Abstract
Morphological differences associated with sex or stage, together with total lipids and carotenoids, were studied in Euphausia superba as possible indicators of physiological condition. E. superba displays sexual dimorphism during growth. A group of mature males, called Males II herein, has a greater abdominal length, suggesting that they are faster swimmers, a feature implying higher metabolic rates and a higher demand for protecting pigments like carotenoids. Mature Males II have proportionally lower lipids but higher total lipid-soluble carotenoids, a counterintuitive finding. Males II also have bigger eyes. Significant regressions with carotenoids were found for wet weight, abdominal length, and eye diameter. On a spatial analysis, population composition reflects reproductive activity. Males II would be in search of females for fecundation and, thus, are dominant in some areas. The PCA analysis of 10 allometric and biochemical variables show a distinct Males II group differing in morphology, carotenoids, and lipid contents. The carotenoid:lipid ratio was highest for Males II, supporting the hypothesis of the role of carotenoids in the activity of the species. Mature males may experience physiological stress during reproduction and probably die shortly afterwards. A relationship between activity, morphometrics, and carotenoid content seems evident, deserving further investigation.
Similar content being viewed by others
Introduction
The Antarctic krill, Euphausia superba, has been the focus of multiple studies in the past because of its central role in the trophic web in the Antarctic Ocean. It has been found that during its ontogenetic development, E. superba undergoes a morphological transformation related primarily to sexual maturity1,2,3,4,5,6,7,8,9.
For E. superba, Miller (1983) found a significant difference between sexes, as well as between mature and immature females, in the slope of the regression between total length and carapace length5. Färber Lorda (1986, 1990) found an ontogenetic morphometric differentiation in E. superba and Thysanoessa macrura, and proposed a Differentiation Index (DI) to classify groups of individuals according to sex or age based on morphological traits7,8. Two groups of males were identified, hereafter referred to as Males I and Males II, which are related to the ratios between total body length, carapace length, and abdominal length8. In order to determine the longevity of E. superba and T. macrura—the dominant and coexisting species during summer in the southern part of the Indian Ocean, a multivariate study including morphological information showed that total lipids and total carotenoids also differ in quantity across development stages, assuming a 4-year life cycle based on morphometric, lipid, and carotenoid data, and a two-year life cycle for T. macrura. Males II have a lower lipid content versus all other sex groups or development stages9. Three age groups were separated when lipofuscines (age pigments) and morphometric variables were analysed, also showing that these groups could be separated based on morphometric measurements alone, indicating a morphological differentiation in Antarctic krill10.
The allometric relationship of total length vs. wet weight for Males II showed a steeper slope and a higher condition coefficient relative to the other sex or development stages; however, a spatial analysis revealed geographic differences beyond those related to sex or development stage11. In another paper, Färber Lorda et al. also showed that, spatially, the composition of populations was a key driver of mean total lipid content and mean DI12. An important implication of these differences is that animals in continuous movement, like krill13, will have faster swimming capacities. Males II have a longer abdomen, suggesting that they are better or faster swimmers8. In E. superba, a stronger but slower beating of pleopods was found in males, as compared to females14. Thus, we assumed a faster swimming capacity in mature Males II, which are expected to be more active in search of females for fecundation during the reproductive season in summer. The population sex ratio was found to be largely influenced by the swimming capacities of males and females, and may contribute to the segregation of different sexes or development stages within swarms of E. superba13,15,16; a faster growth rate was observed in males than in females during the Antarctic summer17. A reanalysis of body shrinkage during winter in E. superba in its natural habitat as compared to animals reared in aquaria18 found that it occurred only in females, but not in males19.
Lipids in krill have been widely studied, finding important differences between sexes11,12,20,21,22,23,24,25,26,27, especially during the reproductive season. However, Cripps et al.28, analysing fatty acids in both phytoplankton and E. superba, concluded that diet is likely more important than sex or development stage to determine fatty acid composition28. Geographic distribution is also related to differences in the biochemical composition of krill, especially as regards lipids and population structure13,25. Bouyancy has been studied in zooplankton, and it is hypothesised that lipids contribute to the buoyancy capacity of many zooplankton species, particularly E. superba. Water temperature plays a central role, as the proportion of lipids in solid phase contributes to buoyancy. The ratio of solid and liquid lipids constitutes a key element for the maintenance of krill at a depth that provides sufficient food and other favourable environmental conditions29.
Krill performs vertical migrations like most zooplankton, which apparently is a defence mechanism to avoid predation and to remain within the depth range where food is available30. This behaviour also confers protection from damaging light irradiance31,32, in addition to the synthesis of carotenoids–mostly astaxanthin, a potent antioxidant33,34,35,36,37,38,39,40,41.
Carotenoids in euphausiids were first studied by Wagner (1939, in Mauchline and Fisher, 1969) using euphausiids from baleen-whale stomach contents42. This author claimed that ß-carotene was converted into vitamin A in the stomach of baleen whales; further work by Kon and Thompson showed that the carotenoid astaxanthin was the dominant pigment in euphausiids, together with vitamin A33,34. Fisher et al. studied the evolution of the pigments vitamin A and astaxanthin in Meganyctiphanes norvegica and Thysanoessa raschii in relation to body weight, reporting higher concentrations in smaller animals43. These authors also reported higher levels in larval stages of both species and higher concentrations of these pigments in the eyes, a finding also supported by Czerpak et al.38. In a seasonal study, Fisher et al. found that both vitamin A and astaxanthin increased with body weight; however, fluctuations were also important, being related to higher phytoplankton availability, especially diatoms, with higher values in spring44. The exposure of krill to different light radiation intensities during vertical migrations and seasonal changes in light intensity under pack-ice produce important seasonal changes in the concentration of carotenoids and the colour of individuals, as shown by Auerswald et al. for E. superba41. However, former studies showed that krill is highly sensitive to ultraviolet radiation, which may lead to DNA damage45 and higher mortality, as shown by Newman et al.46, thus producing an avoidance reaction in E. superba47.
