Abstract
We aim to study the association of hyperlipidemia and statin use with COVID-19 severity. We analysed a retrospective cohort of 717 patients admitted to a tertiary centre in Singapore for COVID-19 infection. Clinical outcomes of interest were oxygen saturation ≤ 94% requiring supplemental oxygen, intensive-care unit (ICU) admission, invasive mechanical-ventilation and death. Patients on long term dyslipidaemia medications (statins, fibrates or ezetimibe) were considered to have dyslipidaemia. Logistic regression models were used to study the association between dyslipidaemia and clinical outcomes adjusted for age, gender and ethnicity. Statin treatment effect was determined, in a nested case–control design, through logistic treatment models with 1:3 propensity matching for age, gender and ethnicity. All statistical tests were two-sided, and statistical significance was taken as p < 0.05. One hundred fifty-six (21.8%) patients had dyslipidaemia and 97% of these were on statins. Logistic treatment models showed a lower chance of ICU admission for statin users when compared to non-statin users (ATET: Coeff (risk difference): − 0.12 (− 0.23, − 0.01); p = 0.028). There were no other significant differences in other outcomes. Statin use was independently associated with lower ICU admission. This supports current practice to continue prescription of statins in COVID-19 patients.
Similar content being viewed by others
Introduction
The COVID-19 pandemic continues to grow around the world, with more than 20 million cases worldwide1. The coronavirus infections, COVID-19, SARS and MERS, are all associated with dysregulated immune and inflammatory processes. Severe cases of COVID-19 are characterised by high circulating pro-inflammatory cytokines concentrations, as well as high neutrophil counts and lymphopenia2,3,4. COVID-19 has been associated with hyperinflammatory states, cardiovascular disease and venous thromboembolism5,6,7.
Inflammation has a potential role in the pathogenesis of dyslipidaemia8. Statins are commonly used to treat hyperlipidemia and its pleiotropic effects have been shown to reduce cytokines in various non-infective conditions9,10. Long term statin therapy correlates with better outcome in the setting of bacterial pneumonia11,12 and influenza13. A randomised controlled trial evaluating atorvastatin as a treatment for influenza showed significantly lower levels of inflammatory cytokines with treatment [NCT02056340].
Medical comorbidities such as diabetes, hypertension and cardiovascular diseases have been identified as risk factors for severe COVID-19 in numerous large case series from China, Italy and the United States14,15,16. Dyslipidaemia has not been identified as an independent risk factor17, although it is associated with diabetes and hypertension, and contributes to cardiovascular diseases. We aimed to study the association of dyslipidaemia with COVID-19 associated inflammation and the correlation between long term statin therapy and disease severity.
Methods
We carried out a retrospective cohort study of patients with confirmed COVID-19 hospitalized at the National Centre of Infectious Diseases (NCID), Singapore. NCID is responsible for managing more than 60% of COVID-19 patients admitted to hospital in Singapore. Patients were identified by primary care and emergency doctors based on case definitions informed by evolving epidemiological risk factors, case detection from active contact tracing, enhanced pneumonia surveillance and diagnostic testing based on doctors’ discretion18. Patients were included if they were hospitalized from 22 January 2020 to 15 April 2020. This cohort was previously studied for associations with diabetes and hypertension pharmacotherapy19 but this analysis is restricted towards studying the association of dyslipidaemia with COVID-19 associated inflammation and the effects of the use of statin with disease severity.
Electronic medical records of hospitalised patients with COVID-19 confirmed by PCR performed on respiratory samples were reviewed to extract information on demographic data on age, gender and ethnicity, presence of comorbidities and concomitant medications, laboratory investigations including full blood count, renal and liver function tests, C-reactive protein (CRP) and lactate dehydrogenase (LDH) and clinical outcomes of COVID-19. Clinical outcomes of interest were hypoxia with oxygen saturation ≤ 94% requiring supplemental oxygen, intensive care unit (ICU) admission and invasive mechanical ventilation (IMV) and death. All study procedures and data collections were performed in accordance with institutional guidelines. The study protocol was reviewed and approved by the Singapore, Ministry of health who provided a waiver of informed consent from study participants for data collection under the Infectious Disease Act as part of national public health research.
