Abstract
High-altitude-adapted ectotherms can escape competition from dominant species by tolerating low temperatures at cooler elevations, but climate change is eroding such advantages. Studies evaluating broad-scale impacts of global change for high-altitude organisms often overlook the mitigating role of biotic factors. Yet, at fine spatial-scales, vegetation-associated microclimates provide refuges from climatic extremes. Using one of the largest standardised data sets collected to date, we tested how ant species composition and functional diversity (i.e., the range and value of species traits found within assemblages) respond to large-scale abiotic factors (altitude, aspect), and fine-scale factors (vegetation, soil structure) along an elevational gradient in tropical Africa. Altitude emerged as the principal factor explaining species composition. Analysis of nestedness and turnover components of beta diversity indicated that ant assemblages are specific to each elevation, so species are not filtered out but replaced with new species as elevation increases. Similarity of assemblages over time (assessed using beta decay) did not change significantly at low and mid elevations but declined at the highest elevations. Assemblages also differed between northern and southern mountain aspects, although at highest elevations, composition was restricted to a set of species found on both aspects. Functional diversity was not explained by large scale variables like elevation, but by factors associated with elevation that operate at fine scales (i.e., temperature and habitat structure). Our findings highlight the significance of fine-scale variables in predicting organisms’ responses to changing temperature, offering management possibilities that might dilute climate change impacts, and caution when predicting assemblage responses using climate models, alone.
Similar content being viewed by others
Introduction
The signature of climate change is now apparent in changes in species’ distributions, as species move in response to warming. For example, the median altitudinal range change is 11 m upwards per decade1. Mountain-dwelling assemblages often include many endemics adapted to narrow niches, isolated in a landscape matrix that limits dispersal2,3,4. These assemblages are particularly susceptible to climate change, as high-altitude zones should experience faster rates of warming5. Upslope migration of organisms into smaller areas6, when habitat allows, increases interspecific competition and reduces population sizes7. Aspect is also influential, as equator-facing slopes experience greater radiation and higher temperatures than slopes facing away from the equator8. These differences in abiotic conditions are reflected in the structure and composition of vegetation on the different mountain aspects, and in species’ distributions and behaviour on mountain slopes9. For example, the red ant Myrmica sabuleti is primarily limited to south-facing slopes in the United Kingdom, where temperatures are sufficiently warm10. Shifts in temperature regimes associated with climate change should affect ectotherms, in particular, as they have fewer physiological adaptations for coping with temperature changes than endotherms11.
Ants (Hymenoptera: Formicidae) are abundant and found on most continents12. Their community composition and role in ecosystem functioning can effect considerable changes in ecosystem structure and function13,14,15,16. They are considered thermophilic (i.e., heat-loving)17, and ant species richness can decrease with altitude (along with decreasing temperature), or exhibit mid-elevational peaks18,20. Cool temperatures should limit ant species’ distributional ranges3, with some species occupying areas only because they can tolerate lower temperatures than more dominant species19,20. Displacement with climate change of cold-adapted species by those that can tolerate warmer temperatures has now been found in two Aphaenogaster ant species in the Appalachian Mountains21. Different temperature tolerances imply that ant species assemblages should change with increasing elevation to either a subset of species that can tolerate low temperatures, or to a distinct assemblage found only at higher elevation sites, and ant assemblages on the pole-facing aspect of mountains could be similar to those found at higher elevations on the equator-facing side. Alternatively, if ants are more influenced by habitat structure, which may differ between the two aspects, ant assemblages should differ significantly between the two mountain aspects.
Microclimates, for example, those created by topography22, vegetation15,23 or soils24, can modify temperature extremes, buffering against climate change. Microclimates may allow species with different thermal tolerances to coexist, increasing diversity25. Thus, any consideration of effects of altitude on species assemblages should include not only the larger-scale measures of altitude and aspect, but also finer scale measures of vegetation and soils, which can also influence ant assemblages26. Furthermore, factors like vegetation cover are amenable to management interventions, allowing for some adaptation to climate change27,28.
In the Soutpansberg, in tropical north-eastern South Africa, the impact of aspect is considerable: climate differs markedly between the north and southern aspects, owing to the mountain’s East–North–East to West–South–West orientation, resulting in arid slopes on the northern aspect characterised by open dry savanna, and thicket or forest on the mesic southern aspect, with herbaceous habitats at the highest elevations18,29. The region is vulnerable to global change, currently experiencing reduced summer rainfall, elevated surface temperatures, and widespread, more frequent droughts23,30. Recent models reveal contraction of species’ ranges at higher elevation from montane regions in two southern African countries4. In our study site, ants show a decrease in species diversity with increasing altitude18, and at higher altitude sites, diversity over time seems to be more variable31. Here, we assess whether ant species composition across elevational gradients changes owing to species turnover or species loss, by assessing the relative contribution of these two components to beta diversity over 6 years. We expect total beta diversity for mountain slopes to increase with increasing elevation, and perhaps also with time. Ants have been found to display patterns of species turnover with change in elevation32,33, so we expect the species turnover component to represent most beta diversity, with little contribution from species nestedness. We also evaluate whether ant assemblages at different elevations and aspects of the Soutpansberg mountain vary with altitude and/or aspect as well as fine-scale vegetation measures.