Shifts in colour have been classified as physiological and morphological changes41. In krill, physiological colour change takes place readily and occurs in specialized cells, chromatophores, which can adopt different colours. Morphological changes could be due to either an increase or a decrease in pigment concentration, the amount of chromatophores present, or a combination of both41. The Babak Law states that the number of chromatophores or the concentration of pigments either increase or decrease in relation to prolonged light periods lasting from days to months48. Crustaceans do not synthesize carotenoids, so they have to feed on them; once consumed, carotenoids are mostly transformed into astaxanthin37.
This study determined the total carotenoid and total lipid contents, along with morphometric measurements, in E. superba individuals of different sex and development stages, from various locations across the Antarctic Ocean. These individuals were collected over a short time during summer, mostly after the reproductive season, a key period for the metabolism of lipids and carotenoids according to previous results by other authors18,26,35.
Material and methods
Samples of Antarctic krill were collected in the southern part of the Indian Ocean during the FIBEX MD 25 cruise on board R. V. Marion Dufresne in the summer (February, 1981), using an RMT net with 5 mm mesh size and 2 mm in the cod end. Samples were collected from each of 15 stations at noon and midnight, following echo-sounding signals; some samples were collected following swarm detections at given stations (Fig. 1). The specimens sampled were individually frozen at − 70 °C on board and transported to the laboratory. Once thawed, individuals were measured to the nearest 0.01 mm under a dissecting microscope. Samples were well-preserved, with no apparent damage and with turgescent eyes. This preservation method also maintained the specimens in good condition to perform the lipid and carotenoid analyses on the same individuals. The measurements performed were eye diameter, carapace length, total length, and wet weight, as described in Färber Lorda7,8,11; abdominal length was determined as the difference between total and carapace length. In the laboratory, each individual was weighed and measured to calculate the Differentiation Index (DI; Färber Lorda, 1986, 1990)7,8 as follows:
DI values for the different sex or stage groups are shown in Table 1.
Afterwards, lipid extraction was performed and total carotenoids were estimated.
Carotenoids and lipids
A pool of animals was first homogenized on a small volume of distilled water, after which the homogenate was split into two portions; lipids were extracted with the Bligh and Dyer49 method in one portion and with acetone in the other. The absorbance peak of both samples was compared on a scan from 300 to 700 nm, showing almost identical profiles and a similar peak with both extraction methods (Blight and Dyer, at 485 nm; acetone, at 490 nm); the experience was repeated three times with the same results. Total carotenoids were evaluated using the lipid extract read in a spectrophotometer at 485 nm; carotenoids concentration was calculated with the formula by Jackowska et al.50 as follows:
where Ca = carotenoids, in µg/gr (wet weight), E = absorbance of the sample, v = Extraction volume, D = width of the spectrophometer cuvette (1 cm), W = wet weight of the sample
Total carotenoids were calculated in µg per individual and in grams per individual. Lipids were first extracted from animals using the method of Bligh and Dyer49, followed by the colorimetric method of Pande et al.51 using tripalmitin as standard, as mentioned in Färber Lorda7,9; total lipids were expressed as mg per animal and as % wet weight. Data were processed and figures were plotted using the Sigmaplot program. These data were partially published elsewhere aiming to understand the longevity of E. superba9. Carotenoid data per individual or per g were tested for normality; they all showed a normal distribution for each sex or development stage, but for the entire sample, only the data expressed as carotenoids per g showed a normal distribution. ANOVA and regression analyses were performed on the data; for ANOVA, α was set at 95% (Table 3). When data were not normally distributed (P < 0.005), a Kruskall Wallis test was performed (P < 0.001) and Mann–Whitney’s U was calculated for each paired group of sex or development stage. When the paired data were normally distributed, the Student’s t-test was run; these tests were performed on the three main variables analysed (Results shown in Table 3). Lipid data expressed as % were arc-sin transformed and a Krukall-Wallis test was performed (P < 0.001). Power regression analyses were performed occasionally on non-normally distributed data; they are shown with their 95% confidence intervals. Cohen’s d Index was calculated to evaluate the effect of size on the ANOVA and regression analyses (Table 3), and Rosenthal’s r was computed on the Kruskall-Wallis nonparametric test, to evaluate the degree of separation between the paired comparison means by sex or development stage: a higher value means that means are more different, i.e., more separated. In order to better understand the morphological and biochemical differences by sex or development stage, as well as differences related to the geographic distribution of the populations sampled, Principal Components Analyses (PCA) were performed with the Statistica program.
Ethical standard
All applicable international, national, and institutional guidelines for the care and use of animals were followed.
Results
Carotenoid levels were highest in Males II, lowest in Juveniles, and intermediate in Males I, Mature Females, and Spent Females, in either per-individual or per-g (Table 2) basis. Males II show low lipid and high carotenoid levels both per individual and per gram (Fig. 2). Carotenoid content in E. superba by sex showed that the morphologically different Males II group had the highest carotenoid content, both per individual and per gram (wet weight). A Kruskall-Wallis rank test showed significant differences between all groups (H4114 = 57.756, P < 0.001) for carotenoids per individual, and the test for the Effect of Size yielded ƞ2 = 0.4715, i.e., 47.15% of the variance is explained by the data. The Mann–Whitney test was used to test for differences between each paired combination of sex or stage groups; the results for carotenoids and lipids are shown in Table 3.
For carotenoids by animal, a non-significant difference was found between Mature Females and Males I, between Mature Females and Spent Females, and between Spent Females and Males I; for the remaining group pairs, differences were significant (Table 3). The effect of size is shown in Table 3; Rosenthal’s r or Cohen’s d for all comparisons, particularly those including Males II, showed a clear separation of groups and a large gap. Only the comparison between Mature and Spent Females yielded a low r, i.e., a small separation between these two groups. For lipids, the Kruskall-Wallis test performed on non-normally distributed data showed significant differences between the 5 groups analysed (H4114 = 45.79, P < 0.001); ƞ2 = 0.3666 indicates that 36.66% of the variance is explained by the data; and the Mann–Whitney test for the comparison of each combination of paired groups showed significant differences between all groups, except for Males II vs. Juveniles and Males II vs. Males I, as shown in Table 3. The effect of size is also shown in Table 3 with the Rosenthal’s r value or the Cohen’s d value.
