Abstract
Motor imagery (MI) is the mental simulation of an action without any overt motor execution. Interestingly, a temporal coupling between durations of real and imagined movements, i.e., the so-called isochrony principle, has been demonstrated in healthy adults. On the contrary, anisochrony has frequently been reported in elderly subjects or those with neurological disease such as Parkinson disease or multiple sclerosis (MS). Here, we tested whether people with MS (PwMS) may have impaired MI when they imagined themselves walking on paths with different widths. When required to mentally simulate a walking movement along a constrained pathway, PwMS tended to overestimate mental movement duration with respect to actual movement duration. Interestingly, in line with previous evidence, cognitive fatigue was found to play a role in the MI of PwMS. These results suggest that investigating the relationship between cognitive fatigue and MI performances could be key to shedding new light on the motor representation of PwMS and providing critical insights into effective and tailored rehabilitative treatments.
Similar content being viewed by others
Introduction
Motor imagery (MI) is an active mental process in which a subject internally simulates a movement without any corresponding motor output. Accumulating evidence indicates functional similarities between actually and mentally executed actions, notably regarding temporal characteristics, neural correlates and autonomic responses1. A plethora of studies shows that actual and mental movements are subject to common motor rules and principles (e.g., Fitts’ law)2,3,4. Indeed, results on healthy adults usually indicate a temporal coupling between the duration of actual and mental movements3, 5,6, the so-called “isochrony principle”2. Congruently, fMRI studies have shown that the neural networks involved in MI overlap considerably with those recruited for actual motor execution7,8,9.
On the contrary, anisochrony, i.e., a temporal discrepancy between actual and mental movements, has been frequently reported in populations of neurological patients, as people after stroke10, with Parkinson disease (PD)11,12 and with multiple sclerosis (MS)13,14,15, gaining increasing attention as a promising additional clinical investigation tool16. Recently, Tacchino et al.17 showed that behavioural performance and brain activations during MI are correlated to disease severity in people with MS (PwMS) and proposed anisochrony as a surrogate behavioural marker of MS evolution, especially in the early stages of the disease.
As for healthy subjects (HS), most investigations on MI of people with neurological diseases have mainly examined upper limb movements with different motor tasks such as sequential pressing of buttons18, finger-to-thumb opposition19, pointing towards targets15,20, hand manipulation17,21, hand grasping and arm-lifting22, and bimanual circle-line coordination23. However, examples of MI involving lower limb are also present24,25,26,27,28,29,30.
Walking is particularly interesting for a broader understanding of MI. In fact, this represents a unique window into the study of MI because subjects are requested to simulate full-body movements and simultaneously update environmental spatial information5. In general, findings indicate that healthy good imagers usually preserve temporal congruence31,32,33 as well as recruiting very similar cerebral networks (e.g. fronto-parietal areas, basal ganglia, brainstem, cerebellum)34,35 when they are asked to actually and mentally execute tasks involving locomotion.
Since impaired locomotion is a frequent and major source of disability in patients with neurological diseases and new techniques are continuously required to improve gait rehabilitation, a better characterization of MI of locomotion in these populations is fundamental. People after stroke seem to retain locomotion MI abilities36 and mental practice appears to be a beneficial intervention for stroke rehabilitation37; nevertheless the existing evidence remains inconclusive because of significantly statistical heterogeneity and methodological flaws38. More contradictory results have been found in patients with PD, mainly due to differences in testing procedures and task instructions27,39,40.
The few studies investigating MI in PwMS are mainly limited to considering upper limb actions13,15,17, showing also how cognitive and mood disorders affect MI28,41,42. Only recently the focus has been shifted to lower limb movements43,44,45. Interestingly, evidence indicates that MI has been shown to improve not only walking, but also balance, fatigue, mood and quality of life in MS43,44,45. However, to date, it remains unclear whether PwMS show anisochrony between actual and mental locomotion as well and, if so, whether this temporal discrepancy is modulated by spatial constraints5,13,15,17 and/or affected by cognitive fatigue.
Investigating the relationship between fatigue and MI performances in PwMS could be key to shedding new light on the motor representation of PwMS and providing insights into new rehabilitative treatments. In fact, fatigue is one of the most debilitating symptoms in MS as it significantly impacts patients’ daily life activities and quality of life46. Given its multifactorial nature, frequently divided into various components such as physical vs. cognitive, fatigue is difficult to define or operationalize, although neurophysiological and fMRI studies have indicated that a cortico-subcortical disconnection could support the central origin of fatigue and explain its complex interaction with other clinical conditions (e.g. mood, cognitive functioning, disability)47. Interestingly, given the significant cognitive effort required to concentrate on MI tasks, loss of attention and declined arousal level due to cognitive fatigue have been found to significantly alter neural signals in the Brain Computer Interface (BCI) system48. As indicated by Deluca et al.49, abnormal cerebral activation in the basal ganglia and frontal lobes (that are also recruited in the MI task) may represent the extra “effort” (i.e., allocation of more neural resources) required to maintain the same level of performance, supporting the notion that increased cerebral activation may reflect the additional effort (i.e., cognitive fatigue) to adequately perform behavioral tasks in PwMS. Therefore, investigation of the complex inter-relationship between cognitive fatigue and MI is very relevant.
Although there is clear and consistent evidence that PwMS may have difficulties in the mental simulation of actions and that several aspects could affect performances, previous results are far from conclusive. Thus, to shed light on this aspect, we asked PwMS to actually and mentally walk along three paths with different widths and we compared their performances to those of HS.
