Abstract
Exploitation of organisms by multiple parasite species is common in nature, but interactions among parasites have rarely been studied. Myrmica ants are rich in parasites. Among others, the ectoparasitic Rickia wasmannii fungus and the parasitic caterpillars of myrmecophilous Phengaris butterflies often infect the same Myrmica colonies. In this study, we examined the effects of R. wasmannii on the adoption, long-term development, and survival of P. alcon. In laboratory conditions, caterpillars introduced into nests of Myrmica scabrinodis uninfected with R. wasmannii survived significantly longer compared to caterpillars introduced into infected nests. In the field, joint infection was less common than expected if both parasites exploited M. scabrinodis colonies independently. Pre-pupal caterpillars of P. alcon were somewhat larger in nests infected with R. wasmannii than those found in uninfected nests. Based on these results it seems that R. wasmannii infection of M. scabrinodis affects the survival and development of P. alcon caterpillars, suggesting competition between these two ant parasites.
Similar content being viewed by others
Introduction
Organisms are often exploited by multiple parasites in nature, but this is rarely highlighted in ecological studies1,2,3. From the point of view of the host, co-infecting parasites may have synergistic, additive, or antagonistic effects on fitness1,4,5. Co-infection may also result in fitness-changing interactions between the parasites2,6, as they may show contest or scramble competition for host resources1, or have more complex interactions7,8,9. When any of the species involved in such interactions are of conservation concern, then an understanding of the effects of multiple infections on both hosts and parasites is vital. While the importance of conserving parasites is being increasingly acknowledged10,11,12, the threats or protection offered by co-infecting parasites has largely been ignored.
Colonies of ants (Hymenoptera: Formicidae) are subject to numerous parasites, ranging from pathogens and endo- and ecto- macroparasites and parasitoids that exploit individuals, to social parasites that exploit entire colonies and their resources13,14,15,16,17,18,19. This makes ant colonies good model organisms for parasitological studies.
Myrmica ants are particularly rich in parasites. Their colonies often provide a home for different social parasites, including other Myrmica species, Lomechusa beetles (Coleoptera: Staphylinidae), Platyarthrus woodlice (Isopoda, Platyarthridae), Cyphoderus springtails (Entomobryomorpha: Paronellidae), larvae of Microdon hoverflies (Diptera: Syrphidae) and the caterpillars of Phengaris (= Maculinea) butterflies (Lepidoptera: Lycaenidae). Myrmica individuals can be infected by parasitoid wasps (Hymenoptera: Eucharitidae and Ichneumonidae) and flies (Diptera: Phoridae), endoparasitic nematodes (Mermithidae, Rhabditidae and Steinernematidae), ecto- and endoparasitic fungi, and various bacteria20,21,22,23. While there is some information on the co-infection levels of Myrmica nests by different species of Phengaris butterflies and Microdon myrmicae24,25 the consequences of these co-infections for the parasites themselves is poorly understood.
Over the last two decades we have investigated two parasites of Myrmica ants in detail (Fig. 1, Supplementary Video S1), socially parasitic Phengaris butterflies and the ectoparasitic fungus Rickia wasmannii (Ascomycota: Laboulbeniales). Both affect their Myrmica host ants, but in different ways. Phengaris caterpillars have a strong negative effect on their host colonies by feeding on their brood and/or by receiving food by trophallaxis that would otherwise go to ant workers or larvae26,27. Rickia wasmannii increases the need for water of the host ants, makes them less aggressive and less bold, increases allo- and auto-grooming frequency, reduces the size of workers and the thickness of their cuticle and, in general, causes earlier death of workers, at least in the lab14,28,29,30,31,32. Despite the fact that these two organisms can co-occur within the same Myrmica colonies33, we know of only a single study addressing the effects of common infection. Csata et al.14 found that M. scabrinodis colonies infected by R. wasmannii adopted a higher proportion of offered larvae of Phengaris teleius and P. alcon compared to uninfected colonies, although for P. alcon differences were mostly due to differences in discovery of caterpillars rather than caterpillar rejection. In that study, the caterpillars had only 120 min to be discovered and transported by the ants to the nest. However, short-term adoption does not mean the integration of caterpillars into the Myrmica colonies and their successful development to adult butterflies34,35.
Illustrations of study organisms. (a) Phengaris alcon female laying eggs on Gentiana pneumonanthe flower buds. (b) Overwintered P. alcon caterpillars in a Myrmica scabrinodis nest. (c) Drawing of an adult Rickia wasmannii thallus. (d) Myrmica scabrinodis worker uninfected with R. wasmannii. (e) Myrmica scabrinodis worker with numerous R. wasmannii thalli on its cuticle; see also Supplementary Video S1. Photo: David R. Nash (a), Ádám Bakos (b, d, e); the drawing (c) is in the public domain, from the original illustrations by Roland Thaxter, courtesy of the Farlow Reference Library of Cryptogamic Botany, Harvard University.
In this study we examined the effects of R. wasmannii on the adoption and long-term development and survival of P. alcon. Specifically, we investigated whether R. wasmannii infection of M. scabrinodis colonies may have an effect on any of the following traits: (1) the discovery time of P. alcon caterpillars; (2) the adoption time of P. alcon caterpillars; (3) the overwintering survival of adopted P. alcon caterpillars; (4) the proportion of P. alcon parasitized nests in the field; (5) the level of P. alcon parasitism in the field; and (6) the size of prepupal P. alcon larvae in the field (Figs. 2, 3, 4, 5, 6, 7, 8 and 9).
The field site at Gyilkos-rét where Myrmica scabrinodis nests and Phengaris alcon eggs and caterpillars were collected. (a) Early June. (b) Mid-August with flowering Gentiana pneumonanthe in the central foreground. See Fig. 5 in Csősz et al.29 for maps of the locality. Photo: András Tartally (a) and Márton József Paulin (b).
Nest design. (a) Plastic box. (b) Plaster floor. (c) Glass plate. (d) Nest chamber with entrance. (e) “Drinker” (a 1.5 mL Eppendorf tube with cut tip, stuffed with a piece of cloth and filled with tap water). (f) Honey-sugar water in a “feeder” (the cap of an 1.5 mL Eppendorf tube). (g) Insect food (dead cockroach). (h) Myrmica scabrinodis workers. (i) Young Phengaris alcon caterpillar with a M. scabrinodis worker. The diameter of the nest is 60 mm. Photo: Ferenc Báthori.
