negatively impacted by growth at e[CO2], the MR cultivars Bolles and RB07 suffered 23% and 25% decreases in stem strength, respectively, at e[CO2].
Though MR wheat was significantly taller than susceptible wheat at e[CO2] (Fig. 1b), this did not provide a yield advantage, as each group had an equivalent increase in grain yield at e[CO2] (Fig. 2). Grain yield per plant was dramatically higher, by 30%, at e[CO2] (P < 0.0001) but there was no significant interaction between CO2 and cultivar or FHB group. The increased yield was predominately due to an additional tiller per plant at e[CO2] (25% increase; P < 0.0001); each plant usually had four tillers at a[CO2], which increased to five tillers per plant at e[CO2]. Yield per tiller had a minor increase of 5% at e[CO2] (P = 0.043). The average seed weight was not impacted by growth at e[CO2], nor significantly different between MR and susceptible groups.
Differences in yield per plant of various Fusarium head blight (FHB) moderately resistant (MR) and susceptible wheat cultivars at ambient CO2 (a[CO2]) and elevated CO2 (e[CO2]), ordered by FHB resistance score at the top of the figure panel. Solid and dashed lines represent the mean yield per plant for each FHB resistance group at a[CO2] and e[CO2], respectively.
Effects of elevated CO2 on grain composition
Growth at e[CO2] significantly impacted grain composition, especially the protein content of grain from FHB resistant wheat cultivars (Fig. 3). While there were differences in protein content among cultivars, the cultivar x [CO2] interaction was not significant (P = 0.07), but there was a significant interaction between the FHB resistance rating × [CO2] (P = 0.031). On average, there was less protein in both MR and susceptible cultivars at e[CO2], but this difference was only significant for the MR cultivars, which had 13% less protein (P = 0.025; Fig. 3a). There was an inverse relationship between the FHB resistance and the change in protein content at e[CO2] (r = − 0.37; P = 0.012; Fig. 3b,c). Furthermore, the overall percent change in protein content at e[CO2] was greater in the MR cultivars compared with susceptible (P = 0.004; Fig. 3c).
Percent grain protein of various Fusarium head blight (FHB) moderately resistant (MR) and susceptible wheat cultivars. Change in protein content determined by evaluating differences in grain protein from wheat grown at elevated CO2 (e[CO2]), compared with wheat grown at ambient (a[CO2]). (a) Grain protein content of susceptible and MR wheat grain grown at a[CO2] and e[CO2], error bars represent standard error. (b) Percent change in protein content at e[CO2] by cultivar and FHB resistance score. Horizonal bars represent group mean protein loss. Wheat cultivars were ordered by FHB resistance score at the top of the figure panel. (c) Percent change in protein content by FHB group. Different letters or asterisks (*) denote statistically significant differences as determined by a Tukey adjusted generalized mixed model analysis of variance (P < 0.05); MR (n = 27), and susceptible (n = 18), for each CO2 growth condition.
While overall the percent change in protein content at e[CO2] was greater in the MR and there was no significant cultivar x [CO2] interaction, there were two notable exceptions in the trend, Rollag and Glenn. The percent change in protein content for Rollag and Glenn, which have a designated resistance score of 3, were on average comparable to the percent change shown for the susceptible cultivars with a score of 6 (Fig. 3b). Although Glenn, which had the greatest level of resilience in protein content, does not contain Fhb1, Bolles which similarly lacks Fhb1 displayed above average change in protein content at e[CO2]. The exacerbated reduction in percent protein content of MR cultivars at e[CO2] was associated with greater starch accumulation (Figs. 3, 4). While there were differences in starch content among cultivars, the cultivar × [CO2] interaction was not significant. However, there was a significant interaction between the degree of FHB resistance × [CO2] (P = 0.014). Again, the susceptible cultivars had no significant change in starch content (3%) when grown at e[CO2], while the MR cultivars had significant accumulation (8%) of starch (P = 0.011; Fig. 4a). The percent change in grain starch content was correlated with increasing FHB resistance (r = 0.39; P = 0.0086; Fig. 4b,c). Furthermore, the overall percent change in starch accumulation at e[CO2] was greater in the MR compared with susceptible cultivars (P = 0.002; Fig. 3c). Changes in protein and starch at e[CO2] were both found to increase with FHB resistance score. Finally, starch and protein content were negatively correlated with one another at e[CO2] (r = − 0.59, P < 0.0001), suggesting that the loss of grain protein was due to dilution by the storage carbohydrate starch.