Methods
Participants
Sample size was determined using values from the study by Tacchino et al.17 which suggested a 0.3 point difference [standard deviation (SD) = 0.3] in Index of Performance (IP), a measure of participants’ mental movement ability, of PwMS. Considering 80% power and 5% (two-sided) level of significance, the planned sample size was 15 PwMS and 15 HS. PwMS were recruited among outpatients at the Rehabilitation Service of Genoa of the Italian Multiple Sclerosis Society (AISM). Inclusion criteria for PwMS were clinically defined MS according to McDonald criteria50, a stable phase of the disease without relapses in the previous 3 months, all disease courses, an Expanded Disability Status Scale (EDSS) ≤ 6.5 (i.e. able to walk with at most one aid, e.g. cane or single crutch)51. The HS were age-matched, did not present any neurological or muscular disorders and had normal or corrected-to-normal vision. Moreover, the subjects belonging to the two groups were included if they were right-handed as determined by the Edinburgh Handedness Inventory52 and showed preserved cognitive functioning as measured by a Symbol Digit Modalities Test53 (SDMT) score > 38. Subjects with a history of severe psychiatric disorders as indicated by Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5) criteria54, blurred vision or cardiovascular and respiratory disorders were excluded. Participants from both groups were assessed with the following tests: the Timed 25-Foot Walk (T25-FW)55 to measure performances in walking speed, the short form of the International Physical Activity Questionnaire (IPAQ)56,57 to gather information about the usual time spent in walking, in vigorous and moderate intensity activity, and in sedentary behaviours, the Dual-tasking questionnaire (DTQ)59 which collects information about everyday difficulties with dual-tasking and the Kinesthetic and Visual Imagery Questionnaire (KVIQ)58 for MI vividness. PwMS were also assessed with the Modified Fatigue Impact Scale (MFIS)60 to gather information about fatigue perception.
All subjects gave written informed consent in accordance with the revised Declaration of Helsinki61. The study was approved by the Ethics Committee of San Martino Hospital, Genoa, Italy. All methods were carried out in accordance with relevant guidelines and regulations.
Procedure
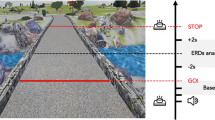
The experimental setup was an adapted version from Personnier et al.5. Participants stood upright behind the starting line; their arms were hanging along the body, and their feet were parallel and slightly apart. They were asked to actually or mentally walk through three 5-m long paths, each with a different width: 20 cm, 35 cm and 50 cm. The different spatial constraints made it possible to test walking under several conditions, from normal to very accurate according to the path’s width. During actual movements, participants were invited to perform the task without walking on the lines limiting the paths at the self-paced speed. During mental movements, participants had to imagine themselves walking at a self-paced speed in the first-person perspective (i.e., as if one were the actor of the action). This has been considered to be closer to the real execution of movement14 since individuals elicit kinaesthetic sensation representations of actions as if they were actually performing them (see Fig. 1 for a representation of the experimental setup).
Experimental setup. Both groups of participants were asked to actually (a) or mentally (b) walk through paths of 5 m of length and delimited at three different widths: 20 cm, 35 cm, 50 cm. Icons were made by Freepik from www.flaticon.com.
The duration of actual and mental movements was recorded by means of an electronic stopwatch (temporal resolution: 10 ms). When the experimenter gave the “Go!” signal, temporal acquisition started, and participants had to begin walking or imagining they were walking. During the actual movements, the temporal acquisition was stopped when the participants’ shoulders clearly crossed the finish line; during mental trials participants were required to verbally indicate “Stop!” when they mentally crossed the finish line. The time elapsed between the “Go!” signal and crossing the line corresponded to the movement duration.
Participants performed 12 walking movements for each path width (20 cm, 35 cm and 50 cm) in each experimental condition (actual and mental); for a total of 72 movements distributed over two different sessions separated by a time interval of 48 h. Within each session, actual and mental trials were presented to the participants in two different blocks. In the first session, the mental movements were performed before the actual ones, while in the second session the actual movements were performed before the mental ones. The three paths were randomly presented to the participants within each block. The two experimental conditions were separated by a 5-min break. In order to familiarize with the task, all participants performed one actual and one mental movement within each path. No information concerning their temporal performance was given to the participants during the practice or the experimental trials.
Statistical analysis
For each actually (A) and mentally (M) executed path, the mean movement duration of locomotion was calculated. In order to better examine participants’ mental movement ability, we computed an Index of Performance (IP) expressed in percentage:
The IP reflects whether participants overestimate (negative values) or underestimate (positive values) movement durations during MI with respect to actual performances, where an IP approaching zero would represent a good motor imagery performance. However, although it is considered a reliable and objective measure of MI accuracy and an indicator of motor system integrity7,62, this index could hinder differences between imagined and actual movement durations when, for instance, some subjects underestimate and others overestimate movement durations during MI. As suggested by5, in order to provide a clearer and more complete analysis of MI performance, we also calculated the absolute values of IP.