Given that species assemblage composition influences the traits of species present and thus the functions they perform, we also investigate patterns in functional diversity with different elevations, vegetation, soil structure and aspect. Functional diversity (FD) is the range and value of species traits present within a community, which influences how those species contribute to ecological function34. Thus FD reflects ecosystem pattern and process35. With global change, there is growing interest in how FD varies across environmental gradients33,36,37. Given that heating could be associated with changes to species composition and the number of species, particularly at altitude31, patterns of FD with altitude and aspect may give insights into whether and how the role of ants in ecosystem functioning might change, or whether species are merely replaced by functional analogues along abiotic or biotic gradients. At environmental extremes, resources are increasingly limited, which can amplify environmental filtering38,39. Such filtering can select for functionally-unique traits which enable species to cope with extreme conditions, with only species possessing the necessary traits persisting. A pattern of increasingly-nested subsets of available functional strategies with rising altitude has recently been found for ants elsewhere33, so we might expect lower FD at higher altitudes.
Most studies are carried out over relatively short time periods, delivering only snapshots of pattern and process. Here, we collected data biannually for six consecutive years, producing one of the largest standardised, spatio-temporal invertebrate assemblage datasets assembled to date. We asked (i) whether ant assemblages at high altitude are subsets of those found at lower altitudes, or whether they are a different set of species, and whether there is evidence of change in beta diversity over the relatively short period of 6 years; (ii) whether ant assemblages respond primarily to abiotic factors like temperature and solar radiation, or primarily to habitat structure, or whether both abiotic and biotic factors influence species composition; and (iii) whether FD displays patterns with elevation, aspect and/or habitat structure.
Materials and methods
Study site
Ants were sampled in the Soutpansberg Mountains, Vhembe Biosphere Reserve, a recognised southern African centre of endemism40. We sampled 11 sites spaced between 160 to 290 m apart (Electronic Supplementary Material Fig. 1) across an elevational transect (beginning at 23° 02′ 16.91ʺ S, 29° 26′ 34.22ʺ E) running north to south, starting at 800 m above sea level (a.s.l.) on the southern aspect, ascending to 1700 m a.s.l., before descending to 800 m a.s.l. on the northern aspect. Erosion-resistant quartzite, conglomerate, sandstone, shale rocks, and basalt characterise the transect41, which has wet summers, dry winters and mean annual precipitation of about 450 mm42.
Ant sampling
Epigaeic ants were sampled each January (wet season) and September (dry season) from 2009 to 2015, at the 11 sites. Each site housed four replicates, spaced 300 horizontal metres apart to avoid pseudo-replication43. Each replicate contained ten pitfall traps (ø 62 mm), 10 m apart, in a grid of two parallel lines (2 × 5). Traps containing a 50% solution of propylene glycol were left open for 5 days in each survey. Ants from traps were then collected and identified to species level where possible, otherwise to genus and then morphospecies. Specimens are lodged in the University of Venda Natural History Collection housed in the Chair on Biodiversity Value and Change.
Vegetation structure sampling
Horizontal habitat (vegetation) structure was quantified using a 1 m2 grid placed over each pitfall trap, and percentage area of bare ground, vegetation, rock and leaf litter was calculated. To quantify vertical habitat structure, we used four rods (1.5 m high) counting the number of vegetation contacts recorded along 25 cm intervals of the rod (0–25 cm, 25–50 cm, 50–75 cm, 75–100 cm, 100–125 cm, 125–150 cm, 150 + cm). These four rods were placed 1.5 m away from the pitfall trap, in a straight line originating at the pitfall trap and passing through each corner of the 1 m2 grid. To summarize vertical and horizontal habitat structure, we performed a Principal Component Analysis (PCA) on the vegetation measures we had gathered for vegetation structure, to produce two main axes for horizontal (i.e., pch1 and pch2) and vertical (pcv1 and pcv2) vegetation structure.
Soil sampling
In January 2010, ten soil samples were taken from each replicate using a soil auger, 7.5 cm in diameter and 10–15 cm deep. The ten samples were mixed, dried, and analysed by BemLab (Pty) Ltd laboratories, South Africa for composition (clay, sand, rock and silt), pH, conductivity, C, K, Na, Ca, Mg, P, and NO3. We then summarised soil characteristics using a PCA, for which the two main axes were pcsoil1 and pcsoil2.
Temperature measurement
Within two replicates at each site, one Thermocron iButton (Semiconductor Corporation, Dallas/Maxin TX and USA), buried 1 cm below the soil recorded temperature at hourly intervals for the entire period of sampling. These readings were used to generate maximum (Tmax), mean (Tmean) and minimum (Tmin) temperature readings for each site and sampling period.
Functional traits
We measured seven morphological traits (Table 1) relating to resource use by ants44,45 using a ZEISS Discovery V12 Modular Stereo Microscope with Motorized 12 × Zoom Module and Zeiss Application Suite V3.024, calibrated using a stage micrometer (Jena, Germany). A minimum of five individuals were measured for each species. Only minor workers were measured. There may be microclimatic variation in species’ sizes, so we aimed to measure specimens from each elevation. For most species, this was not possible, however, as they did not occur across the altitudinal range, or there were too few specimens to assess intraspecific size variation.
Data analysis
Sampling sufficiency
We compiled sample-based rarefaction curves for ants for each altitude at each aspect using the nonparametric Incidence Coverage Estimator (ICE) and Michaelis–Menten richness estimate to determine adequacy of sampling, in the programme EstimateS46. Sampling was deemed adequate when values converged at the highest values (Electronic Supplementary Material Table 1).