Percent grain starch accumulation of various Fusarium head blight (FHB) moderately resistant (MR) and susceptible wheat cultivars. Starch accumulation determined by evaluating differences in starch content from wheat grown at elevated CO2. (a) Grain starch content of susceptible and MR wheat grain grown at ambient (a[CO2]) and elevated (e[CO2]), error bars represent standard error. (b) Percent starch accumulation at e[CO2] by cultivar and FHB resistance score. Horizonal bars represent group mean starch accumulation, error bars represent standard error. Wheat cultivars were ordered by FHB resistance score at the top of the figure panel. (c) Percent starch accumulation by FHB group. Different letters or asterisks (*) denote statistically significant differences as determined by a Tukey adjusted generalized mixed model analysis of variance (P < 0.05); MR (n = 27), and susceptible (n = 18), for each CO2 growth condition.
There were substantial differences in grain water soluble carbohydrate (WSC) content among wheat cultivars, but unlike starch, there was no significant effect of elevated CO2 on glucose, fructose, sucrose, or raffinose (supplemental Fig. 2). Only maltose was significantly higher (10%) at e[CO2] (P = 0.015), but there was no significant interaction between [CO2] and cultivar or FHB resistance. The total WSC content evaluated by HPAEC-PAD was not impacted by elevated CO2.
Fructans are storage carbohydrates made up of polymeric fructose of varying chain length. Both MR and susceptible cultivars had higher fructan content at e[CO2] than at a[CO2] (P < 0.0001; Fig. 5a), but MR cultivars had a significantly greater percent change in fructan content (65%) compared with susceptible wheat (37%; Fig. 5b; P = 0.012).
(a) Fructan concentration of susceptible and moderately resistant (MR) wheat grain grown at ambient CO2 (a[CO2]) and elevated CO2 (e[CO2]), error bars represent standard error. (b) Percent change in fructan content at e[CO2] by Fusarium head blight (FHB) resistance group. An asterisk (*) denotes a statistically significant effect of elevated CO2 on Fructan concentration as determined by Tukey adjusted generalized mixed model analysis of variance (P < 0.05); MR (n = 27), and susceptible (n = 18), for each CO2 growth condition.
Fatty acids are typically very minor components in wheat grain, but each fatty acid assayed substantially differed among cultivars. However, there was no significant impact of elevated CO2 on the ratios of fatty acids and there was no significant interaction between cultivar × [CO2] or FHB group (Supplemental Fig. 3). Palmitic acid was slightly higher in the susceptible cultivars at e[CO2] and lower in MR cultivars, but these differences were not statistically significant (P = 0.098).
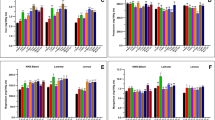
Mineral nutrient content is essential for both plant growth and human nutrition; phosphorus, potassium, magnesium, calcium, manganese, zinc, iron, and copper are all defined as essential dietary nutrients42. Mineral content differed between cultivars and was significantly impacted by growth at e[CO2] (Fig. 6); however, there was no significant cultivar × [CO2] interaction. Growth at e[CO2] resulted in significantly less iron (32%) and copper (13%) in the susceptible cultivars (Fig. 6a; P < 0.01). The MR cultivars had less phosphorus (11%), magnesium (9%), calcium (18%), zinc (13%), iron (32%), and copper (17%) at e[CO2] (Fig. 6b; P < 0.01). There was a significant FHB resistance score × [CO2] interaction for phosphorus (P = 0.005) and magnesium content (P = 0.047); the greater a cultivar’s FHB resistance, the greater the difference in phosphorus (r = − 0.43; P = 0.0039) and magnesium (r = − 0.36; P = 0.0157) contents between ambient vs e[CO2] grown grain. Interestingly, there was more manganese content in both FHB susceptible (12%) and MR cultivars (16%) (Fig. 6; P = 0.0005) at e[CO2].