A Shapiro–Wilks test revealed that all variables were normally distributed (always p > 0.05) and, therefore, parametric tests were used. Independent t tests were performed on SDMT, T25FW, IPAQ, DTQ and KVIQ total scores to assess potential differences between groups in cognitive, locomotion, usual physical abilities, dual-task and MI vividness performance. A two-way analysis of variance (ANOVA), with group as a between-subjects factor (PwMS; HS) and path width as a within-subjects factor (20 cm; 35 cm; 50 cm) was performed on IP values. Furthermore, an ANOVA with group as a between-subjects factor (PwMS; HS), path width (20 cm; 35 cm; 50 cm) and type of movement (actual; mental) as within-subjects factors was performed on actual and mental movement durations. Post-hoc tests (Bonferroni’s correction; p < 0.05) were applied to explore significant effects and interactions. To test any potential association between anisochrony and fatigue perception among PwMS, a correlation analysis was performed between the absolute values of IP (IPabs)5 across different path widths and MFIS (total score and subscale scores). Statistical analyses were run using SPSS 23.
Results
In order to evaluate MI in PwMS, we asked fifteen PwMS (14 with relapsing–remitting course; 1 with secondary progressive course; 4 males; mean EDSS: 2.97 ± 1.86; mean MFIS: 38.53 ± 17.57) and fifteen HS (7 males) to take part in this study. The two groups were age matched (PwMS: 42.67 ± 11.67 years; HS: 41.60 ± 13.35 years; t28 = 0.23; p = 0.82). No significant differences were found between groups in walking speed as indicated by the T25-FW (PwMS: 7.14 ± 1.93 s; HS: 6.23 ± 1.03 s; t28 = 1.59; p = 0.122), in usual physical activity by IPAQ (walking in MET-min/week: PwMS: 1178.10 ± 519.19; HS: 1214.40 ± 208.78; t28 = -0.25; p = 0.803; moderate in MET-min/week: PwMS: 1420 ± 949.13; HS: 1536 ± 586.48; t28 = − 0.40; p = 0.690; vigorous in MET-min/week: PwMS: 1760 ± 1146.22; HS: 1872 ± 422.56; t28 = − 0.35; p = 0.725; total in MET-min/week: PwMS: 4358.10 ± 2366.04; HS: 4622.40 ± 820.83; t28 = − 0.41; p = 0.686; sedentary in h/day: PwMS: 4.4 ± 2.2; HS: 3.4 ± 1.40; t28 = 1.48; p = 0.149), in dual-task abilities as evaluated by DTQ (PwMS: 0.16 ± 0.07; HS 0.13 ± 0.07; t28 = 1.00; p = 0.326), in information processing speed measured by SDMT (PwMS: 48.27 ± 19.34; HS: 51.00 ± 11.56; t28 = − 0.47; p = 0.64) and in MI vividness as tested by KVIQ (PwMS: 126.20 ± 33.28; HS: 137.53 ± 21.47; t28 = − 1.11; p = 0.28). See Table 1 for a summary.
Overestimation of mental movement duration in PwMS
The results of an ANOVA on IP showed a main effect of group [F (1,28) = 12.79, p = 0.001, η2 = 0.314]. IP approached zero at all path widths in HS (− 3.5% ± 4.7%), while PwMS tended to mentally overestimate the duration of the required action (− 27.3% ± 4.7%), reflecting the presence of anisochrony (i.e. mentally executed locomotion lasted longer than actually executed locomotion). No significant differences in the path width factor (p = 0.456, η2 = 0.028) or in group*path width interaction (p = 0.103, η2 = 0.078) were found. However, we observed a trend towards anisochrony in PwMS with the decreasing of the path width (PwMS M ± SE: 20 cm: − 29.6% ± 4.6%, 35 cm: − 27.9% ± 5.1%, 50 cm: − 24.3% ± 5.0%; HS M ± SE: 20 cm: − 1.9% ± 4.6%, 35 cm: − 4.5% ± 5.1%, 50 cm: − 4.0% ± 5.0%), suggesting a progressive increment of both actual and mental durations along with the increase in spatial precision.
Influence of path spatial constraints on both actual and mental movement duration in PwMS
Although IP offered a single and informative measure to examine participants’ mental movement ability, it did not clarify whether and to what extent subjects tended to overestimate or underestimate mental movements across paths with different spatial widths. The ANOVA on actual and mental movement duration further qualified the results found in the IP statistical analysis. A significant group*path width*type of movement interaction was found [F (2,56) = 6.801, p = 0.040, η2 = 0.108] (Fig. 2): post-hoc analysis showed that actual locomotion across all path widths in PwMS was significantly slower compared to that in HS (PwMS M ± SE: 20 cm: 5.68 ± 0.37 s, 35 cm: 5.37 ± 0.32 s, 50 cm: 5.15 ± 0.25 s; HS M ± SE: 20 cm: 4.26 ± 0.37 s, 35 cm: 4.23 ± 0.32 s, 50 cm: 4.23 ± 0.25 s) (always p < 0.05). The same result was found for mental locomotion (PwMS M ± SE: 20 cm: 7.31 ± 0.50 s, 35 cm: 6.81 ± 0.49 s, 50 cm: 6.41 ± 0.41 s; HS M ± SE: 20 cm: 4.36 ± 0.50 s, 35 cm: 4.45 ± 0.49 s, 50 cm: 4.25 ± 0.41 s) (always p < 0.005). Moreover, mental locomotion of PwMS was significantly slower with respect to actual locomotion at all path widths (always p < 0.001). We did not observe the same result in HS (always p > 0.05). In addition, while mental locomotion in PwMS was significantly different across all path widths (always p < 0.005), considering actually executed locomotion, significant differences were only found between the narrower (20 cm) and the wider paths (35 cm and 50 cm) (p < 0.005 in both cases) (see Fig. 2 for a graphical representation of major results).