Ant assemblage beta diversity
We assessed whether ant assemblages at high altitudes were subsets of those found at lower altitudes, or whether they comprised completely different species, and whether there was a signal of change in ant species composition over the 6 years of the study, using nestedness and turnover components of beta diversity47. βsor gives the total variation in composition between assemblages, and is the sum of variation owing to nestedness (βsne) and species turnover (βsim). We first assessed the components of beta diversity on data from traps combined from all years, per site, and then also on sites only on the north and south aspects, using the package “betapart”48 on presence-absence data. Then, using presence-absence data for the different dates of sampling, we plotted a dendrogram to allow visualisation of the different plots at different times, clustering using group averaging. This dataset allowed us to assess decay in similarity between assemblages, by fitting a negative exponential function to increasing assemblage dissimilarity over spatial (i.e., altitude within aspect) and temporal (i.e., total days from the first day of sampling) distances. To assess temporal changes in beta diversity, we allocated sites to one of three categories (low: < 1000 m a.s.l., medium: 1000 – 1400 m a.s.l, and high: > 1400 m).
Ant assemblage composition
We pooled assemblage data for traps within an elevational band and aspect, but kept these separate for sampling season and year. These multivariate abundance data formed the response variable for generalised linear models (GLM), testing for differences in assemblage composition with altitude, aspect, season and interactions between them, as well as measures of habitat (i.e., horizontal and vertical vegetation and soil structure, minimum, maximum and mean temperatures). We assessed these using the R package “mvabund”49 with the functions “manyglm” and “anova.manyglm”, using a negative binomial distribution with log-link. This approach accounts for confounding mean–variance relationships, which are common in abundance data containing many zeros50. Likelihood ratio statistics are summed for each species, yielding a community-level measure for each altitude and aspect category, and the PIT-residual bootstrap method51 derives p-values by resampling 999 rows of the dataset. Models were checked for departure from model assumptions by visually examining plots of Dunn-Smyth residuals against fitted values to identify any non-random patterns. We also performed an unconstrained ordination, using the package “boral” in R52, modelling species abundance data using a negative binomial distribution and plotting the relationships between sites in two dimensions. To help visualise these assemblages, we allocated sites to one of five altitudinal bands, each spanning 180 m. These were: A = 810–990 m; B = 991–1170 m; C = 1171–1350 m; D = 1351–1530 m; E = 1531 m–1710 m; and north (N) and south (S) aspect categories. Thus AS indicates traps within the elevational band of 810 – 990 m a.s.l. on the southern slope, and CN indicates traps between 1171 and 1350 m on the northern aspect.
Functional diversity across an altitudinal gradient
We used FD as measured by Petchey and Gaston53. Trait data were standardized to ensure biological variation within each trait was treated equally (each trait thus had a mean of zero and a standard deviation of one). We gave Weber’s length a double weighting as it reflects not only habitat complexity and metabolic function, but is indicative of body size (key in thermoregulation44,54). We used Gower distance to convert the species by trait matrix to a distance matrix, and clustered the matrix using the “average” method55, which produced the highest cophenetic correlation (0.87) between estimated and original distances arising from the dendrogram. Thus, each species was given a measure of similarity with every other species, based on the functional traits used. Adding a species to an assemblage increases the likelihood of adding new traits, so FD tends to increase with species richness56. To account for this, simulation models were adopted, comparing observed FDs against a null distribution of FD values. This allows calculation of standardised effect size (sesFD), which corrects for increases in FD with added species, providing a reliable measure of degree of functional differentiation within assemblages (sesFD). We calculated sesFD using the analogous ses.pd function in the R package “picante”57. For a site with n species, the simulation models randomly select n species from the total pool without replacement, and formulate an expected FD for that group of species. We used the independent swap method, in which species number was maintained within each sample, but the number of sites in which each species occurred was kept constant, so that species were only included in simulations proportional to their occurrence in the dataset. We ran models 999 times, creating a distribution of 1000 values for each observed value. The mean of this distribution was then subtracted from the observed FD value and divided by the standard deviation of the null distribution, to provide sesFD. Thus sesFD reflects the number of standard deviations in which the observed community is above or below the mean (0) of the simulated communities from the null model58.
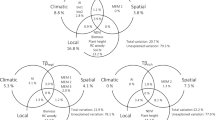
To assess how sesFD changed with environmental variables, we used a general linear mixed model, with sesFD as the dependent variable, and altitude, aspect, vegetation (represented by pch1, pch2, pcv1 and pcv2), soils (pcsoil1 and pcsoil2), temperature (TMax, Tmean and TMin), and interactions between altitude and aspect, as explanatory variables, with site as a random intercept variable, using the package nlme59 in R statistical programme version 3.0. We included an interaction between altitude and aspect because a lower altitude on the south side could have the same sesFD as a higher altitude on the north side, owing to the effects of aspect on temperature. We used the “dredge” function in MuMIn60 to find the best models by specifying that only explanatory variables correlated with Pearson correlation < 0.5 could be included in any single model, and identified the best models as those within two units of that with the lowest AICc, and thus have substantial support of being the best models61. We then used model averaging on these models. We calculated variance explained by fixed (marginal R2), and fixed and random effects (conditional R2, 62) for the best and worst models included in the model averaging, and report on both of these. We checked for constant error variance by plotting residuals against fixed values, and quantile–quantile plots to assess normality of errors for these models.
