Abstract
Although evidence based guidelines recommend optimal use of beta blockers in all patients with chronic heart failure unless contraindicated, they are often underutilized and/or prescribed below the recommended dosage in the majority of patients with heart failure. To our knowledge, however, the optimal use of beta-blockers in chronic heart failure is not investigated in Ethiopia. Therefore, the aim of our study was to investigate the utilization and optimization of beta blockers in the management of patients with chronic heart failure in Ethiopia. A prospective observational study was conducted among ambulatory patients with chronic heart failure in Ethiopia. We included adult patients with a diagnosis of heart failure with a baseline left ventricular ejection fraction < 40% who had been on follow-up for at least 6 months. Patients were recruited into the study during their appointment for medication refilling using simple random sampling technique. All patients were followed for at least 6 months to determine the optimal use of beta blockers. The optimal use of beta blockers was determined according to evidence based guidelines. After explaining the purpose of the study, we obtained written informed consent from all participants. Data were collected through patient interview and review of patients’ medical records. Binary logistic regression analysis was performed to identify factors associated with utilization of beta blockers. A total of 288 patients were included in the study. Out of the total, 67% of the patients were receiving beta blockers. Among the patients who received beta blockers, 34.2% were taking guideline recommended beta blockers while 65.8% were taking atenolol, which is not guideline recommended beta blocker. Among the patients who received guideline recommended beta blockers, only 3% were taking optimal dose. Prior hospitalization [Adjusted Odds ratio (AOR) 0.38, 95% confidence interval (CI) 0.19–0.76], dose of furosemide > 40 mg (AOR 0.39, 95% CI 0.20–0.76), ischemic heart disease (AOR 3.27, 95% CI 1.66–6.45), atrial fibrillation (AOR 4.41, 95% CI 1.38–14.13) were significantly associated with the utilization of beta-blockers. Despite proven benefit, beta blockers were not optimally used in most of the participants in this study. The presence of ischemic heart disease and atrial fibrillation were positively associated with the utilization of beta blockers while hospitalization and higher diuretic dose were negatively associated with the utilization of beta blockers. Clinicians should attempt to use evidence based beta blockers at guideline recommended target doses that have been shown to have morbidity and mortality benefit in chronic heart failure. Moreover, more effort needs to be done to minimize the potentially modifiable risk factors for underutilization of beta blocker in chronic heart failure therapy.
Similar content being viewed by others
Introduction
Heart failure (HF) is a significant contributor to cardiovascular disease burden that affects around 26 million people in the globe1,2. HF is a growing disease burden worldwide owing to increasing elderly population and comorbidity1. The burden of HF has been increasing in sub-Saharan Africa including Ethiopia3,4. Over the past decades, significant advances have been made in the therapies and prevention of HF5. However, HF remained the leading cause of morbidity and mortality6.
Beta-adrenergic receptors are the predominant G protein- coupled receptors subtypes that regulate the cardiac function and physiology7,8. Beta adrenergic receptors are activated by epinephrine and norepinephrine and the activation of adrenergic receptors has detrimental effects in patients with chronic HF8,9. Beta adrenergic receptors play an important role in the pathophysiology of heart disease and are common therapeutic targets sites for beta-blockers9. Beta blockers are drugs that bind to beta-adrenergic receptors and thereby block the binding of norepinephrine and epinephrine to these receptors7,10. The blockage of beta adrenergic receptors plays an important role in delaying the progression of HF. Therefore, the main benefit of beta-blockers in chronic HF is to block deleterious G protein over-activation in the heart7,9,10.
Beta blockers are the core component of standard therapy in chronic HF with reduced ejection fraction11,12. In patients with systolic HF, evidence-based beta-blockers (carvedilol, metoprolol, and bisoprolol) have been proved to have morbidity and mortality benefit in several randomized clinical trials13,14,15,16. The optimal use of beta-blockers has been demonstrated to improve symptoms, reduce hospitalizations, improve left ventricular function, and enhance survival in chronic HF patients with reduced ejection fraction17,18. Despite the proven benefit of beta blockers in chronic HF, they are often underutilized in actual clinical practice19,20.
In compensated HF patients with reduced ejection fraction, beta-blockers should be initiated at low dose and up-titrated slowly (usually doubling the dose every 2–4 weeks ) to a target dose or maximum tolerable dose21. Several studies reported that clinical benefits of beta-blockers in systolic HF are dose-dependent and a better benefit occurred at higher doses22,23,24,25. In the majority of patients with chronic HF, target doses of beta-blockers are achievable and tolerable21,25,26. Hence, beta–blockers should be titrated to attain a target dose or a maximum tolerable dose to get a maximum clinical benefit11,12,27.
