Abstract
Predation of invertebrate pest by natural enemies is a critical contribution of nature to people, because invertebrate pests cause a vast amount of economic damage and pesticides use has many long-term costs. Veteran trees are keystone structures and hotspots for biodiversity, and are a potential source of natural enemies. To explore this, we used a balanced experimental design where we measured predatory beetle diversity and attack marks on three colors of artificial caterpillars placed around 20 veteran oaks and 20 nearby young oaks, in Southern Norway. We predicted that around the veteran oaks there would be a greater diversity of predatory beetles and more invertebrate attacks on artificial caterpillars. Sampling for predatory beetles was conducted in summer 2017 and 2018, and invertebrate attacks were measured in 2018. We found support for the predictions: diversity of predatory beetles was higher around veteran trees and there were more arthropod attack marks on artificial caterpillars placed around veteran trees. Our results indicated that veteran trees are a source of natural enemies. Valuing and protecting veteran trees and their communities is an essential step towards a more sustainable system of management that has the possibility of promoting both the wellbeing of people and biodiversity.
Similar content being viewed by others
Introduction
Biodiversity loss and the associated environmental and social problems are considered some of the central challenges of our time1, in part, because all societies are dependent on the functioning of ecosystems for the support of human existence and wellbeing2. Nature’s contribution to people [NCP] has been suggested as a framework to help societies better understand and relate to the ecosystems on which they depend3. NCP can be defined as all the contributions, both positive and negative, of living nature (diversity of organisms, ecosystems, and their associated ecological and evolutionary processes) to people’s quality of life1.
Invertebrate pests are an example of an NCP with considerable negative impacts on society. Agricultural intensification has exacerbated this problem by simplifying the landscape and reducing biodiversity4, and the pervasive method of using pesticides to control invertebrate pests has many interrelated costs for local people, future generations and biological communities5,6,7,8. One viable alternative to the use of pesticides is to preserve or enhance predation by the natural enemies in a target area9. While pest control with natural enemies is an ancient method, with records of it being implemented as early as 900 AD9, it has recently gained new interest as an beneficial NCP10,11. Additionally, research has shown that landscape complexity is critical for this NCP, because higher levels of habitat heterogeneity have positive effects on the ability of multiple enemies to coexist due to the presence of additional non-pest prey and greater range of microhabitats11,12.
Veteran trees have played a prominent role in many cultures around the world and throughout the ages13. They are ‘keystone structures’ for biological communities14,15, and are an integral aspect in many traditional landscapes and sacred sites13,16,17. Veteran oaks are a hotspot for biodiversity in Northern Europe18,19 and enhance the structural complexity of the landscape20. Large trees in agroforestry systems have been found to enhance functional biodiversity and promote beneficial NCP including invertebrate pest control21. Veteran oaks provide food resources and shelter for a diverse set of species19, including arthropod predators22,23 and may be a source of a natural enemies of invertebrate pests.
However, high levels of biodiversity do not necessarily result in enhanced ecosystem functioning; ecosystem functioning is instead more closely related to a diversity in traits [functional diversity] rather than taxonomic diversity24,25. Therefore, functional diversity can be considered the link between biodiversity and ecosystem functioning. When specifically considering predation by natural enemies, mounting evidence suggests that predation increases when the natural enemies have complementary traits10,11. Natural enemies are complementary when they attack different pest species, have differences in their phenologies and diurnal/nocturnal activities, and have different hunting behaviors11. Although it has been established that veteran trees increase the structural complexity of the landscape15 and are a source of diverse predator22, the link between biodiversity and ecosystem functioning has received less attention and it is unknown to what extent the presences of these trees influences the beneficial NCP of predation by natural enemies.