For the ANOVA on carotenoids expressed as µg per g−1 by sex and stage (Table 1), a significant difference was observed between groups (F4114 = 5.652, P < 0.001), A comparison between all the possible combinations of paired sex or Stage of development groups was performed with the t of Student, all the comparisons with Males II showing significant differences, but not among the other paired comparisons. The d value for the effect of size on each paired group of data is also shown in Table 3; all the comparisons with Males II show a large or very large non-overlap according to the d value. Based on Eta (ƞ2p = 0.8408%, D.F. = 4 and 114), 84.08% of the variance is explained by the data.
The difference between Males I and Males II, in percentage of lipid content (arc-sin transformed data) was significant. In other words, Males II had a significantly lower lipid concentration than Males I (t141 = 3.66; P = 0.004) – a counterintuitive finding, since carotenoids are mostly lipid-soluble. However, on a per-individual basis, a non-significant difference was observed for lipids between the two groups (Kruskall-Wallis: H141 = 1.126, P = 0.289), likely because Males I are smaller.
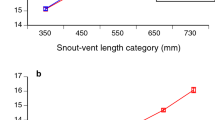
The regression between carapace length and total length by sex or stage, analysed on a larger sample (Fig. 3), showed that Males I apparently evolved towards a shorter carapace and constitute the Males II group, given the lower significance of r2 in both groups. Carotenoids increased in parallel with wet weight, eye diameter, and abdominal length, as shown by the significant regressions (Fig. 4a–c). However, the data were not normally distributed, showing in all cases a clear separation of the Males II group with some mixing with the bigger Mature Females or Males I. Although the regressions between DI and carotenoids per animal for each sex or stage group (Fig. 5) were non-significant, they are shown to emphasize the morphological differences; all other regressions between the different variables by sex or development stage were non-significant due to the large individual variability. The non-significant regressions between DI and carotenoids showed that the lowest DI corresponds to the highest carotenoid concentrations in Males II, whereas higher DI values are related to lower carotenoid content. (In support of these findings, other non-significant regressions are shown in Supplementary Information 1–5).
Linear regression of total length vs carapace length for all sex groups with a larger sample, with the Confidence Intervals. Data were normally distributed (Shapiro–Wilk test); but their variances were not always constant. Juveniles: TL = 1.093 + 2.471 CL, N = 76, r2 = 0.949, Normality: P = 0.276, Homoscedasticity, Failed: P = 0.021. Females: TL = 8.354 + 1.881CL, N = 75, r2 = 0.889, Normality: P = 0.785, Homoscedasticity, Passed: P = 0.115. Males I: TL = 4.417 + 2.293 CL, N = 72, r2 = 0.744, Normality: P = 0.268, Homoscedasticity, Passed: P = 0.328. Males II: TL = 6.162 + 2.445 CL, N = 32, r2 = 0.757, Normality: P = 0.836, Homoscedasticity, Passed: P = 0.245.
(a) Regression of carotenoid content per individual vs. wet weight. A power model was applied and a non-normal distribution was found (P = 0.0424; 95% confidence intervals included). Data for Males II encircled in blue. (b) Regression of carotenoid content per individual vs. abdominal length. A power model was applied and a non-normal distribution was found (P = 0.0470; 95% confidence intervals shown). Data for Males II encircled in blue. (c) Regression of carotenoid content per individual vs. eye diameter. Again, a power model was applied and a non-normal distribution was found (P = 0.0083; 95% confidence intervals shown). Data for Males II encircled in blue.
We used the carotenoid (µg):lipid (mg) ratio as an indicator of the physiological status of individuals (whether good or bad; this is not yet clear). It was highest for Males II and lowest for Mature Females (Fig. 6).
The analysis per transect showed that lower mean DI values are associated with a higher carotenoid content (Fig. 7a, b). The maturity of individuals in transect 64°S was markedly different relative to the other transects, being composed of adults; therefore, we decided to pool together transects 62°S and 63°S for comparison vs. transect 64°S. The pooled 62°S + 63°S transect showed lower mean carotenoid levels and intermediate and highly variable lipid values; however, the mean Differentiation Index was higher than for transect 64°S. The mean carotenoid content per gram was higher for transect 64°S (402.69 ± 14.508) than for transect 62°S + 63°S (330.22 ± 15.65), and differences were statistically significant for carotenoid content per gram (F1117 = 11.124, P = 0.001), mean length (Kruskall-Wallis, H1117 = 29.712, P < 0.001), and mean body weight (F1117 = 28.195, P < 0.001). Only two Male II individuals were found in transect 62°S + 63°S; however, the number of individuals for transect 62°S + 63°S was smaller (49) than for transect 64°S (70). In general, individuals were bigger and mature in transect 64°S. Also, a significant difference between transects was found for the carotenoid:lipid ratio (Kruskall-Wallis, H1117 = 6.358, P = 0.012), being higher for transect 64°S versus transect 62°S + 63°S (18.67 ± 1.38 vs. 14.12 ± 1.31).
When the data were analysed with a Principal Components Analysis (Fig. 8a), we found that the groups were, again, separated by sex rather than by stations or transects, probably because the differences between sexes are more important than differences between station and those associated with specific physical or trophic conditions. Males II is again separated from all other groups. However, only two of all Males II individuals were found at Station 19; all others were found at transect 64°S. The data accounted for 81.17% of the variance. The first factor was associated with the variables Wet Weight (WW), Total Length (TL), Abdominal Length (LA), and Eye Diameter (ED), and partially with Carotenoids per Individual (CAI) on the positive side, and with Carapace Length (CL) on the negative side (mostly size variables). Factor 2 is associated with Carotenoids per gram (CaG) and Carotenoid:Lipid ratio (Ca/Li) on the positive side of the axis, and with the Differentiation Index (DI) on the negative side, with lipids shared in the two axes. All Males II were grouped in a position opposite to that of the DI variable and near CaI (carotenoids per animal) and Ca/Li. The direction and distance of the variables is shown in Fig. 8b; as mentioned above, all size variables were located on the negative side of Factor 1. DI is positioned near lipids (Li), as expected, and opposite to Ca/Li and CaG; the separation of the Males II group is determined by these two variables (CaG and Ca/Li). The contributions of the ten variables are shown in Table 4.
