Abstract
Metabolic syndrome (MetS) has increasingly gained importance as the main risk factor for cardiovascular diseases and type II diabetes mellitus. Various natural compounds derived from plants are associated with beneficial effects on the incidence and progression of MetS. This study aimed to evaluate the effects of Capsicum annuum on factors related to MetS by assessing randomized controlled trials (written in English). We searched the online databases of PubMed, Embase, Google scholar, and Cochrane Library up to April 2020. ‘Patient/Population, Intervention, Comparison and Outcomes’ format was used to determine whether intervention with C. annuum supplementation compared with placebo supplementation had any effect on the components of MetS among participants. We considered standardized mean differences (SMD) with 95% confidence intervals (CI) as effect size measures using random-effects model. Analysis of the included 11 studies (n = 609) showed that C. annuum supplementation had significant effect on low density lipoprotein-cholesterol [SMD = − 0.39; 95% CI − 0.72, − 0.07; P = 0.02; prediction interval, − 1.28 to 0.50] and marginally significant effect on body weight [SMD = − 0.19; 95% CI − 0.40, 0.03; P = 0.09]. However, larger and well-designed clinical trials are needed to investigate the effects of C. annuum on MetS.
Similar content being viewed by others
Introduction
Metabolic syndrome (MetS), also known as syndrome X, is a clinical condition characterized by abdominal obesity, insulin resistance, hypertension, and dyslipidemia1. MetS has increasingly gained importance as the main risk factor for cardiovascular disease (CVD) and type II diabetes mellitus (T2D), which can lead to mortality2. Risk factors for CVD and T2D include hypertension, dyslipidemia, and high fasting glucose, which, in fact, characterize MetS3,4,5. Since the prevalence of the MetS is among a quarter of the global population, it has become a problem not only in the Western world but also in developing countries6.
MetS has been defined by various healthcare organizations, with minor differences in the definitions7. Four components, namely, obesity, hypertension, hyperglycemia, and dyslipidemia, are commonly used to describe this condition3. However, these factors could be improved to prevent MetS; this can be done by adopting healthier lifestyle habits such as dietary modification. Particularly, various natural compounds derived from plants are associated with beneficial effects on the incidence and progression of MetS8.
Capsicum annuum is a pungent spice, which is also known as red pepper or chili pepper. The capsaicinoid in the spice is responsible for its pungency. The main capsaicinoid is capsaicin, which is characterized by its chemopreventive, antioxidant, anti-inflammatory, hypolipidemic, thermogenic, and weight-reducing effects9. Recently, a non-pungent compound, capsinoid (e.g., capsiate, dihyrocapsiate), was discovered. Though few systematic reviews were conducted to provide an overview of the effects of C. annuum on factors related to MetS10,11, no meta-analysis (the quantitative summary of different or conflicting results) has been conducted in this regard. Therefore, the aim of this study was to evaluate the effects of C. annuum on factors related to MetS by focusing on relevant and related randomized controlled trials (RCTs). We determined whether an intervention with C. annuum supplementation compared with placebo supplementation had any effect on the components of MetS among the participants.
Results
Identification and selection of studies
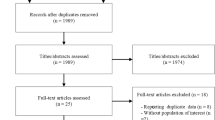
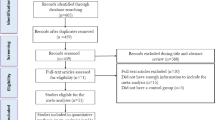
A total of 327 studies were identified from database search and three additional articles were identified from manual search. There were 273 articles after removing duplicate results; 251 trials were subsequently excluded by two authors based on their titles and abstracts. The remaining 22 articles were reviewed for dual full-text screening. After the review, a total of 6 trials were excluded. Thereafter, two papers were excluded due to insufficient data presentation12,13 and two more were excluded due to inappropriate interventions14,15, leaving a total of 12 studies, which were included in the systematic review. In meta-analysis, one study that has patients as subjects was excluded16. The whole selection process is presented in the Preferred Reporting Items for Systematic Reviews and Meta-Analyses flow diagram (Fig. 1).
Description of included studies
A total of 12 studies were included in the systematic review. The characteristics and findings of the included studies are summarized in Table 1. Most studies included both sexes, except for three studies that included only females16,17,18 and one study that included only males19. All studies were conducted on adults aged over 18 years; one study was conducted on middle-aged people and elderly (over 50 years)20. Out of the 12 studies, five were conducted in the United States18,19,21,22,23, three in Korea17,24,25, two in Japan20,26, one in Australia27, and one in China16. Ten of the studies were parallel-arm studies16,17,19,20,21,22,23,24,25,26 whereas the other two were crossover studies18,27. Ten studies were conducted on individuals with body mass index (BMI) above 25 kg/m216,17,18,19,21,22,23,24,25,27, and two studies were conducted on healthy individuals with BMI below 25 kg/m220,26. Six studies focused on intervention with purified capsaicinoids or capsinoids capsules19,20,21,22,23,26, three studies were on pepper powder or juice supplementation16,18,27, and another three were on fermented red pepper paste supplementation17,24,25. The duration of the interventions in the included studies varied from 4 to 12 weeks.