Graphical representation of the group*path width*type of movement interaction. Asterisks indicate significant differences (p < 0.05). Mental locomotion was significantly different across all path widths (ps < 0.05) in PwMS, but not in HS. Considering actually executed locomotion, significant differences were only found between the narrower (20 cm) and the wider paths (35 cm and 50 cm) in PwMS. Vertical lines represent standard error.
Role of cognitive fatigue on PwMS’ performances
In PwMS, the correlation between IPabs and the total score of MFIS was significant at all path widths (20 cm: r = 0.719, p = 0.002; 35 cm: r = 0.362, p = 0.011; 50 cm: r = 0.707, p = 0.003): the higher the reported fatigue perception, the higher the anisochrony. More interestingly, IPabs significantly correlated with the MFIS cognitive subscale at all path widths (20 cm: r = 0.752, p = 0.001; 35 cm: r = 0.584, p = 0.022; 50 cm: r = 0.673, p = 0.006), suggesting a role of cognitive fatigue on PwMS’ performances (see Fig. 3). No correlations with physical (20 cm: r = 0.439, p = 0.102; 35 cm: r = 0.479, p = 0.071; 50 cm: r = 0.512, p = 0.051) or psychosocial (20 cm: r = 0.472, p = 0.075; 35 cm: r = 0.354, p = 0.195; 50 cm: r = 0.408, p = 0.131) subscales were found.
Discussion
MI is a dynamic state during which the representation of an action is internally reproduced within working memory without any overt output2. The aim of the present study was to test MI in people with Multiple Sclerosis (PwMS). Although there is clear and consistent evidence that PwMS may have difficulties in the mental simulation of actions, previous results are far from being conclusive. Thus, a critical advance of these studies was necessary. Here, we investigate if PwMS show anisochrony in a task involving lower limbs and if the dependency on spatial constraints was present. In addition, due to the major role of fatigue in the life of PwMS, the relationship between cognitive fatigue and MI performances of patients was investigated. To this aim, fifteen PwMS with mild disability and 15 age-matched healthy subjects (HS) were required to either actually walk or imagine themselves walking on three paths of the same length, but different widths. When asked to mentally simulate a walking movement along a constrained pathway, PwMS tended to overestimate mental movement duration with respect to actual movement duration, thus confirming the presence of anisochrony. Moreover, the investigation of movement durations revealed that a progressive increment of both actual and mental durations occurred with the augmentation of spatial precision; this increment was more consistent for the mental task at the narrowest paths. Interestingly, in line with previous evidence, cognitive fatigue was found to play a role in the MI of PwMS.
The analysis of actual movement durations across different path widths showed that PwMS were slower than HS. Although no statistical differences were found between groups in T25-FW, this result was expected because MS typically influences postural control and body equilibrium as of the first stages of the disease; thus, ambulation was more challenging for PwMS with respect to HS, especially when walking on a path with spatial constraints was required. This effect is also present in mental locomotion; in fact, the mental simulation of movements reflects the updating of motor representation in line with disease evolution. Thus, the slower the actual movements, the slower mental simulation. Interestingly, PwMS and HS reported to spent almost the same time in walking or in other physical activities, as indicated by IPAQ scores. This further corroborated our results suggesting that PwMS could not have less experience of walking to refer when performing the MI tasks and, thus, anisochrony could not be attributed to differences in physical practice or, conversely, sedentary behaviours.
Furthermore, the temporal requirement of the task (i.e. at a self-paced speed) may explain why here we found that the mental actions were slower than those actually executed in the MS group, similarly to the study using the task of squeezing a ball17 and differently from that requiring pointing movements15. In the latter, PwMS imagined faster than actual performances, probably as a consequence of two concurrent factors. One is that the difficulty of the task may have led to slowness in the actual movement. The other is they could adopt a simulation strategy so to execute the task as accurately and fast as possible, as they were asked to. This would have prevented them from full integrating the spatial constraints (i.e., path width) during their mental processes and may have resulted in a sped up mental representation63. In the present study, the lack of temporal constraints may have induced PwMS to dedicate a longer time to accurately simulating the full-body movements and simultaneously updating the environmental spatial information5. Moreover, the cognitive demand of the task could be further burdened by the need to verbally indicate when the shoulders mentally crossed the finish line in order to stop the temporal acquisition. Although it is a simple request, for PwMS it could represent a sort of dual-task condition64 with consequent slower mental performances65. It seems that imagining themselves walking while considering path constraints (additional cognitive task), that require dividing attentional capacities is more difficult than a single task66. However, as indicated by DTQ score, PwMS in our study did not show any difficulties in managing dual-task activities. Thus, anisochrony in MS group could not be explained in terms of DT cost. Also, a factor that may contribute to MI performance is the presence of cognitive impairment67, one of the most disturbing and debilitating disorder in MS, present in 43–70% of adults with MS and documented in all MS subtypes68,69. Information processing speed, working memory, attention and executive functions are the major cognitive domains affected70,71. Therefore, the representation of a mentally reproduced action requires a preserved cognitive functioning14, particularly in terms of speed information processing, which involves the ability to both maintain and manipulate information for a certain period. However, in our study, PwMS performed similarly to HS in SDMT, known to be the best predictor of MS cognitive impairment53. However, to date, a neuropsychological assessment covering attention, memory, visuo-spatial and executive functions may be adequate to both detect subtle cognitive deficits and to disentangle their possible role in MI.