Continuous and categorical variables are presented as median (interquartile range) and frequency (%), respectively. We used linear regression models to assess the association between each of the complete blood count variables and inflammatory markers with dyslipidaemia status adjusting for age, gender and ethnicity. To assess the possible treatment effect of statin use on these outcomes we used a nested case–control design, wherein after excluding patients with diabetes and hypertension, we estimated the statin treatment effect through logistic treatment models with 1:3 propensity matching for age, gender and ethnicity. All statistical tests were two-sided, and statistical significance was taken as p < 0.05. All statistical analyses were performed using Stata version 15.
Results
Within our cohort of 717 patients, one hundred fifty-six (21.8%) patients had dyslipidaemia. Individuals with dyslipidaemia were older (62.5 years, IQR 55–68 years versus 37 years, IQR 27–52 years) and more likely to be of Malay ethnicity (18.6% versus 8.9%). Approximately 24–59% of patients had coexisting diabetes, hypertension and atherosclerotic cardiovascular disease defined as history of ischemic heart disease, stroke or peripheral vascular disease. In terms of inflammatory markers, those with dyslipidaemia were more likely to have higher CRP, LDH, procalcitonin, white cell count and neutrophil count but lower lymphocyte count. Patients with dyslipidaemia were more likely to require supplemental oxygen, ICU admission and IMV. The risk of death was higher (p < 0.05). See Tables 1 and 2.
Dyslipidaemia was associated with higher white cell count and neutrophil count but not the other inflammatory markers. See Tables 1 and 2.
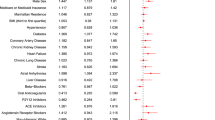
Of those who had hyperlipidemia, 151 (96.7%) were on statins, 12 (7.7%) were on fibrates and 10 (6.4%) were on ezetimibe. In the nested case–control analysis after excluding patients with diabetes and hypertension, 40 patients were on statins and 509 were non-statin users. The baseline characteristics of these 40 patients is described in Table 3. Approximately 22.5% (9/40) patients had baseline atherosclerotic cardiovascular disease. None of the patients had renal disease or other comorbid diseases. Logistic treatment models using propensity matching showed a lower chance of ICU admission for statin users when compared to non-statin users (Average treatment effect of statins (ATET) Coeff (risk difference): − 0.12 (− 0.23, − 0.01); p = 0.028). There were no other significant differences in other outcomes (Table 4).
Discussion
We found that statins was associated with better outcomes in COVID-19. Similar results have been reported from a large study from Hubei province, China wherein they found that statin use was associated with a lower risk of mortality in COVID-19 infections20. In another retrospective study, atorvastatin also associated with a lower risk of death in COVID-19 patients admitted to the intensive care unit21. In a study involving nursing home residents, statin use was associated with higher chances of asymptomatic infection22.
Lipids and cholesterol-rich membrane microdomains facilitates the interaction between the surface glycoprotein S of SARS-CoV and the cellular receptor angiotensin-converting enzyme 2 (ACE2)23. Cholesterol has been implicated to have a possible role in the increased risk of infection in the elderly patients wherein higher tissue cholesterol has been shown to increase the endocytic entry of SARS-CoV-2 along with increased trafficking of angiotensin converting enzyme-2 (ACE-2) in a preprint24. After cellular entry, RNA viruses require intracellular cholesterol and fatty acids for further replication. For e.g. it has been demonstrated that during the initial phase of dengue virus infection, there is an increase in intracellular cholesterol concentration. This is associated with an increase in low density lipoprotein (LDL) concentrations in cells and a concomitant increase in the enzymatic activity of 3-hydroxy-3-methyl-glutaryl-CoA (HMG-CoA) reductase inside the cells25,26. Three decades ago, Mabuchi et al., reported that statins can effectively reduce LDL concentrations through HMG-CoA reductase inhibition27 and in the last three decades statins have become the most widely prescribed lipid lowering medication. In COVID-19, statins may help to reduce viral entry and viral transmission by inhibition of the HMG-CoA reductase in the cells which will make less cholesterol available inside cells and tissues.
