Abstract
The production of electricity and heat in Poland is the reason why the commercial power industry is the largest emitter of CO2. At the same time, significant amounts of solid by-products of combustion, which can be used to bind CO2 by mineral carbonation, are generated during the production processes. The article presents the results of research on mineral sequestration of CO2 (suspension-CO2) using fluidized bed combustion (FBC) fly ashes from hard coal combustion. The analyzed fluidized bed combustion (FBC) fly ashes were characterized by a significant free CaO content (1.7–6.8%) and a high CO2 binding potential ranging from 9.7 to 15.7%. In the case of fluidized bed combustion (FBC) fly ashes suspensions, the basic product of the carbonation process is calcium carbonate, which is clearly indicated by the results of the phase composition determination of solidified suspensions of fluidized bed combustion (FBC) fly ashes. The degree of carbonation, i.e. the degree of CO2 binding, calculated on the basis of the calcium carbonate content, in the analyzed suspensions was up to 1.1%. Mineral carbonation also reduces the leachability of pollutants such as: Zn, Cu, Pb, Ni, As, Hg, Cd, Cr, Cl, and SO42-. The pH is also reduced from about 12 to about 9. Aqueous suspensions of fluidized bed combustion (FBC) fly ashes with introduced CO2 can potentially be used in underground mining. These activities are in line with the concepts of Carbon Capture and Utilization and the idea of circular economy.
Similar content being viewed by others
Introduction
The recent anthropogenic greenhouse gas emissions are the highest in history and are probably the main cause of the climate change observed since the mid-20th century - the so-called global warming. A particular role is played by CO2 emissions from fossil fuel combustion and industrial processes, which in the period from 1970 to 2010 accounted for approximately 78% of the total increase in greenhouse gas emissions1.
The commercial power industry is the largest emitter of carbon dioxide in Poland (Table 1). This is due to the fact that coal is the primary fuel used in the power industry, which is conditioned by the abundant resources of this energy source in Poland. Due to these conditions, it will be difficult to reduce CO2 emissions from energy production. One of the possibilities to reduce CO2 emissions is mineral sequestration using waste generated in the same energy production process.
The mineral sequestration of CO2 is a method that, due to the stable binding of CO2, is an alternative to geological storage.
Mineral sequestration is a method of reducing CO2 emissions, in which fly ashes from coal combustion play a special role. They are produced in the same energy production process; mineral sequestration may allow their economic use.
The waste products which should primarily be taken into account when it comes to CO2 binding are the fly ashes with a high content of free CaO2.
Studies on the suitability of fly ashes from coal combustion for reducing CO2 emissions by mineral carbonation are carried out worldwide by different authors using different process conditions (Table 2).
Unfortunately, the existing literature lacks information on the type of boilers, which directly affects the properties of by-products of coal combustion.
The analyses are carried out using fly ashes and water suspensions and dry fly ashes in direct and indirect carbonation.
FBC fly ashes are waste materials that, due to the high content of free CaO, can be used to reduce CO2 emissions by mineral carbonation. The high CaO content in fluidized ashes is due to the fact that the coal combustion process is integrated with the desulfurization process. Fluidized ashes contain, in addition to components derived from coal combustion, desulfurization products and sorbent residues.
Mineral carbonation studies carried out using fly ashes from combustion in fluidized bed boilers indicate the high potential of this waste (Table 3). However, there has been relatively little research focused on this topic. The work presented in this paper contributes to the research in this area by analyzing fly ashes from coal combustion.
The article presents the results of studies on the use of mineral sequestration using aqueous suspensions of fluidized bed combustion (FBC) fly ashes from hard coal combustion in order to reduce CO2 emissions.
The research results presented in the article are a new approach to the analysis of CO2 binding by aqueous fly ashes suspensions as a method of reducing the emission of this harmful greenhouse gas as well as allow the recovery of fly ashes from coal combustion in fluidized bed boilers. As shown in Table 3, little research focused on the use of fly ashes from fluidized bed boilers.
In an attempt to reduce pollution to the atmosphere, while retaining efficient fuel combustion, more and more power plants and heat and combined heat and power plants install fluidized bed boilers.
In recent years, the number of boilers in power plants and combined heat and power plants using fluidized bed combustion has increased. In 1998 in Poland, there were only two fluidized-bed boilers used in the commercial power industry, while there were already 11 in 2003 and 27 in 20163.
In Poland, FBC fly ashes are now used mainly in underground mining, in the form of aqueous suspensions, and in road construction4.
Fly ashes from coal combustion in the form of water suspension have been used in underground mining since the 1980s e.g. for sealing underground voids6.
The use of FBC fly ashes for CO2 binding by mineral carbonation and depositing them in underground mines5,6,7 is in line with the idea of closed-loop recycling.