Beside spatial constraints, fatigue also seems to influence mental performances as demonstrated by a strong correlation between fatigue perception as measured by MFIS and MI ability. Fatigue is likely to affect somato-sensory perception and involve perturbation of the body schema72,73, strongly enhancing perception of effort and limiting the endurance of sustained physical and mental activities74. MS related fatigue had such a negative impact on the quality of life, daily activities, psychological well-being and relationships with friends. Furthermore, although cognitive fatigue could be related to other different cognitive states as drowsiness, loss of attention, decreased arousal, lower focus level75, our results are in line with other studies that confirmed a significant relationship between cognitive fatigue and MI performance48,76,77. A fatigued body after either continuous or intermittent exercises may affect MI ability, since it reproduces both forward and inverse prediction models, that are crucial to generate temporally accurate and vivid motor representations. In line with77, anisochrony in walking could thus originate from the central integration of proprioceptive afferents under a fatigued state that may impact, in turn, motor predictions of movements involving lower limbs.
At neural level, in MS related fatigue an abnormal recruitment of the primary motor cortex (PMC) and supplementary motor cortex (SMC), two areas involved in movement control, has been recently shown. In addition, microstructural damage in several fronto-connected associative white matter (WM) tracts (i.e., corona radiata, cingulum, anterior thalamic radiation, corpus callosum, forceps minor, superior longitudinal fasciculus, inferior fronto-occipital fasciculus, thalamus, cerebral peduncle and basal ganglia)47 has also been highlighted, thus supporting the hypothesis that fatigue is subtended by both a motor and non-motor network dysfunction46. Furthermore, the anterior cingulate cortex (ACC), which is extensively connected with other cortical areas and associated to alertness and attention, has been found to be altered in PwMS. Thus, its dysfunction in MS might represent the substrate of cognitive fatigue and/or the cognitive component of MS-related counterparts of fatigue46,78. Several studies have interpreted the increased cerebral activity as a rearrangement, a “compensation” occurring in PwMS in an attempt to make up for the neural dysfunction and, thus, responsible for the development of fatigue symptoms46. As suggested by49, it is possible that while compensation may indeed be occurring, that factor itself might lead to cognitive fatigue. Equally, cognitive fatigue might require the recruitment of additional brain areas to compensate for the extra ‘effort’ required for a continued performance. Although this study lacked neuroimaging data of actual and mental tasks, preventing us from drawing conclusions about the neural mechanism between compensation and cognitive fatigue, we can speculate a pivotal role of cognitive fatigue that might influence the way in which PwMS experienced the mental task, probably affecting its internal representation.
Moreover, many findings have frequently pointed out the similarities between the profile of deficits in MS and that observed in healthy elderly people79,80,81. In the study by Personnier et al.5, in which the temporal features of mental locomotion in normal aging were investigated, the authors found that elderly people increased anisochrony when the spatial precision required to perform the task increased. Our study is in line with findings from Personnier et al.5 confirming a tendency to anisochrony in PwMS, further corroborated by the investigation of movement durations. We found that a progressive increment of both actual and mental durations occurred with the augmentation of spatial precision; this increment was more consistent for the mental task at the narrowest paths.
Although current results do not have direct therapeutic implications, they provide some critical hints for MI use in the clinical and rehabilitation context. In fact, to date, a plethora of studies demonstrated that mental practice through MI leads to motor improvements by constituting a potential tool for motor learning and relearning, and for rehabilitation, especially for people with physical disabilities14,82. Specifically for PwMS, it seems that spatial constraints and cognitive fatigue need to be correctly weighted according to the individual rehabilitative intervention. Since walking is typically one of the first functions to be affected by MS with a high impact on the quality of life of the patients, a better understanding of how PwMS are able to imagine walking could contribute to improving understanding of their action representation and eventually proposing new rehabilitative tools based on the mental practice of balance and locomotion14. Improvements in motor task execution (e.g., increased gait speed) following MI training are believed to be due to the development and refining of the internal representation of the motor task via activation of the movement-related neural network83. Furthermore, mental practice with MI offers a unique and attractive opportunity to improve locomotor skills through a safe and self-paced training for people with severe disability that, due to their walking difficulties, can be done only in short bursts, especially in the early phase of rehabilitation. Given the potential benefits of MI in neurological rehabilitation, further studies are needed to explore its contribution to the extent of motor gains in a population with physical disabilities.
References
Munzert, J., Lorey, B. & Zentgraf, K. Cognitive motor processes: The role of motor imagery in the study of motor representations. Brain Res. Rev. 60, 306–320 (2009).
Decety, J. & Jeannerod, M. Mentally simulated movements in virtual reality: Does Fitts ’ s law hold in motor imagery ?. Behav. Brain Res. 72, 127–134 (1996).
Decety, J., Jeannerod, M. & Prablanc, C. The timing of mentally represented actions. Behav. Brain Res. 34, 35–42 (1989).
Parsons, L. M. Temporal and kinematic properties of motor behavior reflected in mentally simulated action. J. Exp. Psychol. Hum. Percept. Perform. 20, 709–730 (1994).
Personnier, P., Kubicki, A., Laroche, D. & Papaxanthis, C. Temporal features of imagined locomotion in normal aging. Neurosci. Lett. 476, 146–149 (2010).