Mineral content of susceptible (a) and moderately resistant (MR) (b) wheat grain grown at ambient CO2 (a[CO2]) and elevated CO2 (e[CO2]). Error bars represent standard error and asterisks (*, **) denote statistically significant differences in the mineral content of wheat grown at a[CO2] and e[CO2] by Fusarium head blight (FHB) resistance group, as determined by a Tukey adjusted generalized mixed model analysis of variance (P < 0.05, P < 0.01, respectively); MR (n = 27), and susceptible (n = 18), for each CO2 growth condition.
After identifying numerous differences between FHB resistant groups at e[CO2] within the univariate generalized linear model approach, a multivariate discriminant analysis was performed to investigate separation of the variables as a collective entity. The analysis attempts to classify each cultivar to a given FHB group using the previously described results. The goal was to determine how well individual observations can be properly separated into groups and to identify the relative importance of key plant characteristics for FHB resistance level classification/identification. The function coefficients, presented in the Pooled Within Canonical Structure tables for each independent variable (Fig. 7), compare the relative association and influence of each independent variable in determining the discriminant score of the canonical variable (Canonical1).
Discriminant analysis applied to the Fusarium head blight (FHB) susceptible and moderately resistant (MR) cultivars at (a) ambient CO2 (a[CO2]), and (b) elevated CO2 (e[CO2]). Groups are separated along Cannonical1 by the function coefficients of the independent variables presented in each table, found within the figure panel (a,b). Blue objects represent susceptible cultivars, red MR. The inner circle represents the 95% confidence region of the group mean, and the outer circle represents the area containing 50% of the group population. Rays represent the independent variables relative association with the canonical variable along the Canonical1 axis.
At a[CO2], susceptible and MR groups were quite similar and difficult to differentiate, with a misclassification rate of 20% (Fig. 7a). The characteristics with the most discriminating power at a[CO2] were zinc, heading date, calcium, and maltose content, in order of relative importance. The eigenvalue at a[CO2], the amount of variance explained by the associated discriminant function, was only 0.7, and the entropy R2, the measure of fit, was 0.34; properly classifying cultivars into FHB resistance groups at current atmospheric conditions was prone to misclassification.
At e[CO2], susceptible and MR groups separated and were easily differentiated, with a misclassification rate of only 2% (Fig. 7b). The plant characteristics with the most discriminating power at e[CO2] were: plant height, yield per plant, heading date, palmitic acid, yield per tiller, potassium, maltose, calcium, oleic acid, and total WSC, in order of relative importance. The eigenvalue at e[CO2] was 4.5, with an entropy R2 of 0.87; a perfect fit has an entropy R2 of 1. The MR and susceptible cultivars were distinct from one another at e[CO2], and cultivars were accurately classified into FHB resistance groups. The high degree of group separation at e[CO2] (Fig. 7b) is consistent with the markedly different responses of MR and susceptible cultivars to growth at e[CO2]. The results of the discriminant analysis are consistent with the hypothesis of the study, showing that growth at e[CO2] more severely impacted FHB MR cultivars compared with susceptible.
Discussion
In the present study, we demonstrated that the deleterious effects of elevated CO2 on wheat nutrition is more severe for FHB moderately resistant (MR) cultivars. To our knowledge, this is the first report of a correlation between the degree of plant pathogen resistance and grain nutritional content loss in response to elevated CO2. This is of significant importance because future wheat growers may be dissuaded from planting MR cultivars due to reduced end-use quality and marketability from growth at elevated CO2. Therefore, our results reveal the vital importance to identify wheat cultivars that can maintain nutritional integrity and FHB resistance in future climate conditions.
Currently, only a few QTLs have been validated to confer stable moderate FHB resistance, but no trait, or combination of traits, providing complete FHB resistance has been discovered24,43. The introgression of FHB resistance from Sumai 3 and other Chinese germplasm, the predominate sources of FHB resistance in North American wheat, may have resulted in the unintended reduction of nutritional content, which is further exacerbated at e[CO2].