(a) Projection of the cases on the PCA analysis of the data analysed by station, and by sex or stage of development. A separation between sexes is evident, and again, Males II (encircled in red) forms a group clearly separated from the rest of the data. Variables are represented by pink triangles with abbreviated green letters; cases, by blue circles with their station number, and sex (Ju: Juveniles, MF: Mature Females, SF: Spent Females, MI: Males I, MII: Males II). The first two components explain 81.17% of the variance. (b) Biplot of the variables with their distance and direction. N = 119 with 10 active variables (in Green). All variables are positioned on the negative side of factor 1 and on both sides of Factor 2, explaining 81.17% of the variance.
Discussion
The results reported here showed that individuals with a potentially greater motile capacity, like Males II, showed a higher carotenoid content either per individual or per gram of wet weight (Table 1). This suggests that Males II probably require protecting pigments like carotenoids, or that this development stage has a faster lipid metabolism. Virtue et al. (1996)52 found that mature E. superba males had lower lipid levels, especially regarding triacylglycerol, a short-term storage lipid, during the reproductive season. Also, mature males did not survive after capture (100% death rate), contrasting with other sex groups that showed a better survival performance, such as females. From this study, these authors inferred that mortality rates in krill differ between males and females due to energetic constraints on reproduction. In our study, total lipids in Males II was equivalent to levels mentioned previously for mature males, but the role of high carotenoid levels is uncertain. Males II have bigger eyes (Fig. 4c); thus, we can assume that they accumulate more carotenoids32,33,35. However, the role of carotenoids in the physiology of Males II is still unclear, i.e., whether high carotenoid levels are a sign of stress or of good health. Other studies have assumed that a higher carotenoid content reflects better food quality37,44; however, the very low lipid levels found in Males II suggests that they are not in good physiological condition. Interestingly, carotenoid data for the total sample differs markedly when analysed on a per-individual or a per-gram basis (Table 1). This finding shows the importance of individual development stages and/or a great individual variability, probably also derived from the local trophic conditions37,44. An annual cycle of carotenoid composition in Euphausia pacifica collected from the Saanich Inlet, British Columbia, showed that the three major carotenoid pigments, making up to 85–90% of the total, are astaxanthin and its mono- and di-esters. The relative composition of the three pigments remained constant throughout the study. Temporal variations in individual and weight-specific levels appeared to be related to season and sexual maturity. These results suggested that E. pacifica can only store astaxanthin and its two esters in a specific ratio37.
The large size of E. superba facilitates the detection of measurable differences in the timing of spawning between males and females. We assumed that males die after fecundation and females die after spawning; this would result in a smaller proportion of males in winter and an approximately 1:1 ratio in summer53. This resembles the hypothesis put forward by Virtue et al.52, but considers females as well. A reduced swimming velocity was observed in recently spawned females58, which likely contributes to the segregation by sex within swarms after spawning and affects the sex ratio within a swarm.
The vertical migration on Meganyctiphanes norvegica was found to differ between males and females54; as females need more food during the reproductive season, they migrate closer to the surface to obtain it and attain reproductive success. This mechanism may also occur in the case of the Antarctic krill, E.superba; therefore, another possible explanation should be considered to explain the differences in sex ratio between the stations sampled in our study. The sex ratio may change in our samples simply because males were swimming deeper, and thus were not always sampled in our cruise. Although these measurements were highly variable, differences in longevity between males and females may also be an important factor17. Males face a high level of stress, with the probable synergistic effect of various conditions, including low lipid but high carotenoid levels and intense activity when searching for females to fertilize them, also considering that the low lipid level will not provide males with the buoyancy needed to maintain their position in the water column29. As a result, males would be using more energy to keep their pace at the same speed and depth as the more buoyant (because of their high lipid level) females. Under this scenario, an aspect worth investigating is whether the higher carotenoid content in Males II is an indicator of good adaptation. As mentioned by Kils13, krill live in a sort of physiological extreme, at the edge of their energetic capacity, which could easily lead to their death. E. superba can swim at speeds of more than two body lengths per second55 and can perform horizontal migrations, even against the prevailing current flows56,57,58; thus, its physiology is more accelerated than that of other crustaceans. The respiration rate of E. superba has been found to be directly proportional to individual weight, thus requiring an exponent b in the equation: Respiration rate = a weight b, where b is closer to 1 vs. other crustaceans, instead of being closer to 0.755.
Krill accumulate carotenoids in the eyes35,36,44, a likely explanation for the higher carotenoid content in Males II, which have bigger eyes. Taken together, the higher carotenoid content, bigger abdomen, and bigger eyes seemingly provide advantages to mature Males II in search of females to fertilize. However, it is unclear whether these are really advantages or just an anticipation of their death. These findings lead to several questions: why does higher carotenoid content occur in animals with lower lipid content? Is this a consequence of a higher metabolic rate in Males II?
Another possible explanation could be the presence of carotenoproteins in crustaceans as proposed by Ceccaldi60 and Ceccaldi et al.61, which could explain the co-occurrence of low lipids and higher carotenoids. Czeczuga62 reported the presence of carotenoproteins in E. superba, but their importance and role has not been studied yet. Also, a circadian rhythm of carotenoids has been observed in the eyes and hepatopancreas of Penaeus japonicus63,64, but has never been studied in E. superba. Lipofuscins reflect the physiological age of an animal; they were studied by Karnaukhov65,66 in molluscs, and by Etteshank and others10,67,68 in Antarctic krill. Carotenoids are included, at least partially, in lipofuscin macromolecules, and are supposed to constitute an oxygen reserve for animals living under low-oxygen conditions66, but its function is still unclear in both crustaceans and molluscs. Further studies on the role of carotenoids and carotenoproteins in E. superba are necessary to better understand their function.