Potential sources of bias
The Risk of Bias (ROB) assessments for individual studies are presented in Fig. 2. Three out of the 12 included studies had a high ROB, whereas the remaining nine studies had some ROB concerns. Bias arising from the randomization process was low in three studies16,20,25, eight studies had some ROB concerns17,19,21,22,23,24,26,27, and one study had a high ROB18 because the only information in this article was a sentence stating that the study was randomized. Bias due to deviations from the intended intervention were low in all studies except for one17, in which allocation concealment was not reported. Bias due to missing outcome data was low in all the 12 studies. Two studies16,20 were rated with a low ROB in the measurement of outcomes, whereas 10 articles did not indicate whether the outcome assessors were aware of the intervention the study participants underwent. Two studies, in which only subgroup results were presented without the overall results17 and the results of follow-up were not reported25, were rated with a high ROB in the selection of the reported result.
The effects of C. annuum supplementation on obesity
The forest plot for overall random effects of C. annuum supplementation on anthropometric parameters in participants is shown in Fig. 3. Studies were grouped into those involving participants with low (< 25 kg/m2) and high (≥ 25 kg/m2) BMI. The meta-analysis on body weight involved five results from four studies that included participants with high BMI versus two studies that included participants with low BMI. The overall pooled statistics showed that the body weight of participants (seven results from six studies) were marginally reduced [standardized mean differences (SMD) = − 0.19; 95% confidence intervals (CI) − 0.40, 0.03; P = 0.09]. There was no statistically significant heterogeneity between the studies in the analysis of body weight [I2 = 0% and P = 0.46]. The effect of C. annuum was not significantly different between subgroups according to BMI. Five effect sizes from five studies on BMI and nine effect sizes from seven studies on body fat were included in the analysis of the impact of C. annuum (Fig. 3). The SMD of the overall pooled BMI and body fat was − 0.33 [95% CI − 1.03, 0.37; P = 0.36] and − 0.15 [95% CI − 0.35, 0.05; P = 0.13], respectively; the results did not decrease significantly. However, considerable heterogeneity was detected in the analysis of BMI [I2 = 85% and P < 0.01]. Sensitivity analyses of the effect of C. annuum supplementation on BMI showed that removal of any study did not alter the significance of the pooled effect size (Supplementary Fig. S1). However, removal of some studies changed the significance of the heterogeneity. Heterogeneity between studies regarding the pooled effect size of BMI reduced to 0% when the study of Cha et al.24 was removed. Each study’s contribution to the heterogeneity is presented by the Baujat plot in Supplementary Fig. S1.
The effects of C. annuum supplementation on hypertension
Overall, three effect sizes from three studies on diastolic blood pressure (DBP) and four effect sizes from four studies on systolic blood pressure (SBP) were included in the analysis of the impact of C. annuum (Fig. 4). C. annum supplementation had no significant effect on DBP [SMD = − 0.16; 95% CI − 0.49, 0.17; P = 0.34] and no significant between-study heterogeneity was observed in the analysis [I2 = 0% and P = 0.54]. Although C. annum supplementation had no significant effect on SBP [SMD = 0.22; 95% CI − 0.44, 0.88; P = 0.52], a significant heterogeneity was observed in the analysis of SBP [I2 = 81% and P < 0.01]. Sensitivity analyses of the effect of C. annuum supplementation on SBP showed that removing any study did not alter the significance of the pooled effect size but changed the heterogeneity to 0% when the study of Cha et al.24 was removed (Supplementary Fig. S2).
The effects of C. annuum supplementation on hyperglycemia
Three studies investigated the outcomes and impact C. annuum supplementation on glucose levels (Fig. 5). C. annuum supplementation had no significant effect on glucose levels [SMD = − 0.58; 95% CI − 1.62, 0.45; P = 0.27] but the analysis showed considerable heterogeneity [I2 = 91% and P < 0.01] between studies. Sensitivity analyses of the effect of C. annuum supplementation glucose levels showed that removal of any study did not alter the significance of the pooled effect size but lowered the heterogeneity to 0% when the study of Cha et al.24 was removed (Supplementary Fig. S3).