Assessing differences in the predation by natural enemies is not straight forward26. Attacks by predators on invertebrate prey are usually cryptic and rarely leave any evidence of the event, and visual observations are seldom possible and complicated by presence of the observer27. Other methods such as examination of predator gut contents, or radioactive labelling of the prey have difficulties distinguishing between “real” predation and scavenging or secondary predation28. An alternative method of measuring predation intensity is with the use of artificial caterpillars. The technique involves fashioning prey from malleable, non-hardening material, deploying them in the field and then measuring depressions left on the models by predators29,30. The marks left on the artificial caterpillars from attempted predation event can be used to identify the predator30. Additionally, different colors of artificial caterpillars can be deployed to mimic different prey species27,31. The level of identification of predator attack marks has varied between studies, but identification at a coarse taxonomic level (bird, mammal, or invertebrates) has been shown to be the most prudent approach29. Invertebrate attacks on artificial caterpillars have been attributed to ants, ground beetles, predatory bugs, predatory wasps, parasitoid wasps, spiders27 and in some cases non-predatory insects26,27,31. Although the method of deploying artificial caterpillars to measure predation rates has limitations, it has been found to be suitable for measuring predation rates in comparative studies27.
In order to measure the contribution of veteran oaks to the beneficial NCP of predation by natural enemies, we employed a balanced experimental design. We matched twenty veteran oaks with twenty nearby young oaks, which were taken to represent the background levels, and measured the diversity of predatory beetles and the number of invertebrate attack marks on artificial caterpillars placed around the trees. We included trees from open landscapes and forests to span the variation that is observed in veteran oak distribution in Northern Europe, but we were primarily focused on the dichotomy between the veteran and the young tree. The study had two main predictions related to this dichotomy: first, there would be a greater diversity of beetle predators around veteran trees, and second, there would be more attacks on artificial caterpillars deployed around veteran trees. The first prediction was based on the fact that veteran oaks have high diversity of arthropods associated with them19. We defined diversity as including both taxonomic and functional diversity, and for our measure of functional diversity we chose to focus on traits that were identified as being complementary for natural enemies10,22,32. The next prediction was rooted in the first: a community of predators with diverse complementary traits (ie: predators with different hunting strategies, prey species and phenologies) will have high predation levels. If these predictions are supported, veteran trees should be considered as enhancing predation by providing a source of natural enemies.
Results
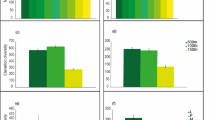
Over the course of the two summers we captured a total of 465 beetle species of which 173 were predators. Both species richness and functional diversity of complementary traits were higher around veteran than young trees (Table 1, P = 0.01 and 0.037, respectively). Independent of the type of tree, there was a yearly and seasonal effect on both measures of diversity, which decreased through the season and was significantly lower at the end of the summer in 2018 (Table 1, Fig. 1).
Estimates from the models that predicted species richness (left plot) and functional diversity (right plot) of predatory beetles (± SEM; n = 238 from 40 trees). Species richness was modeled with a generalized liner mixed effect model with Poisson error distribution, and functional diversity was calculated as functional dispersion (FDis) and modeled with a Liner mixed effect model with Gaussian error distribution. Both measures of diversity were higher around veteran trees (P = 0.01, 0.037, top), and independent of the type of tree there was a yearly and seasonal effect where diversity decreased through the season and was significantly lower at the end of 2018 (P = 0.001, 0.017, bottom). Window trap samples were collected once a month from May to August 2018, at the same time as the artificial caterpillars were collected. A summary of the models is presented the Table 1 (Ill. by Matthew Cooper).
In total we placed out 720 artificial caterpillars, however we found that many (52%) of them were either missing or damaged to the point of obscuring other attack marks. Despite this, the remaining caterpillars were well balanced within the experimental setup. We found that arthropods attacked 47% of the remaining artificial caterpillars (n = 345). The optimal model that predicted the number of arthropod attack marks on the artificial caterpillars included the type of tree, the color of the caterpillars, the position of caterpillars and a seasonal effect (Table 1, Fig. 2). Attack rates were higher around veteran trees than young trees (P = 0.007). Green caterpillars were attacked less than caterpillars with other colors (P = 0.005), and all caterpillars were attacked more when they were placed at ground level (P = 0.016). There was also a decrease in attacks towards the end of the summer (P = 0.041).
Estimated invertebrate attack rates on the artificial caterpillars from the Generalized liner mixed effect model with Negative binomial error distribution (± SEM; n = 345). Artificial caterpillars were subjected to a 30-day exposer, being collected and replaced once a month from May to August 2018, at the same time as the window trap samples were collected. A summary of the model is presented the Table 1 (Ill. by Matthew Cooper).