According to evidence-based guidelines11,12, only carvedilol, metoprolol, and bisoprolol are recommended beta blockers in chronic HF with reduced ejection fraction. The recommended daily target doses of evidence based beta-blockers are 200 mg metoprolol, 10 mg bisoprolol, and 50 mg carvedilol28. Despite high rates of beta-blockers’ target doses are achieved in randomized clinical trials, the rates of target dose achievement in clinical practice remained low26,29. Suboptimal use of beta-blockers has been associated with poor treatment outcome in HF patients26,30.
Bet-blockers are well tolerated in the majority of patients with chronic HF31,32. More importantly, the target doses of beta-blockers are tolerable in the majority of HF patients with careful titration28,33. Hence, beta-blockers should be used at optimal dose in all patients with chronic HF unless contraindicated11,12. Conversely, they are often underutilized and/or prescribed below the recommended dosage in the majority of patients with HF24,32,34. To our knowledge, however, the optimal use of beta-blockers therapy in chronic HF is not investigated in Ethiopia. Therefore, our study investigated the utilization and optimization of beta blockers therapy in the management of patients with chronic HF in Ethiopia.
Material and methods
Study design and study setting
We conducted a prospective observational study at ambulatory care clinic of Jimma University Medical Center. Jimma University medical center is a teaching and referral hospital that provides service for both outpatient and inpatient. It is a major public hospital in southwest Ethiopia that serves for about 15 million people in the catchment area.
Study participants
We included adult patients aged > 18 years with a diagnosis of HF with a baseline left ventricular ejection fraction < 40% who had been on follow up for at least 6 months. Patients with precautions and contraindications to the use of beta-blockers including hypotension (< 90/60mmg), bradycardia (< 60 beats per minute), asthma, decompensated heart failure and those patients with incomplete medical record were excluded from the study.
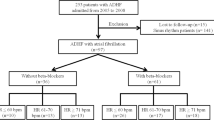
A sample of 355 was calculated using a single population proportion formula assuming 50% rate of optimal use of beta-blockers among patients with HF, 95% confidence level, 5% margin of error, and 10% of contingency for nonresponse rate. From a total of 355 participants approached, 67 patients were excluded from the study due to precaution/contraindication to beta-blockers [55] and incomplete medical record [12].
Data collection procedure
We recruited patients into the study during their appointment for medication refilling using simple random sampling technique. We obtained written informed consent from all participants after we had explained the purpose of the study Data regarding socio-demographics were retrieved using face to face interview. Respective patients clinical and treatment related characteristics were retrieved from patients’ medical record using data abstraction checklist. In order to determine whether the doses of beta blockers are up-titrated to a target dose/ maximum tolerable dose or not, all patients were followed for at least 6 months.
The optimal use of beta blockers was determined according to evidence based guidelines11,28. Patients were said to be optimally treated if they received optimal dose of guideline-recommended beta blockerss (metoprolol/carvedilol). Beta blockers were said to be underutilized if they were not used by patients in the absence of contraindication. Patients were considered as receiving guideline-recommended beta-blocker therapy if they received a prescription of carvedilol, or metoprolol while they were considered as receiving non-recommended beta blocker if they received a prescription of atenolol. The dose of beta blockers was said to be optimal if it was given at guideline-recommended target dose or a maximum tolerable dose is given for the patients. Whereas, the dose was said to be suboptimal if the patient was taking any dose of beta blockers below the target dose in the absence of contraindications for up-titration. For metoprolol, the optimal dose was considered if the dose was = 200 mg daily or maximum tolerable dose and for carvedilol, the dose was said to be optimal if it was ≥ 50 mg daily or maximum tolerable dose.
Data analysis
We used EPI data management (version 4.2.0) and the Statistical Package for the Social Science (SPSS version 21.0) to record and analyze the data, respectively. We computed the frequency; mean (standard deviation), and median (interquartile) of categorical and continuous variables using descriptive analysis. We checked multicollinearity among predictor variables using variance inflation factor (VIF) and none was collinear. Univariable logistic regression analysis was performed to determine the association of each independent variable with utilization of beta blockers. Furthermore, multivariable binary logistic regression model was done to identify predictors of beta blockers utilization. A p value of < 0.05 was considered statistically significant in all analyses.