Results
A total of 102,496 ants representing 35 genera and 122 species were caught in pitfall traps over 6 years of sampling. Of these, 102 species had five or more individuals, and provided reliable, standardised trait data for use in FD analysis. Few species were present in sufficient abundance across sites along the elevational gradient to allow specification of a size per altitude. Furthermore, interspecific variation was greater than intraspecific variation, so we used mean measures for ants across all sites as traits.
Vegetation structure
For horizontal habitat structure, the first axis (pch1) captured 41%, the second (pch2) 30% of the variation, whilst for vertical habitat structure, these two axes captured 37% (pcv1) and 24% (pcv2) of variation. The first principal component axis for horizontal habitat structure (pch1) correlated positively with bare ground and negatively with vegetation cover. The second (pch2) correlated positively with leaf litter cover and negatively with rock cover (Electronic Supplementary Material Fig. 2a). For vertical habitat structure, pcv1 increased with vertical structure, particularly at levels between 50 and 125 cm. The second axis, pcv2, correlated positively with canopy cover above 100 cm in height, but negatively with cover below 75 cm (Electronic Supplementary Material Fig. 2b).
Soil characteristics
The PCA for soils explained 61% of the variation, the first axis explaining 46%, the second, 15%. The first principal component axis (pcsoil1) was positively correlated with acidic soil and negatively with basic soils. The second principal component axis (pcsoil2) was positively correlated with sites that had sandy soil and negatively with clay (Electronic Supplementary Material Fig. 3).
Beta diversity of ant assemblage composition: nestedness or turnover
Beta diversity was primarily driven by species turnover. For north and south sites together, total beta diversity was 0.682, the component explained by nestedness was only 6.1% (0.042), with turnover explaining the remaining 93.9% (0.641). When only sites on the northern aspect were compared, total beta diversity was 0.510, the nestedness component was 0.069 (13.5%), the remaining 86.5% (0.441) was species turnover. On the southern aspect, total beta diversity was similar to that of the northern aspect, at 0.500; the nestedness component was lower as a proportion than for the northern aspect, however, at 0.043 (8.7%) and turnover was 91.3% (0.453).
Beta-decay models show increasing total beta diversity, βsor with elevation, although the rate of increase slowed with increasing elevation (Fig. 1a). The trend of increasing βsor over space was statistically significant (βslope = 0.00084, n = 8645, pseudo r2 = 0.26, p = 0.01). The component of beta diversity owing to nestedness declined (Fig. 1b), whilst that associated with turnover increased (Fig. 1c), with altitude.
The relationship between ant species (a) total beta diversity, βsor; comprised of its (b) nestedness-resultant component, βnes, and (c) turnover component, βsim for the Soutpansberg, South Africa. The red and blue lines represent the mixed model predictions for north and south aspects, based on data for all years, with each data point representing a comparison between the lowest elevation sites (810 m) and subsequent higher elevations.
A comparison of beta decay over time found no significant patterns for low (p = 0.78) or medium (p = 0.61) altitude ant communities over the 6 years of our study, but there was a significant signal of change in beta diversity over time for high altitude sites, although the explanatory power was low (βslope = 5.4 × 10–5, n = 1127, pseudo r2 = 0.01, p = 0.01, Electronic supplementary Fig. 4).
Ant assemblages relative to aspect and known altitudinal thresholds
Ant assemblages tended to be distinct between the two aspects, and altitude and the first component of horizontal vegetation structure was also significant (global test statistic = 57.8, resid. df = 123; p < 0.001; Table 2, Fig. 2). Cluster analysis of βsor (Electronic Supplementary Material Fig. 5) shows that communities stayed within their altitudinal ranges over time. At lower elevations (i.e., < 1530 m), ant assemblages were distinct between aspects, with assemblages found on the northern aspect quite different to those found on the south. Within those aspect groups, species composition also varied with elevation, but more markedly on the southern aspect, particularly for species above 1170 m (Fig. 2). At the highest elevations, however, aspect no longer delineated a difference between ant assemblages, with high altitude assemblages being significantly different from those at lower altitudes on either aspect (Fig. 2).
Functional diversity (sesFD) across an altitudinal gradient
The best models explaining functional differentiation amongst ant species across the altitudinal gradient over the 6 years of our study did not include altitude, but included aspect and other factors associated with fine-scale habitat measures, such as horizontal and vertical vegetation structure, mean or minimum temperature. One model included season, and another included a measure of soil (Table 3). The best model obtained from model averaging found that sesFD was significantly lower on the southern aspect relative to the north, and was higher with increasing mean and minimum temperatures; however, the microclimate provided by vegetation in this setting also appears to be key: functional differentiation decreased with pch1 and pcv2, in other words, functional diversity increased with higher vegetation cover in the lower stratum and decreased when vegetation structure at higher strata became more complex. sesFD also increased with soil sandiness. The best-averaged model was:
For the best model, fixed effects explained 39.8% of the variation, random effects (i.e., site) explained almost no extra variation. The worst model of those within 2 AICc of the best explained 38.6% of the variation, again, random effect explained almost no additional variation. A plot of aspect and altitude shows the variation in sesFD with altitude (not significant) and aspect (significantly lower on the southern aspect; Fig. 3).