Papaxanthis, C. & Schieppati, M. Imagined and actual arm movements have similar durations when performed under different conditions of direction and mass. Exp. Brain Res. 143, 447–452 (2002).
Saiote, C. et al. Resting-state functional connectivity and motor imagery brain activation. Hum. Brain Mapp. 37, 3847–3857 (2016).
Lotze, M. & Halsband, U. Motor imagery. J. Physiol. Paris 99, 386–395 (2006).
Bonzano, L., Roccatagliata, L., Ruggeri, P., Papaxanthis, C. & Bove, M. Frontoparietal cortex and cerebellum contribution to the update of actual and mental motor performance during the day. Sci. Rep. 6, 1–11 (2016).
Dettmers, C., Benz, M., Liepert, J. & Rockstroh, B. Motor imagery in stroke patients, or plegic patients with spinal cord or peripheral diseases. Acta Neurol. Scand. 126, 238–247 (2012).
Abbruzzese, G., Avanzino, L., Marchese, R. & Pelosin, E. Action observation and motor imagery: Innovative cognitive tools in the rehabilitation of Parkinson’s disease. Park. Dis. 2015, 1–7 (2015).
Dominey, P., Decety, J., Broussolle, E., Chazot, G. & Jeannerod, M. Motor imagery of a lateralized sequential task is asymmetrically slowed in hemi-Parkinson’s patients. Neuropsychologia 33, 727–741 (1995).
Heremans, E. et al. Cued motor imagery in patients with multiple sclerosis. Neuroscience 206, 115–121 (2012).
Malouin, F. & Richards, C. L. Mental practice for relearning locomotor skills. Phys. Ther. 90, 240–251 (2010).
Tacchino, A. et al. Imagined actions in multiple sclerosis patients: Evidence of decline in motor cognitive prediction. Exp. Brain Res. 229, 561–570 (2013).
Rienzo, F. D., Collet, C. & Hoyek, N. Impact of neurologic deficits on motor imagery: A systematic review of clinical evaluations. Neuropsychol. Rev. 24, 116–147 (2014).
Tacchino, A. et al. Motor imagery as a function of disease severity in multiple sclerosis: An fMRI study. Front. Human Neurosci. 11, 1–10 (2018).
Cunnington, R., Iansek, R., Johnson, K. A. & Bradshaw, J. L. Movement-related potentials in Parkinson ’ s disease Motor imagery and movement preparation. Brain 120, 1339–1353 (1997).
Heremans, E. et al. Motor imagery ability in patients with early-and mid-stage Parkinson disease. Neurorehabil. Neural Repair 25, 168–177 (2011).
Sirigu, A. et al. The mental representation of hand movements after parietal cortex damage. Am. Assoc. Adv. Sci. 273, 1564–1568 (1996).
Ruby, P. & Decety, J. Effect of subjective perspective taking during simulation of action: A PET investigation of agency. Nat. Neurosci. 4, 546–550 (2001).
Kobelt, M., Wirth, B. & Schuster-Amft, C. Muscle activation during grasping with and without motor imagery in healthy volunteers and patients after stroke or with Parkinson’s disease. Front. Psychol. 9, 597 (2018).
Morioka, S. et al. Motor-imagery ability and function of hemiplegic upper limb in stroke patients. Ann. Clin. Transl. Neurol. 6, 596–604 (2019).
Alkadhi, H. et al. What disconnection tells about motor imagery: Evidence from paraplegic patients. Cereb. Cortex 15, 131–140 (2004).
Bakker, M. et al. Motor imagery of foot dorsiflexion and gait: Effects on corticospinal excitability. Clin. Neurophysiol. 119, 2519–2527 (2008).
Cramer, S. C., Orr, E. L. R., Cohen, M. J. & Lacourse, M. G. Effects of motor imagery training after chronic, complete spinal cord injury. Exp. Brain Res. 177, 233–242 (2007).
Helmich, R. C., Bloem, B. R. & Toni, I. Motor imagery evokes increased somatosensory activity in Parkinson’ s disease patients with tremor. Hum. Brain Mapp. 33, 1763–1779 (2012).
Heremans, E., D’Hooge, A., De Bondt, S., Helsen, W. & Feys, P. The relation between cognitive and motor dysfunction and motor imagery ability in patients with multiple sclerosis. Mult. Scler. J. 18, 1303–1309 (2012).
Kim, J.-H., Cho, Y.-S., Park, J.-S. & Kim, W.-J. Effect of motor imagery training and electromyogram-triggered neuromuscular electrical stimulation on lower extremity function in stroke patients: A pilot trial. J. Phys. Ther. Sci. 29, 1931–1933 (2017).
Marusic, U. et al. Motor imagery during action observation of locomotor tasks improves rehabilitation outcome in older adults after total hip arthroplasty. Neural Plast. 2018, 20 (2018).
Van Der Meulen, M., Allali, G., Rieger, S. W., Assal, F. & Vuilleumier, P. The influence of individual motor imagery ability on cerebral recruitment during gait imagery. Hum. Brain Mapp. 35, 455–470 (2014).
Personnier, P., Ballay, Y. & Papaxanthis, C. Mentally represented motor actions in normal aging: III. Electromyographic features of imagined arm movements. Behav. Brain Res. 206, 184–191 (2010).
Saimpont, A., Malouin, F., Tousignant, B. & Jackson, P. L. The influence of body configuration on motor imagery of walking in younger and older adults. Neuroscience 222, 49–57 (2012).