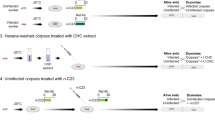
Survival of caterpillars in the mini colonies, and temperature at which they were kept. Before overwintering (drop in temperature after 110 days), nests are divided into those that were infected by Rickia wasmannii and uninfected nests. All surviving caterpillars were weighed at the start of overwintering, and the survivorship of those above (Large) and below (Small) the mean mass in infected and uninfected nests are then shown separately. Shaded regions are 95% confidence intervals around the survivorship, estimated from a proportional hazards survival model.

taken from digital camera (by Anna Ágnes Somogyi, with 5 mm square paper background), showing 22 P. alcon caterpillars, including one clearly dead and two small caterpillars (which will pupate next year), and one P. alcon pupa. (b) Background and all objects other than live prepupal caterpillars removed and scale bar added (based on 5 mm squares) in Adobe Photoshop. (c) Tracing of perimeter of prepupal caterpillars in Fiji. (d) Export of details of each prepupal caterpillar from Fiji as a CSV file (here shown in Microsoft Excel), including calculated area in mm2.
Summary of the procedure for measuring the size of prepupal Phengaris alcon caterpillars. (a) Original image
Histograms showing the distribution of number of prepupal caterpillars of Phengaris alcon in nests of Myrmica scabrinodis that are either infected by Rickia wasmannii (red bars) or uninfected (blue bars). Superimposed on the histogram are the fitted negative binomial probability density plots for infected (red line) and uninfected (blue line) nests.
Box-plots with overlaid raw data showing the sizes of prepupal caterpillars of Phengaris alcon (measured as caterpillar area on field photographs) in nests of Myrmica scabrinodis with (red symbols) or without (blue symbols) Rickia wasmannii infection, and with (triangles) or without (circles) P. alcon pupae present. Nests are sorted along the x-axis by decreasing mean caterpillar size separately for infected and uninfected nests.
Results
Adoption and survival of caterpillars in the lab
For those caterpillars that were adopted, no significant difference between infected and uninfected colonies was observed in discovery time (F1,35 = 0.245, p = 0.623), adoption time (F1,35 = 1.28, p = 0.266), or mass of caterpillars at time of introduction (F1,35 = 0.497, p = 0.486; Fig. 6). These quantities also did not differ between the original colony fragments (F11,35 = 1.12, p = 0.374; F11,35 = 1.33, p = 0.249; F11,35 = 1.62, p = 0.135 respectively).
Of the 35 caterpillars introduced into uninfected mini M. scabrinodis colonies, six (17%) survived the overwintering process, but none of the caterpillars introduced into infected colonies survived this long (Fig. 4), leading to a significant difference in survival probability (Wald χ2 = 5.77, df = 1, p = 0.0162). There was also a significant effect of original colony fragment on survival probability (nested within R. wasmannii infection; Wald χ2 = 25.1, df = 12, p = 0.0144). For those caterpillars that were re-weighed in November, there was also a significantly higher survival for those in uninfected nests (Wald χ2 = 10.87, df = 1, p = 0.0010), and a positive association between mass and survival (Wald χ2 = 38.3, df = 1, p < 0.0001), but there was also a significant interaction between R. wasmannii infection and mass (Wald χ2 = 8.89, df = 1, p = 0.0042), with smaller caterpillars in infected nests showing disproportionately high mortality (Fig. 4). In addition, there was a significant effect of original colony fragment on survival probability (nested within R. wasmannii infection; Wald χ2 = 28.4, df = 12, p = 0.0047).
Patterns of P. alcon and R. wasmannii infection of M. scabrinodis in the field
Of the 41 nests of M. scabrinodis found within 2 m of a Gentiana pneumonanthe (Dicotyledonopsida: Gentianaceae) plant, 15 (37%) were identified as infected with R. wasmannii in the field. When workers from these were examined under the microscope in the laboratory, the number of thalli of R. wasmannii found on workers was highly variable between individuals (i.e., aggregated, as is typical for macroparasites36). Fitting a generalized linear model with negative binomial errors to the data on number of thalli per individual from infected nests (fitted dispersion parameter θ = 4.39) showed a highly significant difference between nests in the level of infection (Wald χ2 = 245.05, df = 1, p < 0.0001), and also a strong effect of estimated worker age (Wald χ2 = 70.3, df = 4, p < 0.0001), with older workers having a higher load of thalli (Fig. 7), but no association with the presence or absence of P. alcon caterpillars (Wald χ2 = 0.719, df = 1, p = 0.396). Only one callow worker (age class 1) was sampled, which bore no thalli.
Twenty six of the 41 M. scabrinodis nests (63%) were parasitized by P. alcon, with between 1 and 44 caterpillars per nest. This aggregative distribution was again well-described by a negative binomial distribution, with dispersion parameter θ = 2.80 (Fig. 8), and there was no association between infection with R. wasmannii and the number of caterpillars P. alcon present (Wald χ2 = 0.1.94, df = 1, p = 0.163). Joint infection and parasitism of nests was less common than expected if both parasites acted independently (χ2 = 4.37, df = 1, p = 0.037).
The size (area on photographs) of P. alcon prepupal caterpillars in each nest varied considerably between nests (variance explained = 45%, Wald p = 0.009; Fig. 9), but was significantly larger in nests that also contained one or more pupae (F1,19.9 = 16.46, p = 0.025). There was no association between the total number of P. alcon within a nest and the size of prepupal P. alcon caterpillars (F1,17 = 0.005, p = 0.944). Caterpillars were somewhat larger in nests infected with R. wasmannii (Least squared mean ± SE: 39.4 ± 2.57 mm2) than those that were uninfected (32.1 ± 1.12 mm2), although not significantly so (F1,20.5 = 1.75, p = 0.201). It is notable that a simple analysis of caterpillar sizes without taking between-nest differences into account would have led to the conclusion that prepupal caterpillars are significantly larger in infected nests of M. scabrinodis (t279 = 4.2, p < 0.0001).
When the number of prepupal caterpillars and pupae was divided by the number of nests, this index of infection level37 was higher in uninfected (10.12) than in infected (4.69) nests.
Discussion
Based on our results, it is clear that R. wasmannii infection of M. scabrinodis affects the survival and development of P. alcon caterpillars, which suggests competition1,2 between these two ant associates, although this effect is subtle.