Discussion
Species composition change with altitude was driven primarily by species turnover, with a low contribution from nestedness. Thus each elevation has its own unique set of species, with the higher elevations, in particular, having unique sets of species. In addition to ant assemblages varying with both altitude and aspect, horizontal vegetation cover also influenced species composition (Table 2). We observed that the primary driver of species assemblages is altitude, which may be unsurprising, given that ectothermic species are more vulnerable to temperature fluctuations than endotherms, and species found at higher elevations occur in naturally isolated altitudinal niches4,63. Elsewhere, ant species occurring at higher altitudes have been found to occur at those elevations because they can tolerate cold temperatures better than more dominant species found at lower elevations either through physiology or behaviour3,21. Further assessment of the ability of ant species in this study to withstand temperature extremes would help to confirm or refute these findings, particularly if lower-altitude species are particularly cryophobic. Climate warming might pose a threat to these high elevation species, which could be outcompeted by species from lower elevations.
The highest sites on both sides of the mountain clustered together in the ordination, distinct from those at lower elevations on either side of the mountain. In the ordination, ant assemblages at middle and lower elevation sites also showed some separation according to altitude, but within their aspects (Fig. 2; Table 2). Habitat structure differed considerably between the two aspects at all altitudes except at the highest sites, where the vegetation structure was similar between north- and south-facing slopes. That the highest sites clustered together could also be partly explained by proximity: sites at the top of the mountain were closer to each other than those lower down, resulting in less dispersal limitation.
Partitioning of beta diversity suggests that the species found at these higher elevations are distinct from those lower down, and not a subset of lower elevation assemblages. This is of particular concern within the context of global climate change as this subset of species could go extinct. This is further emphasized by the decrease in nestedness with elevational distance, highlighting the uniqueness of these higher elevation sites. Over the 6 year period, assemblages do not seem to have changed, except perhaps those at higher elevations. Six years is a relatively short period over which to observe changes in species assemblages, and this explains the low explanatory power of time (r2 = 0.01). Nevertheless, this trend was significant and raises flags for the importance of monitoring high (and low) altitude assemblages over the long term.
Patterns of functional diversity across an altitudinal gradient
Interestingly, although species composition was well explained by altitude, the functional diversity of those species was not. This suggests that as species composition changes with altitude, the functional traits represented amongst these species responds to other environmental factors, in this case, primarily habitat structure, along with temperature and soils.
Overall trends in FD across ecological gradients are beginning to emerge in the literature, although patterns vary33,36,39,64. For ant assemblages at altitude, our models found that FD (measured as sesFD; Fig. 3, Table 3) was lower on the southern aspect, but did not show a clear pattern with altitude. FD did vary with mean and minimum temperature, which vary with altitude, but also with habitat structure. Habitat structure, measured as horizontal and vertical structure emerged as significantly related to sesFD. As horizontal habitat structure linked to vegetation cover, increased, so too did sesFD (because pch1 was negatively correlated with horizontal habitat structure). Yet sesFD was negatively correlated with pcv1, which was a measure of vertical habitat structure, particularly that at > 50 cm height. Thus, it seems as if horizontal structure at low levels creates habitat complexity for ground-dwelling ants, so increasing functional differentiation (sesFD), but plant cover over 50 cm does not contribute to habitat structure, but may serve only to reduce ground temperature, which would be expected to be negative for ectothermic species. Plant cover above 50 cm in height is associated with increased shading and woody thickening, and is associated with relatively little ground cover. The implications of our findings are that studies focused on large scale measures like altitude may risk missing fine-scale changes associated with heterogeneous habitats. Habitat structure buffers changes in temperature and models based on temperature alone could overestimate the impact of climate change65,66. Fine-scale data collection is often laborious and costly, yet these data may make valuable contributions to large-scale models that would otherwise overlook microclimatic effects23. Adaptive Dynamic Global Vegetation Models can be used to simulate state variables causally linked to these fine scale variables67. These models also suggest that there could be a considerable lag in how vegetation responds to climate change, further confounding models based on temperature alone68. The importance of microclimate in dictating fine-scale biodiversity is increasingly recognised9,69,70,71,72. Here, we find that FD is well explained by finer-scale measures of habitat.
That FD, and to some extent species composition, are influenced by fine-scale factors implies that predicting species’ responses to climate change is complicated by habitat structure and may not be well predicted by only broad-scale predictors such as temperature and elevation66. This has implications for management: if vegetation structure can be maintained and managed, ectothermic species may be less impacted than expected, and it may be possible to ameliorate some of the inexorable effects of climate change, with careful conservation strategies. At the same time, drought intensity and frequency are predicted to increase73,74, with a risk that precipitation-dependent vegetation may die off.
At the regional scale, our findings shed light on the relative importance of high-altitude sites in Afromontane systems, which harbour distinct species assemblages. Vhembe Biosphere has recently received calls for all areas of high altitude to be declared core conservation zones, and our findings support this directive. Given that altitude is a major predictor of assemblage composition, as macro-climate warms, species should move upslope, as organisms attempt to remain within ambient temperatures to which they are adapted6. However, given that the climatically coolest areas are at the apex of mountains, such zones are by definition small in area. Therefore, with gradual movement up a gradient in response to warming, available habitat for a given species will likely shrink incrementally over time, ultimately jeopardising persistence6. Species at the lowest elevations may not be replaced, but lowest elevation communities may simply suffer species attrition75. Here, we found that ant species assemblages at each elevation are distinct, that nestedness makes very little contribution to beta diversity. In the context of global change, microclimates may be key to modulating the potentially deleterious impacts to high-altitude processes at a range of scales.