Reduced grain protein has also been associated with the introgression of the FHB resistance QTLs Fhb1 and Fhb5, as well as the stacking of other Fhb QTLs; these decreases in protein content were found to be dependent on the donor and recipient genetic background25,26. However, sources of Fhb1 from various Chinese donor cultivars were found to have no negative impact on agronomic traits in winter wheat, but the degree of FHB resistance in the recipient cultivar varied significantly, likely due to additional minor QTLs introgressed from the donor44. Improved FHB resistance is also associated with a number of minor loci from resistant donor lines, mostly genes expressing a variety of plant defense related kinases, nucleotide-binding and leucine rich repeats45,46. The introgression of these disease resistant traits from non-adapted donors can cause significant linkage drag, negatively affecting agronomic traits46,47. Nevertheless, it will be essential for breeders to know if Fhb1 is associated with the more severe loss in grain protein content at elevated CO2. The limited number of MR cultivars without Fhb1 used in this study does not allow for accurate correlations between the Fhb1 QTL and the quality penalty under elevated CO2. In future studies we are addressing this critical question utilizing near isogenic lines with and without the Fhb1 QTL. It may be necessary to use FHB resistance genes not derived from the Sumai 3 cultivar, such as Fhb7, which encodes a glutathione-S-transferase to detoxify trichothecene mycotoxins43. This recently identified source of resistance may provide plant breeders an alternative method of protecting wheat from FHB while improving nutritional climate resilience.
Disease resistance traits often have trade-offs, negative pleiotropic effects on important agronomic characteristics such as yield, nutritional quality, and resistance to other diseases or pests48. The allocation of plant resources for self-protection necessarily reduces available resources for growth or reproduction, and even the presence of resistance genes generally results in a loss of fitness49. The decline of grain nutritional content in the MR cultivars at elevated CO2 may be due to this defense trade-off paradigm, particularly as FHB resistance is often tightly associated with traits which reduce crop performance and grain quality, and the introgression of these traits may enhance disease resistance at the cost of reduced yield and less grain protein26,50.
However, breeding efforts currently manage to maintain acceptable gain quality standards in MR cultivars. Rising CO2 may exacerbate the negative effects on agronomic traits, making cultivar selection to maintain grain quality and FHB resistance more difficult in the future. The significant decrease in grain protein content at e[CO2] in the MR cultivars (Fig. 3) is highly concerning, as FHB resistance traits may become a nutritional liability in future climate conditions. Farmer cultivar selection is typically focused on yield, grain quality, loss avoidance, and marketability. If MR cultivars have declining grain quality and marketability with rising CO2, farmers may abandon disease resistant lines for more susceptible cultivars, significantly increasing the risk of FHB outbreaks.
Furthermore, elevated atmospheric CO2 may increase MR wheat susceptibility to FHB and mycotoxin contamination due to the substantial changes in nutrient composition. Starch is a known inducer of trichothecene mycotoxins by Fusarium fungal pathogens, and changes in grain nutrient composition at elevated CO2 have been observed to increase pathogen trichothecene mycotoxin production10,51. Furthermore, virulence-associated genes are often linked to major nitrogen regulatory transcription factors and toxin production is considerably influenced by the available nitrogen of the host52,53. However, FHB resistance is also significantly correlated with increased plant height54,55,56, thus further research is needed to empirically determine the full effects of these physiological changes on host susceptibility to FHB.
Previous reports have shown that wheat grown at elevated CO2 has higher grain carbohydrate content and lower relative amounts of proteins, minerals, and lipids13,14. The decline of grain protein in the MR cultivars (Fig. 3) was directly correlated with the accumulation of storage carbohydrates, starch, and fructans (Figs. 4, 5), which is consistent with the dilution effect of excess carbohydrates. Increased nitrogen application may reduce these dilution effects. However, complex biological partitioning mechanisms are also likely involved6,13,15,16. The uniform decrease in iron concentrations across all FHB groups grown at e[CO2], regardless of starch accumulation, suggests a more complex metabolic response.