The effects of C. annuum supplementation on dyslipidemia
The forest plot for the overall effect of C. annuum supplementation on triacylglycerol (TG), which included results of three studies, is shown in Fig. 6. The pooled statistics showed that the SMD of TG was − 0.96 [95% CI − 2.67, 0.74; P = 0.27], which was not significantly different from that of the control group. The effect of C. annuum supplementation on TG showed considerable heterogeneity between studies [I2 = 95% and P < 0.01]. Sensitivity analyses of the effect of C. annuum supplementation on TG showed that removal of any study did not alter the significance of the pooled effect size but heterogeneity was lowered to 0% when the study of Cha et al.24 was removed (Supplementary Fig. S4). The forest plot for the overall effect of C. annuum supplementation on high density lipoprotein (HDL)-cholesterol, low density lipoprotein (LDL)-cholesterol, and total-cholesterol is shown in Fig. 6. C. annuum supplementation had no significant effects on HDL-cholesterol [SMD = 0.05; 95% CI − 0.28, 0.37; P = 0.78] and no significant between-study heterogeneity was noted in the analysis [I2 = 0% and P = 0.88]. Overall, four effect sizes from four studies were included in the analysis of the impact of C. annuum supplementation on total cholesterol and LDL-cholesterol. C. annuum supplementation had significant effects on LDL-cholesterol [SMD = − 0.39; 95% CI − 0.72, − 0.07; P = 0.02; prediction interval, − 1.28 to 0.50] compared to placebo supplementation. However, C. annuum supplementation had no significant effects on total-cholesterol [SMD = − 0.47; 95% CI − 1.06, 0.12; P = 0.12]. Statistical heterogeneity was detected in the analysis of total-cholesterol [I2 = 71% and P = 0.02] and LDL-cholesterol [I2 = 13% and P = 0.33]. Sensitivity analyses of the effect of C. annuum supplementation total-cholesterol showed that removal of any study did not alter the significance of the pooled effect size but lowered heterogeneity to 0% when the study of Cha et al.24 was removed (Supplementary Fig. S5).
Adverse events
All RCTs for C. annuum supplements reported no serious adverse effects and no events leading to withdrawal. However, three studies from the intervention groups reported adverse events, including leg cramps19, dyspepsia23, bowel irregularities23, diarrhea16,23, skin rash23, heat sensation in the oral cavity16, and skin wheals16 , whereas there were no adverse effects in the placebo groups. Six studies reported no adverse events during the study18,20,21,22,25,27, and three studies did not report any information regarding adverse events17,24,26.
Discussion
The aim of the present study was to investigate the effects of C. annuum on the factors related to MetS by focusing on the results of RCTs. Globally, MetS is considered as the main risk factor for CVD and T2D2. Several researches have been performed to investigate strategies such as dietary modification, which have a beneficial role in the prevention of MetS. A wide variety of herbs are considered as complementary and alternative treatments for MetS because they have beneficial effects on MetS and fewer side effects than conventional drugs. A systematic review and meta-analysis were performed to show the effects of green tea, Nigella sativa, and Irvingia gabonensis on MetS28,29.
In the present study, we found that C. annuum has marginally significant effects on body weight [SMD = − 0.19; 95% CI − 0.40, 0.03; P = 0.09]. Capsaicin is known to promote negative energy balance by increasing satiety and suppressing hunger, reducing energy and fat intake, and inducing thermogenesis30,31,32. Capsinoids, including capsiate and dihydrocapsiate, are also known to exert beneficial effects on energy balance. Reinbach et al., reported that CH-19 sweet pepper (non-pungent) reduces energy intake during positive energy balance. Additionally, CH-19 sweet pepper is associated with increased oxygen consumption, diet-induced thermogenesis, and an activated sympathetic nervous system (SNS)33,34. Furthermore, a meta-analysis revealed that capsiate increases energy expenditure (EE), but capsaicin has no overall effect on EE; however, both capsaicin and capsiate enhance fat oxidation35.
The effects of C. annuum on weight management are due to the activation of transient receptor potential vanilloid type 1 (TRPV1) receptors. In a preclinical study, dose-dependent administration of a TRPV1 agonist, capsaicin, prevented adipogenesis in stimulated 3T3-L1-preadipocytes, and oral administration of capsaicin prevented obesity in males36. Non-pungent capsiate is also an exogenous agonist of TRPV1 receptors and is associated with improved body metabolism including glucose homeostasis and obesity37. TRPV1 activation leads to activation of the SNS38, and inhibition of food intake by SNS activation accounts for reduced body fat stores and weight loss38. Some herbs and their active compounds, including several piperine analogs from Piper nigrum, can also reduce weight through TRPV1-dependent mechanisms39,40,41.