Interestingly, there was no significant difference between infected and uninfected M. scabrinodis mini colonies in the discovery time of pre-adopted P. alcon caterpillars (Fig. 6). This is in contrast with the results of Csata et al.14, who showed lower discovery of P. alcon caterpillars within 120 min by uninfected than infected colonies, but is in harmony with the finding that R. wasmannii infection does not affect the locomotory behaviour of M. scabrinodis workers38. A potential reason for such a contrast is the longer time allowed in our experiment (see below), or differences in the populations studied. We also found no significant difference in adoption time between infected and uninfected mini colonies (Fig. 6). Csata et al.14 did not measure discovery and adoption time separately, but found no difference in the overall time between introduction and adoption between uninfected and infected colonies for those caterpillars that were adopted. It is noteworthy that in 39 out of 48 cases where caterpillars were adopted in our study (81%), it took longer than 120 min since initial introduction. However, all but one of the 70 introduced caterpillar were discovered within this period (Fig. 6).
The pattern of survival of adopted caterpillars was significantly different in uninfected and infected mini colonies, with somewhat higher survival in uninfected colonies both before and after (but not during) overwintering (Fig. 4). For those caterpillars that survived to enter the wintering phase, there was a strong effect of both infection of its host mini colonies by R. wasmannii and size of each caterpillar on its chance of survival, and these effects were synergistic, such that small caterpillars in infected mini colonies had a considerably lower survival probability than that predicted based on their size and the infection status of their host nests alone (Fig. 4). When we examined colonies of M. scabrinodis in the field, we found that the number of colonies simultaneously infected with R. wasmannii and parasitized by P. alcon was lower than expected if the two parasites were acting independently of each other. This pattern has also previously been found for co-infection of Myrmica colonies by different Phengaris species and/or Microdon myrmicae24,25.We also found that prepupal caterpillars of P. alcon were larger in nests where some caterpillars had already pupated, and there was a tendency for prepupal caterpillars to be larger in infected than uninfected ant nests (Fig. 9).
Putting these pieces of evidence together suggests that infection of an ant colony by R. wasmannii has a disproportionately large effect on the survival and development of smaller caterpillars of P. alcon, but rather little effect on larger caterpillars; this is what results in the somewhat larger sizes of prepupal caterpillars in infected nests. Increased mortality among small caterpillars would also lead to the lower-than-chance occurrence of co-coinfection observed in field colonies, although from our spring surveys it was not possible to demonstrate that this mortality has taken place. In general, we found evidence supporting an overall negative effect of R. wasmannii on P. alcon caterpillars, although these effects were relatively minor, and did not prevent R. wasmannii infected M. scabrinodis from raising a relatively high number of P. alcon in this population. Much lower numbers of P. alcon (and lower proportions of infected nests) are frequently found in other populations, including those where M. scabrinodis is the only host25. The only putatively positive effect of R. wasmannii infection on P. alcon was the apparent larger size of its prepupal caterpillars in infected nests compared to uninfected nests (Fig. 9), although this is likely an artefact due to the removal of smaller caterpillars from the population. However, it cannot be ruled out that the lack of competition that this could produce would increase adult fecundity or mating success39,40. In this regard it is relevant to note that we found no relationship between the number of P. alcon and size of prepupal caterpillars in this population, as might be expected if there was strong competition41.
It is clear that there is a large amount of between-host-colony variation in survival and growth of P. alcon caterpillars, and the putative effects of R. wasmannii on P. alcon (or vice versa). Some of this probably reflects the resources available to the ant colony (and hence to its parasites), and it is likely that colony size (and hence worker force and brood availability) are also important34,42. Nevertheless, the significant effect of original colony on survivorship of caterpillars of P. alcon under controlled conditions also suggests that other intrinsic properties of colonies are also important. Such a property could be the between-colony variation in cuticular hydrocarbon profiles, which has been shown to have a major effect on adoption and survival of Phengaris caterpillars25,42,43,44. It is known that R. wasmannii infection changes the cuticular hydrocarbon profile and the overall hydrocarbon abundance of M. scabrinodis14. Based on our results, however, it seems that there is unlikely to be an effect of R. wasmannii on adoption of P. alcon caterpillars, which is the stage where matching of hydrocarbons with those of the host is critical42,45,46, but subsequent survival and growth may be impacted by the fungus. Phengaris alcon caterpillars can successfully switch host species during larval development47,48, and this may be facilitated by the ability of larvae to change their own hydrocarbon profiles44,49 but may also depend on other colony characteristics47.
Between-colony differences in aggression and susceptibility to parasitism are also likely to be important, which may be linked to colony social structure50 as well as to coevolutionary arms races between parasites and hosts42. We still do not know whether these effects are largely genetically or environmentally determined51,52,53,54,55, but in any case the large between-colony variation suggests that large numbers of colonies need to be sampled to examine the interactions between Myrmica ants and their parasites25.
Although our results suggest an overall negative effect of R. wasmannii on the development of P. alcon caterpillars, since P. alcon is a rather virulent parasite of Myrmica nests42,56, while R. wasmannii is much less virulent14,30,31,32, the possibility exists that infection by R. wasmannii could be beneficial for M. scabrinodis nests if P. alcon is common in a population. The reported negative effects of R. wasmannii on its Myrmica host are also mostly found in laboratory studies, and their importance under field conditions is unclear. The seasonal differences in R. wasmannii infection57 could be considered when laboratory experiments are translated to field conditions. While increased allo- and auto-grooming frequency30, the smaller worker size29, and the less aggressive and more timid behaviour14,31 associated with R. wasmannii infection may have an effect on resource acquisition and territorial disputes in the field, it is difficult to imagine that these will have as large an effect on fitness as the direct consumption of brood or diversion of resources from sexual progeny by P. alcon. Increased water-loss and earlier mortality of R. wasmannii infected M. scabrinodis workers32 is also unlikely to be a major problem in the field, where M. scabrinodis is typically found in marshy meadows where humidity is high and water abundant.
In general, our study supports the idea that competition between parasites for host resources may have complex outcomes for both parasites and hosts1,2. The immediate resources used by the two parasites are different, but both draw on the overall resources of the ant colony. Our study was not designed to examine host fitness, and so it is unclear whether the combined effects of the two parasites were additive, synergistic, or antagonistic. Further experiments focussing on host fitness would be necessary to examine this aspect of the interactions. Theoretical models1,2,7, greenhouse and laboratory experiments3,5, and examples from biological control4,6 suggest that synergistic or additive virulence effects are most likely58,59, so the possibility of a conditional60 antagonistic effect, as outlined above, is intriguing. Such effects are likely to be more common than hitherto documented61; for example, such a conditional effect has recently been proposed for a microsporidian parasite of Daphnia62. There is no evidence for direct interaction between the two parasites in our study. Despite R. wasmannii being able to also infect myrmecophilous arthropods in Myrmica nests63, we have never observed infection of P. alcon by R. wasmannii, either in the course of this study, or when examining numerous other P. alcon caterpillars in Hungary and Denmark. Hence, contest (interference) competition is unlikely to be the major form of competition between the two parasites, whereas all the indirect evidence we have points to scramble (exploitation) competition for host resources, which contrasts with the intraspecific contest competition that has been demonstrated for P. alcon41. In other parasitic systems it has been argued that the type of interaction between parasites should be reflected in the association in infection levels between parasites within a host population2. Recent comparative studies have found more positive than negative associations between multi-parasite infections in hosts58,59, which has been interpreted as facilitation of infection of one parasite by the presence of another e.g.64. It has been suggested that such interactions are mostly amensal—where one parasite has a positive effect on the other, but there is no measurable reciprocal effect2,65,66. Whether this represents contest or scramble competition has been interpreted differently by different authors, but in this study we found a negative association between the two parasites, but also clear evidence of an asymmetry in their effects on each other, with P. alcon apparently being strongly affected by R. wasmannii, but not vice versa.