Discussion
In the present study, we found that our predictions that there would be higher predatory beetle diversity (both taxonomic and functional) and invertebrate attack rates around veteran oaks were supported. We also found that the diversity of predatory beetles and invertebrate attack rates decreased in parallel through the season, indicating that they were interrelated. Veteran trees have been identified as a hotspot for invertebrate diversity in Northern Europe19 and have a greater diversity of beetles associated with them than younger trees15. Building on these finding, our results are the first to show that both the diversity of natural enemies and invertebrate predation rates were higher around veteran trees than young trees, given an otherwise similar habitat. Our results considered in conjunction with each other, provide strong evidence to support the conclusion that veteran trees are a source of natural enemies.
Habitat heterogeneity is important for predation by natural enemies, as it increases the possibility of multiple invertebrate enemies to coexist11,12 and has been linked to a greater diversity of invertebrate predators22,33. The coexistence of natural enemies with complementary traits has been found to be the most important predictor of pest control by natural enemies10. Müller et al.17 attributes the finding that veteran trees had greater diversity of beetles associated with them than young trees to the structural heterogeneity proved by the veteran trees. As the size and age of a tree increases so does its structural heterogeneity, and this increases the number of micro-habitats available for shelter and potential prey species34,35. In our study it is likely that the structural heterogeneity proved by the veteran trees allowed for the coexistence of natural enemies and promoted complementary trait diversity, which lead to the higher predation rates that we observed around veteran trees. It should be noted however, that we did not determine the identity of the invertebrate predators attacking the artificial caterpillars, and it is therefore unknown to what extent our findings were a result of attacks from specialized predators that are dependent on veteran trees and more generalist predators that were attracted to veteran trees for shelter and as a source of prey. It is likely that it was the later, as it has been shown that ground beetles are important predators in Northern European forests26,36,37. In either case however, veteran trees provide resources that are beneficial for invertebrate predators and are lacking or of lower quality in younger trees17.
Although this study focused on the dichotomy between veteran and young trees at rather small spatial scales (200 m or less), other research has established that the immediate surroundings and the wider landscape around the veteran trees can influence invertebrate diversity22,38,39,40,41 and that large scale anthropogenic factors such as urbanization gradients influence predation rates by natural enemies12,26. In contrast, research within agroecosystems has found that strips of non-agriculture area with resources for natural enemies reduce pests and crop plant damage independent of landscape complexity42. It is clear that veteran trees should not be considered independent units because invertebrate diversity has been shown to be influenced by habitat connectivity at spatial scales up to 25 km22,38,39, but the interaction between local and regional landscape effects with predation around veteran trees needs more research.
The findings that green artificial caterpillars were attacked less by arthropod predators and that artificial caterpillars were attacked more when they were placed at ground level are interesting results that have implications for pest control with natural enemies and future research with artificial caterpillars. It is likely that green caterpillars were less detectable due to lower contrast with the background36 and that ground dwelling predators were responsible for the increase in attack marks on artificial caterpillars placed at ground level. It has been found that visual signals have a strong influence on hunting arthropods43,44,45, and artificial prey coloration had an effect on invertebrate attacks in Northern Europe36 but less so in the tropics31. This difference may be due to the dominance of ants in tropics, which are more chemically oriented and therefore less influenced by the prey coloration31. Certain ant species such as Lasius brunneus and L. ferrugineus, are associated with tree hollows in Europe46, but ants were only observed in high numbers at three of the study trees (one veteran and two young). On the other hand, predation by beetles has been observed to be influenced by prey coloration, although this has been found to vary between even related species47, and ground beetles are likely the dominant invertebrate predator attacking artificial caterpillars in Northern European forests26,36,37. The conclusion that beetles were the dominant predator attacking the artificial caterpillars in our study is further supported by the fact that we found ground beetles in our traps, and as discussed previously, invertebrate attack rates paralleled the trapping data of predatory beetles. It would, however, be beneficial to know the identity of the invertebrate predators so that these findings could be more clearly integrated into an understanding of predation by natural enemies.