As a result of the energy production process from coal extracted in mines are generated - fly ashes and CO2, which can potentially be used in suspension technology and deposited in mines5,6,7, thus closing the waste and CO2 circulation (Fig. 1).
It is the implementation of cleaner production technologies.
The mineral sequestration in fly ashes-water suspensions is a particularly interesting method because the basic product formed in the process - CaCO3 is a component of the hardened suspension. Therefore, fly ashes from the energy sector can be recovered, while CO2 can be disposed of, and thus the problem of using by-products of the combustion process – fly ashes is solved5.
However, the use of suspensions subjected to the mineral sequestration, which could potentially be used in mining, requires knowledge of the degree of binding of CO2 based on determining the increase in the amount of CaCO3 as the basic product of the carbonation process. Determining the amount of CaCO3 produced in the carbonation process allows estimating how much CO2 can be permanently bound in hardened suspensions and successfully used in underground mining technologies.
The problem of reducing CO2 emissions is also very important due to the increasing EUA prices, which in February 2017 stood at EUR 4.90 and in September 2019 at EUR 27.038.
Experimental
Research methodology
FBC fly ashes used in the study were obtained from the combustion of aqueous suspensions with the following fly ashes to water weight ratios: PF 1−0.9; PF 2−0.6; PF 3–0.8; PF 4–0.7.
When determining the degree of mineral sequestration of CO2, the phase composition was tested using differential thermal analysis and the calcite content in the suspension was determined by the thermogravimetric analysis.
A complementary study on the leachability of pollutants from fly ashes-water suspensions was carried out in order to determine effects of their exposure to CO2. The suspensions were exposed to CO2 in installation consisting of research chambers, recording equipment, CO2 cylinders, and a regulator5,7.
The analysis of phase composition (DTA) and the analysis of calcium carbonate content (TG) were carried out in the atmosphere with the heating rate of 10 °C·min−1.
The microstructural analysis of fly ashes-water suspensions was carried out using a JEOL scanning electron microscope equipped with an Oxford Instruments EDS 540 system.
In determining the impact of CO2 on the properties of waste-aqueous suspensions, a series of tests was performed to determine the leachability of: chlorides (Cl−), sulfates (SO42−), arsenic (As), chromium (Cr) (total), cadmium (Cd), copper (Cu), lead (Pb), nickel (Ni), zinc (Zn) and mercury (Hg), chemical oxygen demand (COD), and pH.
The content of arsenic, chromium, cadmium, copper, lead, nickel, zinc, arsenic, and mercury in aqueous solutions was analyzed using two methods: ICP AES and plasma mass spectrometry. The chemical oxygen demand (COD) was tested in accordance with the PN-74 C-04578/03 standard. Chloride content was determined using Volhard method and sulfate content using atomic emission spectrometry with inductively coupled plasma. The obtained results were compared with values defined in the PN-G-11011 standard: Materials for Backfilling and Caulking of Cavings – Requirements and Tests (due to the fact that fly ashes from fluidized bed boilers are primarily used in the mining industry).
Materials used in the study
FBC fly ashes from coal combustion characterized by a high content of CaO and free CaO (Table 4) were selected for the analysis.
The study used fly ashes (FBC fly ashes) from the following fluidized bed boilers:
CFB boiler, hard coal-fired boiler, fluid circulation boiler, atmospheric furnace boiler, natural circulation boiler, and single drum boiler − PF 1,
OF type, hard coal-fired, two-pass boiler, atmospheric furnace with circulating fluidized bed − PF 2,
OF type, circulating fluidized bed − PF 3.
The analyzed fly ashes were characterized by high content of CaO and free CaO (Table 3).
For the analyzed fly ashes, the maximum capacity of binding was calculated using Steinour formula9:
The results
Phase composition and calcium carbonate content
To determine the usefulness of the analyzed suspensions for sequestration of CO2 through mineral carbonation, the thermogravimetric and differential analyses were carried out.
In order to confirm the occurrence of mineral carbonation processes, and thereby CO2 binding by fly ashes-water suspensions, their phase composition was analyzed, and the content of calcium carbonate, the main product of carbonation, was determined.
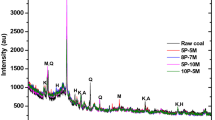
The DTA curves of suspensions with FBC fly ashes PF 1 (Fig. 2) show four distinct endothermic effects with the maximums at:
82 °C, which can be attributed to the presence of ettringite,
124 °C, characteristic for the hydrated phases (silicates, aluminates, and calcium sulfoaluminates),
441 °C associated with Ca(OH)2,
711 °C associated with decomposition of calcium carbonate and the exothermic effect with the maximum at 568 °C, probably associated with the presence of unburned carbon.