The observed increased grain yield per plant at elevated CO2 (Fig. 2) was consistent with previous reports57,58. Additionally, global wheat yields are predicted to significantly increase with rising CO2, though these gains are highly dependent on growing temperatures, water availability, and nitrogen fertilization5,12,59. Furthermore, typical high planting densities in wheat may increase plant height and lodging risk due to increased intra and inter-plant competition60. Increased nitrogen fertilization to maintain grain protein and improve yield at elevated CO2 was found to be only somewhat effective; however, increased nitrogen fertilizer rates did increase internode length, plant height, and ultimately lodging risk61,62. Attempts to ameliorate grain nutritional losses with increased fertilization may simply lead to more lodging, thus a better focus may be identifying climate resilient wheat cultivars.
Increased wheat plant height can lead to a greater risk of crop lodging, stem bending and crop collapse, which can substantially reduce yields and lead to fungal contamination, but is highly dependent on cultivar and planting density63. Plant height dramatically increased in the MR cultivars (Fig. 1) and was the most discriminating characteristic for defining FHB groups at e[CO2] (Fig. 7). Furthermore, wheat stem strength was lower in MR cultivars RB07 and Bolles at e[CO2], potentially exacerbating the risk of crop lodging. The current study is inherently limited in fully characterizing the impact of elevated CO2 on wheat in future field conditions. Growth in enclosed chambers can alter and downregulate photosynthesis, stifle root structure and volume, and the lack of wind induced mechanical stress may change the rate of lignin deposition64,65. Further research at a free air carbon enrichment (FACE) facility is needed to elucidate cultivar specific lodging risk at elevated CO2.
The loss of grain protein at elevated CO2 is not simply an issue of human nutrition. The end use suitability of wheat flour is intractably linked to grain protein content. Low protein flour (8–11%) is generally used for cakes and pastries while high protein flours (> 14%) are used for breads and pastas5,66. Wheat grown at elevated CO2 often has significantly less grain protein, which reduces the baking quality, compromising the final food product7,17,18. Increased starch content at elevated CO2 has also been associated with reduced baking quality as well as starch damage during the milling process7. Future farmers and food processors can anticipate substantially increased wheat yields of lower quality grain with poorer dough rheology, reduced loaf volume and baking quality19. The identification of wheat cultivars that can maintain nutritional integrity and FHB resistance in future climate conditions are critically needed to ensure the safety and security of our food supply.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Shiferaw, B. et al. Crops that feed the world 10. Past successes and future challenges to the role played by wheat in global food security. Food Secur. 5, 291–317 (2013).
UN-FAO. (Food and Agriculture Organization of the United Nations (FAO), www.fao.org/faostat, 2021).
Vaughan, M., Backhouse, D. & Del Ponte, E. Climate change impacts on the ecology of Fusarium graminearum species complex and susceptibility of wheat to Fusarium head blight: A review. World Mycotoxin J. 9, 685–700 (2016).
Chakraborty, S. Migrate or evolve: Options for plant pathogens under climate change. Glob. Change Biol. 19, 1985–2000. https://doi.org/10.1111/gcb.12205 (2013).
Asseng, S. et al. Climate change impact and adaptation for wheat protein. Glob. Change Biol. 25, 155–173 (2019).
Taub, D. R. & Wang, X. Why are nitrogen concentrations in plant tissues lower under elevated CO2? A critical examination of the hypotheses. J. Integr. Plant Biol. 50, 1365–1374 (2008).
Panozzo, J. et al. Elevated carbon dioxide changes grain protein concentration and composition and compromises baking quality. A FACE study. J. Cereal Sci. 60, 461–470 (2014).
Soba, D. et al. Metabolic effects of elevated CO2 on wheat grain development and composition. J. Agric. Food Chem. 67, 8441–8451 (2019).
Liu, C. & der Fels-Klerx, V. Quantitative modeling of climate change impacts on mycotoxins in cereals: A review. Toxins 13, 276 (2021).
Hay, W. T. et al. Changes in wheat nutritional content at elevated [CO2] alter Fusarium graminearum growth and mycotoxin production on grain. J. Agric. Food Chem. 68, 6297–6307. https://doi.org/10.1021/acs.jafc.0c01308 (2020).
Goswami, R. S. & Kistler, H. C. Heading for disaster: Fusarium graminearum on cereal crops. Mol. Plant Pathol. 5, 515–525. https://doi.org/10.1111/J.1364-3703.2004.00252.X (2004).