To the best of our knowledge, only a few meta‐analysis studies have investigated the anti‐obesity effects of C. annuum. After ingestion of capsaicin or capsinoids, EE increases and the respiratory quotient decreases, causing elevated fat oxidation42. Additionally, Zsiborás et al. showed that these metabolic effects of capsaicin and capsinoids are significant in individuals with a BMI greater than 25 kg/m242. Another meta-analysis also indicated that both capsaicin- and capsiate-augmented EE enhance fat oxidation, especially in high doses. The findings of the meta-analysis by Whiting et al. suggest that reduced energy intake from daily consumption of capsaicinoids contributes to weight management43. Golzarand et al. in another meta-analysis reported a significant weight loss after consumption of capsaicin supplements (− 0.50 kg; 95% CI − 0.90 to − 0.11) but body fat percentage did not change significantly compared to baseline values (0.11%, 95% CI − 0.22 to 0.43, P = 0.51)44.
Unfortunately, we could not establish a significant effect of C. annuum on TG (P = 0.27) and HDL-cholesterol levels (P = 0.78), and total cholesterol (P = 0.12); however, LDL-cholesterol (P = 0.02) were affected by C. annuum supplementation. The China Health and Nutrition Survey showed that frequent consumption of spicy food items including chili pepper is associated with improved blood lipid profiles and decreased risk for CVD45. Further, a preclinical study indicated that red chili pepper ethanol extract exhibits hypolipidemic effects. This was demonstrated through decreased levels of total cholesterol, LDL-cholesterol, TG, and very low density lipoprotein, with simultaneous increased HDL-cholesterol levels in female albino Wistar rats undergoing high fat diets46. Dietary capsaicinoids reduced total plasma cholesterol, non-HDL cholesterol, and TG in hamsters undergoing a high-cholesterol diets, suggesting decreased cholesterol absorption47. Additionally, short-term supplementation of capsaicin decreases LDL-cholesterol and increases HDL-cholesterol in vivo48.
CVD, a leading cause of death, now accounts for approximately one third of all deaths globally49. According to the World Health Organization, most CVD cases can be prevented by ameliorating risk factors such as high levels of blood lipids. Our present study showed that C. annuum can help reduce the risk of CVD by decreasing LDL-cholesterol levels. The well-known anti-oxidant effect of peppers doubles these beneficial effects50,51. This is because increased oxidative stress has a crucial role in the development of CVD and dietary consumption of antioxidants has been associated with reduced risk of CVD52,53. Both anti-hyperlipidemic and anti-oxidant effects of C. annuum are expected to effectively reduce the risk of CVD.
As an agonist for TRPV1 receptors, it has been suggested that capsaicin or capsiate can improve glucose metabolism by decreasing inflammation, increasing adiponectin levels, and lowering glucose and insulin levels37. Although we did not observe any significant effects of C. annuum on blood glucose levels, a lot of studies reported the ameliorating effect of capsaicin or capsiate on glucose metabolism via various mechanisms including improved insulin sensitivity, increased insulin levels, and gut micro biota modulation in diabetic animal models54,55,56. Though C. annuum has been shown to have beneficial effects on the blood pressure of preclinical hypertensive rats57, we did not note any significant effects of C. annuum supplementation on DBP and SBP in meta-analysis of the present study. Therefore, well‐designed clinical trials are needed for further evidence on hypolipidemic, hypoglycemic, and hypotensive effects of C. annuum, which have been identified in previous preclinical studies.
The strengths of the present study include consideration of all clinical trials that investigated the health-related effects of C. annuum on the components of MetS including obesity, hypertension, hyperglycemia, and dyslipidemia. However, some limitations need to be considered. First, the effects of C. annuum on the components of MetS were investigated using a small number of included studies; thus, evidence to support the effects was not enough. We could not assess subgroup analysis, meta-analysis of variance or meta-regression, and publication bias. Second, the present meta-analysis included many studies with ROB. Third, there was significant between-study heterogeneity in the present study which may be explained by differences in the methods of C. annuum intervention, study design, study population, etc. Finally, we did not register this systematic review in PROSPERO, an international prospective register of systematic reviews. For future studies, registering systematic reviews in prospective registers such as PROSPERO is recommended to avoid unintended duplication and to ensure transparent research process.