Methods
Terminology
To allow easier distinction between the two types of Myrmica parasites used in this study, we refer to M. scabrinodis nests containing P. alcon caterpillars as “parasitized”, while M. scabrinodis colonies and workers with R. wasmannii are termed “infected”. Similarly, “unparasitized” is used about nests without P. alcon and “uninfected” about nests without R. wasmannii. The nests containing neither R. wasmannii nor P. alcon are referred to as “healthy”. “Colony fragments” from wild M. scabrinodis nests were collected to give workers for the “mini colonies” for the lab work (see details: Adoption and survival of caterpillars in the lab). “Larvae” is used to refer to ant larvae, while the larvae of the butterflies are called “caterpillars”. We use the term “social parasite” to refer to any parasite that lives on the resources of a social insect colony rather than the individual members of that colony67 rather than in the narrow sense of an ant species that exploits another ant species19.
Study species, population and site
The sample site was in northern Hungary at Gyöngyös: Sár-hegy: Gyilkos-rét (47°48' N, 19°58' E; 352 m a.s.l). It is a small (ca. 0.4 ha), marshy meadow with tall-sedge and dense stands of Gentiana pneumonanthe, the initial foodplant of P. alcon, surrounded by oak forest (Fig. 2).
We sampled nests of M. scabrinodis (Fig. 1, Supplementary Video S1), which is a common ant species on this meadow that is, as far as is known, the only host of P. alcon and R. wasmannii at this site25,68. Numerous healthy, infected, parasitized or co-infected M. scabrinodis nests can be found at Gyilkos-rét25, making this site ideal for this study. To confirm the identity of putative M. scabrinodis nests, 5–10 workers from each nests were examined under a 40 × magnifying hand lens in the field, and then transferred to vials of 67.5% ethanol to confirm identification in the lab by AT, using a Leica MZ125 microscope (Wetzlar, Germany) at 10–160 × magnification and keys by Radchenko and Elmes69.
Rickia wasmannii (Fig. 1, Supplementary Video S1) is one of the most widespread ectoparasitic70 Laboulbeniales fungal species in Europe, infecting ten Myrmica species and some of their arthropod associates from Turkey to Portugal63,71,72,73. Several effects of Laboulbeniales fungi on their hosts are known, which are primarily negative3,14,29,30,31,32,74 but can also be indirectly positive75. Research in recent years has made R. wasmannii one of the best known ant-parasitic Laboulbeniales species. The presence of R. wasmannii on worker ants was checked along with the identification of the ants (see above) in the field by AT, and later confirmed by FB in the laboratory based on thallus morphology76,77.
While the Alcon blue butterfly, P. alcon (Fig. 1, Supplementary Video S1) is currently considered as “least concern” in the latest edition of the European red list of butterflies78, it is classified as “near-threatened” in Hungary79, and there is concern that its numbers have decreased rapidly in Europe since the last assessment80. Its caterpillars start their development feeding on seeds of different Gentiana host plant species. In the final instar phase, the caterpillars leave the initial food plants and mimic the odour42,43,44 and the sound81 of certain Myrmica species, so as to be “adopted” and raised by the ants82. These ‘cuckoo’ caterpillars are mostly fed by host workers with trophallactic regurgitations but can also feed directly on ant brood26. The abundant P. alcon population at Gyilkos-rét belongs to the G. pneumonanthe-using, hygrophilic form (P. alcon H)25. This population typically flies in July and the caterpillars leave the host plants for adoption in August, depending on annual weather conditions (AT, pers. obs.).
Adoption and survival of caterpillars in the lab
Because M. scabrinodis is a polygynous species69, it is easy to collect colony fragments with a few (but not all) queens, hundreds of workers, and brood without extirpating the “mother” colonies. We collected 7 infected and 7 uninfected colony fragments on the 4th August. Ants were kept in plastic boxes (16.5 cm × 11.5 cm × 6 cm) treated with Fluon (ICI, London, GB) on their inner walls to prevent them from escaping. Inside these boxes, we created nest chambers (5.5 cm × 4.5 cm × 1 cm) with plaster floors, covered with glass plates. The ants were kept at room temperature (22 ± 1 °C) under a natural light cycle, and fed with frozen cockroaches on Thursdays and with 20–20% honey–sugar water solution on Mondays and Thursdays prior to wintering. Water was available ad libitum.
Five small artificial “mini” M. scabrinodis colonies were derived from each of the 7 infected and 7 uninfected colony fragments and used for the tests, resulting in 35 infected and 35 uninfected mini colonies in total. Each mini colony was set up without either queen or brood, and contained 20 workers. They were kept under the same conditions as their mother colonies, except that their plastic boxes were round (60 mm diam.), and the nest chambers were smaller (2 cm × 1.5 cm × 1 cm; Fig. 3). If any of the 20 workers died during the course of the experiment, they were replaced by others from their mother colony fragment.
For wintering (12 November–24 April, see below for details), the nests were moved to a climate-controlled chamber where the temperature was gradually decreased to 7 ± 2 °C, and then increased back to 22 ± 1 °C at the end of winter (Fig. 4).
To obtain pre-adopted P. alcon caterpillars, 15 stems of G. pneumonanthe bearing eggs of P. alcon were collected from strong plants in the field, and moved to the lab on 4 August. In the lab, stems were kept in a glass of water placed in a plastic basin. They were kept fresh for 2–3 weeks while the caterpillars emerged. One young fourth instar P. alcon caterpillar, freshly dropped from their G. pneumonanthe initial host plants, was weighed to the nearest 0.1 mg using an OHAUS Pioneer PA64C analytical balance (Parsippany, NJ) and introduced to each mini colony using a fine brush. The time spent between introduction and the first contact with a worker (discovery time) as well as the time between the first contact and adoption (adoption time) were recorded. The survival of the caterpillars was monitored weekly (on Thursdays), except during wintering, when they were disturbed less frequently. Surviving caterpillars were weighed again before the start of the wintering period, on 12 November (week 14). This study was run until the 24 April (week 34), when only one caterpillar was still alive (in an uninfected mini colony; Fig. 4).