In our small observational cohort, we observed a significant trend towards higher white cell counts and neutrophil counts in patients with dyslipidaemia. A key pathological process that leads to cardiovascular disease is inflammation. Statins have been shown to have significant pleiotropic, anti-inflammatory and immunomodulatory effects28,29,30,31,32,33,34,35,36, independent of its ability to reduce low-density lipoprotein36. Even in rheumatological disease statins are known to modulate the inflammatory response37. Additional to its beneficial effects in cardiovascular disease, statins may be beneficial in patients with bacterial sepsis38,39, community acquired pneumonia40 and influenza13. Severe outcomes in COVID-19 is associated with higher markers of inflammation and a “cytokine storm”41,42,43. Statins have the potential to block the molecular mechanisms, including NF-κB and NLRP3 inflammasomes and TLR signalling which are responsible for the "cytokine storm" in severe COVID-19 patients44,45,46.
COVID-19 has been associated with significant cardiovascular complications due to direct effects of SARs-CoV-2 virus with significant effects of the virus on the expression and function of ACE-2 in the vasculature and evidence of coronary endothelial dysfunction and endothelialitis seen in multiple vascular beds in fatal patients with COVID-1947,48. Cases of acute coronary events (acute myocardial infarction and thromboembolism) triggered in patients with no underlying history of ischemic heart disease have been reported worldwide49,50,51. Statins are known to be effective in the prevention of endothelial dysfunction and downstream, atherosclerotic pathways and to prevent coronary artery disease52. It has been suggested that dyslipidaemia patients maybe at higher risk of atherosclerotic events after recovery from COVID-19. Similar exacerbations have been reported in influenza infections. It has been suggested that statins should be intensified to reduce cardiovascular risk post COVID-19 infection53,54.
Hence, we suggest that statins should be continued in dyslipidaemia patients who develop COVID-19 infection . This is especially important in COVID-19 as severe disease is related to cardiovascular comorbidities during infection and increased cardiovascular risk post recovery.
Our study has several limitations. We did not have the quantitative lipid profile on admission to study detailed correlations with the lipid phenotype. We did not have the values of the previous lipid profile including maximum LDL-Cholesterol, so we could not study correlations of cholesterol load with COVID-19 severity. Our cohort was small and adverse outcome rates, were low. This will affect the generalisability of our findings. Lastly, there was a risk of channelling bias as patients on statins were more likely to have more severe cardiovascular disease than those without.
Conclusion
We found dyslipidaemia patients had a significant trend towards a higher innate immune response shown by higher white cell counts and neutrophil counts. Statin use was independently associated with lower requirement for ICU admission. This supports current practice to continue prescription of statins in hyperlipidemia and other metabolic disorders in COVID-19 patients. The effect of statin use and intensification on COVID-19 disease severity and atherosclerotic events after recovery needs to be studied in larger observational studies or ideally in randomised controlled trials.
Data availability
The datasets are available from the corresponding author upon reasonable request.
References
Situation Reports [Internet]. Situation Reports. 2020 [cited 2020May12]. Available from: https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200511-covid-19-sitrep-112.pdf?sfvrsn=813f2669_2.
Liu, J. et al. Longitudinal characteristics of lymphocyte responses and cytokine profiles in the peripheral blood of SARS-CoV-2 infected patients. EBioMedicine 55, 102763 (2020).
Konig, M. F. et al. Preventing cytokine storm syndrome in COVID-19 using α-1 adrenergic receptor antagonists. J. Clin. Invest. 130, 139642 (2020).
Chen, X. et al. Detectable serum severe acute respiratory syndrome coronavirus 2 viral load (RNAemia) is closely correlated with drastically elevated interleukin 6 level in critically ill patients with coronavirus disease. Infect. Dis. Clin. https://doi.org/10.1093/cid/ciaa449 (2019).