La Fougere, C. et al. Real versus imagined locomotion: A [18F]-FDG PET-fMRI comparison. Neuroimage 50, 1589–1598 (2010).
Miyai, I. et al. Cortical mapping of gait in humans: A near-infrared spectroscopic topography study. Neuroimage 14, 1186–1192 (2001).
Malouin, F. et al. Motor imagery for optimizing the reacquisition of locomotor skills after cerebral damage. Neurophysiol. Found. Ment. Mot. Imag. 20, 161–176 (2010).
Braun, S. M., Beurskens, A. J., Borm, P. J., Schack, T. & Wade, D. T. The effects of mental practice in stroke rehabilitation: A systematic review. Arch. Phys. Med. Rehabil. 87, 842–852 (2006).
Li, R.-Q., Li, Z.-M., Tan, J.-Y., Chen, G.-L. & Lin, W.-Y. Effects of motor imagery on walking function and balance in patients after stroke: A quantitative synthesis of randomized controlled trials. Complement. Ther. Clin. Pract. 28, 75–84 (2017).
Snijders, A. H. et al. Gait-related cerebral alterations in patients with Parkinson’s disease with freezing of gait. Brain 134, 59–72 (2010).
Cohen, R. G., Chao, A., Nutt, J. G. & Horak, F. B. Freezing of gait is associated with a mismatch between motor imagery and motor execution in narrow doorways, not with failure to judge doorway passability. Neuropsychologia 49, 3981–3988 (2011).
Azin, M., Zangiabadi, N., Tabrizi, Y. M., Iranmanesh, F. & Baneshi, M. R. Deficiency in mental rotation of upper and lower-limbs in patients with multiple sclerosis and its relation with cognitive functions. Acta Med. Iran. 54, 510–517 (2016).
Tabrizi, Y. M., Mazhari, S., Nazari, M. A., Zangiabadi, N. & Sheibani, V. Abnormalities of motor imagery and relationship with depressive symptoms in mildly disabling relapsing-remitting multiple sclerosis. J. Neurol. Phys. Ther. 38, 111–118 (2014).
Seebacher, B., Kuisma, R., Glynn, A. & Berger, T. Effects and mechanisms of differently cued and non-cued motor imagery in people with multiple sclerosis: A randomised controlled trial. Mult. Scler. J. 20, 1352458518795332 (2018).
Seebacher, B., Kuisma, R., Glynn, A. & Berger, T. Exploring cued and non-cued motor imagery interventions in people with multiple sclerosis: A randomised feasibility trial and reliability study. Arch. Physiother. 20, 1–19 (2018).
Kahraman, T., Savci, S., Ozdogar, A. T., Gedik, Z. & Idiman, E. Physical, cognitive and psychosocial effects of telerehabilitation-based motor imagery training in people with multiple sclerosis: A randomized controlled pilot trial. J. Telemed. Telecare 20, 1357633X18822355 (2019).
Bisecco, A. et al. Fatigue in multiple sclerosis: The contribution of resting-state functional connectivity reorganization. Mult. Scler. J. 24, 1696–1705 (2018).
Bisecco, A. et al. Fatigue in multiple sclerosis: The contribution of occult white matter damage. Mult. Scler. 22, 1676–1684 (2016).
Talukdar, U., Hazarika, S. M. & Gan, J. Q. Motor imagery and mental fatigue: Inter-relationship and EEG based estimation. J. Comput. Neurosci. 46, 55–76 (2019).
DeLuca, J., Genova, H. M., Hillary, F. G. & Wylie, G. Neural correlates of cognitive fatigue in multiple sclerosis using functional MRI. J. Neurol. Sci. 270, 28–39 (2008).
Thompson, A. J. et al. Diagnosis of multiple sclerosis: 2017 revisions of the McDonald criteria. Lancet Neurol. 17, 162–173 (2018).
Kurtzke, J. F. Rating neurologic impairment in multiple sclerosis: An expanded disability status scale (EDSS). Neurology 33, 1444 (1983).
Oldfield, R. C. The Assessment and Analysis of Handedness: The Edinburgh InventoryVol 9 97–113 (Pergamon Press, Oxford, 1971).
Langdon, D. W. et al. Recommendations for a Brief International Cognitive Assessment for Multiple Sclerosis (BICAMS). Mult. Scler. J. 18, 891–898 (2012).
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders 5th edn. (American Psychiatric Association Press, New York, 2013).
Motl, R. W. et al. Validity of the timed 25-foot walk as an ambulatory performance outcome measure for multiple sclerosis. Mult. Scler. J. 23, 704–710 (2017).
Craig, C. L. et al. International physical activity questionnaire: 12-country reliability and validity. Med. Sci. Sports Exerc. 35, 1381–1395 (2003).
Mannocci, A. et al. International Physical Activity Questionnaire: Validation and assessment in an Italian sample. Ital. J. Public Health 7, 369–376 (2010).
Malouin, F. et al. The Kinesthetic and Visual Imagery Questionnaire (KVIQ) for assessing motor imagery in persons with physical disabilities: A reliability and construct validity study. J. Neurol. Phys. Ther. 31, 20–29 (2007).
Evans, J. J., Greenfield, E., Wilson, B. A. & Bateman, A. Walking and talking therapy: Improving cognitive-motor dual-tasking in neurological illness. J. Int. Neuropsychol. Soc. 15, 112–120 (2009).
Kos, D. et al. Evaluation of the modified fatigue impact scale in four different European countries. Mult. Scler. J. 11, 76–80 (2005).