Ethics approval and informed consent
Approval for this study was obtained from the institutional review board of Jimma University, College of Health and Medical Sciences. We fully explained the purpose and protocol of the study to all participants included in the study and written informed consent was obtained from each participant. The personal information was entirely confidential and protected. All methods were performed in accordance with the approved institutional guidelines.
Results
Sociodemographic and clinical characteristics
A total of 288 patients were included in the study. The mean [± standard deviation (SD)] age of the patients was 52.7 ± 14.7 years and 55.9% were males. Two-thirds of the participants were rural dwellers and more than half (57.3%) were unable to write and read (Table 1).
Clinical related characteristics
In this study, the majority of patients were in New York Heart Association (NYHA) class III (52.1%) and II (43.4%). More than half (58%) of the participants had been hospitalized one or more times during the last year. Two-thirds (67%) of the participants had two or more comorbidities. Ischemic heart disease (52.8%), hypertension (29.5%), and valvular heart disease (16.3%) were commonly identified comorbid diseases. The mean arterial blood pressure (MAP) was 87.6 (± 10.2) mmHg and the mean ejection fraction (EF) was 27.5 (± 6.5) % (Table 2).
Treatment related characteristics
The mean (SD) duration of treatment was 2.8 (2.1) years. The mean (± SD) number of medications per patient was 4.3 ± 1.1 and 43.1% of the participants were taking five more medications. Angiotensin converting enzyme inhibitors (82.6%), loop diuretics (77.8%), and beta-blockers (67%) were most frequently used HF medications. More than half (58%) of the patients were receiving a combination of angiotensin converting enzyme inhibitors and beta blockers (Table 3).
Utilization and dosing of beta-blockers in heart failure patients
Out of the total, 193(67%) participants received beta-blockers. Among the patients who received beta-blockers, only 66(34.2%) were taking evidence based beta-blockers while 127(65.8%) were taking atenolol, which is not evidence based beta blocker. Among the patients who took evidence based beta-blockers (66), only 2(3%) were receiving optimal dose. Of the total patients who received evidence based beta blockers, 7(10.6%) reached greater than or equals to 50% of the guideline recommended target dose. The mean daily doses of metoprolol, carvedilol, and atenolol that were taken by the participants were 29.8 mg, 19.1 mg, and 30.3 mg, respectively (Table 4).
Factors associated with the utilization of Beta-blockers
We performed a univariable logistic regression analysis to compare HF patients who were receiving beta-blockers and not receiving beta-blockers using the socio-demographic, clinical and treatment-related characteristic. Accordingly, hospitalization (Crude Odds ratio (COR) 0.42, 95% confidence interval (CI) 0.25–0.71), dose of furosemide > 40 mg (COR 0.35, 95% CI 0.20–0.63), ischemic heart disease (COR 2.31, 95% CI 1.40–3.82), atrial fibrillation (COR 5.91, 95% CI 1.78–19.63) and number of medication ≥ 5 (COR 1.68, 95% CI 1.01–2.79) were significantly associated with the use of beta-blockers. Furthermore, variables with P < 0.25 in the univariable analysis were re-entered into the multivariable logistic regression model. The full model containing all predictors was statistically significant (Chi-square = 48.610, degree of freedom (df) = 8, P < 0.001). According to multivariable logistic regression analysis, hospitalization (Adjusted Odds ratio (AOR) 0.38, 95% confidence interval (CI) 0.19–0.76), dose of furosemide > 40 mg (AOR 0.39, 95% CI 0.20–0.76), ischemic heart disease (AOR 3.27, 95% CI 1.66–6.45), atrial fibrillation (AOR 4.41, 95% CI 1.38–14.13) were significantly associated with the use of beta-blockers (Table 5).
Discussion
Although beta-blockers were traditionally thought to be contraindicated, they have consistently been shown to reduce morbidity and mortality in chronic HF with reduced ejection fraction29,35. Evaluation of utilization and optimization of beta blockers in chronic heart failure management is crucial to designing programs for future intervention. In the current study, we therefore investigated the utilization and optimization of beta-blockers among patients with HF.