Ainsworth, E. A. & Long, S. P. 30 years of free-air carbon dioxide enrichment (FACE): What have we learned about future crop productivity and its potential for adaptation?. Glob. Change Biol. 27, 27–49 (2021).
Högy, P. & Fangmeier, A. Effects of elevated atmospheric CO2 on grain quality of wheat. J. Cereal Sci. 48, 580–591 (2008).
DaMatta, F. M., Grandis, A., Arenque, B. C. & Buckeridge, M. S. Impacts of climate changes on crop physiology and food quality. Food Res. Int. 43, 1814–1823 (2010).
Fernando, N. et al. Elevated CO2 alters grain quality of two bread wheat cultivars grown under different environmental conditions. Agric. Ecosyst. Environ. 185, 24–33 (2014).
Broberg, M. C., Högy, P. & Pleijel, H. CO2-induced changes in wheat grain composition: Meta-analysis and response functions. Agronomy 7, 32 (2017).
Fernando, N. et al. Rising CO2 concentration altered wheat grain proteome and flour rheological characteristics. Food Chem. 170, 448–454 (2015).
Arachchige, P. M. S. et al. Wheat (Triticum aestivum L.) grain proteome response to elevated [CO2] varies between genotypes. J. Cereal Sci. 75, 151–157 (2017).
Walker, C. K. et al. Adaptive traits do not mitigate the decline in bread wheat quality under elevated CO2. J. Cereal Sci. 88, 24–30 (2019).
Thompson, M. et al. Effect of elevated carbon dioxide on plant biomass and grain protein concentration differs across bread, durum and synthetic hexaploid wheat genotypes. J. Cereal Sci. 87, 103–110 (2019).
Bencze, S. et al. Rising atmospheric CO2 concentration may imply higher risk of Fusarium mycotoxin contamination of wheat grains. Mycotoxin Res. 33, 229–236 (2017).
Váry, Z., Mullins, E., McElwain, J. C. & Doohan, F. M. The severity of wheat diseases increases when plants and pathogens are acclimatized to elevated carbon dioxide. Glob. Change Biol. 21, 2661–2669 (2015).
Cuperlovic-Culf, M. et al. Effects of atmospheric CO2 level on the metabolic response of resistant and susceptible wheat to Fusarium graminearum infection. Molecular Plant-Microbe Interactions, MPMI-06-18-0161-R (2019).
Zhu, Z. et al. Breeding wheat for resistance to Fusarium head blight in the Global North: China, USA and Canada. Crop J. 7, 730–738 (2019).
Brar, G. S., Pozniak, C. J., Kutcher, H. R. & Hucl, P. J. Evaluation of Fusarium head blight resistance genes Fhb1, Fhb2, and Fhb5 introgressed into elite Canadian hard red spring wheats: Effect on agronomic and end-use quality traits and implications for breeding. Mol. Breed. 39, 44 (2019).
McCartney, C. A. et al. The evaluation of FHB resistance QTLs introgressed into elite Canadian spring wheat germplasm. Mol. Breed. 20, 209–221. https://doi.org/10.1007/s11032-007-9084-z (2007).
Anderson, J. et al. Registration of ‘Linkert’spring wheat with good straw strength and adult plant resistance to the Ug99 family of stem rust races. J. Plant Reg. 12, 208–214 (2018).
Anderson, J. et al. Registration of’Ulen’wheat. Crop Sci. 46, 979–981 (2006).
Busch, R., McVey, D., Rauch, T., Baumer, J. & Elsayed, F. Registration of Wheaton wheat. Crop Sci. 24, 622–622 (1984).
Anderson, J. A. et al. Registration of ‘Bolles’ hard red spring wheat with high grain protein concentration and superior baking quality. J. Plant Reg. 12, 215–221 (2018).
Mergoum, M. et al. Registration of’Glenn’wheat. Crop Sci. 46, 473–475 (2006).
Anderson, J. et al. Registration of 'Lang-MN' hard red spring wheat. Plant Registrations (2021).
Anderson, J. et al. Registration of ‘RB07’wheat. J. Plant Regist. 3, 175–180 (2009).