Patterns of P. alcon and R. wasmannii infection of M. scabrinodis in the field
Myrmica scabrinodis nests within 2 m of G. pneumonanthe plants (the approximate foraging zone of Myrmica workers83) were located on 23 June. The nests were carefully opened and searched for fully-grown P. alcon larvae and pupae. The number of larvae and pupae were noted and if any were present, photographs taken in the field in order to measure their size (Fig. 5). At least 15 workers from each colony were collected and transferred to the laboratory in vials of 67.5% ethanol. Host ant identity was confirmed and workers were examined for presence of R. wasmannii under a Leica MZ125 microscope at 10–160 × magnification. Any P. alcon were then carefully replaced in the field nest, and the nesting material restored as far as possible.
Measuring prepupal caterpillars
The collected P. alcon caterpillars (and pupae) from each nest were placed onto a sheet of 5-mm square paper, and organized such that they were well separated (Fig. 5, Supplementary Note S2). Photographs of caterpillars from each nest were taken using a Nikon D3200 camera (Tokyo, Japan) with Nikon AF-S Micro NIKKOR 40 mm 1:2.8 lens, hand-held directly above the caterpillars. Photographs were taken in the shade using auto-focus in shutter-priority mode, with a fixed shutter speed of 1/60 s and ISO of 400, which resulted in apertures of between f-3.3 and f-8 under field conditions. The subjectively sharpest image from each nest was used for subsequent estimation of caterpillar sizes.
The sizes of the fully developed (prepupal) caterpillars in each nest were estimated using the Fiji image processing software84. Since they were relatively rare, the sizes of pupae (n = 6) and small (2-year developing) caterpillars (n = 8) were not estimated, and neither were sizes of damaged or clearly dead caterpillars (n = 10). Before measuring, the photographs were edited using Adobe Photoshop 2020 (San Jose, CA) to remove small traces of soil, seeds and weeds, and to create a scale bar based on the graph-paper background. The edited images were opened in Fiji84, where the area of each caterpillar was calculated based on the scale bar, using a macro script (Supplementary plugin S3). The results were saved in CSV files for statistical analysis. See Supplementary Note S2 for more details of this process.
Determination of age of workers and R. wasmannii thallus load
Altogether 225 M. scabrinodis workers (collected randomly from the centre of the nest) from colonies recorded as infected in the field (15 from each colony) were screened for thalli of R. wasmannii using a Leica MZ125 microscope at 10–160 × magnification. All fungal thalli were counted on the whole ant body. All workers collected from colonies (15 from each colony) recorded as uninfected in the field were also screened, but no thalli were found. According to the coloration of the cuticle, each worker ant from infected colonies was assigned to one of five age categories following Cammaerts-Tricot85.
Statistical analysis
The mass, discovery time, and adoption time of caterpillars were compared between infected and uninfected mini colonies using linear mixed models (LMMs), with original colony treated as random variable. The time variables were right-skewed and log-transformed for this analysis. Survival of caterpillars introduced into infected and uninfected colonies was compared using a proportional hazards survival model, with original colony nested within presence or absence of R. wasmannii. An additional survival analysis was carried out for those caterpillars that had survived until wintering and had hence been weighed on 12 November (week 14). Here caterpillar mass was also included as a covariate, together with its interaction with R. wasmannii infection status, and once again original colony nested within presence or absence of R. wasmannii.
The number of P. alcon caterpillars was compared between infected and uninfected nests in the field using a generalized linear model with negative binomial errors. The number of thalli of R. wasmannii on workers was compared between parasitized and unparasitized nests of M. scabrinodis using a generalized linear mixed model (GLMM) with negative binomial errors, which also included worker age as a covariate and colony as a random factor. The independence of infection by R. wasmannii and parasitism by P. alcon in the field was tested using a Chi-squared test for a 2 × 2 contingency table.
The size of prepupal caterpillars of P. alcon in infected and uninfected colonies was compared using a LMM, which also included a dummy variable coding whether pupae were present in the colony, and the total number of P. alcon (larvae and pupae) present, with colony as a random factor.
All analyses were carried out using JMP Pro v. 15.2.1 (SAS corporation, Cary, NC), except GLMMs, which were carried out using the glmer.nb() function of the R package lme4 version 1.1-2686.
Data availability
The data generated and analyzed during this study are available for download as Supplementary data S4.
Change history
09 January 2022
The original online version of this Article was revised: Supplementary Information files 1-4 included redundant captions of the Supplementary Information files and have subsequently been removed. As a result, the remaining Supplementary Information files have been renumbered. In addition, the labels of Supplementary note S2, Supplementary plugin S3, and Supplementary data S4 were incorrectly given as Supplementary Information 5, Supplementary Information 6, and Supplementary Information 7.
References
Frank, S. A. Models of parasite virulence. Q. Rev. Biol. https://doi.org/10.1086/419267 (1996).
Dobson, A. P. The population dynamics of competition between parasites. Parasitology https://doi.org/10.1017/S0031182000057401 (1985).
Haelewaters, D. et al. Mortality of native and invasive ladybirds co-infected by ectoparasitic and entomopathogenic fungi. PeerJ https://doi.org/10.7717/peerj.10110 (2020).
Shapiro-Ilan, D. I., Bruck, D. J. & Lacey, L. A. Principles of Epizootiology and Microbial Control. In Insect Pathology 29–72 (Elsevier, 2012). https://doi.org/10.1016/B978-0-12-384984-7.00003-8.
Renkema, J. M. & Cuthbertson, A. G. S. Impact of multiple natural enemies on immature Drosophila suzukii in strawberries and blueberries. Biocontrol https://doi.org/10.1007/s10526-018-9874-8 (2018).
Furlong, M. & Pell, J. Interactions between entomopathogenic fungi and other arthropods natural enemies. In Insect-Fungal Associations, Ecology and Evolution (eds Vega, F. & Blackwell, M.) 51–73 (Oxford University Press, 2005).
Lafferty, K. D. Interacting parasites. Science https://doi.org/10.1126/science.1196915 (2010).
Price, S. L. et al. Recent findings in fungus-growing ants: evolution, ecology, and behavior of a complex microbial symbiosis. In Genes, Behaviors and Evolution of Social Insects (eds Azuma, N. & Higashi, S.) 255–280 (Hokkaido University Press, 2003).