In our study, 67% of the patients received beta blockers, which is in line with a study conducted in France, 65%36. Besides, 58% of the patients received a combination of beta blockers and angiotensin converting enzyme inhibitors, which is also consistent with France study, 61%36. Despite beta blockers are recommended to be used in all patients with HF with reduced ejection fraction unless contraindicated, one-third of the participants were not receiving beta blockers without any reason in the present study. In agreement with our study, beta blockers were underutilized in other similar studies20,26,36,37. Surprisingly, among the patients who received beta blockers, only one-third received guideline-recommended beta blockers. Based on the evidence based clinical practice guidelines11,12,28, metoprolol, carvediolol, and bisoprolol are the only recommended beta blockers in HF with reduced ejection fraction. In contrast, two-thirds of the participants were receiving another beta blocker (atenolol), which is not guideline-recommended beta blocker. The possible justification for the high prescription rate of atenolol might be due to the affordability issue as the evidence based beta blockers (metoprolol/carvedilol) are by far more expensive than atenolol. Furthermore, it could be attributed to the lack of availability of the evidence based beta blockers, absence of HF treatment guideline, and lack of awareness of medical practitioners about the differences between the evidence based beta blockers and other beta blockers in our setting.
In our study, the presence of ischemic heart disease and atrial fibrillation were positively associated with the utilization of beta blockers. This could be justified that atrial fibrillation and ischemic heart disease are additional indications of beta blockers38,39,40. Hence, this may initiate medical practitioners to prescribe beta-blockers. Beta blockers have been shown to reduce hospitalization in several studies17,41,42,43. Similarly, hospitalization was negatively associated with the use of beta blockers in the current study. Moreover, high dose of furosemide was negatively associated with the utilization of beta blockers. Consistent with our finding, the increased dose of furosemide has been reported to affect the use of beta blockers in chronic HF management44. Therefore, medical practitioners should prescribe diuretics with careful consideration taking into account their negative impact on the use of guideline-recommended drugs that have survival benefit. More importantly, the dose of furosemide needs to be optimized to allow utilization of beta blockers.
For the better outcome, beta blockers should be titrated up to a guideline-recommended target dose or maximum tolerable dose12,21,34. More importantly, the target doses are achievable in the majority of patients with chronic heart HF31,33. Despite guideline recommendations, most of the participants were receiving suboptimal dose in our study. In agreement to our study, most of the patients were receiving suboptimal dose in other similar studies20,36,41. In the present study, only 10.6% reached greater than or equals to 50% of the guideline-recommended target dose, which is quite different from the study conducted in France, 56%26. This could be attributed to the difference in medical practitioners’ expertise and the poor awareness of dose titration practice of beta blockers as observed in this study. Therefore, more effort needs to be done to up-titrate beta blockers to target dose or maximum tolerable dose.
Strength and limitation of the study
The strength of our study is the prospective collection of data, which enables for accurate evaluation of the data related to treatment, clinical and laboratory parameters. Our study is the first to have full report on treatment optimization of beta blockers in Ethiopia. Hence, this finding may enhance future intervention to optimize beta-blockers use in patients with chronic HF.
Although we have attempted to assess various factors that might influence the utilization of beta blockers, we did not assess the impact of health professionals’ level of knowledge on the utilization of beta blockers. This study may not provide adequate evidence regarding the cause-effect relationship of beta-blocker utilization and its risk factors due to the inherent characteristics of cross-sectional study. Our study is a single center study; therefore, it might not be generalized to the general population. Moreover, as the findings of this study could be affected by the differences in participants’ characteristics, disease distribution, healthcare infrastructure, and methods employed, they should be extrapolated to other countries with caution.
Conclusion
Despite the proven benefit in chronic HF, beta blockers were not optimally used in most of the participants in this study. The presence of ischemic heart disease and atrial fibrillation were positively associated with the utilization of beta blockers while hospitalization and higher diuretic dose were negatively associated with the utilization of beta blockers. Clinicians should attempt to use evidence based beta blockers at guideline recommended target doses that have been shown to have morbidity and mortality benefit in chronic heart failure. Furthermore, more efforts need to be done to minimize the potentially modifiable risk factors for underutilization of beta blocker in chronic heart failure therapy. More importantly, the dose of furosemide needs to be optimized to allow utilization of beta blockers.
Data availability
The dataset of this article is accessible on reasonable request from the corresponding author.
References
Savarese, G. & Lund, L. H. Global public health burden of heart failure. Cardiac Fail. Rev. 3, 7 (2017).
Benjamin, E. J. et al. Heart disease and stroke statistics-2018 update: a report from the American Heart Association. Circulation 137, e67 (2018).
Agbor, V. N. et al. Heart failure in sub-Saharan Africa: a contemporaneous systematic review and meta-analysis. Int. J. Cardiol. 257, 207–215 (2018).
Nyaga, U. F. et al. Data on the epidemiology of heart failure in Sub-Saharan Africa. Data Brief 17, 1218–1239 (2018).