Riphagen, S., Gomez, X., Gonzalez-Martinez, C., Wilkinson, N. & Theocharis, P. Hyperinflammatory shock in children during COVID-19 pandemic. Lancet 395, 1607–1608 (2020).
Clerkin, K. J. et al. COVID-19 and cardiovascular disease. Circulation 141, 1648–1655 (2020).
Artifoni, M. et al. Systematic assessment of venous thromboembolism in COVID-19 patients receiving thromboprophylaxis: incidence and role of D-dimer as predictive factors. J. Thromb. Thrombolysis. 50, 211 (2020).
Schonbeck, U. & Libby, P. Inflammation, immunity, and HMG-CoA reductase inhibitors: statins as antiinflammatory agents?. Circulation 109, 18–26 (2004).
Wassmann, S. et al. Rapid effect of 3-hydroxy-3-methylglutaryl coenzyme A reductase inhibition on coronary endothelial function. Circ. Res. 93, e98-103 (2003).
Fang, C. H., Li, J. J. & Hui, R. T. Statins, like aspirin, should be given as early as possible in patients with acute coronary syndrome. Med. Hypotheses 64, 192–6 (2005).
Novack, V. et al. The effects of statin therapy on inflammatory cytokines in patients with bacterial infections: a randomized double-blind placebo controlled clinical trial. Intensive Care Med. 17, 1255–1260 (2009).
Mortensen, E. M., Restrepo, M. I., Anzueto, A. & Pugh, J. The effect of prior statin use on 30-day mortality for patients hospitalized with community-acquired pneumonia. Respir. Res. 17, 82 (2005).
Vandermeer, M. L. et al. Association between use of statins and mortality among patients hospitalized with laboratory-confirmed influenza virus infections: a multistate study. J. Infect. Dis. 205, 13–19 (2012).
Guan, W. J. et al. Clinical characteristics of coronavirus disease 2019 in China. New Engl. J. Med. 382, 1708–1720 (2020).
Richardson, S. et al. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City area. JAMA 323, 2052–2059 (2020).
Mancia, G., Rea, F., Ludergnani, M., Apolone, G. & Corrao, G. Renin-angiotensin-aldosterone system blockers and the risk of covid-19. N. Engl. J. Med. 382, 2431–2440 (2020).
Chow, N. et al. Preliminary estimates of the prevalence of selected underlying health conditions among patients with coronavirus disease 2019—United States, February 12–March 28, 2020. MMWR Morb. Mortal Wkly. Rep. 69, 382–386 (2020).
Ng, Y. X. et al. Evaluation of the effectiveness of surveillance and containment measures for the first 100 patients with COVID-19 in Singapore—January 2–February 29, 2020. MMWR Morb. Mortal Wkly. Rep. 69, 307–311 (2020).
Dalan, R. et al. The association of hypertension and diabetes pharmacotherapy with COVID-19 severity and immune signatures: an observational study. Eur. Heart J. Cardiovasc. Pharmacother. 2020, 098. https://doi.org/10.1093/ehjcvp/pvaa098 (2020).
Zhang, X. J. et al. In-hospital use of statins is associated with a reduced risk of mortality among individuals with COVID-19. Cell Metab. 32, 176–187 (2020).
Rodriguez-Nava, G. et al. Atorvastatin associated with decreased hazard for death in COVID-19 patients admitted to an ICU: a retrospective cohort study. Crit. Care 24, 429 (2020).
De Spiegeleer, A. et al. The effects of ARBs, ACEis, and statins on clinical outcomes of COVID-19 infection among nursing home residents. J. Am. Med. Dir. Assoc. 21, 909–914 (2020).
Glende, J. et al. Importance of cholesterol-rich membrane microdomains in the interaction of the S protein of SARS-coronavirus with the cellular receptor angiotensin-converting enzyme 2. Virology 381, 215–221 (2008).