Telfer, S. et al. Species interactions in a parasite community drive infection risk in a wildlife population. Science https://doi.org/10.1126/science.1190333 (2010).
Carlson, C. J. et al. A global parasite conservation plan. Biol. Conserv. https://doi.org/10.1016/j.biocon.2020.108596 (2020).
Colwell, R. K., Dunn, R. R. & Harris, N. C. Coextinction and persistence of dependent species in a changing world. Annu. Rev. Ecol. Evol. Syst. https://doi.org/10.1146/annurev-ecolsys-110411-160304 (2012).
Gagne, R. B. et al. Parasites as conservation tools. Conserv. Biol. https://doi.org/10.1111/cobi.13719 (2021).
Csősz, S. & Majoros, G. Ontogenetic origin of mermithogenic Myrmica phenotypes (Hymenoptera, Formicidae). Insectes Soc. https://doi.org/10.1007/s00040-008-1040-3 (2009).
Csata, E. et al. Lock-picks: fungal infection facilitates the intrusion of strangers into ant colonies. Sci. Rep. https://doi.org/10.1038/srep46323 (2017).
Pearson, B. & Raybould, A. F. The effects of antibiotics on the development of larvae and the possible role of bacterial load in caste determination and diapause in Myrmica rubra (Hymenoptera: Formicidae). Sociobiology 31, 77–90 (1998).
Schmid Hempel, P. Evolutionary Parasitology—The Integrated Study of Infections, Immunology, Ecology, and Genetics (Oxford University Press, 2011).
Donisthorpe, J. K. The Guests of British Ants—Their Habits and Life Histories (George Routledge And Sons, Limited, 1927).
Hölldobler, B. E. & Wilson, E. O. The Ants (The Belknap Press of Harvard University Press, 1990).
Buschinger, A. Social parasitism among ants: A review (Hymenoptera: Formicidae). Myrmecol. News 12, 219–235 (2009).
Quevillon, L. E. The Ecology, Epidemiology, and Evolution of Parasites Infecting Ants (Hymenoptera: Formicidae) (Pennsylvania State University, 2018).
Quevillon, L. E. & Hughes, D. P. Pathogens, parasites, and parasitoids of ants: a synthesis of parasite biodiversity and epide-miological traits. BioRxiv https://doi.org/10.1101/384495 (2018).
Di Salvo, M. et al. The microbiome of the Maculinea-Myrmica host-parasite interaction. Sci. Rep. https://doi.org/10.1038/s41598-019-44514-7 (2019).
Witek, M., Barbero, F. & Markó, B. Myrmica ants host highly diverse parasitic communities: from social parasites to microbes. Insectes Soc. https://doi.org/10.1007/s00040-014-0362-6 (2014).
Witek, M. et al. Interspecific relationships in co-occurring populations of social parasites and their host ants. Biol. J. Linn. Soc. https://doi.org/10.1111/bij.12074 (2013).
Tartally, A. et al. Patterns of host use by brood parasitic Maculinea butterflies across Europe. Philos. Trans. R Soc. B Biol. Sci. https://doi.org/10.1098/rstb.2018.0202 (2019).
Wardlaw, J. C., Thomas, J. A. & Elmes, G. W. Do Maculinea rebeli caterpillars provide vestigial mutualistic benefits to ants when living as social parasites inside Myrmica ant nests? Entomol. Exp. Appl. https://doi.org/10.1046/j.1570-7458.2000.00646.x (2000).
Thomas, J. A. & Wardlaw, J. C. The capacity of a Myrmica ant nest to support a predacious species of Maculinea butterfly. Oecologia https://doi.org/10.1007/BF00317247 (1992).
Csata, E., Billen, J., Bernadou, A., Heinze, J. & Markó, B. Infection-related variation in cuticle thickness in the ant Myrmica scabrinodis (Hymenoptera: Formicidae). Insectes Soc. https://doi.org/10.1007/s00040-018-0628-5 (2018).
Csősz, S., Rádai, Z., Tartally, A., Ballai, L. E. & Báthori, F. Ectoparasitic fungi Rickia wasmannii infection is associated with smaller body size in Myrmica ants. Sci. Rep. https://doi.org/10.1038/s41598-021-93583-0 (2021).
Csata, E., Erős, K. & Markó, B. Effects of the ectoparasitic fungus Rickia wasmannii on its ant host Myrmica scabrinodis: Changes in host mortality and behavior. Insectes Soc. https://doi.org/10.1007/s00040-014-0349-3 (2014).
Báthori, F., Rádai, Z. & Tartally, A. The effect of Rickia wasmannii (Ascomycota, Laboulbeniales) on the aggression and boldness of Myrmica scabrinodis (Hymenoptera, Formicidae). J. Hymenopt. Res. https://doi.org/10.3897/jhr.58.13253 (2017).
Báthori, F., Csata, E. & Tartally, A. Rickia wasmannii increases the need for water in Myrmica scabrinodis (Ascomycota: Laboulbeniales; Hymenoptera: Formicidae). J. Invertebr. Pathol. https://doi.org/10.1016/j.jip.2015.01.005 (2015).
Tartally, A. Myrmecophily of Maculinea Butterflies in the Carpathian Basin (Lepidoptera: Lycaenidae), PhD thesis, https://dea.lib.unideb.hu/dea/handle/2437/78921 (University of Debrecen, Hungary, 2008)
Elmes, G. W., Wardlaw, J. C., Schönrogge, K., Thomas, J. A. & Clarke, R. T. Food stress causes differential survival of socially parasitic caterpillars of Maculinea rebeli integrated in colonies of host and non-host Myrmica ant species. Entomol. Exp. Appl. https://doi.org/10.1111/j.0013-8703.2004.00121.x (2004).
Nash, D. R., Als, T. D. & Boomsma, J. J. Survival and growth of parasitic Maculinea alcon caterpillars (Lepidoptera, Lycaenidae) in laboratory nests of three Myrmica ant species. Insectes Soc. https://doi.org/10.1007/s00040-011-0157-y (2011).
Wilson, K., Grenfell, B. T. & Shaw, D. J. Analysis of aggregated parasite distributions: a comparison of methods. Funct. Ecol. https://doi.org/10.2307/2390169 (1996).
Tartally, A., Nash, D. R., Varga, Z. & Lengyel, S. Changes in host ant communities of Alcon Blue butterflies in abandoned mountain hay meadows. Insect Conserv. Divers. https://doi.org/10.1111/icad.12369 (2019).