Finally, it should be noted that a large portion of the artificial caterpillars placed out during this study were found to be missing or damaged to the point of obscuring other attack marks. It does not appear that this influenced the results related to invertebrate attacks because the remaining caterpillars were well balanced within the experimental setup. We presume that the missing and damaged caterpillars were a result of bird attacks29. Our study was not designed to measure avian predation and the relatively short distance between the two types of trees likely allowed birds to easily move between them. The influence of veteran trees on avian predation is clear avenue of future research, but a different study design would be needed to further explore this relationship.
Conclusions
Our results that species richness and complementary trait diversity of invertebrate predators responded in parallel through the season with predation rates and were higher around the veteran trees clearly indicate that veteran trees are a source of natural enemies. Veteran trees are valuable because of their cultural significance16 and their importance for biodiversity19. They increase the structural complexity of landscapes35 and based on our results, their communities may contribute to invertebrate pest control. These results give clear incentive to protect veteran trees and their associated communities. Veteran trees also provide additional contributions of benefits to people that are both economic and cultural48,49,50. Hartel et al.51 goes on to state that wood-pastures with veteran scattered trees provide a model ecosystem for the sustainable integration of food production and biodiversity conservation. Protecting and valuing veteran trees and their communities and reintegrating them into agricultural systems is an essential step towards a more sustainable system of management and has the possibility of enhancing the wellbeing of people while promoting biodiversity.
Material and methods
In order to test our predictions, we established a balanced experimental design where we chose 20 veteran oaks in the central distribution of oaks in Southern Norway from the Norwegian database of veteran oaks51 and matched them with 20 young oaks from the nearby surroundings. We used stratified random sampling to include veteran trees in forest and open landscapes (n = 12 and 8, respectively), had trunks with circumferences of 2 m or greater (measured at the height of 130 cm) and had young oaks in the immediate surroundings. Young oaks were within 200 m of the focal veteran oak, had similar immediate surroundings (e.g. openness, sun exposure and surrounding tree species) and were at least 50 m from any other veteran oak. The mean circumference of the veteran oaks was 283 cm (200–405 cm) and mean circumference of the young oaks was 74.5 cm (25–148 cm). The trees were originally identified within 500 × 500 m blocks, which we will refer to as the sampling blocks51. Twelve pairs of trees were nested in clusters of three within a sampling block, but all tree pairs were more than 100 m apart. All trees were within a 30 km radius of the city of Larvik.
To measure the functional and taxonomic diversity of predatory beetles, we sampled the beetle communities around the focal tree with flight intercept traps over the course of summer 2017 and 2018. The traps were made of two intersecting 20 × 40 cm windows with a funnel below leading to a vial containing propylene glycol, water (4:1 mixture) and a drop of detergent used as a surfactant. The traps were hung from a branch in the canopy of the focal tree and were placed out in May and emptied once a month until August (Fig. 3).
A figure of the study design that we used to measure the contribution of veteran oak invertebrate comminutes to predation by natural enemies. We measured predatory beetle diversity with window traps and predation rates with attack marks left on artificial caterpillars around 20 pairs of young and veteran oaks in Southern Norway. The window traps were active through the summer in 2017 and 2018 and artificial caterpillars were placed out in the summer 2018. The artificial caterpillars were secured to a natural attachment sites 2–4 m from the trunk of the focal tree (Ill. by Matthew Cooper).
Predation rates were measured with artificial caterpillars made from plasticine (JOVIE) formed into 20–30 mm long and 5 mm thick cylinders by hand. A metal wire (Ø 1.1 mm) extending from the core of each caterpillar were used for attachment. Six artificial caterpillars were placed 2–4 m from the trunk of the focal tree and split equally by two positions: 1.5–2 m and at ground level (0–10 cm). The caterpillars were attached to a natural site of attachment (branch or stem). Although it has been shown that the plant species had little effect on invertebrate predation of artificial caterpillars31, the caterpillars was either attached to a branch of the focal tree or to a branch of a nearby deciduous tree. Three colors (green, brown and black) were chosen to mimic the variety of lepidoptera larvae commonly found in Scandinavian forests (Fig. 3). Artificial caterpillars were subjected to a 30-day exposer, being collected and replaced once a month from May to August 2018, at the same time as the window trap samples were collected. The location of the caterpillars remained the same through the experiment, but to avoid bias the colors were randomized within the location so that the order was not same between sampling periods.