World Medical Association General Assembly. Declaration of Helsinki. Ethical principles for medical research involving human subjects. World Med. J. 54, 20 (2008).
Rienzo, F. D., Collet, C. & Hoyek, N. Impact of neurologic deficits on motor imagery: A systematic review of clinical evaluations. Neuropsychol. Rev. https://doi.org/10.1007/s11065-014-9257-6 (2014).
Guillot, A. & Collet, C. Duration of mentally simulated movement: A review. J. Mot. Behav. 37, 10–20 (2005).
Leone, C., Patti, F. & Feys, P. Measuring the cost of cognitive-motor dual tasking during walking in multiple sclerosis. Mult. Scler. J. 21, 123–131 (2015).
Veldkamp, R. et al. Structured cognitive-motor dual task training compared to single mobility training in persons with multiple sclerosis, a multicenter RCT. J. Clin. Med. 8, 2177 (2019).
Muci, B., Keser, I., Meric, A. & Karatas, G. K. What are the factors affecting dual-task gait performance in people after stroke?. Physiother. Theory Pract. 00, 1–8 (2020).
Butchard-Macdonald, E., Paul, L. & Evans, J. J. Balancing the demands of two tasks: An investigation of cognitive-motor dual-tasking in relapsing remitting multiple sclerosis. J. Int. Neuropsychol. Soc. 24, 247–258 (2018).
Rao, S. M., Leo, G. J., Bernardin, L. & Unverzagt, F. Cognitive dysfunction in multiple sclerosis: I. Frequency, patterns, and prediction. Neurology 41, 685–691 (1991).
Amato, M. P., Prestipino, E. & Bellinvia, A. Identifying risk factors for cognitive issues in multiple sclerosis. Expert Rev. Neurother. https://doi.org/10.1080/14737175.2019.1590199 (2019).
Chiaravalloti, N. D. & Deluca, J. Cognitive impairment in multiple sclerosis. Lancet Neurol. 7, 1139–1151 (2008).
Grzegorski, T. & Losy, J. Cognitive impairment in multiple sclerosis—a review of current knowledge and recent research. Rev. Neurosci. 28, 845–860 (2017).
Kanekar, N., Santos, M. J. & Aruin, A. S. Anticipatory postural control following fatigue of postural and focal muscles. Clin. Neurophysiol. 119, 2304–2313 (2008).
Paillard, T. Effects of general and local fatigue on postural control: A review. Neurosci. Biobehav. Rev. 36, 162–176 (2012).
Chaudhuri, A. & Behan, P. O. Fatigue in neurological disorders. Lancet (Lond., Engl.) 363, 978–988 (2004).
Cao, T., Wan, F., Wong, C. M., da Cruz, J. N. & Hu, Y. Objective evaluation of fatigue by EEG spectral analysis in steady-state visual evoked potential-based brain-computer interfaces. Biomed. Eng. Online 13, 1–13 (2014).
Lorey, B. et al. The embodied nature of motor imagery: The influence of posture and perspective. Exp. Brain Res. 194, 233–243 (2009).
Kanthack, T. F. D., Guillot, A., Clémençon, M., Debarnot, U. & Di Rienzo, F. Effect of physical fatigue elicited by continuous and intermittent exercise on motor imagery ability. Res. Q. Exerc. Sport 20, 1–14 (2020).
Rocca, M. A. et al. Default-mode network dysfunction and cognitive impairment in progressive MS. Neurology 74, 1252–1259 (2010).
Deluca, J., Chelune, G. J., Tulsky, D. S. & Lengenfelder, J. Is speed of processing or working memory the primary information processing deficit in multiple sclerosis?. J. Clin. Exp. Neuropsychol. 26, 550–562 (2004).
Lajoie, Y., Teasdale, N., Bard, C. & Fleury, M. Attentional demands for walking: Age-related changes. Adv. Psychol. https://doi.org/10.1016/S0166-4115(96)80011-2 (1996).
Reicker, L. I., Tombaugh, T. N., Walker, L. & Freedman, M. S. Reaction time: An alternative method for assessing the effects of multiple sclerosis on information processing speed. Arch. Clin. Neuropsychol. 22, 655–664 (2007).
Saimpont, A., Malouin, F. & Jackson, P. L. Motor imagery and aging. J. Mot. Behav. 45, 21–28 (2013).
Nicholson, V., Watts, N., Chani, Y. & Keogh, J. W. Motor imagery training improves balance and mobility outcomes in older adults: A systematic review. J. Physiother. 65, 200–207 (2019).
Acknowledgements
We would like to thank all the people with MS followed as outpatients at the Rehabilitation Service of Genoa of the Italian Multiple Sclerosis Society (AISM) for their participation to this research. A special thanks to Maria Madera and Giulia Bignone for their help in patient’s recruitment.
Funding
This study received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
A.T., M.B., G.B. and J.P. developed the study concept; data collection and clinical assessment was performed by L.P. and E.P.; M.M.B. enrolled patients; J.P. planned and performed the data analysis; J.P. and A.T. drafted the manuscript. All authors provided critical revisions and approved the final version of the manuscript for submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Podda, J., Pedullà, L., Monti Bragadin, M. et al. Spatial constraints and cognitive fatigue affect motor imagery of walking in people with multiple sclerosis. Sci Rep 10, 21938 (2020). https://doi.org/10.1038/s41598-020-79095-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-79095-3
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.