Abstract
This systematic review and meta-analysis aimed to evaluate the impact of COVID-19 on pregnant women. We searched for qualified studies in PubMed, Embase, and Web of Science. The clinical characteristics of pregnant women with COVID-19 and their infants were reported as means and proportions with 95% confidence interval. Eleven studies involving with 9032 pregnant women with COVID-19 and 338 infants were included in the meta-analysis. Pregnant women with COVID-19 have relatively mild symptoms. However, abnormal proportions of laboratory parameters were similar or even increased, compared to general population. Around 30% of pregnant women with COVID-19 experienced preterm delivery, whereas the mean birth weight was 2855.9 g. Fetal death and detection of SARS-CoV-2 were observed in about 2%, whereas neonatal death was found to be 0.4%. In conclusion, the current review will serve as an ideal basis for future considerations in the treatment and management of COVID-19 in pregnant women.
Similar content being viewed by others
Introduction
The recent outbreak of the coronavirus disease 2019 (COVID-19) pandemic has called for a prompt response from the scientific community. As of July 30, 2020, confirmed infections have amounted up to over 17 million cases, with casualties reaching an alarming number of over 667,0001. The fact that the disease is actively spreading in the world implies that we must prepare for the worst; as a consequence, labs worldwide have pooled their efforts to identify possible therapeutic methods, estimate future progression trends of the pandemic, and sort out the most vulnerable from existing data in order to prepare patient-specific measures.
In this context, many research papers have shed light on the varying effect of COVID-19 depending on patient characteristics, including age and smoking2,3,4,5,6,7,8. Another important population that deserves meticulous consideration during the COVID-19 pandemic is the pregnant. Pregnancy is a state that is particularly susceptible to infectious diseases, and it is unsurprising that viral infections may affect pregnancy outcomes; previous literature has revealed that viral respiratory illnesses may lead to a higher risk of obstetric complications and adverse perinatal outcomes9,10, primarily due to changes in the immune response11. Furthermore, previous literature on the impact of the 2009 H1N1 influenza virus or the Zika virus suggest that infectious diseases may increase complications and even exhibit fatal effects on pregnant women12,13. Such acknowledgement has led to vigorous investigation from many research groups on the impact of COVID-19 in pregnant patients14,15,16; yet the need to accumulate, organize and analyze such data is evident, given the urgency of the situation.
To that end, this study aims to systematically review previous literature on the impact of COVID-19 on pregnant women. Several important issues, such as perinatal outcome or vertical transmission, are additionally raised in the prognosis of COVID-19 in the pregnant population. Based on ample pre-existing evidence, the current paper attempts to unravel meaningful factors that may aid medical personnel in dealing with such issues and to discover symptoms or phenomena that are specific to the pregnant population.
Results
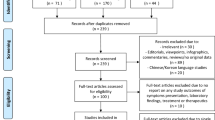
A total of 3765 records were identified from searches of three databases, and 1720 duplicates were excluded. After removing 1865 studies during title and abstract screening, 180 were selected for full-text review. Thereafter, 169 articles were excluded for the following reasons: reviews and expert opinions (n = 55); case reports with less than 10 cases (n = 52); irrelevant studies (n = 17); studies without confirmation of COVID-19 by polymerase chain reaction (PCR) tests (n = 9); studies only including the infants born to mothers without confirmed COVID-19 (n = 7); and overlapping studies (n = 29). Hence, eleven studies on 9032 pregnant women with COVID-19 and 338 neonates were ultimately included for meta-analysis17,18,19,20,21,22,23,24,25,26,27.
The main characteristics of the included studies are listed in Table 1. All studies were conducted in China. The median age and gestational age at admission of the study population ranged from 28 to 34 years and from 31.3 to 37.3 weeks, respectively. The Cesarean delivery rate varied across studies from 18.2 to 100%. The quality score ranged from 8 to 10.