In an attempt to determine the longevity of E. superba using multivariate analyses, including the Differentiation Index and lipid and carotenoid content, further work by Färber Lorda9 sorted out morphologically different groups, with Males II clearly separated in a different cohort. Astaxanthin is the main carotenoid in krill, being a potent antioxidant36,37,38 that could provide protection when a higher metabolic rate is necessary in stages like Males II. However, krill do not synthetize carotenoids, but get them from pigment-rich food. Males II are probably feeding well, but their faster metabolic rate and fecundation needs preclude the accumulation of lipids. A work by Auerswald et al.41 described changes in pigment content between summer and winter, concluding that carotenoids serve as protection from solar radiation and are adapted to the changing seasonal and diel light conditions. In our study, during summer and away from the ice edge, animals need additional protection provided by pigments like carotenoids. However, in this paper we found that high carotenoid levels are apparently related not only to light intensity, but also to the activity of animals. We still do not know if the carotenoid levels observed in this study are typical during summer, when animals are not under the pack ice, nor the role of these higher carotenoid levels in Males II.
Most regression analyses were non-significant; however, the different regressions, whether non-significant or significant, demonstrate the uniqueness of the Males II group; they do show that this group differentiates from all others in either morphological or biochemical variables. Our sampling took place during the summer and over a relatively short period of time; thus, it reflects summer conditions. In Males II, carotenoid content was higher in both per-individual and per-gram basis. The Cohen factor for the effect of size on the data showed differences in per-gram carotenoid content between Males II and all other sex groups or stages; Males II was the statistically significant different group in all cases, with the lowest overlap on the data, according to the Cohen’s d test. The approach in this study is spatial, not seasonal. The significant differences found between transects in terms of total length, wet weight, total carotenoids per individual, and the carotenoid:lipid ratio evidence a major role of carotenoids in mature individuals, with higher mean values for transect 64°S likely related to the reproductive condition and the greater motility associated with it. However, when the Principal Components Analysis was performed on the data by station, groups were separated only for the different sex or stage groups, but not by transect or station. Males II were present only at the southernmost transect, except for two individuals in Station 19. This station was located at the rim of a gyre, which may have caused some mixing with the other transects12. Males II formed a clearly distinct group (Fig. 7b). The analysis by transect is hard to interpret, since each station shows individuals with markedly different characteristics, mainly as regards differences between sex and development stages. For example, lipids and their corresponding DI have opposite values, i.e., high values in Females and low in Males II, with both present in the same stations regardless of the local conditions. Nonetheless, the separation of the Males II group was evident. The PCA analysis shows the uniqueness of the Males II group, with 10 variables included, and determine the unique characteristics of this group. The variables that best determine the group are the Carotenoid:Lipid ratio and the carotenoid content per gram, and the fact that Males II are placed on the opposite side of the variable DI, which determines the body proportions in each group of sex or development stage.
Various aspects of our study point to the greater motility of Males II, probably in search of females for fecundation, including the bigger abdomen and larger eye diameter, which translate into higher accumulation of carotenoids35,43. This group showed a unique morphometry in all the regressions analysed (see also SI 1–5), in addition to higher carotenoid and lower lipid contents. The carotenoid:lipid ratio is highest for Males II and lowest for Mature Females, another indication of a unique physiology in Males II. These findings, together with the lower DI in Males II (Table 1), support the hypothesis of a central role of carotenoids in the physiology of Male II individuals, since a lower DI means more active animals if we follow the hypothesis of a greater activity level in Males II.
Geographically, samples collected in the southern transect had a significantly higher carotenoid content per gram, and were composed of more mature individuals, including a higher number of Males II, characterised by significantly greater total length and wet weight, and a greater mean carotenoid:lipid ratio, all of which are likely related to greater motility. A high DI is associated with lower carotenoid content per individual, especially in Males I. There is no proof of a direct relationship between lower DI and higher carotenoid content, but an interesting relationship is shown. Using data normalised by weight, a higher respiration rate was recently found in mature male individuals that show DI values lower than 3.5 (Tarling et al., in preparation), which supports our findings. A comparison of the three most important species of krill was conducted, showing that E. superba has the greatest carapace and pleopod length relative to the other species studied, as well as a reduction of carapace length relative to total length in males (Tarling et al., submitted). This study suggests a potential relationship of carotenoids with the activity of individuals. The implication of these findings are that a lower DI is equivalent to a greater energetic expenditure, which probably explains our results.
Hypotheses
-
During the reproductive season, Males II are under stress associated with reproduction. They are more active when searching for females, and use an excess energy that is not compensated for by food intake. As a result, Males II show a low lipid content and higher carotenoid levels (which could be present as carotenoproteins;60,61), the latter functioning as protective pigments derived from the accelerated metabolism in this development stage. Males II die after fecundation, as proposed by other authors38,53.
-
The biochemical composition of Males II reflects an accelerated metabolism during the reproductive season, with lower lipid but higher carotenoid levels, the latter being the protective pigment for an accelerated metabolism; animals subsequently recover in the rich summer environment when primary production is high, re-maturing repeatedly69,70,71.
-
A combination of the two hypotheses above: krill will die if they do not encounter favourable trophic conditions, or they will re-start the maturation process when they find favourable trophic conditions, during their continuous displacements by either active swimming or advection (which may also play a role in these scenarios, with less energy expenditure when advected). Krill will make good use of the energy-saving formation of swarms72,73, considering the characteristics of this highly adaptive species that shows an outstanding plasticity in its adaptions18,59.
Conclusions
In E. superba, the Males II group showed a unique morphometry and always appeared as a separate group in all the signficant and non-significant regressions plotted.