In conclusion, the present study evaluated of the effects of C. annuum on factors related to MetS by focusing on the results of RCTs. Our results showed that C. annuum supplementation had significant effect on LDL-cholesterol and marginally significant effect on body weight. Larger and well-designed clinical trials are needed to investigate the efficacy and safety of this dietary supplement in the treatment of MetS.
Methods
This systematic review and meta-analysis was reported in accordance with the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) guidelines58 (Supplementary Appendix 1).
Search strategy
The databases of PubMed, Embase, Google scholar, and Cochrane Library were searched up to April 2020 to identify relevant published articles. We used the following search terms to identify relevant titles and abstracts: “Capsicum annuum OR chili pepper OR chilli OR pepper NOT black pepper”. Moreover, only the studies conducted in clinical settings and written in English were considered. Additional articles were identified via a manual search of the reference lists of related original articles, reviews, and meta-analyses. After removing duplicate results using the Endnote software, the titles and abstracts were screened by two authors (Hwan-Hee Jang and Young-Min Lee) using Rayyan QCRI. The relevant studies were reviewed for dual full-text screening. Disagreements were resolved through discussion.
Inclusion criteria
The meta-analysis was performed using the ‘Patient/Population, Intervention, Comparison and Outcomes’ format to determine whether an intervention with C. annuum supplementation (I) compared with placebo supplementation (C) had any effect on the components of MetS (O) among participants (P). The outcomes of interest were as follows: obesity (BMI, body weight, and body fat), hypertension (SBP and DBP), hyperglycemia (blood glucose), and dyslipidemia (TG, total-cholesterol, LDL-cholesterol, and HDL-cholesterol). Parallel or crossover RCTs were included, and observational studies and review articles were excluded. Papers written in English and accessibility of the full-text publication were required for inclusion. Two authors independently reviewed data from all the studies that fulfilled the inclusion criteria and any conflicts was resolved by consensus.
Data extraction and ROB assessment
Two reviewers extracted and recorded the following data from each included study: authors and year of publication, study design, study population (proportion of women, age, number of participants, and BMI), intervention (dose, type, and duration), the outcomes (BMI, body fat, blood glucose, blood pressure, blood cholesterol, etc.) (Table 1). The ROB assessment was independently conducted by two reviewers using the Cochrane ROB 2.0 tool for parallel RCTs59. This tool considers selection bias, performance bias, detection bias, attrition bias, and reporting bias. Any disagreements were resolved by consensus.
Statistical analysis
We used SMD with 95% CI as effect size measures. If not reported17,18,19,20,25,26, the mean differences were calculated by subtracting the baseline mean from the post-intervention mean; the standard deviation (SD) differences were estimated using the following formula: SDdiff = √SDB2 + SDF2 − 2 × Corr × SDB × SDF, where SDB is the baseline SD and SDF is the SD of the final measures in the study60. The correlation value was conservatively set at 0.5 to calculate the change in SD61. Due to clinical heterogeneity of the studies, including differences in study design, doses, and intervention, a meta-analysis of quantitative data was conducted using random-effects model. A forest plot was mapped to indicate the pooled SMD and the 95% CI. Between-study heterogeneity was tested by using forest plots visually. Thereafter, both the Q homogeneity test and I2 statistics were used to evaluate the statistical heterogeneity quantitatively. Generally, there is considerable heterogeneity when p value for Q statistics is less than 0.1 and I2 is more than 50%62. Sensitivity analysis was conducted to investigate the effect of each study on the pooled effect size. All analyses were performed using R Statistical Software version 4.0.2 (Foundation for Statistical Computing, Vienna, Austria, URL https://www.r-project.org/).
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
McCracken, E., Monaghan, M. & Sreenivasan, S. Pathophysiology of the metabolic syndrome. Clin. Dermatol. 36, 14–20 (2018).
Ford, E. S. Risks for all-cause mortality, cardiovascular disease, and diabetes associated with the metabolic syndrome: A summary of the evidence. Diabetes Care 28, 1769–1778 (2005).
Cornier, M.-A. et al. The metabolic syndrome. Endocr. Rev. 29, 777–822 (2008).
Akbari-Fakhrabadi, M., Heshmati, J., Sepidarkish, M. & Shidfar, F. Effect of sumac (Rhus Coriaria) on blood lipids: A systematic review and meta-analysis. Complement. Ther. Med. 40, 8–12 (2018).
Namazi, N. et al. The impact of cinnamon on anthropometric indices and glycemic status in patients with type 2 diabetes: A systematic review and meta-analysis of clinical trials. Complement. Ther. Med. 43, 92–101 (2019).
Saklayen, M. G. The global epidemic of the metabolic syndrome. Curr. Hypertens. Rep. 20, 12 (2018).