The meta-analysis results of symptoms of pregnant patients with COVID-19 are shown in Table 2. Among the pregnant patients infected by severe acute respiratory coronavirus 2 (SARS-CoV-2), fatigue was the most prevalent symptoms (54.5%), followed by cough (50.1%) and fever (27.6%). Other common symptoms such as dyspnea, myalgia, and sore throat were observed in about 21%, 16%, and 11% of pregnant women with COVID-19, respectively. The prevalence of diarrhea was less than 10%. In terms of laboratory findings, approximately 48%, 43% and 36% of infected pregnant women had elevated CRP, lymphopenia, and leukocytosis, respectively. The results presented in Table 3 show maternal baseline comorbidities. The prevalence of hypertension (including pregnancy-induced hypertension) and diabetes (including gestational diabetes) was 3.7 and 4.2%, respectively, whereas 4.7% of pregnant women with COVID-19 had asthma.
The pregnancy and perinatal outcomes of pregnant patients who were infected by SARS-CoV-2 are presented in Table 4. Around 30% of pregnant women with COVID-19 experienced preterm delivery, whereas premature rupture of membranes and fetal distress were observed in about 2%. The mean birth weight was 2855.9 g (95% CI 2634.9–3076.9 g) and the prevalence of small-for-gestational-age births was estimated as 17.4% (95% CI 0–56.0%). Mean Apgar scores at 1 min and 5 min were 8.8 (95% CI 8.6–9.0) and 9.2 (95% CI 8.3–10.1), respectively. Fetal death was observed in about 2%, whereas neonatal death was found to be 0.4%.
In the present study, detection of SARS-CoV-2 was observed in about 2% of the population; a total of five newborns were reported as SARS-CoV-2 positive. Among them, three newborns with vaginal delivery received swab specimen tests on the first day after birth, and one newborn with cesarean delivery was tested on the seventh day. While four SARS-CoV-2 positive newborns were roomed-in and breastfed, data on one neonate was unavailable.
Discussion
The first notable finding of this study is the difference in common COVID-19 symptoms between pregnant patients and non-pregnant patients. Well-known symptoms of COVID-19 include fever, cough, and dyspnea; in a previous study on non-pregnant COVID-19 patients, the proportion of those who show each symptom was shown to be 83%, 82%, and 31%, respectively28. In our study of pregnant women, the proportions decreased to 28%, 51%, and 21%, indicating relatively mild symptoms. This result was in line with another previous study by Liu et al. that compared pregnant and non-pregnant COVID-19 patients, where more pregnant patients were classified as mild or common29.
Milder symptoms in pregnant COVID-19 patients may be explained by the younger average age compared to the general COVID-19 patient population; additionally, as there was much fewer comorbidities, symptoms might have appeared to be less profound in the pregnant population. In fact, chronic diseases such as hypertension and diabetes were less observed in our study than in previous studies not restricted to pregnant women; the prevalence of hypertension, diabetes and chronic kidney disease were found to be 3.7%, 4.2% and 0.2% in our study, whereas those in a study with 5700 patients with COVID-19 were 56.6%, 33.8% and 5.0%, respectively30.
Unlike common symptoms, various abnormalities of laboratory parameters showed a similar or even increased trend in pregnant women with COVID-19 compared to general patients. A previous meta-analysis using patients with COVID-19 reported that proportions of leukocytosis, lymphopenia, and elevated CRP levels were 17%, 43%, and 58%, respectively31, while those in our study were 36%, 43%, and 48%. This gap between the pregnant and general patient population was probably attributable to changes in the immune response in pregnancy11.
Pregnancy is regarded as an immunocompromised status in some aspects, especially since maternal immunity is altered to tolerate fetal antigens by suppressing cell-mediated immunity32. However, the number of immune cells, such as macrophages, natural killer (NK) cells and regulatory T cells, can increase even in normal pregnancy33. Accordingly, pregnancy yields a unique immunity status32 and may experience increased susceptibility to certain intracellular pathogens, including bacteria and viruses34. Furthermore, it has been reported that the number of leukocytes (mostly accounting for neutrophils) increased early in gestation and remained elevated, which can be explained by the increased corticosteroid and estrogen levels35.
Of utmost importance is the effect of COVID-19 infection in pregnant women; in this regard, pregnancy outcomes were observed in the current study. In total, 29% of the study sample exhibited preterm delivery, a strikingly high number compared to the norm, which was reported to be between 5 and 18%36. In previous research on pregnant patients in past coronaviridae outbreaks, namely severe acute respiratory syndrome (SARS) and Middle East respiratory syndrome (MERS), the proportions of pregnant patients that experienced preterm delivery were 29% and 32%, respectively, which were approximately similar to the ratio in pregnant COVID-19 patients37.