Anderson, J. et al. Registration of ‘Rollag’spring wheat. J. Plant Regist. 9, 201–207 (2015).
Anderson, J. A. et al. Registration of ‘Sabin’wheat. J. Plant Regist. 6, 174–179 (2012).
Anderson, J. et al. Registration of ‘Shelly’hard red spring wheat. J. Plant Regist. 13, 199–206 (2019).
Dien, B. S. et al. Chemical composition and response to dilute-acid pretreatment and enzymatic saccharification of alfalfa, reed canarygrass, and switchgrass. Biomass Bioenerg. 30, 880–891 (2006).
Boratyński, J. Colorimetric method for the determination of ketoses using phenol-acetone-boric acid reagent (PABR). Anal. Biochem. 137, 528–532 (1984).
Hartman, L. & Lago, R. C. A. Rapid preparation of fatty acid methyl esters from lipids. Lab. Pract. 22, 475–476 (1973).
Miller, C. N. et al. Elucidation of the genetic basis of variation for stem strength characteristics in bread wheat by Associative Transcriptomics. BMC Genomics 17, 1–11 (2016).
Shahbazi, F. & Nazari Galedar, M. Bending and shearing properties of safflower stalk. J. Agric. Sci. Technol. 14, 743–754 (2012).
US-HHS & USDA. (Washington, DC, www.health.gov, 2015).
Wang, H. et al. Horizontal gene transfer of Fhb7 from fungus underlies Fusarium head blight resistance in wheat. Science 368 (2020).
Li, T. et al. Effects of the Fhb1 gene on Fusarium head blight resistance and agronomic traits of winter wheat. Crop J. 7, 799–808 (2019).
Bent, A. F. & Mackey, D. Elicitors, effectors, and R genes: The new paradigm and a lifetime supply of questions. Annu. Rev. Phytopathol. 45, 399–436 (2007).
Brar, G. S. et al. Genetic factors affecting Fusarium head blight resistance improvement from introgression of exotic Sumai 3 alleles (including Fhb1, Fhb2, and Fhb5) in hard red spring wheat. BMC Plant Biol. 19, 1–19 (2019).
Von der Ohe, C., Ebmeyer, E., Korzun, V. & Miedaner, T. Agronomic and quality performance of winter wheat backcross populations carrying non-adapted Fusarium head blight resistance QTL. Crop Sci. 50, 2283–2290 (2010).
Nelson, R., Wiesner-Hanks, T., Wisser, R. & Balint-Kurti, P. Navigating complexity to breed disease-resistant crops. Nat. Rev. Genet. 19, 21–33 (2018).
Brown, J. & Rant, J. Fitness costs and trade-offs of disease resistance and their consequences for breeding arable crops. Plant. Pathol. 62, 83–95 (2013).
Zhuang, Y., Gala, A. & Yen, Y. Identification of functional genic components of major Fusarium head blight resistance quantitative trait loci in wheat cultivar Sumai 3. Mol. Plant Microbe Interact. 26, 442–450 (2013).
Oh, M. et al. Transcription factor ART1 mediates starch hydrolysis and mycotoxin production in Fusarium graminearum and F. verticillioides. Mol. Plant Pathol. 17, 755–768 (2016).
Snoeijers, S. S., Pérez-García, A., Joosten, M. H. A. J. & De Wit, P. J. G. M. The effect of nitrogen on disease development and gene expression in bacterial and fungal plant pathogens. Eur. J. Plant Pathol. 106, 493–506. https://doi.org/10.1023/a:1008720704105 (2000).
Walkowiak, S. & Subramaniam, R. A nitrogen-responsive gene affects virulence in Fusarium graminearum. Can. J. Plant Path. 36, 224–234 (2014).
Draeger, R. et al. Identification of QTLs for resistance to Fusarium head blight, DON accumulation and associated traits in the winter wheat variety Arina. Theor. Appl. Genet. 115, 617–625 (2007).
Steiner, B. et al. Molecular mapping of resistance to Fusarium head blight in the spring wheat cultivar Frontana. Theor. Appl. Genet. 109, 215–224 (2004).