Attack marks on the artificial caterpillars were documented in the field and verified in the lab. They were identified as being made by either arthropods, birds, small mammals or an unknown source based on a key provided by Low et al.29, and counted for each taxonomic group. In total we placed out 720 artificial caterpillars, but 375 were found to be either missing or unidentifiable, presumably due to bird attacks29. Despite this, the remaining caterpillars were well balanced within the experimental setup. There were 162 caterpillars remaining around the veteran trees and 183 around the young trees, 187 reaming at the high and 158 at the low location, and regarding the different colors there were 110 black, 116 brown and 119 green caterpillars. However, the number of caterpillars remaining through the sampling period did decrease as the summer progressed: we collected 177 caterpillars in the first period, 96 in the second period and 72 in the last period.
All beetles collected in the flight intercept traps were identified to the species level following the taxonomy of The Norwegian Biodiversity Information Centre52 by an expert. Following the protocol set by Wetherbee et al.24 species were classified as predators based on both adult and larvae diets, and adult trait information (body length, relative eye size and peak activity date) was collected from literature or calculated from available material (Table 2). Functional diversity was subsequently calculated based on all traits. Since functional diversity indices are sensitive to missing trait information, we verified that at least 80% of all species in the functional groups had trait information53. All species that were excluded as a result of lack of data were rare in the data set (abundance less than 5). We chose to use functional dispersion (FDis) to measure functional diversity because it accounts for species abundances, it can be calculated for multiple traits, and species richness has limited effect on it54. FDis is a measure of dispersion in trait space and is calculated as the mean distance of species to the centroid of the community and is weighted by abundances54.
Prior to statistical analysis, we followed the steps for data exploration outlined by Zuur et al.55. Statistical analysis was carried out in R version 3.4.056. Species richness and FDis were calculated with the dbFD function in the ‘FD’ package54,56. FDis was calculated using a Gower dissimilarity matrix and the "cailliez" correction method54,57. All models were created with the function glmmTMB from the package ‘glmmTMB’58. The following predictor variables were initially included in all models: whether the tree was veteran or young, the sampling period (early, mid or late), whether the tree was in an open landscape or a forest, a land use gradient, the tree cover density in a 50 and 100 m radius of the focal tree, and the circumference of the focal tree. Additionally, the sampling year was included in the beetle diversity models and the color and location of the artificial caterpillars were included in the invertebrate attack model. The best model was chosen with backward model selection based on Akaike information criterion (AIC) and non-significant predictors were removed (P > 0.05)55. We also determined the best error distribution and random effect structure by comparing the AIC of candidate models using the AICtab function in the package ‘bbmle’59. We compared three different random effect structures to deal with spatial correlation between the tree pairs (the veteran/young tree pair, the sampling blocks and a crossed random effect of tree pair and sampling block) and a model with no random effect, and found that a random intercept model with sampling block as the random effect was the best random effect structure for all models. The coefficient of determination (pseudo R2) was calculated for the models using the r.squaredGLMM function in the MuMIn package60.
We modeled species richness of predatory beetles with a Generalized liner mixed effect model with Poisson error distribution. We found that arthropod attack marks were overdispersed, and used a Generalized liner mixed effect model with Negative binomial error distribution with the “NB2” parameterization (variance = µ(1 + µ/k) to deal with the additional dispersion58,61. Although FDis is bound between 0 and 1, in our dataset it was approximately normally distributed, so we used a Linear mixed model with Gaussian error distribution to model it. The data also appeared to have slight zero inflation, but the Linear mixed effect model had a lower AIC and was chosen as our final model62. The final models were checked for patterns in the residuals, influential observations, and spatial and temporal structure that was not accounted for by the model55. The following packages were also used for data manipulation, statistical analysis and graphical visualization: ‘lattice’63, ‘ggplot2’64, ‘dplyr’65.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
IPBES. Intergovernmental science-policy platform on biodiversity and ecosystem services. ttps://ipbes.net (2018).
Kadykalo, A. N. et al. Disentangling ‘ecosystem services’ and ‘nature’s contributions to people’. Ecosyst. People 15(1), 269–287 (2019).
Díaz, S. et al. Assessing nature’s contributions to people. Science 359(6373), 270–272 (2018).