Wang, H., Yuan, Z., Pavel, M. A. & Hansen, S. B. Cholesterol and COVID19 lethality in elderly. bioRxiv https://doi.org/10.1101/2020.05.09.086249v2 (2020).
Soto-Acosta, R. et al. The increase in cholesterol levels at early stages after dengue virus infection correlates with an augment in LDL particle uptake and HMG-CoA reductase activity. Virology 442, 132–147. https://doi.org/10.1016/j.virol.2013.04.003 (2013).
Scicali, R., Di Pino, A., Piro, S. & Rabuazzo, A. M. Purrello F (2020) May statins and PCSK9 inhibitors be protective from COVID-19 in familial hypercholesterolemia subjects?. Nutr. Metab. Cardiovasc. Dis. 30, 1068–1069. https://doi.org/10.1016/j.numecd.2020.05.003 (2020).
Mabuchi, H. et al. Effect of an inhibitor of 3-hydroxy-3-methyglutaryl coenzyme A reductase on serum lipoproteins and ubiquinone-10-levels in patients with familial hypercholesterolemia. N. Engl. J. Med. 305, 478–482 (1981).
Bifulco, M. & Gazzerro, P. Statins in coronavirus outbreak: It’s time for experimental and clinical studies. Pharmacol. Res. 156, 104803 (2020).
Ridker, P. M. et al. Long-term effects of pravastatin on plasma concentration of C-reactive protein. The Cholesterol and Recurrent Events (CARE) Investigators. Circulation 100, 230 (1999).
McCarey, D. W. et al. Trial of atorvastatin in rheumatoid arthritis (TARA): double-blind, randomised placebo-controlled trial. Lancet 363, 2015 (2004).
Albert, M. A., Danielson, E., Rifai, N. & Ridker, P. M. PRINCE Investigators. Effect of statin therapy on C-reactive protein levels: the pravastatin inflammation/CRP evaluation (PRINCE): a randomized trial and cohort study. JAMA 286, 64 (2001).
Zhang, J. et al. Statins directly suppress cytokine production in murine intraepithelial lymphocytes. Cytokine 61, 540–545 (2013).
Iwata, A. et al. Inhibitory effect of statins on inflammatory cytokine production from human bronchial epithelial cells. Clin. Exp. Immunol. 168, 234–240 (2012).
van der Meij, E. et al. A clinical evaluation of statin pleiotropy: statins selectively and dose-dependently reduce vascular inflammation. PLoS ONE 8, e53882 (2013).
Duan, H. Y. et al. Effect of atorvastatin on plasma NT-proBNP and inflammatory cytokine expression in patients with heart failure. Genet. Mol. Res. 14, 15739–15748 (2015).
Zeiser, R. Immune modulatory effects of statins. Immunology 154, 69–75 (2018).
Erkan, D. et al. A prospective open-label pilot study of fluvastatin on proinflammatory and prothrombotic biomarkers in antiphospholipid antibody positive patients. Ann. Rheum Dis. 73, 1176–1180 (2014).
Liappis, A. P., Kan, V. L., Rochester, C. G. & Simon, G. L. The effect of statins on mortality in patients with bacteremia. Clin. Infect. Dis. 33, 1352–1357 (2001).
Ma, Y. et al. Systematic review and meta-analysis on the association between outpatient statins use and infectious disease-related mortality. PLoS ONE 7, e51548 (2012).
Mortensen, E. M., Restrepo, M. I., Anzueto, A. & Pugh, J. The effect of prior statin use on 30-day mortality for patients hospitalized with community-acquired pneumonia. Respir. Res. 6, 82 (2005).
Huang, C. et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 395, 497–506 (2020).
Chen, L. et al. Analysis of clinical features of 29 patients with 2019 novel coronavirus pneumonia. Zhonghua Jie He He Hu Xi Za Zhi. 43, E005 (2020).
Xu, Z. et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir. Med. https://doi.org/10.1016/S2213-2600(20)30076-X (2020).
Rodrigues-Diez, R. R. et al. Statins: could an old friend help in the fight against COVID-19?. Br. J. Pharmacol. https://doi.org/10.1111/bph.15166 (2020).