Wieser, M. et al. Pharmacological therapy of heart failure with reduced ejection fraction. Therapeutische Umschau Revue Therapeutique 75, 180–186 (2018).
Komajda, M. et al. Incremental benefit of drug therapies for chronic heart failure with reduced ejection fraction: a network meta-analysis. Eur. J. Heart Fail. 20, 1315–1322 (2018).
Wang, J., Gareri, C. & Rockman, H. A. G-protein-coupled receptors in heart disease. Circ. Res. 123, 311403 (2018).
Capote, L. A., Mendez Perez, R. & Lymperopoulos, A. GPCR signaling and cardiac function. Eur. J. Pharmacol. 763, 143–148 (2015).
Florea, V. G. & Cohn, J. N. The autonomic nervous system and heart failure. Circ. Res. 114, 1815–1826 (2014).
Lymperopoulos, A., McCrink, K. A. & Brill, A. Impact of CYP2D6 genetic variation on the response of the cardiovascular patient to carvedilol and metoprolol. Curr. Drug Metab. 17, 30–36 (2015).
Ponikowski, P. et al. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC). Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur. J. Heart Fail. 18, 891–975 (2016).
Yancy, C. W. et al. 2017 ACC/AHA/HFSA focused update of the 2013 ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Failure Society of America. J. Am. Coll. Cardiol. 70, 776–803 (2017).
Merit-HF Study Group. Effect of metoprolol CR/XL in chronic heart failure: Metoprolol CR/XL Randomised Intervention Trial in Congestive Heart Failure (MERIT-HF). Lancet (London, England) 353, 2001–2007 (1999).
Packer, M. et al. Effect of carvedilol on the morbidity of patients with severe chronic heart failure: results of the carvedilol prospective randomized cumulative survival (COPERNICUS) study. Circulation 106, 2194–2199. https://doi.org/10.1161/01.cir.0000035653.72855.bf (2002).
Domanski, M. J. et al. A comparative analysis of the results from 4 trials of beta-blocker therapy for heart failure: BEST, CIBIS-II, MERIT-HF, and COPERNICUS. J. Card Fail 9, 354–363 (2003).
Investigators, C. I. The Cardiac Insufficiency Bisoprolol Study II (CIBIS-II): a randomised trial. Lancet (London, England) 353, 9–13 (1999).
Manurung, D. & Trisnohadi, H. B. Beta blockers for congestive heart failure. Acta Med. Indones 39, 44–48 (2007).
Sallach, J. A. & Goldstein, S. Use of beta-blockers in congestive heart failure. Ann. Med. 35, 259–266 (2003).
Berthelot, E., Eicher, J., Salvat, M., Seronde, M. & de Groote, P. Medical inertia in the optimization of heart failure treatment after discharge and its relationship to outcome. Health Care Curr. Rev. 6, 2 (2018).
Loop, M. S. et al. Low Utilization of Beta-Blockers Among Medicare Beneficiaries Hospitalized for Heart Failure With Reduced Ejection Fraction. J. Card Fail 16, 31107–31102 (2018).
Basile, J. N. Titration of beta-blockers in heart failure. How to maximize benefit while minimizing adverse events. Postgrad. Med. 113, 63–70 (2003).
Packer, M. Does a target dose or a target heart rate matter to outcomes when prescribing β-blockers to patients with chronic heart failure?. Circ. Cardiovasc. Qual. Outcomes 11, e004605 (2018).
Driscoll, A., Srivastava, P., Toia, D., Gibcus, J. & Hare, D. L. A nurse-led up-titration clinic improves chronic heart failure optimization of beta-adrenergic receptor blocking therapy–a randomized controlled trial. BMC Res Notes 7, 1756–0500 (2014).
Gheorghiade, M. et al. Medication dosing in outpatients with heart failure after implementation of a practice-based performance improvement intervention: findings from IMPROVE HF. Congest. Heart Fail. 18, 9–17 (2012).
Schmidt, S. et al. Treatment with higher dosages of heart failure medication is associated with improved outcome following cardiac resynchronization therapy. Eur. Heart J. 35, 1051–1060 (2014).
Solal, A. C. et al. Optimization of heart FailUre medical Treatment after hospital discharge according to left ventricUlaR Ejection fraction: the FUTURE survey. Arch. Cardiovasc. Dis. 105, 355–365 (2012).
Cowie, M. R. & Komajda, M. Quality of physician adherence to guideline recommendations for life-saving treatment in heart failure: an International Survey. Cardiac Fail. Rev. 3, 130 (2017).