Alberti, K. et al. Harmonizing the metabolic syndrome: A joint interim statement of the international diabetes federation task force on epidemiology and prevention; national heart, lung, and blood institute; American heart association; world heart federation; international atherosclerosis society; and international association for the study of obesity. Circulation 120, 1640–1645 (2009).
Rochlani, Y., Pothineni, N. V., Kovelamudi, S. & Mehta, J. L. Metabolic syndrome: Pathophysiology, management, and modulation by natural compounds. Ther. Adv. Cardiovasc. Dis. 11, 215–225 (2017).
Srinivasan, K. Biological activities of red pepper (Capsicum annuum) and its pungent principle capsaicin: A review. Crit. Rev. Food Sci. Nutr. 56, 1488–1500 (2016).
van den Driessche, J. J., Plat, J. & Mensink, R. P. Effects of superfoods on risk factors of metabolic syndrome: A systematic review of human intervention trials. Food Funct. 9, 1944–1966 (2018).
Sanati, S., Razavi, B. M. & Hosseinzadeh, H. A review of the effects of Capsicum annuum L. and its constituent, capsaicin, in metabolic syndrome. Iran. J. Basic Med. Sci. 21, 439 (2018).
Hoeger, W., Harris, C., Long, E. & Hopkins, D. Four-week supplementation with a natural dietary compound produces favorable changes in body composition. Adv. Ther. 15, 305–314 (1998).
Kroff, J. et al. The metabolic effects of a commercially available chicken peri-peri (African bird’s eye chilli) meal in overweight individuals. Br. J. Nutr. 117, 635–644 (2017).
Lejeune, M. P., Kovacs, E. M. & Westerterp-Plantenga, M. S. Effect of capsaicin on substrate oxidation and weight maintenance after modest body-weight loss in human subjects. J. Br. J. Nutr. 90, 651–659 (2003).
Nagasukeerthi, P., Mooventhan, A. & Manjunath, N. K. Short-term effect of add on bell pepper (Capsicum annuum var. grossum) juice with integrated approach of yoga therapy on blood glucose levels and cardiovascular functions in patients with type 2 diabetes mellitus: A randomized controlled study. Complement. Ther. Med. 34, 42–45. https://doi.org/10.1016/j.ctim.2017.07.011 (2017).
Yuan, L.-J. et al. Capsaicin-containing chili improved postprandial hyperglycemia, hyperinsulinemia, and fasting lipid disorders in women with gestational diabetes mellitus and lowered the incidence of large-for-gestational-age newborns. Clin. Nutr. 35, 388–393 (2016).
Kim, Y. et al. Hypoxanthine levels in human urine serve as a screening indicator for the plasma total cholesterol and low-density lipoprotein modulation activities of fermented red pepper paste. Nutr. Res. 30, 455–461. https://doi.org/10.1016/j.nutres.2010.06.014 (2010).
Nieman, D. C., Cialdella-Kam, L., Knab, A. M. & Shanely, R. A. Influence of red pepper spice and turmeric on inflammation and oxidative stress biomarkers in overweight females: A metabolomics approach. Plant Foods Hum. Nutr. 67, 415–421. https://doi.org/10.1007/s11130-012-0325-x (2012).
Galgani, J. E. & Ravussin, E. Effect of dihydrocapsiate on resting metabolic rate in humans. Am. J. Clin. Nutr. 92, 1089–1093. https://doi.org/10.3945/ajcn.2010.30036 (2010).
Yokoyama, K. et al. Effects of capsinoids on daily physical activity, body composition and cold hypersensitivity in middle-aged and older adults: A Randomized Study. Nutrients 12, 212 (2020).
Lee, Y., Cha, Y. S., Park, Y. & Lee, M. PPARγ2 C1431T polymorphism interacts with the antiobesogenic effects of Kochujang, a Korean fermented, soybean-based red pepper paste, in overweight/obese subjects: A 12-week, double-blind randomized clinical trial. J. Med. Food 20, 610–617. https://doi.org/10.1089/jmf.2016.3911 (2017).
Rogers, J. et al. Capsaicinoids supplementation decreases percent body fat and fat mass: Adjustment using covariates in a post hoc analysis. BMC Obes. 5, 22. https://doi.org/10.1186/s40608-018-0197-1 (2018).
Snitker, S. et al. Effects of novel capsinoid treatment on fatness and energy metabolism in humans: Possible pharmacogenetic implications. Am. J. Clin. Nutr. 89, 45–50. https://doi.org/10.3945/ajcn.2008.26561 (2009).