In addition, data on neonates born from COVID-19 patients showed varying tendencies compared to the non-infected. Average Apgar scores at 1 min and 5 min were recorded to be an adequate 8.8 and 9.2, respectively, while average body weight was 2855.9, which is considered normal. On the other hand, proportions of fetal distress was 15.1%, displaying dissimilarity to those of non-inflicted cases, which were 6.8%38,39. While the rate of fetal death in China has been reported to be 0.43%, the present study population exhibited a rate of 2.4%, showing a significant gap as well40. Finally, there was one case (0.4%) of neonatal death in our study, which indicated that the presence of COVID-19 in the mother did not seem to result in a higher probability of neonatal death.
Vertical transmission is another crucial issue, primarily as newborns possess an underdeveloped protective system against external sources of potential harm. Yet controversy had existed regarding whether SARS-CoV-2 can be transmitted from the mother to the fetus within the uterus. Research on previous coronavirus outbreaks fail to provide definite evidence for or against vertical transmission in pregnant patients; on the other hand, in other respiratory viruses such as influenza or respiratory syncytial virus (RSV), cases of vertical transmission have been reported41,42. In the present study, a total of five newborns were reported as SARS-CoV-2 positive, suggesting that vertical transmission of COVID-19 may not be negligible.
In addition, a case report showed that virus-specific antibodies (IgG in 5 and IgM in 2 among 6 neonates) were detected in serum samples of the neonates born to pregnant COVID-19 patients, although SARS-CoV-2 itself was undetected by PCR tests43. Detection of IgM was of interest because unlike IgG, which can be transferred passively across the placenta from the mother to fetus44, IgM is known to be too large to cross the placenta45 and might be produced by the fetus itself; accordingly, positive IgM in neonates might indicate past exposure, implying the possibility of vertical transmission of SARS-CoV-2. However, in a study of Ben-Hur et al., transplacental passage of IgM was detected in cases of severe inflammation46. Detection of IgM in neonates should be interpreted with caution regarding whether the IgM in neonates was produced by fetus after vertical transmission or transferred from the mother.
The present meta-analysis bears a few limitations that should be considered in the interpretation of results. Detailed clinical manifestations were not available in the selected studies. In addition, the associations between clinical features and outcomes were not provided. Nevertheless, as a systematic review and meta-analysis on pregnant COVID-19 patients, the current review will serve as an ideal basis for future considerations in the treatment and management of COVID-19 in pregnant women.
Methods
Literature search strategy
Two researchers separately searched PubMed, Embase, and Web of Science for studies on clinical characteristics of pregnant women with COVID-19 and their neonates, published between 1 January 2020 and 20 July 2020. The following search terms were used [(Pregnan * OR gestation * OR maternal OR fetal OR perinatal OR obstetric * OR neonate * OR infant * OR newborn*)] AND [(“coronavirus disease 2019” OR “coronavirus disease-19” OR “COVID-19” OR “2019-nCoV” OR “SARS-CoV-2” OR “novel coronavirus”)]. There was no restriction on language of publication. Duplicates and obviously irrelevant studies were excluded through initial screening of titles and abstracts, and the remaining articles were further reviewed according to inclusion and exclusion criteria. A flow diagram summarizing the study selection process is shown in Fig. 1.
Inclusion and exclusion criteria
The following criteria were used to identify eligible studies: (1) describing the clinical characteristics of pregnant women with COVID-19 and their neonates; (2) in which COVID-19 diagnosis was confirmed by PCR tests; and (3) using an observational study design. Exclusion criteria were: (1) reviews and expert opinions; (2) in vitro or animal studies; (3) studies on infants born to mothers without confirmed COVID-19 infection; or (4) case series or case report with less than 10 cases. In instances of overlapping data, only the most recent and comprehensive data were included in the meta-analysis.