The morphologically unique Males II also showed higher carotenoid and lower lipid contents, both per individual and per gram of wet weight. Geographically, population composition plays an important role in total carotenoid and total lipid contents, which depend on their particular morphology and reflect different swimming capacities.
The large size of E. superba allows obtaining measurable differences between individuals and sexes on both morphometric and biochemical composition. The high carotenoid:lipid ratio reflects the unique physiology of Males II and the likely protective role of carotenoids in euphausiids.
The Principal Components Analysis on 10 variables shows mostly differences between sex stages in morphometrics and the biochemical composition of individuals, particularly for Males II.
In view of previous results, we consider that the most realistic hypothesis to explain the higher carotenoid and lower lipid contents observed in our study is seemingly that of stressed animals that will die after fecundation.
The carotenoid:lipid ratio shows higher values in Males II, suggesting a distinctive physiological profile during the reproductive season.
However, too many uncertainties remain, limiting the strength of the conclusions about the physiological role of the higher carotenoid content in Males II.
Data availability
All data are available from the authors upon request.
References
Mauchline, J. & Fisher, L. R. The biology of Euphausiids. Adv. Mar. Biol.7, 454 pp (1969).
Mauchline, J. The biology of mysids and euphausiids. Adv. Mar. Biol.18, 370–680 (1980).
Makarov, R. R., & Denys, C. J. Stages of sexual maturity of Euphausia superba Dana. SCAR, SCOR; IABO, ACMRR, Biomass Handbook, 11, 13 pp. (1979).
Siegel, V. Relationships of various length measurements of Euphausia superba Dana. Meeresfrosch.29, 114–117 (1982).
Miller, D. G. M. Variation in body length measurement of Euphausia superba Dana. Pol. Biol.2, 17–20 (1983).
Endo, Y. Allometric differences observed on the same sized mature males of the Antarctic krill (Euphausia superba Dana). Bull. Plank. Soc. Jap.36, 5–10 (1989).
Färber-Lorda, J. Etudes Biologiques, Energétiques et Biochimiques du krill antarctique (Euphausia superba et Thysanoessa macrura), récolté au cours de la campagne FIBEX (fevrier 1981). PH. D. Thesis. 214 pp (1986). - Title in English: Biological, Energetic and Biochemical studies on Antarctic krill (Euphausia superba and Thysanoessa macrura), collected during the FIBEX cruise (Febbruary 1981). PH. D. Thesis, 214 pp (1986).
Färber-Lorda, J. Somatic length relationships and ontogenetic morphometric differentiation of Euphausia superba and Thysanoessa macrura of the southwest Indian Ocean during summer (February 1981). Deep-Sea Res.37, 1135–1143 (1990).
Färber-Lorda, J. Multivariate approach to the morphological and biochemical differentiation of Antarctic krill (Euphausia superba and Thysanoessa macrura). Deep-Sea Res.38, 771–779 (1991).
Ettershank, G. A new approach to the assessment of longevity in in the Antarctic krill Euphausia superba. J. Crust. Biol.4(Spec 1), 295–305 (1984).
Färber-Lorda, J. Length Vs, weight relationships and coefficient of condition of Euphausia superba and Thysanoessa macrura of the SW Indian ocean during summer. Mar. Biol.118, 645–650 (1994).
Färber-Lorda, J., Beier, E. & Mayzaud, P. Spatial approach to the morphological and biochemical differentiation of Antarctic krill. J. Mar. Syst.78, 518–524 (2009).
Kils, W. The swimming behaviour, swimming performance and energy balance of Antarctic krill Euphausia superba. Biomass Sci. Ser.3, 121 (1981).
Johnson, M. L. & Tarling, G. Influence of individual state on swimming capacity and behaviour of Antarctic krill Euphausia superba. Mar. Ecol. Prog. Ser.366, 99–110 (2008).
Watkins, J. L., Buchholz, F., Priddle, J., Morris, D. J. & Rickettes, C. Variation in reproductive status of Antarctic krill swarms; evidence for a size-related sorting mechanism?. Mar. Ecol. Prog. Ser.82, 163–174 (1992).
Krafft, B. A., Skaret, G. & Knutsen, T. An Antarctic krill (Euphausia superba) hotspot: population characteristics, abundance and vertical structure explored from a krill fishing vessel. Polar Biol.38, 1687–1700 (2015).
Kawaguchi, S. et al. Male krill grow fast and die young. Mar. Ecol. Prog. Ser.345, 199–210 (2007).
Ikeda, T. & Dixon, P. Body shrinkage as a possible overwintering mechanism of the Antarctic krill, Euphausia superba Dana. J. Exp. Mar. Biol. Ecol.62, 143–151 (1982).
Tarling, G. A. et al. Growth and shrinkage in Antarctic krill Euphausia superba is sex-dependent. Mar. Ecol. Prog. Ser.547, 61–78 (2016).
Raymont, J. E. G., Srinivasagam, R. T. & Raymont, J. K. B. Biochemical studies on marine zooplankton. IX. The biochemical composition of Euphausia superba. J. Mar. Biol. Assoc. U. K.51, 581–588 (1971).
Bottino, N. R. Lipid composition of two species of Antarctic krill: Euphausia superba and E. crystallorophias. Com. Biochem. Physiol.50, 479–484 (1975).
Clarke, A. The biochemical composition of krill Euphausia superba, Dana, from South Georgia. J. Exp. Mar. Biol. Ecol.43, 221–236 (1980).
Clarke, A. Lipid content and composition of Antarctic krill Euphausia superba Dana. J. Crust. Biol.4(Spec 1), 285–294 (1984).
Pond, D., Watkins, J., Priddle, J., & Sargent, J. Variation in the lipid content and composition of Antarctic krill Euphausia superba at South Georgia. Mar. Ecol., Prog. Ser. 117, 49–57 (1995).
Mayzaud, P., Albessard, E. & Cuzin-Roudy, J. Changes in lipid composition of the Antarctic krill Euphausia superba in the Indian sector of the Antarctic Ocean: influence of geographical location, sexual maturity stage and distribution among organs. Mar. Ecol. Prog. Ser.173, 149–162 (1998).