Cha, Y. S. et al. Kochujang, fermented soybean-based red pepper paste, decreases visceral fat and improves blood lipid profiles in overweight adults. Nutr. Metab. 10, 24. https://doi.org/10.1186/1743-7075-10-24 (2013).
Lim, J. H. et al. Supplementation with Aspergillus oryzae-fermented kochujang lowers serum cholesterol in subjects with hyperlipidemia. Clin. Nutr. 34, 383–387. https://doi.org/10.1016/j.clnu.2014.05.013 (2015).
Nirengi, S. et al. Assessment of human brown adipose tissue density during daily ingestion of thermogenic capsinoids using near-infrared time-resolved spectroscopy. J. Biomed. Opt. 21, 091305 (2016).
Ahuja, K. D., Robertson, I. K., Geraghty, D. P. & Ball, M. J. The effect of 4-week chilli supplementation on metabolic and arterial function in humans. Eur. J. Clin. Nutr. 61, 326–333. https://doi.org/10.1038/sj.ejcn.1602517 (2007).
Payab, M. et al. Effect of the herbal medicines in obesity and metabolic syndrome: A systematic review and meta-analysis of clinical trials. Phytother. Res. 34, 526–545 (2020).
Lee, J., Chung, M., Fu, Z., Choi, J. & Lee, H.-J. The effects of Irvingia gabonensis seed extract supplementation on anthropometric and cardiovascular outcomes: A systematic review and meta-analysis. J. Am. Coll. Nutr. 39, 388–396 (2020).
Westerterp-Plantenga, M., Smeets, A. & Lejeune, M. Sensory and gastrointestinal satiety effects of capsaicin on food intake. Int. J. Obes. Relat. Metab. Disord. 29, 682–688 (2005).
Reinbach, H. C., Smeets, A., Martinussen, T., Møller, P. & Westerterp-Plantenga, M. Effects of capsaicin, green tea and CH-19 sweet pepper on appetite and energy intake in humans in negative and positive energy balance. Clin. Nutr. 28, 260–265 (2009).
Ludy, M.-J. & Mattes, R. D. The effects of hedonically acceptable red pepper doses on thermogenesis and appetite. Physiol. Behav. 102, 251–258 (2011).
Hachiya, S. et al. Effects of CH-19 Sweet, a non-pungent cultivar of red pepper, on sympathetic nervous activity, body temperature, heart rate, and blood pressure in humans. Biosci. Biotechnol. Biochem. 71, 671–676 (2007).
Ohnuki, K. et al. CH-19 sweet, a non-pungent cultivar of red pepper, increased body temperature and oxygen consumption in humans. Biosci. Biotechnol. Biochem. 65, 2033–2036 (2001).
Ludy, M.-J., Moore, G. E. & Mattes, R. D. The effects of capsaicin and capsiate on energy balance: Critical review and meta-analyses of studies in humans. Chem. Senses 37, 103–121 (2012).
Zhang, L. L. et al. Activation of transient receptor potential vanilloid type-1 channel prevents adipogenesis and obesity. Circ. Res. 100, 1063–1070 (2007).
Zsombok, A. Vanilloid receptors—do they have a role in whole body metabolism? Evidence from TRPV1. J. Diabetes Complicat. 27, 287–292 (2013).
Bray, G. Reciprocal relation of food intake and sympathetic activity: Experimental observations and clinical implications. Int. J. Obes. Relat. Metab. Disord. 24, S8-17 (2000).
Boaz, M., Kaufman-Shriqui, V., Sherf-Dagan, S., Salem, H. & Navarro, D. A. Frequently used medicinal herbs and spices in weight management: A review. Funct. Foods Health Dis. 10, 305–324 (2020).
Christie, S., Wittert, G. A., Li, H. & Page, A. J. Involvement of TRPV1 channels in energy homeostasis. Front. Endocrinol. 9, 420. https://doi.org/10.3389/fendo.2018.00420 (2018).
Westerterp-Plantenga, M., Diepvens, K., Joosen, A. M., Bérubé-Parent, S. & Tremblay, A. Metabolic effects of spices, teas, and caffeine. Physiol. Behav. 89, 85–91 (2006).
Zsiborás, C. et al. Capsaicin and capsiate could be appropriate agents for treatment of obesity: A meta-analysis of human studies. Crit. Rev. Food Sci. Nutr. 58, 1419–1427 (2018).
Whiting, S., Derbyshire, E. & Tiwari, B. Could capsaicinoids help to support weight management? A systematic review and meta-analysis of energy intake data. Appetite 73, 183–188 (2014).