Csata, E., Bernadou, A., Rákosy-Tican, E., Heinze, J. & Markó, B. The effects of fungal infection and physiological condition on the locomotory behaviour of the ant Myrmica scabrinodis. J. Insect Physiol. https://doi.org/10.1016/j.jinsphys.2017.01.004 (2017).
Baylis, M. & Pierce, N. E. Lack of compensation by final instar larvae of the myrmecophilous lycaenid butterfly, Jalmenus evagoras, for the loss of nutrients to ants. Physiol. Entomol. https://doi.org/10.1111/j.1365-3032.1992.tb01186.x (1992).
Elgar, M. A. & Pierce, N. E. Mating success and fecundity in an ant-tended lycaenid butterfly. In Reproductive Success: Studies of Individual Variation in Contrasting Breeding Systems 59–75 (Chicago University Press, 1988).
Thomas, J. A., Elmes, G. W. & Wardlaw, J. C. Contest competition among Maculinea rebeli butterfly larvae in ant nests. Ecol. Entomol. https://doi.org/10.1111/j.1365-2311.1993.tb01082.x (1993).
Nash, D. R., Als, T. D., Maile, R., Jones, G. R. & Boomsma, J. J. A mosaic of chemical coevolution in a large blue butterfly. Science https://doi.org/10.1126/science.1149180 (2008).
Schlick-Steiner, B. C. et al. A butterfly’s chemical key to various ant forts: intersection-odour or aggregate-odour multi-host mimicry? Naturwissenschaften https://doi.org/10.1007/s00114-004-0518-8 (2004).
Schönrogge, K. et al. Changes in chemical signature and host specificity from larval retrieval to full social integration in the myrmecophilous butterfly Maculinea rebeli. J. Chem. Ecol. https://doi.org/10.1023/B:JOEC.0000013184.18176.a9 (2004).
Als, T. D., Nash, D. R. & Boomsma, J. J. Geographical variation in host-ant specificity of the parasitic butterfly Maculinea alcon in Denmark. Ecol. Entomol. https://doi.org/10.1046/j.1365-2311.2002.00427.x (2002).
Als, T. D., Nash, D. R. & Boomsma, J. J. Adoption of parasitic Maculinea alcon caterpillars (Lepidoptera: Lycaenidae) by three Myrmica ant species. Anim. Behav. https://doi.org/10.1006/anbe.2001.1716 (2001).
Tartally, A., Somogyi, A. Á., Révész, T. & Nash, D. R. Host ant change of a socially parasitic butterfly (Phengaris alcon) through host nest take-over. Insects https://doi.org/10.3390/insects11090556 (2020).
Thomas, J. A., Elmes, G. W., Schönrogge, K., Simcox, D. J. & Settele, J. Primary hosts, secondary hosts and ‘non-hosts’: common confusions in the interpretation of host specificity in Maculinea butterflies and other social parasites of ants. In Studies on the Ecology and Conservation of Butterflies in Europe (eds. Settele, J., Kühn, E. & Thomas, J. A.) vol. 2 99–104 (Pensoft, 2005).
Thomas, J. A. et al. Mimetic host shifts in an endangered social parasite of ants. Proc. Biol. Sci. https://doi.org/10.1098/rspb.2012.2336 (2013).
Fürst, M. A., Durey, M. & Nash, D. R. Testing the adjustable threshold model for intruder recognition on Myrmica ants in the context of a social parasite. Proc. R. Soc. B Biol. Sci. https://doi.org/10.1098/rspb.2011.0581 (2012).
Maák, I. et al. Habitat features and colony characteristics influencing ant personality and its fitness consequences. Behav. Ecol. https://doi.org/10.1093/beheco/araa112 (2021).
Chapman, B. B., Thain, H., Coughlin, J. & Hughes, W. O. H. Behavioural syndromes at multiple scales in Myrmica ants. Anim. Behav. https://doi.org/10.1016/j.anbehav.2011.05.019 (2011).
Martin, S. J., Helanterä, H. & Drijfhout, F. P. Is parasite pressure a driver of chemical cue diversity in ants? Proc. R. Soc. B Biol. Sci. https://doi.org/10.1098/rspb.2010.1047 (2011).
Nehring, V., Evison, S. E. F., Santorelli, L. A., D’Ettorre, P. & Hughes, W. O. H. Kin-informative recognition cues in ants. Proc. R. Soc. B Biol. Sci. https://doi.org/10.1098/rspb.2010.2295 (2011).
Van Zweden, J. S. et al. Blending of heritable recognition cues among ant nestmates creates distinct colony gestalt odours but prevents within-colony nepotism. J. Evol. Biol. https://doi.org/10.1111/j.1420-9101.2010.02020.x (2010).
Nash, D. R. & Andersen, A. Maculinea-sommerfugle og stikmyrer på danske heder—coevolution i tid og rum. Flora og Fauna 121, 133–141 (2015).
Haelewaters, D., Boer, P., Gort, G. & Noordijk, J. Studies of Laboulbeniales (Fungi, Ascomycota) on Myrmica ants (II): variation of infection by Rickia wasmannii over habitats and time. Anim. Biol. https://doi.org/10.1163/15707563-00002472 (2015).
Dallas, T. A., Laine, A.-L. & Ovaskainen, O. Detecting parasite associations within multi-species host and parasite communities. Proc. R. Soc. B Biol. Sci. https://doi.org/10.1098/rspb.2019.1109 (2019).
Herczeg, D., Ujszegi, J., Kásler, A., Holly, D. & Hettyey, A. Host–multiparasite interactions in amphibians: a review. Parasit. Vectors https://doi.org/10.1186/s13071-021-04796-1 (2021).
Bronstein, J. L. Conditional outcomes in mutualistic interactions. Trends Ecol. Evol. https://doi.org/10.1016/0169-5347(94)90246-1 (1994).
Zhang, Z., Yan, C. & Zhang, H. Mutualism between antagonists: Its ecological and evolutionary implications. Integr. Zool. https://doi.org/10.1111/1749-4877.12487 (2021).
Rogalski, M. A., Stewart Merrill, T., Gowler, C. D., Cáceres, C. E. & Duffy, M. A. Context-dependent host-symbiont interactions: Shifts along the parasitism-mutualism continuum. Am. Nat. https://doi.org/10.1086/716635 (2021).
Pfliegler, W. P., Báthori, F., Haelewaters, D. & Tartally, A. Studies of Laboulbeniales on Myrmica ants (III): myrmecophilous arthropods as alternative hosts of Rickia wasmannii. Parasite https://doi.org/10.1051/parasite/2016060 (2016).
Chouvenc, T., Efstathion, C. A., Elliott, M. L. & Su, N.-Y. Resource competition between two fungal parasites in subterranean termites. Naturwissenschaften https://doi.org/10.1007/s00114-012-0977-2 (2012).