Data availability
Intended archive: ResearchGate.
References
Chen, I.-C., Hill, J. K., Ohlemüller, R., Roy, D. B. & Thomas, C. D. Rapid range shifts of species associated with high levels of climate warming. Science (80-). 333, 1024–1026 (2011).
Beniston, M. Climatic change in mountain regions: A review of possible impacts. Clim. Chang. 5, 5–31 (2003).
Bishop, T. R., Robertson, M. P., van Rensburg, B. J. & Parr, C. L. Coping with the cold: Minimum temperatures and thermal tolerances dominate the ecology of mountain ants. Ecol. Entomol. 42, 105–114 (2017).
Bentley, L. K., Robertson, M. P. & Barker, N. P. Range contraction to a higher elevation: The likely future of the montane vegetation in South Africa and Lesotho. Biodivers. Conserv. 28, 131–153 (2019).
Pepin, N. et al. Elevation-dependent warming in mountain regions of the world. Nat. Clim. Chang. 5, 424–430 (2015).
Peters, R. L. & Darling, J. D. S. The greenhouse effect and nature reserves. Bioscience 35, 707–717 (1985).
MacArthur, R. & Wilson, E. The Theory of Island Biogeography (Princeton University Press, Princeton, 1967).
Soliveres, S., DeSoto, L., Maestre, F. T. & Olano, J. M. Spatio-temporal heterogeneity in abiotic factors modulate multiple ontogenetic shifts between competition and facilitation. Perspect. Plant Ecol. Evol. Syst. 12, 227–234 (2010).
Suggitt, A. J. et al. Habitat microclimates drive fine-scale variation in extreme temperatures. Oikos 120, 1–8 (2011).
Thomas, J. A., Rose, R. J., Clarke, R. T., Thomas, C. D. & Webb, N. R. Intraspecific variation in habitat availability among ectothermic animals near their climatic limits and their centres of range. Funct. Ecol. 13, 55–64 (1999).
Porter, W. P. & Gates, D. M. Thermodynamic equilibria of animals with environment. Ecol. Monogr. 39, 227–244 (1969).
Del Toro, I., Ribbons, R. R. & Pelini, S. L. The little things that run the world revisited: A review of ant-mediated ecosystem services and disservices (Hymenoptera: Formicidae). Myrmecol. News 17, 133–146 (2012).
Wilson, E. The little things that run the world (the importance and conservation of invertebrates). Conserv. Biol. 1, 344–346 (1987).
Folgarait, P. J. Ant biodiversity and its relationship to ecosystem functioning: A review. Biodivers. Conserv. 7, 1221–1244 (1998).
Seymour, C. & Joseph, G. Ecology of Smaller Animals Associated with Savanna Woody Plants. in Savanna Woody Plants And Large Herbivores (eds. Scogings, P. & Sankaran, M.) 183–212 (2019).
Palmer, T. M. et al. Breakdown of an ant-plant mutualism follows the loss of large herbivores from an African savanna. Science (80-). 319, 192–195 (2008).
Hölldobler, B. & Wilson, E. The Ants (Harvard University Press, Cambridge, 1990).
Munyai, T. C. & Foord, S. H. Temporal patterns of ant diversity across a mountain with climatically contrasting aspects in the tropics of Africa. PLoS ONE 10, 1–16 (2015).
Dunn, R. R., Parker, C. R. & Sanders, N. J. Temporal patterns of diversity: Assessing the biotic and abiotic controls on ant assemblages. Biol. J. Linn. Soc. 91, 191–201 (2007).
Urban, M. C., Tewksbury, J. J. & Sheldon, K. S. On a collision course: Competition and dispersal differences create no-analogue communities and cause extinctions during climate change. Proc. R. Soc. B Biol. Sci. 279, 2072–2080 (2012).
Warren, R. J. & Chick, L. Upward ant distribution shift corresponds with minimum, not maximum, temperature tolerance. Glob. Chang. Biol. 19, 2082–2088 (2013).
Suggitt, A. J. et al. Extinction risk from climate change is reduced by microclimatic buffering. Nat. Clim. Change 8, 2 (2018).
Joseph, G. S. et al. Microclimates mitigate against hot temperatures in dryland ecosystems: termite mounds as an example. Ecosphere Article e01509 (2016).
Baudier, K. M., Mudd, A. E., Erickson, S. C. & O’Donnell, S. Microhabitat and body size effects on heat tolerance: Implications for responses to climate change (army ants: Formicidae, Ecitoninae). J. Anim. Ecol. 84, 1322–1330 (2015).
Zellweger, F., Roth, T., Bugmann, H. & Bollmann, K. Beta diversity of plants, birds and butterflies is closely associated with climate and habitat structure. Glob. Ecol. Biogeogr. 26, 898–906 (2017).
Mauda, E. V., Joseph, G. S., Seymour, C. L., Munyai, T. C. & Foord, S. H. Changes in landuse alter ant diversity, assemblage composition and dominant functional groups in African savannas. Biodivers. Conserv. 27, 947–965 (2018).
Andrew, N. R., Miller, C., Hall, G., Hemmings, Z. & Oliver, I. Aridity and land use negatively influence a dominant species’ upper critical thermal limits. PeerJ 2019, 1–20 (2019).