Gámez-Virués, S. et al. Landscape simplification filters species traits and drives biotic homogenization. Nat. Commun. 6(1), 8568 (2015).
Wilson, C. & Tisdell, C. Why farmers continue to use pesticides despite environmental, health and sustainability costs. Ecol. Econ. 39(3), 449–462 (2001).
Pimentel, D. et al. Environmental and economic costs of pesticide use. Bioscience 42(10), 750–760 (1992).
Horrigan, L., Lawrence, R. & Walker, P. How sustainable agriculture can address the environmental and human health harms of industrial agriculture. Environ Health Perspect 110(5), 445–456 (2002).
Popp, J., Pető, K. & Nagy, J. Pesticide productivity and food security. A review. Agron. Sustain. Dev. 33(1), 243–255 (2013).
Barbosa, P. Conservation Biological Control (Academic Press, Cambridge, 1998).
Greenop, A., Woodcock, B., Wilby, A., Cook, S. & Pywell, R. Functional diversity positively affects prey suppression by invertebrate predators: A meta-analysis. Ecology 99(8), 1771–1782 (2018).
Snyder, W. E. Give predators a complement: Conserving natural enemy biodiversity to improve biocontrol. Biol. Control 135, 73–82 (2019).
Perez-Alvarez, R., Nault, B. A. & Poveda, K. Effectiveness of augmentative biological control depends on landscape context. Sci. Rep. 9(1), 8664 (2019).
Hageneder, F. The Heritage of Trees: History, Culture and Symbolism (Floris Books, 2001).
Tews, J. et al. Animal species diversity driven by habitat heterogeneity/diversity: The importance of keystone structures. J. Biogeogr. 31, 79–92 (2004).
Müller, J., Jarzabek-Müller, A., Bussler, H. & Gossner, M. M. Hollow beech trees identified as keystone structures for saproxylic beetles by analyses of functional and phylogenetic diversity. Anim. Conserv. 17(2), 154–162 (2013).
Jim, C. Urban heritage trees: Natural-cultural significance informing management and conservation. In Greening Cities Advances in 21st Century Human Settlements (eds. Tan, P. & Jim, C.) (Springer, Berlin, 2017).
Hu, L., Li, Z., Liao, W. & Fan, Q. Values of village fengshui forest patches in biodiversity conservation in the Pearl River Delta, China. Biol. Conserv. 144(5), 1553–1559 (2011).
Skarpaas, O., Blumentrath, S., Evju, M. & Sverdrup-Thygeson, A. Prediction of biodiversity hotspots in the Anthropocene: The case of veteran oaks. Ecol. Evol. 7(19), 7987–7997 (2017).
Siitonen, J., Ranius, T. The Importance of Veteran Trees for Saproxylic Insects. In Europe's Changing Woods and Forests: From Wildwood to Managed Landscapes (eds. Kirby, K. & Watkins, C.) 140–53 (CAB International, Wallingford, 2015).
Lindenmayer, D. B. et al. New policies for old trees: Averting a global crisis in a keystone ecological structure. Conserv. Lett. 7(1), 61–69 (2014).
Tscharntke, T. et al. Multifunctional shade-tree management in tropical agroforestry landscapes—A review. J. Appl. Ecol. 48(3), 619–629 (2011).
Wetherbee, R., Birkemoe, T., Skarpaas, O. & Sverdrup-Thygeson, A. Hollow oaks and beetle functional diversity: Significance of surroundings extends beyond taxonomy. Ecol. Evol. 10(2), 819–831 (2020).
Pilskog, H., Birkemoe, T., Framstad, E. & Sverdrup-Thygeson, A. Effect of habitat size, quality, and isolation on functional groups of beetles in hollow oaks. Insect Sci. 16, 1–8 (2016).
Lefcheck, J. & Duffy, J. E. Multitrophic functional diversity predicts ecosystem functioning in experimental assemblages of estuarine consumers. Ecology 96(11), 2973–2983 (2015).
Heemsbergen, D. A. et al. Biodiversity effects on soil processes explained by interspecific functional dissimilarity. Science 306(5698), 1019–1020 (2004).