Mayzaud, P., Albessard, E. & Cuzin-Roudy, J. Changes in lipid composition of the Antarctic krill Euphausia superba in the Indian sector of the Antarctic Ocean. J. Mar. Syst.78, 525–535 (1998).
Falk-Petersen, S., Hagen, W., Kattner, G., Clarke, A. & Sargent, J. Lipids, trophic relationship, and biodiversity in Arctic and Antarctic krill. Can. J. Fish. Aquat. Sci.57, 178–191 (2000).
Cripps, G. C. & Hill, H. J. Changes in lipid composition of copepods and Euphausia superba associated with diet and environmental conditions at the marginal ice zone. Deep-Sea Res.145, 1357–1381 (1998).
Pond, D. W. The physical properties of lipids and their role in controlling the distribution of zooplankton in the oceans. J. Plank. Res.34, 443–453 (2012).
Fernandez, M. A. & Färber-Lorda, J. Zooplankton and the oceanography of the eastern tropical pacific: a review. Prog. Oceanogr.69, 318–359 (2006).
Hunstman, A. G. Limiting factors of marine animals 1. The lethal effects of sunlight. Contr. Can. Biol. 2, 83–87 (1924).
Williamson, C. E., Sanders, R. W., Moeller, R. E. & Stutzman, P. L. Utilization of subsurface resources for zooplankton reproduction: implications for diel vertical migration theory. Limnol. Oceanogr.41, 224–233 (1996).
Kon, S. K. & Thompson, S. Y. Preformed vitamin A in northern krill. Biochem. J.45, 31 (1949).
Kon, S. K. & Thompson, S. Y. Preformed vitamin A in crustacean. Archs. Biochem.24, 233–234 (1949).
Czerpak, R., Jackowska, H. & Mical, A. Qualitative analysis of carotenoids in particular parts of body of males and females of Euphausia superba Dana (Crustacea). Pol Polar Res.1, 139–145 (1980).
Maoka, T., Katsuyama, M., Kaneko, N. & Matsuna, T. Stereochemical investigation of carotenoids in the Antarctic krill Euphausia superba. Bull. Jap. Soc. Sci. Fish.5, 1671–1673 (1985).
Funk, V. A. & Hobson, L. A. Temporal variations in the carotenoid composition and content of Euphausia pacifica Hansen in the Saanich Inlet British Columbia. J. Exp. Mar. Biol. Ecol.148, 93–104 (1991).
Virtue, P., Johannes, R. E., Nichols, P. D. & Young, J. W. Biochemical composition of Nyctiphanes australis and its possible use as an aquaculture feed source: lipids, pigments and fluoride content. Mar. Biol.122, 12–128 (1995).
Takaichi, S., Matsui, K., Nakamura M., Muramatsu, M., & Hanad, S. Fatty acids of astaxanthin esters in krill determined by mild mass spectrometry. Comp. Biochem. Physiol. Part B. Biochem. Mol. Biol. 136, 317–322 (2003).
Grynbaum, M. D. et al. Unambiguous detection of astaxanthin and astaxanthin fatty acid esters in krill (Euphausia superba, Dana). J. Sep. Sci.28, 1685–1693 (2005).
Auerswald, L., Freier, U., Lopata. A., Meyer, B. Physiological and morphological colour change in Antarctic krill, Euphausia superba: a field study in Lazarev Sea. The J. Exp. Biol.211, 3850–3858 (2008).
Wagner, K. H. Vitamin A und ß-carotin des fin blau, und spermwals. J. A. Barth, Leipzig (1939). (Vitamin A and β‐Carotene in fin whale, blue whale and sperm whale.)
Fisher, L. R., Kon, S. K. & Thompson, S. Y. Vitamin A and carotenoids in certain invertebrates. I. Marine Crustacea. J. Mar. Biol. Ass. U. K.31, 229–258 (1952).
Fisher, L, R., Kon, S. K. & Thompson, S. Y. Vitamin A and carotenoids in certain invertebrates. II. Studies of seasonal variations in some marine Crustacea. J. Mar. Biol. Ass. U. K.33, 589–612 (1954).
Jarman, S., Elliott, N., Nicol, S., McMinn, A. & Newman, S. The base composition of krill genome and its potential susceptibility to damage by UV-B. Antarct. Sci.11, 23–26 (1999).
Newman, S. J., Nicol, S., Ritz, D. & Marchant, H. Susceptibility of Antarctic krill (Euphausia superba Dana) to ultraviolet radiation. Pol. Biol.22, 50–55 (1999).
Newman, S. J., Nicol, S. & Ritz, D. Behavioural reactions of Antarctic krill (Euphausia superba Dana) to ultraviolet and photosynthetically active radiation. J. Exp. Mar. Biol. Ecol.297, 203–217 (2003).
Babak, E. Über den Einfluss des Lichtes auf die Vermehrung der Hautchromatophoren. Arch. Ges. Physiol. 149, 462–470 (1913). (About the influence of light on the increase in skin-chromatophores).
Bligh, E. G. & Dyer, W. J. A rapid method for total lipid extraction and purification. Can. J. Biochem. Physiol.37, 911–917 (1959).
Jackowska, H., Czerpak, R. & Mical, A. Carotenoids of Antarctic krill (Euphausia superba Dana, Euphausia chrystallorophias, Holt et Tattersall) in dependence on age, structure and sex. Pol. Arch. Hydrobiol.27, 291–303 (1980).
-Pande, S. V., Parvin Khan, R. Venkitasubramanian, T. A. Micro determination of lipids and serum total fatty acids. Analyt. Biochem. 6, 414–423 (1963).
Virtue, P., Nichols, P. D., Nicol, S. & Hosie, G. Reproductive trade-off in male Antarctic krill Euphausia superba. Mar. Biol.126, 521–527sssss (1996).