Yancy, C. W. et al. 2013 ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J. Am. Coll. Cardiol. 62, e147–e239 (2013).
Bhatt, A. S., DeVore, A. D., DeWald, T. A., Swedberg, K. & Mentz, R. J. Achieving a maximally tolerated beta-blocker dose in heart failure patients: is there room for improvement?. J. Am. Coll. Cardiol. 69, 2542–2550 (2017).
Franke, J. et al. Optimization of pharmacotherapy in chronic heart failure: is heart rate adequately addressed?. Clin. Res. Cardiol. 102, 23–31 (2013).
Krum, H. et al. Tolerability of beta-blockers in elderly patients with chronic heart failure: the COLA II study. Eur. J. Heart Fail 8, 302–307 (2006).
Kiel, R. G. & Deedwania, P. The safety and tolerability of beta blockers in heart failure with reduced ejection fraction: is the current underutilization of this evidence-based therapy justified?. Expert Opin. Drug Saf. 14, 1855–1863 (2015).
Gottlieb, S. S. et al. Tolerability of beta-blocker initiation and titration in the Metoprolol CR/XL Randomized Intervention Trial in Congestive Heart Failure (MERIT-HF). Circulation 105, 1182–1188 (2002).
Barywani, S. B., Ergatoudes, C., Schaufelberger, M., Petzold, M. & Fu, M. L. Does the target dose of neurohormonal blockade matter for outcome in Systolic heart failure in octogenarians?. Int. J. Cardiol. 187, 666–672 (2015).
Funck-Brentano, C. Beta-blockade in CHF: from contraindication to indication. Eur. Heart J. Suppl. 8, C19–C27 (2006).
de Groote, P. et al. Is the gap between guidelines and clinical practice in heart failure treatment being filled? Insights from the IMPACT RECO survey. Eur. J. Heart Fail. 9, 1205–1211 (2007).
Maison, P. et al. Utilisation of medications recommended for chronic heart failure and the relationship with annual hospitalisation duration in patients over 75 years of age. A pharmacoepidemiological study. Eur. J. Clin. Pharmacol. 61, 445–451 (2005).
Ozieranski, K. et al. Effect of beta-blockers on 1-year survival and hospitalizations in patients with heart failure and atrial fibrillation: results from ESC-HF Pilot and ESC-HF Long-Term Registry. Pol. Arch. Internal Med. 128, 649–657 (2018).
January, C. T. et al. 2019 AHA/ACC/HRS focused update of the 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation: A report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. J. Am. Coll. Cardiol. 74, 104–132 (2019).
Fihn, S. D. et al. 2014 ACC/AHA/AATS/PCNA/SCAI/STS focused update of the guideline for the diagnosis and management of patients with stable ischemic heart disease: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines, and the American Association for Thoracic Surgery, Preventive Cardiovascular Nurses Association, Society for Cardiovascular Angiography and Interventions, and Society of Thoracic Surgeons. J Am Coll Cardiol 64, 1929–1949 (2014).
Ouwerkerk, W. et al. Determinants and clinical outcome of uptitration of ACE-inhibitors and beta-blockers in patients with heart failure: a prospective European study. Eur. Heart J. 38, 1883–1890 (2017).
Ramahi, T. M. Beta blocker therapy for chronic heart failure. Am. Fam. Physician 62, 2267–2274 (2000).
Chatterjee, S. et al. Benefits of β blockers in patients with heart failure and reduced ejection fraction: network meta-analysis. BMJ 346, f55 (2013).
Rodriguez-Cillero, C. et al. Potentially inappropriate use of furosemide in a very elderly population: An observational study. Int. J. Clin. Pract. 71, 12975 (2017).
Acknowledgements
We would like to express our gratitude to the data collectors and working staff members of Jimma University for their impressive commitments and cooperation. Our gratefulness extended to the HF patients for their volunteer involvement in the study.
Funding
This research is financially supported by Jimma University.
Author information
Authors and Affiliations
Contributions
Y.N. conceptualized and designed the study, analyzed and interpreted the data and drafted the original manuscript, S.A., G.D., and K.G. involved in the manuscript evaluation. All authors have reviewed and approved the final version of the manuscript for submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Niriayo, Y.L., Asgedom, S.W., Demoz, G.T. et al. Treatment optimization of beta-blockers in chronic heart failure therapy. Sci Rep 10, 15903 (2020). https://doi.org/10.1038/s41598-020-72836-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-72836-4
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.