Ollier, M. et al. QTL mapping and successful introgression of the spring wheat-derived QTL Fhb1 for Fusarium head blight resistance in three European triticale populations. Theor. Appl. Genet. 133, 457–477 (2020).
O’Leary, G. J. et al. Response of wheat growth, grain yield and water use to elevated CO2 under a Free-Air CO2 Enrichment (FACE) experiment and modelling in a semi-arid environment. Glob. Change Biol. 21, 2670–2686 (2015).
Högy, P. et al. Effects of elevated CO2 on grain yield and quality of wheat: Results from a 3-year free-air CO2 enrichment experiment. Plant Biol. 11, 60–69. https://doi.org/10.1111/j.1438-8677.2009.00230.x (2009).
Kimball, B. A. Crop responses to elevated CO2 and interactions with H2O, N, and temperature. Curr. Opin. Plant Biol. 31, 36–43 (2016).
Khan, A. et al. Optimization of plant density and nitrogen regimes to mitigate lodging risk in wheat. Agron. J. 112, 2535–2551 (2020).
Pleijel, H., Broberg, M. C., Högy, P. & Uddling, J. Nitrogen application is required to realize wheat yield stimulation by elevated CO2 but will not remove the CO2-induced reduction in grain protein concentration. Glob. Change Biol. 25, 1868–1876 (2019).
Wei, F.-Z., Li, J.-C., Wang, C.-Y., Qu, H.-J. & Shen, X.-S. Effects of nitrogenous fertilizer application model on culm lodging resistance in winter wheat. Acta Agron. Sin. 34, 1080–1085 (2008).
Stapper, M. & Fischer, R. Genotype, sowing date and plant spacing influence on high-yielding irrigated wheat in southern New South Wales. II. Growth, yield and nitrogen use. Aust. J. Agric. Res. 41, 1021–1041 (1990).
Gardiner, B., Berry, P. & Moulia, B. Wind impacts on plant growth, mechanics and damage. Plant Sci. 245, 94–118 (2016).
Ainsworth, E. A. & Long, S. P. What have we learned from 15 years of free-air CO2 enrichment (FACE)? A meta-analytic review of the responses of photosynthesis, canopy properties and plant production to rising CO2. New Phytol. 165, 351–372 (2005).
Delcour, J. A. et al. Wheat gluten functionality as a quality determinant in cereal-based food products. Annu. Rev. Food Sci. Technol. 3, 469–492 (2012).
Acknowledgements
We would like to thank Erin Walter for ICP analysis and instrument calibration. Victoria Nguyen for the sample preparation and analysis of grain starch and fructan content. Nathan Kemp, Jennifer Teresi, and Keegan McConnel for plant care, growth chamber monitoring, and grain harvest. Finally, Gary Grose, for assistance in grain compositional analysis. This work was supported by the U.S. Department of Agriculture, Agricultural Research Service.
Funding
This work was funded by the United States Department of Agriculture. Mention of trade names or commercial products in this publication is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture. Authors have no conflicts of interest to declare. USDA is an equal opportunity provider and employer.
Author information
Authors and Affiliations
Contributions
W.T.H. planned, designed, and coordinated the research efforts of the study. He was also the primary manuscript author and performed the statistical analysis. J.A.A. designed plant treatments, including wheat cultivar selection. S.P.M. assisted in writing and editing the manuscript. M.P.H.-E. designed the experimental procedures for determining wheat grain protein content. G.W.S. and K.D.U. designed and performed the analysis of wheat straw characteristics. M.J.B. designed and performed the analysis of wheat grain water soluble carbohydrate and starch content. K.M.D. and K.L.A. designed, performed, and validated the wheat grain mineral analysis. M.A.B. designed and performed the analysis of wheat grain fatty acid content. M.M.V. supervised research efforts and assisted in the design, coordination, and writing of the manuscript. All Authors reviewed, edited, and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hay, W.T., Anderson, J.A., McCormick, S.P. et al. Fusarium head blight resistance exacerbates nutritional loss of wheat grain at elevated CO2. Sci Rep 12, 15 (2022). https://doi.org/10.1038/s41598-021-03890-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-03890-9
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.