Lawton, J. H. & Hassell, M. P. Asymmetrical competition in insects. Nature https://doi.org/10.1038/289793a0 (1981).
Price, P. W. Evolutionary Biology of Parasites (Princeton University Press, 1980).
Nash, D. R. & Boomsma, J. J. Communication between hosts and social parasites. In Sociobiology of Communication (eds D’Ettorre, P. & Hughes, D. P.) 55–80 (Oxford University Press, 2008).
Tartally, A., Szűcs, B. & Ebsen, J. R. The first records of Rickia wasmannii Cavara, 1899, a myrmecophilous fungus, and its Myrmica Latreille, 1804 host ants in Hungary and Romania (Ascomycetes: Laboulbeniales; Hymenoptera: Formicidae). Myrmecol. News 10, 123 (2007).
Radchenko, A. G. & Elmes, G. W. Myrmica (Hymenoptera: Formicidae) ants of the Old World. vol. 6 (Fauna Mundi 3, 2010).
Tragust, S., Tartally, A., Espadaler, X. & Billen, J. Histopathology of Laboulbeniales (Ascomycota: Laboulbeniales): ectoparasitic fungi on ants (Hymenoptera: Formicidae). Myrmecol. News 23, 81–89 (2016).
Haelewaters, D., Boer, P. & Noordijk, J. Studies of Laboulbeniales (Fungi, Ascomycota) on Myrmica ants: Rickia wasmannii in the Netherlands. J. Hymenopt. Res. https://doi.org/10.3897/JHR.44.4951 (2015).
Espadaler, X. & Santamaria, S. Ecto- and endoparasitic fungi on ants from the Holarctic Region. Psyche, 2012, 168478. https://doi.org/10.1155/2012/168478 (2012).
Báthori, F., Pfliegler, W. P., Zimmerman, C.-U. & Tartally, A. Online image databases as multi-purpose resources: discovery of a new host ant of Rickia wasmannii Cavara (Ascomycota, Laboulbeniales) by screening AntWeb.org. J. Hymenopt. Res, 61, 85-94. https://doi.org/10.3897/jhr.61.20255 (2017).
Riddick, E. W. Ectoparasitic mite and fungus on an invasive lady beetle: parasite coexistence and influence on host survival. Bull. Insectol. 63, 13–20 (2010).
Konrad, M., Grasse, A. V, Tragust, S. & Cremer, S. Anti-pathogen protection versus survival costs mediated by an ectosymbiont in an ant host. Proc. Biol. Sci. https://doi.org/10.1098/rspb.2014.197620141976 (2015).
De Kesel, A., Haelewaters, D. & Dekoninck, W. Myrmecophilous Laboulbeniales Ascomycota in Belgium. Sterbeeckia 34, 3–6 (2016).
Haelewaters, D. The first record of Laboulbeniales (Fungi, Ascomycota) on Ants (Hymenoptera, Formicidae) in The Netherlands. Ascomycete.org 4, 65-69 (2012).
van Swaay, C. et al. European Red List of Butterflies (Publications Office of the European Union, 2010).
Gergely, P. & Hudák, T. Revision of threatened butterfly species in Hungary (Lepidoptera: Rhopalocera). Lepidopterol. Hungarica https://doi.org/10.24386/lephung.2021.17.1.27 (2021).
Wallis de Vries, M. Code rood voor het gentiaanblauwtje. Vlinders 4, 5–8 (2017).
Barbero, F., Thomas, J. A., Bonelli, S., Balletto, E. & Schönrogge, K. Queen ants make distinctive sounds that are mimicked by a butterfly social parasite. Science https://doi.org/10.1126/science.1163583 (2009).
Thomas, J. A., Elmes, G. W., Wardlaw, J. C. & Woyciechowski, M. Host specificity among Maculinea butterflies in Myrmica ant nests. Oecologia https://doi.org/10.1007/BF00378660 (1989).
Elmes, G. W. et al. The ecology of Myrmica ants in relation to the conservation of Maculinea butterflies. J. Insect Conserv. https://doi.org/10.1023/A:1009696823965 (1998).
Schindelin, J. et al. Fiji: an open-source platform for biological-image analysis. Nat. Methods https://doi.org/10.1038/nmeth.2019 (2012).
Cammaerts-Tricot, M.-C. Ontogenesis of the defence reactions in the workers of Myrmica rubra L. (Hymenoptera: Formicidae). Anim. Behav. https://doi.org/10.1016/0003-3472(75)90058-5 (1975).
Bates, D., Mächler, M., Bolker, B. & Walker, S. Fitting linear mixed-effects models using lme4. J. Stat. Softw. https://doi.org/10.18637/jss.v067.i01 (2015).
Acknowledgements
We thank Emese Nagyné Kapczár for secretarial help and project administration, and the staff of the Bükk National Park for supporting this research. We are grateful to Ádám Bakos and Márton József Paulin for the photos of Figures 1 and 2. Danielle Castronova, archivist at the Harvard University Herbaria & Libraries, is acknowledged for scanning the original Thaxter drawing of R. wasmannii for Figure 1. A.T. was supported by a ‘János Bolyai’ Scholarship of the Hungarian Academy of Sciences, by the EFOP-3.6.1-16-2016-00022 project (co-financed by the European Union and the European Social Fund), and by the ÚNKP-18-4, ÚNKP-19-4, and ÚNKP-20-5 New National Excellence Program of the Ministry of Human Capacities. A.Á.S. was supported by the EFOP-3.6.1-16-2016-00022 project. D.H. receives support from the Research Foundation—Flanders (Junior Postdoctoral Fellowship No. 1206620N).
Author information
Authors and Affiliations
Contributions
A.T., A.Á.S, F.B., N.S., and A.M. contributed to the study conception and design. Data preparation and data collection was performed by A.T., N.S., A.Á.S, F.B., A.M., and Á.F.-M. Data analyses were performed by D.R.N. The first draft of the manuscript was written by A.T., D.R.N., D.H., A.Á.S, F.B., and N.S. All authors commented on revised versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tartally, A., Szabó, N., Somogyi, A.Á. et al. Ectoparasitic fungi of Myrmica ants alter the success of parasitic butterflies. Sci Rep 11, 24031 (2021). https://doi.org/10.1038/s41598-021-02800-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-02800-3
This article is cited by
-
Laboratory experiments of Stigmatomyces majewskii (Laboulbeniales: Laboulbeniaceae) infection on Drosophila suzukii (Diptera: Drosophilidae)
Applied Entomology and Zoology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.