Ferrante, M., Lo Cacciato, A. & Lovei, G. L. Quantifying predation pressure along an urbanisation gradient in Denmark using artificial caterpillars. Eur. J. Entomol. 111(5), 649–654 (2014).
Lovei, G. L. & Ferrante, M. A. Review of the sentinel prey method as a way of quantifying invertebrate predation under field conditions. Insect Sci. 24(4), 528–542 (2017).
Kidd, N. A., Jervis, M. A. Population dynamics in Insects as Natural Enemies (ed. Jervis, M. A.) 435–523 (Springer, 2005).
Low, P. A., Sam, K., McArthur, C., Posa, M. R. C. & Hochuli, D. F. Determining predator identity from attack marks left in model caterpillars: Guidelines for best practice. Entomol. Exp. Appl. 152(2), 120–126 (2014).
Howe, A., Lovei, G. & Nachman, G. Dummy caterpillars as a simple method to assess predation rates on invertebrates in a tropical agroecosystem. Entomol. Exp. Appl. 131, 325–329 (2009).
Sam, K., Remmel, T. & Molleman, F. Material affects attack rates on dummy caterpillars in tropical forest where arthropod predators dominate: An experiment using clay and dough dummies with green colourants on various plant species. Entomol. Exp. Appl. 157(3), 317–324 (2015).
Moretti, M. et al. Handbook of protocols for standardized measurement of terrestrial invertebrate functional traits. Funct. Ecol. 31(3), 558–567 (2017).
Mico, E. et al. Contrasting functional structure of saproxylic beetle assemblages associated to different microhabitats. Sci. Rep. 10(1), 1520 (2020).
Ranius, T. & Jansson, N. The influence of forest regrowth, original canopy cover and tree size on saproxylic beetles associated with old oaks. Biol. Conserv. 95(1), 85–94 (2000).
Parmain, G. & Bouget, C. Large solitary oaks as keystone structures for saproxylic beetles in European agricultural landscapes. Insect Conservation and Diversity 11, 100–115 (2018).
Ferrante, M., Barone, G., Kiss, M., Bozóné-Borbáth, E. & Lövei, G. L. Ground-level predation on artificial caterpillars indicates no enemy-free time for lepidopteran larvae. Community Ecol. 18(3), 280–286 (2017).
Lövei, G. L. & Sunderland, K. D. Ecology and behavior of ground beetles (Coleoptera: Carabidae). Annu. Rev. Entomol. 41(1), 231–256 (1996).
Sverdrup-Thygeson, A., Skarpaas, O. & Ødegaard, F. Hollow oaks and beetle conservation: The significance of the surroundings. Biodivers. Conserv. 19(3), 837–852 (2010).
Sverdrup-Thygeson, A., Skarpaas, O., Blumentrath, S., Birkemoe, T. & Evju, M. Habitat connectivity affects specialist species richness more than generalists in veteran trees. For. Ecol. Manag. 403, 96–102 (2017).
Gough, L. A., Birkemoe, T. & Sverdrup-Thygeson, A. Reactive forest management can also be proactive for wood-living beetles in hollow oak trees. Biol. Conserv. 180, 75–83 (2014).
Hagge, J. et al. Congruent patterns of functional diversity in saproxylic beetles and fungi across European beech forests. J. Biogeogr. 46(5), 1054–1065 (2019).
Tschumi, M., Albrecht, M., Entling, M. H. & Jacot, K. High effectiveness of tailored flower strips in reducing pests and crop plant damage. Proc. R. Soc. B. 282(1814), 20151369 (2015).
Bowdish, T. I. & Bultman, T. L. Visual cues used by mantids in learning aversion to aposematically colored prey. Am. Midl. Nat. 129(2), 215–222 (1993).
Kauppinen, J. & Mappes, J. Why are wasps so intimidating: Field experiments on hunting dragonflies (Odonata: Aeshna grandis). Anim. Behav. 66(3), 505–511 (2003).
Prudic, K. L., Stoehr, A. M., Wasik, B. R. & Monteiro, A. Eyespots deflect predator attack increasing fitness and promoting the evolution of phenotypic plasticity. Proc. R. Soc. B. 282(1798), 20141531 (2015).
Micó, E. Saproxylic insects in tree hollows. In Saproxylic Insects: Diversity, Ecology and Conservation (ed. Ulyshen, M. D.) 693–727 (Springer, Berlin, 2018).