Study selection, data extraction and quality assessment
Two investigators separately selected publications and extracted data, and discrepancies were resolved by consensus. The following information was extracted from each study: name of the first author, publication year, study setting, study design, patient age, gestational age at admission, percentage of Caesarean section, and diagnosis criteria of COVID-19. Also, prevalence of clinical symptoms, comorbidities, and maternal and fetal outcomes were extracted, along with mean and standard deviation of birth weight and Apgar scores. In the case of outcomes with lack of data, we utilized and additionally analyzed studies that had been excluded due to overlap; for example, the meta-analysis of leukocytosis was performed using additional studies47,48,49,50,51, as the included studies did not have data for leukocytosis. The methodologic quality of the selected studies was evaluated using the Joanna Briggs Institute critical appraisal checklist for case series by two researchers, independently52. This checklist contains 10 questions for assessing the risk of bias in the design and conduct of the study, and the higher the scores, the higher the study quality.
Statistical analysis
The clinical characteristics of pregnant women with COVID-19 and their neonates were reported as means and proportions with 95% confidence interval (CI). Statistical heterogeneity was assessed using I2 statistics. To avoid bias from studies with a zero-event rate, proportions were transformed via the Freeman–Tukey Double ArcSine method. A random-effects model (DerSimonian-Laird method) was applied to consider the heterogeneity within and between studies and to give a more conservative estimate of statistical confidence. All statistical analyses were performed using R software (version 3.6.0) with meta package53. The review was written based on Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines.
References
Johns Hopkins University & Medicine. Coronavirus COVID-19 Global Cases by the Center for Systems Science and Engineering at Johns Hopkins University. (accessed 30 July 2020); https://coronavirus.jhu.edu/map.html.
Rosenthal, D. M., Ucci, M., Heys, M., Hayward, A. & Lakhanpaul, M. Impacts of COVID-19 on vulnerable children in temporary accommodation in the UK. Lancet Public Health 5, e241–e242 (2020).
Molloy, E. J. & Bearer, C. F. COVID-19 in children and altered inflammatory responses. Pediatr Res. 88, 340–341 (2020).
Yuan, Y., Wang, N. & Ou, X. Caution should be exercised for the detection of SARS-CoV-2, especially in the elderly. J. Med. Virol. (in press).
Gardner, W., States, D. & Bagley, N. The coronavirus and the risks to the elderly in long-term care. J. Aging Soc. Policy. 2020, 1–6 (2020).
Berlin, I., Thomas, D., Le Faou, A. L. & Cornuz, J. COVID-19 and smoking. Nicotine Tob. Res. 22, 1650–1652 (2020).
Vardavas, C. I. & Nikitara, K. COVID-19 and smoking: a systematic review of the evidence. Tob. Induc. Dis. 18, 20 (2020).
Brake, S. J. et al. Smoking upregulates angiotensin-converting enzyme-2 receptor: a potential adhesion site for novel coronavirus SARS-CoV-2 (Covid-19). J. Clin. Med. 2020, 9 (2020).
Chen, Y. H. et al. Pneumonia and pregnancy outcomes: a nationwide population-based study. Am. J. Obstet. Gynecol. 207, 288.e1–7 (2012).
Wong, S. F. et al. Pregnancy and perinatal outcomes of women with severe acute respiratory syndrome. Am. J. Obstet. Gynecol. 191, 292–297 (2004).
Mehta, N., Chen, K., Hardy, E. & Powrie, R. Respiratory disease in pregnancy. Best Pract. Res. Clin. Obstet. 29, 598–611 (2015).
Siston, A. M. et al. Pandemic 2009 influenza A (H1N1) virus illness among pregnant women in the United States. JAMA 303, 1517–1525 (2010).
Moore, C. A. et al. Characterizing the pattern of anomalies in congenital Zika syndrome for pediatric clinicians. JAMA Pediatr. 171, 288–295 (2017).
Liu, H. et al. Why are pregnant women susceptible to COVID-19? An immunological viewpoint. J. Reprod. Immunol. 139, 103122 (2020).
Karimi-zarchi, M. et al. Vertical transmission of Coronavirus Disease 19 (COVID-19) from infected pregnant mothers to neonates: a review. Fetal Pediatr. Pathol. 2020, 1–5 (2020).