Oliver, I., Dorrough, J., Doherty, H. & Andrew, N. R. Additive and synergistic effects of land cover, land use and climate on insect biodiversity. Landsc. Ecol. 31, 2415–2431 (2016).
Hahn, N. Floristic diversity of the Soutpansberg, Limpopo Province, South Africa (University of Pretoria, Pretoria, 2006).
Davis, C. & Vincent, K. Climate Risk and Vulnerability: A Handbook for Southern Africa. (2017).
Joseph, G. S. et al. Stability of Afromontane ant diversity decreases across an elevation gradient. Glob. Ecol. Conserv. 17, e00596 (2019).
Longino, J. T. & Colwell, R. K. Density compensation, species composition, and richness of ants on a neotropical elevational gradient. Ecosphere 2, 1–20 (2011).
Bishop, T. R., Robertson, M. P., van Rensburg, B. J. & Parr, C. L. Contrasting species and functional beta diversity in montane ant assemblages. J. Biogeogr. 42, 1776–1786 (2015).
Tilman, D. Functional Diversity. In Encyclopedia of Biodiversity Vol. 3 (ed. Levin, S. A.) 109–121 (Academic Press, New York, 2001).
Díaz, S. & Cabido, M. Vive la différence: Plant functional diversity matters to ecosystem processes. Trends Ecol. Evol. 16, 646–655 (2001).
Seymour, C. L., Simmons, R. E., Joseph, G. S. & Slingsby, J. A. On bird functional diversity: Species richness and functional differentiation show contrasting responses to rainfall and vegetation structure in an arid landscape. Ecosystems 18, 971–984 (2015).
Joseph, G. S. et al. Termite mounds mitigate against 50 years of herbivore-induced reduction of functional diversity of savanna woody plants. Landsc. Ecol. 30, 2161–2174 (2015).
Díaz, S., Cabido, M. & Casanoves, F. Plant functional traits and environmental filters at a regional scale. J. Veg. Sci. 9, 113–122 (1998).
Modiba, R. V., Joseph, G. S., Seymour, C. L., Fouché, P. & Foord, S. H. Restoration of riparian systems through clearing of invasive plant species improves functional diversity of Odonate assemblages. Biol. Conserv. 214, 46–54 (2017).
Van Wyk, A. & Smith, G. Regions of Floristic Endemism in Southern Africa: A Review with Emphasis on Succulents (Umdaus press, Umdaus, 2001).
Mostert, T., Bredenkamp, G., Klopper, H. & Al, E. Major vegetation types of the Soutpansberg conservancy and the Blouberg nature reserve, South Africa. Koedoe 50, 32–48 (2008).
Mucina, L. & Rutherford, M. C. The vegetation of South Africa, Lesotho and Swaziland. (2011).
McKillup, S. Statistics Explained: An Introductory Guide for Life Scientists (Cambridge University Press, Cambridge, 2011).
Yates, M., Andrew, N., Binns, M. & Gibb, H. Morphological traits: predictable responses to macrohabitats across a 300 km scale. PeerJ 2, e271 (2014).
Schofield, S. F., Bishop, T. R. & Parr, C. L. Morphological characteristics of ant assemblages (Hymenoptera: Formicidae) differ among contrasting biomes. Myrmecol. News 23, 129–137 (2016).
Colwell, R. K. EstimateS: Statistical estimation of species richness and shared species from samples. (2006).
Baselga, A. Partitioning the turnover and nestedness components of beta diversity. Glob. Ecol. Biogeogr. 19, 134–143 (2010).
Baselga, A. & Orme, C. D. L. Betapart: An R package for the study of beta diversity. Methods Ecol. Evol. 3, 808–812 (2012).
Wang, Y., Naumann, U., Eddelbuettel, D. & Warton, D. mvabund: statistical methods for analysing multivariate abundance data. R package version 3.13.1. (2018).
Warton, D. I., Foster, S. D., Death, G., Stoklosa, J. & Dunstan, P. K. Model-based thinking for community ecology. Plant Ecol. 216, 669–682 (2015).
Warton, D. I., Thibaut, L. & Wang, Y. A. The PIT-trap—A “model-free” bootstrap procedure for inference about regression models with discrete, multivariate responses. PLoS ONE 12, 1–19 (2017).
Hui, F. boral: Bayesian Ordination and Regression AnaLysis. R package version 1.6.1. (2018).
Petchey, O. L. & Gaston, K. J. Functional diversity: Back to basics and looking forward. Ecol. Lett. 9, 741–758 (2006).
Weber, N. The biology of the fungus-growing ants. Part IV. Additional new forms. Rev. Entomol. 9, 154–206 (1938).
Laliberté, E. & Shipley, B. FD: measuring functional diversity from multiple traits, and other tools for functional ecology. R package 1.0–11. (2011).
Petchey, O. L. & Gaston, K. J. Extinction and the loss of functional diversity. Proc. Biol. Sci. 269, 1721–1727 (2002).
Kembel, S. et al. Picante: R tools for integrating phylogenies and ecology. Bioinformatics 26, 1463–1464 (2010).
Gotelli, N. J. & Rohde, K. Co-occurrence of ectoparasites of marine fishes: a null model analysis. Ecol. Lett. 5, 86–94 (2002).
Pinheiro, J., Bates, D., DebRoy, S., Sarkar, D. & R Core Team, . nlme: Linear and Nonlinear Mixed Effects Models. (2016).