Reiss, C. S. Age, growth, mortality, and recruitment of antarctic krill, Euphausia superba. In: Siegel, V. (Ed.). Springer. Biology and Ecology of Antarctic krill. Adv. Polar Ecol. Vol. 1. 101–144 (2016).
Tarling, G. A. Sex-dependent diel vertical migration in northern krill Meganyctiphanes norvegica and its consequences for population dynamics. Mar. Ecol. Prog. Ser.260, 173–188 (2003).
Kils, U. Swimming speed and escape capacity of Antarctic krill Euphausia superba. Meeresforsch.27, 264–266 (1979).
Hamner, W. M. Aspects of schooling of Euphausia superba. J. Crust. Biol. 4 (Special Issue): 67, 74 570 (1984). doi 10.1163/1937240x84x00507
Hamner, W. M. & Hamner, P. P. Behavior of Antarctic krill (Euphausia superba): schooling, foraging, and antipredatory behavior. Can. J. Fish. Aquat. Sci.57, 192–202. https://doi.org/10.1139/cjfas-57-S3-192 (2000).
Tarling, G. A. & Thorpe, E. Instantaneous movement of krill swarms in the antarctic circumpolar current. Limnol. Oceanogr.59, 872–886. https://doi.org/10.4319/lo.2014.59.3.0872 (2014).
Kils, U. Swimming and feeding of Antarctic krill, Euphausia superba. Some outstanding energetics and dynamics, some unique morphological details. Workshop on Krill biology. (1983).
Ceccaldi, H. J. Recherches sur la biologie des associations entre protéines et caroténoîdes chez les Crustacés Décapodes: aspects métaboliques et moléculaires. PH. D. Thesis 479 pp. (1968). (Study on the biology of the relationships between protein and carotenoids in Decapod Crustaceans : metabolic and molecular aspects).
Zagalsky, P. F., Ceccaldi, H. J. & Daumas, R. Comparative studies on some decapod crustacean carotenoproteins. Comp. Biochem. Physiol.34, 579–607 (1970).
Otazu Abrill, M., & Ceccaldi, H. J. Variations circadiennes des pigments caroténoides dans les yeux et l'hépatopancréas de Penaeus japonicus (Crustacé, Décapode). C.R. Soc. Biol.172, 684-690 (1978). (Diel variations of carotenoid pigments in the eyes and hepatopancreas of Penaeus japonicus (Crustacea, Decapoda
Ceccaldi, H.J. Problèmes posés par les variations chronobiologiques de la physiologie des Crustacés. Bull. Soc. Ecophysiol. 16, 87 – 95 (1981). (Issues related to the chrono-biological variations in the physiology of Crustaceans).
Czeczuga, B. Studies on carotenoproteins in animals. Euphausia superba Dana1852 (Crustacea, Euphausiacea). Pol. Polar Res. 5:121–127 (1984b).
Karnaukhov, V. N. On the nature and function of yellow aging pigment lipofuscin. Exp. Cell. Res.80, 479–483 (1973).
Karnaukhov, V. N. & Fedorov, G. G. The role of carotenoids and vitamin A in animal’s adaptation to high altitude. Comp. Biochem. Physiol.57, 377–381 (1977).
McGaffin, A. F. et al. Validation and quantification of extractable age pigments for determining the age of Antarctic krill (Euphausia superba). Mar Biol. https://doi.org/10.1007/s00227-011-1688-5 (2011).
Bluhm, B. A., Brey, T., Klages, M. & Arntz, W. E. Occurrence of the autofluorescent pigment, lipofuscin, in polar crustaceans and its potential as an age marker. In Ecological Studies in the Antarctic Sea Ice Zone (eds Arntz, W. E. & Clarke, A.) (Springer, Berlin, 2002).
Thomas, P. G. & Ikeda, T. Sexual regression, shrinkage, re-maturation and growth of spent female Euphausia superba in the laboratory. Mar. Biol.95, 357–363 (1987).
Brown, M., Kawaguchi, S., King, R., Virtue, P. & Nicol, S. Flexible adaptation of the seasonal krill maturity cycle in the laboratory. J. Plankt. Res.33, 821–826 (2011).
Kawaguchi, S., Yoshida, T., Finley, L., Cramp, P. & Nicol, S. The krill maturity cycle: a conceptual model of the seasonal cycle in Antarctic krill. Polar Biol.30, 689–698 (2007).
Tarling, G. A. et al. Variability and predictability of Antarctic krill swarm structure. Deep-Sea Res.I(56), 1994–2012 (2009).
Ritz, D. A. Is social aggregation in aquatic crustaceans a strategy to conserve energy?. Can. J. Fish. Aquat. Sci.57, 59–67 (2000).
Rosenthal, R. & Rubin, D. B. r equivalent: A Simple Effect Size Indicator. Psych. Mets.8, 492–496 (2003).
Cohen, J. Statistical Power Analysis for the Behavioral Sciences 2nd edn. (Lawrence Erlbaum Associates, Publishers, Hillsdale, NJ, 1988).
Acknowledgments
We wish to thank P. Mayzaud (CNRS, Villefranche-Sur- Mer, France) for his advice on the analyses of lipids and other biochemical work. To J. Cuzin-Roudy (CNRS, Villefranche-Sur-Mer, France) for the use of her measurement microscope. To Ofir Molina (CICESE) for assistance with the figures. Thanks also to Enrique Villa Diharce from CIMAT, Guanajuato, México, for his assistance with the statistical analyses, and to Cesar Almeda Jáuregui (CICESE) for his assistance with principal components analyses. María Elena Sánchez-Salazar edited the English manuscript.
Author information
Authors and Affiliations
Contributions
J. F. L. conducted all the experimental work under the supervision of H. J. C., who contributed to this work with his counsel and advisory on methods and ideas.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Färber Lorda, J., Ceccaldi, H.J. Relationship of morphometrics, total carotenoids, and total lipids with activity and sexual and spatial features in Euphausia superba. Sci Rep 10, 13177 (2020). https://doi.org/10.1038/s41598-020-69780-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-69780-8
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.