Chen, S. et al. Clinical analysis of pregnant women with 2019 novel coronavirus pneumonia. J. Med. Virol. (in press).
Dória, M., Peixinho, C., Laranjo, M., Mesquita Varejão, A. & Silva, P. T. Covid-19 during pregnancy: a case series from an universally tested population from the north of Portugal. Eur. J. Obstet. Gynecol. Reprod. Biol. 250, 261–262 (2020).
Ellington, S. et al. Characteristics of women of reproductive age with laboratory-confirmed SARS-CoV-2 infection by pregnancy status—United States, January 22–June 7, 2020. MMWR Morb. Mortal Wkly. Rep. 69, 769–775 (2020).
Liu, Y., Chen, H., Tang, K. & Guo, Y. Clinical manifestations and outcome of SARS-CoV-2 infection during pregnancy. J. Infect. (in press).
Lumbreras-Marquez, M. I., Campos-Zamora, M., Lizaola-Diaz de Leon, H. & Farber, M. K. Maternal mortality from COVID-19 in Mexico. Int. J Gynaecol Obstet. 150, 266–267 (2020).
Mohr-Sasson, A. et al. Laboratory characteristics of pregnant compared to non-pregnant women infected with SARS-CoV-2. Arch. Gynecol. Obstet. 302, 629–634 (2020).
Nayak, A. H. et al. Impact of the coronavirus infection in pregnancy: a preliminary study of 141 patients. J. Obstet. Gynaecol. India 70, 256–261 (2020).
Pereira, A. et al. Clinical course of coronavirus disease-2019 in pregnancy. Acta Obstet. Gynecol. Scand. 99, 839–847 (2020).
Savasi, V. M. et al. Clinical findings and disease severity in hospitalized pregnant women with coronavirus disease 2019 (COVID-19). Obstet. Gynecol. 136, 252–258 (2020).
Sentilhes, L. et al. COVID-19 in pregnancy was associated with maternal morbidity and preterm birth. Am. J. Obstet. Gynecol. (in press).
Vivanti, A. J. et al. Retrospective description of pregnant women infected with severe acute respiratory syndrome coronavirus 2, France. Emerg. Infect. Dis. 26, 2069–2076 (2020).
Yan, J. et al. Coronavirus disease 2019 in pregnant women: a report based on 116 cases. Am. J. Obstet. Gynecol. 223, 111.e1-111.e14 (2020).
Chen, N. et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 395, 507–513 (2020).
Liu, H. et al. Clinical and CT imaging features of the COVID-19 pneumonia: focus on pregnant women and children. J. Infect. 80, e7–e13 (2020).
Richardson, S. et al. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York city area. JAMA 323, 2052–2059 (2020).
Rodriguez-Morales, A. J. et al. Clinical, laboratory and imaging features of COVID-19: a systematic review and meta-analysis. Travel Med. Infect. Dis. 34, 101623 (2020).
Mor, G. & Cardenas, I. The immune system in pregnancy: a unique complexity. Am. J. Reprod. Immunol. 63, 425–433 (2020).
Mor, G., Cardenas, I., Abrahams, V. & Guller, S. Inflammation and pregnancy: the role of the immune system at the implantation site. Ann. N. Y. Acad. Sci. 1221, 80–87 (2011).
Jamieson, D. J., Theiler, R. N. & Rasmussen, S. A. Emerging infections and pregnancy. Emerg. Infect. Dis. 12, 1638–1643 (2020).
Pitkin, R. M. & Witte, D. L. Platelet and leukocyte counts in pregnancy. JAMA 242, 2696–2698 (1979).
WHO. Preterm Birth. (accessed 5 April 2020); https://www.who.int/news-room/fact-sheets/detail/preterm-birth.
Mascio, D. et al. Outcome of Coronavirus spectrum infections (SARS, MERS, COVID 1-19) during pregnancy: a systematic review and meta-analysis. Am. J. Obstet. Gynecol. MFM. 2, 100107 (2020).
Hou, L. et al. Cross sectional study in China: fetal gender has adverse perinatal outcomes in mainland China. BMC Pregnancy Childbirth. 14, 372 (2014).