Castiglione, V., Chiriacò, M., Emdin, M., Taddei, S. & Vergaro, G. Statin therapy in COVID-19 infection. Eur. Heart J. Cardiovasc. Pharmacother. 6, 258–259 (2020).
Dashti-Khavidaki, S. & Khalili, H. Considerations for statin therapy in patients with COVID-19. Pharmacotherapy 40, 484–486 (2020).
Evans, P. C. et al. Endothelial dysfunction in COVID-19: a position paper of the ESC working group for atherosclerosis and vascular biology, and the ESC council of basic cardiovascular science. Cardiovasc. Res. 2020, cvaa230. https://doi.org/10.1093/cvr/cvaa230 (2020).
Ackermann, M. et al. Pulmonary vascular endothelialitis, thrombosis and angiogenesis in COVID-19. N. Engl. J. Med. https://doi.org/10.1056/NEJMoa2015432 (2020).
Li, G. et al. Cardiovascular disease during the COVID-19 pandemic: think ahead, protect hearts, reduce mortality. Cardiol. J. https://doi.org/10.5603/CJ.a2020.0101 (2020).
Tedeschi, D., Rizzi, A., Biscaglia, S. & Tumscitz, C. Acute myocardial infarction and large coronary thrombosis in a patient with COVID-19. Catheter. Cardiovasc. Interv. https://doi.org/10.1002/ccd.29179 (2020).
Shams, A., Ata, F., Mushtaq, K., Munir, W. & Yousaf, Z. Coronary thrombosis in a young male with COVID-19. IDCases 21, 00923. https://doi.org/10.1016/j.idcr.2020.e00923 (2020).
Crunkhorn, S. Statin therapy improves endothelial dysfunction. Nat. Rev. Drug Discov. https://doi.org/10.1038/d41573-020-00137-6 (2020).
Peretz, A., Azrad, M. & Blum, A. Influenza virus and atherosclerosis. QJM 112, 749–755 (2019).
Vuorio, A., Watts, G. F. & Kovanen, P. T. Familial hypercholesterolaemia and COVID-19: triggering of increased sustained cardiovascular risk. J. Intern. Med. 287, 746–747. https://doi.org/10.1111/joim.13070 (2020).
Acknowledgements
The study is funded through NMRC COVID-19 research fund (COVID19RF-001). RD is supported in part by Ministry of Health, Clinician Scientist Award [MOH-000014]; and National Medical Research Council Centre Grant [NMRC/CG/017/2013].
Author information
Authors and Affiliations
Contributions
R.D., W.Y.T.T. conceptualized the study, performed data analysis, data interpretation, literature review and wrote the manuscript. D.E.K.C.: data interpretation and critical review of manuscript. D.C.L. and B.E.Y. conceptualized the study, data interpretation and critical review of manuscript. All authors reviewed the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tan, W.Y.T., Young, B.E., Lye, D.C. et al. Statin use is associated with lower disease severity in COVID-19 infection. Sci Rep 10, 17458 (2020). https://doi.org/10.1038/s41598-020-74492-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-74492-0
This article is cited by
-
Targeting proprotein convertase subtilisin/kexin type 9 (PCSK9): from bench to bedside
Signal Transduction and Targeted Therapy (2024)
-
Cardiopulmonary exercise test combined with red blood cell distribution width to predict cardiovascular complication of thoracic surgery
Scientific Reports (2024)
-
An RNA-immunoprecipitation via CRISPR/dCas13 reveals an interaction between the SARS-CoV-2 5'UTR RNA and the process of human lipid metabolism
Scientific Reports (2023)
-
All-cause mortality in COVID-19 patients receiving statin therapy: analysis of veterans affairs database cohort study
Internal and Emergency Medicine (2022)
-
Identifying FDA-approved drugs with multimodal properties against COVID-19 using a data-driven approach and a lung organoid model of SARS-CoV-2 entry
Molecular Medicine (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.