Harmon, J. P., Losey, J. E. & Ives, A. R. The role of vision and color in the close proximity foraging behavior of four coccinellid species. Oecologia 115(1), 287–292 (1998).
Hartel, T., Réti, K. O. & Craioveanu, C. Valuing scattered trees from wood-pastures by farmers in a traditional rural region of Eastern Europe. Agric. Ecosyst. Environ. 236, 304–311 (2017).
Hougner, C., Colding, J. & Söderqvist, T. Economic valuation of a seed dispersal service in the Stockholm National Urban Park, Sweden. Ecol. Econ. 59(3), 364–374 (2006).
Lindenmayer, D. B. & Laurance, W. F. The ecology, distribution, conservation and management of large old trees. Biol. Rev. 92(3), 1434–1458 (2017).
ARKO. Hule eiker—et hotspot-habitat Sluttrapport under ARKO-prosjektets periode II. https://www.miljodirektoratet.no/globalassets/publikasjoner/dirnat2/attachment/2557/nina-rapport-710_hotspot-hule-eiker_sverdrup-thygeson_2011.pdf. The Norwegian Institute for Nature Research (2011).
NBIC. Norwegian Biodiversity Information Centre. https://www.biodiversity.no (2018).
Majekova, M. et al. Evaluating functional diversity: Missing trait data and the importance of species abundance structure and data transformation. PLoS ONE 11(2), 1–17 (2016).
Laliberte, E. & Legendre, P. A distance-based framework for measuring functional diversity from multiple traits. Ecology 91(1), 299–305 (2010).
Zuur, A., Ieno, E., Walker, N., Saveliev, A. & Smith, G. Mixed Effect Models and Extensions in Ecology with R (Springer, New York, 2009).
R Development Core Team. R: A language and environment for statistical computing. 3.4.0 ed. (R Foundation for Statistical Computing, Vienna, 2017).
Cailliez, F. The analytical solution of the additive constant problem. Psychometrika 48(2), 305–308 (1983).
Brooks, M. E. et al. glmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modeling. R J. 9(2), 378–400 (2017).
Bolker, B. M. Tools for General Maximum Likelihood Estimation. CRAN.R. R package version 1.0.20 (2017).
Barton, K. MuMIn: Multi-Model Inference. R package version 1421. https://CRAN.R-project.org/package=MuMIn (2018).
Hardin, J. W. & Hilbe, J. M. Generalized Linear Models and Extensions (Stata Press, College Station, 2007).
Bolker BM. Linear and generalized linear mixed models. In Ecological Statistics: Contemporary Theory and Application (eds. Fox, G. A., Negrete-Yankelevich, S., Sosa, V. J.) (Oxford University Press, Oxford, 2015).
Sarkar, D. Lattice:Multivariate Data Visualization with R (Springer, New York, 2008).
Wickham, H. ggplot2: Elegant Graphics for Data Analysis (Springer, New York, 2016).
Wickham H. François R. Henry L. Müller K. dplyr: A Grammar of Data Manipulation. R package version 078. https://CRAN.R-project.org/package=dplyr (2018).
Acknowledgements
We would like to thank Johan Kjorven, Sarah DeGennaro and Alexius Folk for assistance in the field, Sindre Ligaard for identifying the beetles and Matthew Cooper for making the illustrations. We would also like to thank the landowners for allowing permission to access their land, without their support this research would not have been possible.
Author information
Authors and Affiliations
Contributions
R.W., T.B. and A.S. conceived the ideas and designed methodology. R.W. collected and analyzed the data and led the writing of the manuscript. All authors contributed critically to the drafts and gave final approval for publication.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wetherbee, R., Birkemoe, T. & Sverdrup-Thygeson, A. Veteran trees are a source of natural enemies. Sci Rep 10, 18485 (2020). https://doi.org/10.1038/s41598-020-75723-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-75723-0
This article is cited by
-
Role of shade trees in conserving beneficial arthropods of biocontrol importance in tea ecosystem
Agroforestry Systems (2024)
-
A novel morphological phenotype does not ensure reduced biotic resistance on an oceanic island
Biological Invasions (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.