Gillam-Krakauer, M. & Gowen Jr., C. W. Birth Asphyxia. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing, 2020. (accessed 5 April 2020); https://www.ncbi.nlm.nih.gov/books/NBK430782/.
UNICEF. Neonatal Mortality. (accessed 5 April 2020); https://data.unicef.org/topic/child-survival/neonatal-mortality.
Takahashi, N., Kitajima, H., Kusuda, S., Morioka, I. & Itabashi, K. Pandemic (H1N1) 2009 in neonates, Japan. Emerg. Infect. Dis. 17, 1763–1765 (2011).
Manti, S. et al. Vertical transmission of respiratory syncytial virus infection in humans. Pediatr. Pulmonol. 52, E81-84 (2017).
Zeng, H. et al. Antibodies in infants born to mothers with COVID-19 pneumonia. JAMA. 323, 1848–1849 (2020).
Kohler, P. F. & Farr, R. S. Elevation of cord over maternal IgG immunoglobulin: evidence for an active placental IgG transport. Nature 210, 1070–1071 (1966).
Burrell, C. J., Howard, C. R. & Murphy, F. A. Fenner and White’s Medical Virology 65–76 (Academic Press, New York, 2017).
Ben-Hur, H. et al. Transport of maternal immunoglobulins through the human placental barrier in normal pregnancy and during inflammation. Int. J. Mol. Med. 16, 401–407 (2005).
Chen, R. et al. Safety and efficacy of different anesthetic regimens for parturients with COVID-19 undergoing Cesarean delivery: a case series of 17 patients. Can. J. Anaesth. 67, 655–663 (2020).
Ferrazzi, E. et al. Vaginal delivery in SARS-CoV-2 infected pregnant women in Northern Italy: a retrospective analysis. BJOG. 127, 1116–1121 (2020).
Gong, X. et al. CT characteristics and diagnostic value of COVID-19 in pregnancy. PLoS ONE 15, e0235134 (2020).
Liu, H. H. et al. Clinical and CT imaging features of the COVID-19 pneumonia: focus on pregnant women and children. J. Infect. 80, E7–E13 (2020).
Qiancheng, X. et al. Coronavirus disease 2019 in pregnancy. Int. J. Infect Dis. 95, 376–383 (2020).
Joanna Briggs Institute. JBI Reviewer’s Manual. (accessed 5 April 2020); https://wiki.joannabriggs.org/display/MANUAL/Appendix+7.3+Critical+appraisal+checklists+for+case+series.
R Core Team. R: a language and environment for statistical computing. R Foundation for Statistical Computing. https://www.R-project.org (2020).
Acknowledgements
None.
Funding
We did not receive any funding.
Author information
Authors and Affiliations
Contributions
All the authors have made substantial contributions to the conception of the study. J.Y., W.K., K.E.L. and H.S.G. contributed to designing the study. J.Y., W.K. and K.E.L. contributed to acquisition and analysis of data. J.Y., W.K., J.M.H., N.L., H.Y.Y., and H.S.G. contributed to interpretation of data. J.Y., W.K., J.M.H., H.Y.Y. and N.L. contributed to drafting of the manuscript. K.E.L. and H.S.G. contributed to critical revision of the manuscript. All authors approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yee, J., Kim, W., Han, J.M. et al. Clinical manifestations and perinatal outcomes of pregnant women with COVID-19: a systematic review and meta-analysis. Sci Rep 10, 18126 (2020). https://doi.org/10.1038/s41598-020-75096-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-75096-4
This article is cited by
-
Neonatal outcomes of maternal prenatal coronavirus infection
Pediatric Research (2024)
-
Pregnancy in the time of COVID-19: towards Fetal monitoring 4.0
BMC Pregnancy and Childbirth (2023)
-
Tracking excess of maternal deaths associated with COVID-19 in Brazil: a nationwide analysis
BMC Pregnancy and Childbirth (2023)
-
Timing and severity of COVID-19 during pregnancy and risk of preterm birth in the International Registry of Coronavirus Exposure in Pregnancy
BMC Pregnancy and Childbirth (2022)
-
Infants infected with SARS-CoV-2 and newborns born to mother diagnosed with COVID-19: clinical experience
Irish Journal of Medical Science (1971 -) (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.