Kamil Barton. MuMIn: Multi-Model Inference. R package version 1.43.17. (2020).
Burnham, K. P. & Anderson, D. R. Model Selection and Multimodel Inference: A Practical Information-Theoretic Approach (2nd ed). Ecological Modelling vol. 172 (2002).
Nakagawa, S. & Schielzeth, H. A general and simple method for obtaining R2 from generalized linear mixed-effects models. Methods Ecol. Evol. 4, 133–142 (2013).
Didham, R., Kapos, V. & Ewers, R. Rethinking the conceptual foundations of habitat fragmentation research. Oikos 121, 161–170 (2012).
Niu, K. et al. Fertilization decreases species diversity but increases functional diversity: A three-year experiment in a Tibetan alpine meadow. Agric. Ecosyst. Environ. 182, 106–112 (2014).
Joseph, G. S. et al. Elephants, termites and mound thermoregulation in a progressively warmer world. Landsc. Ecol. 33, 731–742 (2018).
Bishop, T. R. et al. Thermoregulatory traits combine with range shifts to alter the future of montane ant assemblages. Glob. Chang. Biol. 25, 2162–2173 (2019).
Prentice, I. C. et al. Dynamic global vegetation modeling: Quantifying terrestrial ecosystem responses to large-scale environmental change. Terrest. Ecosyst. Chang. World https://doi.org/10.1007/978-3-540-32730-1_15 (2007).
Pfeiffer, M., Kumar, D., Martens, C. & Scheiter, S. Climate change will cause non-analogue vegetation states in Africa and commit vegetation to long-term change. Biogeosci. Discuss. https://doi.org/10.5194/bg-2020-179 (2020).
Potter, K. A., Woods, H. A. & Pincebourde, S. Microclimatic challenges in global change biology. Glob. Chang. Biol. 19, 2932–2939 (2013).
Scheffers, B. R., Edwards, D. P., Diesmos, A., Williams, S. E. & Evans, T. A. Microhabitats reduce animal’s exposure to climate extremes. Glob. Chang. Biol. 20, 495–503 (2014).
Bonachela, J. A. et al. Termite mounds can increase the robustness of dryland ecosystems to climatic change. Science (80-). 347, 651–655 (2015).
Joseph, G. S. et al. Landuse change in savannas disproportionately reduces functional diversity of invertebrate predators at the highest trophic levels: Spiders as an example. Ecosystems 21, 930–942 (2018).
Hoerling, M. & Kumar, A. The perfect ocean for drought. Science (80-). 299, 691–694 (2003).
Diffenbaugh, N. S. & Field, B. S. Changes in ecologically critical terrestrial climate conditions. Science (80-). 341, 486–493 (2013).
Colwell, R. K., Brehm, G., Cardelús, C. L., Gilman, A. C. & Longino, J. T. Global warming, elevational range shifts, and lowland biotic attrition in the wet tropics. Science (80-). 322, 258–261 (2008).
Fowler, H., Forti, L., Brandão, C. & et al. Ecologia nutricional de formigas. in Ecologia Nutricional de Insetos E Suas Implicações No Manejo de Pragas 131–223 (1991).
Davidson, D., Cook, S. & Snelling, R. Liquid-feeding performances of ants (Formicidae): Ecological and evolutionary implications. Oecologia 139, 255–266 (2004).
Sarty, M., Abbott, K. & Lester, P. Habitat complexity facilitates coexistence in a tropical ant community. Oecologia 149, 465–473 (2006).
Kaspari, M. Body size and microclimate use in Neotropical granivorous ants. Oecologia 96, 500–507 (1993).
Weiser, M. & Kaspari, M. Ecological morphospace of New World ants. Ecol. Entomol. 31, 131–142 (2006).
Gibb, H. et al. Does morphology predict trophic position and habitat use of ant species and assemblages?. Oecologia 177, 519–531 (2015).
Gibb, H. & Cl, P. Does structural complexity determine the morphology of assemblages? An experimental test on three continents. PLoS ONE 8, e64005 (2013).
Acknowledgements
We thank the DST-NRF Centre of Excellence for Invasion Biology, through the South African Research Chairs Initiative Chair on Biodiversity Value and Change in the Vhembe Biosphere Reserve, hosted by the University of Venda. We thank Norbert Hahn for creation of the maps used for Electronic Supplementary Material Fig. 1. We thank four reviewers and the handling editor for their useful and constructive comments which improved the manuscript.
Author information
Authors and Affiliations
Contributions
M.M., G.J., S.F., and C.S. conceived ideas, design and G.J. led writing. T.M. and M.M. collected data. C.S., G.J. and M.M. analysed data. All authors contributed to writing of the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Muluvhahothe, M.M., Joseph, G.S., Seymour, C.L. et al. Repeated surveying over 6 years reveals that fine-scale habitat variables are key to tropical mountain ant assemblage composition and functional diversity. Sci Rep 11, 56 (2021). https://doi.org/10.1038/s41598-020-80077-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-80077-8
This article is cited by
-
Anthropogenic landscape change and amphibian diversity in tropical montane biodiversity hotspots: insights from satellite remote sensing in the Madagascar highlands
Environment, Development and Sustainability (2023)
-
Interactive effects of rangeland management and rainfall on dung beetle diversity
Biodiversity and Conservation (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.