Golzarand, M., Toolabi, K. & Aghasi, M. Effect of green tea, caffeine and capsaicin supplements on the anthropometric indices: A meta-analysis of randomized clinical trials. J. Funct. Foods 46, 320–328 (2018).
Yu, K. et al. Association of spicy food consumption frequency with serum lipid profiles in older people in China. J. Nutr. Health Aging 22, 311–320 (2018).
Al-Jumayi, H. A., Elhendy, H. A. & Darwish, A. M. Biological effects of red chili pepper (Capsicum annuum) consumption on high fat diet female Albino Rats. Pak. J. Biol. Sci. PJBS 23, 150–158 (2020).
Liang, Y. T. et al. Capsaicinoids lower plasma cholesterol and improve endothelial function in hamsters. Eur. J. Nutr. 52, 379–388 (2013).
Lee, C. Y. J., Kim, M., Yoon, S. W. & Lee, C. H. Short-term control of capsaicin on blood and oxidative stress of rats in vivo. Phytother. Res. 17, 454–458 (2003).
WHO. Cardiovascular diseases (CVDs). http://www.who.int/mediacentre/factsheets/fs317/en (2016).
Howard, L., Talcott, S., Brenes, C. & Villalon, B. Changes in phytochemical and antioxidant activity of selected pepper cultivars (Capsicum species) as influenced by maturity. J. Agric. Food Chem. 48, 1713–1720 (2000).
Materska, M. & Perucka, I. Antioxidant activity of the main phenolic compounds isolated from hot pepper fruit (Capsicum annuum L.). J. Agric. Food Chem. 53, 1750–1756 (2005).
Aune, D. et al. Dietary intake and blood concentrations of antioxidants and the risk of cardiovascular disease, total cancer, and all-cause mortality: A systematic review and dose-response meta-analysis of prospective studies. Am. J. Clin. Nutr. 108, 1069–1091 (2018).
Taleb, A. et al. Antioxidant effects and mechanism of silymarin in oxidative stress induced cardiovascular diseases. Biomed. Pharmacother. 102, 689–698 (2018).
Kwon, D. Y. et al. Capsiate improves glucose metabolism by improving insulin sensitivity better than capsaicin in diabetic rats. J. Nutr. Biochem. 24, 1078–1085 (2013).
Zhang, S., Ma, X., Zhang, L., Sun, H. & Liu, X. Capsaicin reduces blood glucose by increasing insulin levels and glycogen content better than capsiate in streptozotocin-induced diabetic rats. J. Agric. Food Chem. 65, 2323–2330 (2017).
Hui, S. et al. Capsaicin improves glucose tolerance and insulin sensitivity through modulation of the gut microbiota-bile acid-FXR axis in type 2 diabetic db/db mice. Mol. Nutr. Food Res. 63, 1900608 (2019).
Yang, D. et al. Activation of TRPV1 by dietary capsaicin improves endothelium-dependent vasorelaxation and prevents hypertension. J. Cell Metab. 12, 130–141 (2010).
Liberati, A. et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. J. Clin. Epidemiol. 62, e1–e34 (2009).
Higgins, J. P. et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 343, d5928 (2011).
Higgins, J. & Green, S. Cochrane Handbook for Systematic Reviews of Interventions Version 5.1. 0. (The Cochrane Collaboration, London, 2011).
Fukuta, H., Goto, T., Wakami, K. & Ohte, N. Effects of drug and exercise intervention on functional capacity and quality of life in heart failure with preserved ejection fraction: A meta-analysis of randomized controlled trials. Eur. J. Prev. Cardiol. 23, 78–85 (2016).
Higgins, J. P. et al. Cochrane Handbook for Systematic Reviews of Interventions (Wiley, Hoboken, 2019).
Acknowledgements
This research was supported by a research program for Agricultural Science & Technology Development from National Institute of Agricultural Science (NAS)-Rural Development Administration (RDA), South Korea (Grant number: PJ01420101), and by a research grant from Seoul Women’s University, South Korea (Grant number 2020-0389).
Author information
Authors and Affiliations
Contributions
Conceptualization, Y.-M.L.; data curation, H.-H.J. and Y.-M.L.; writing—original-draft preparation, H.-H.J. and Y.-M.L.; writing—review and editing, Y.-M.L. and J.-H. L; project administration, S.-H. L. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jang, HH., Lee, J., Lee, SH. et al. Effects of Capsicum annuum supplementation on the components of metabolic syndrome: a systematic review and meta-analysis. Sci Rep 10, 20912 (2020). https://doi.org/10.1038/s41598-020-77983-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-77983-2
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.