The analysis of suspensions of PF 1 fly ashes treated with CO2 has not shown the presence of an endothermic effect associated with the presence of silicates, aluminates, and calcium sulfoaluminates. As can be seen from the DTA curves (Fig. 2), there is a very slight decrease in the content of Ca(OH)2 and a slight increase in the effect associated with the decomposition of CaCO3 for suspensions treated with CO2, which confirms the low degree of carbon dioxide binding.
Based on the DTA curves (Fig. 3), the following phases were found in suspensions with PF 2 fly ashes (clean and treated with CO2):
Ettringite and C-S-H (effects with maximums at 92, 108, 145, and 140 °C);
Unburned carbon (with maximums at 501 and 523 °C)
Calcium carbonate (with the maximum at 697 and 705 °C).
The tested suspensions prepared from PF 3 fly ashes (Fig. 4) are characterized by the presence of the following phases:
Hydrated phases of silicates, aluminates, and calcium sulfoaluminates (effects with maximums at 121 and 122 °C);
Unburned carbon (with the maximum at 527 and 533 °C);
Calcium carbonate (with the maximum at 750 and 752 °C).
All of the analyzed suspensions contain hydrated phases of silicates, aluminates, calcium sulfoaluminates, and calcium carbonate. In addition, suspensions with PF 1 and PF 2 fly ashes contain unburned carbon.
The microstructural analysis confirmed the results of thermogravimetric analysis. The image of the PF 2 sample (Fig. 5) is dominated by the hydration products occurring on the surface of ashes particles; the surface is covered with ettringite crystals; visible C-S-H phase.
In Fig. 6 (sample PF 3) the surface of the sample is covered with a layer of prismatic ettringite crystals underlain by gel forms of silicates and aluminates.
Effect of CO2 on pollutants leachability
Due to the fact that FBC fly ashes are used primarily in mines in the suspension technology, the obtained results were compared with requirements contained in the PN-G-11011 standard: Materials for Backfilling and Caulking of Cavings – Requirements and Tests (Table 5).
Concentrations of pollutants determined by leachability tests mostly do not exceed the limit values specified in the PN-G-11011 standard: Materials for Backfilling and Caulking of Cavings – Requirements and Tests. The only exceptions are sulfates whose value exceeds the limit of 500 mg/dm3 (for suspensions with PF 2 and PF 3) and pH values (for PF1 suspension).
A reduction of leachability in the majority of the analyzed pollutants has been observed in all of the analyzed suspensions. The exception is the leachability of SO4, which increased for the PF 2 and PF 3 fly ashes.
Discussion
The degree of CO2 binding in the analyzed suspensions was calculated on the basis of thermogravimetric analysis (Table 6, Figs. 1−3) based on the difference in calcium carbonate content using following formula10:
Where: CO2uptake− the extent of carbonation, CO2initials− initial carbonate content of the sample, CO2final− final carbonate content of the sample.
\(C{O}_{2uptake}\)
\({{\rm{CO}}}_{2{\rm{initials}}}\)
\({{\rm{CO}}}_{2{\rm{final}}}\).
The highest content of CaCO3 and the same degree of carbonation has been observed in the case of suspensions with PF 2 fly ashes, while the lowest degree of binding was shown by PF1.
The leachability is the result of interdependent processes11. The basic reaction of carbonation, the reaction of Ca(OH)2 with carbon dioxide, as a result of which calcite is formed, lowers the pH. In the case of the analyzed fly ashes, it lowers the pH from about 11 to about 8 (Table 5).
The reduction of leachability of Cr and Pb (found in all suspensions) is probably due to the pH change, which after carbonation is close to the pH of the solution with the minimum solubility of Cr and Pb. The leachability is also reduced by the sorption of metals in the newly formed minerals. They can also form complexes with iron and aluminium hydroxides and oxides. The reduction of leachability of Pb and Cr ions may also be caused by the formation of new oxides or sulfates12.
In the case of the reduction of leachability of Zn, Cr, and Pb, recorded for all analyzed suspensions based on the presence of C-S-H, it can be explained by their immobilization by C-S-H13,14.
The increase in leachability of SO42− (suspensions of PF 1, PF 2, and PF 3) may be due to carbonation which causes decomposition of ettringite and results in the formation of well-soluble CaSO415.
The reduced leachability of arsenic is explained by adsorption and coprecipitation to form solid solution with calcite16. The reduction of Cu leachability is explained by copper carbonate formation17.
In the case of carbonation, an important factor in lowering leachability of some heavy metal ions (Cd, Zn, Mn, Co, Ni, Pb, or Sr) and ions of SO42− is their sorption on calcite, leading to coprecipitation18,19.
The use of FBC fly ashes in mining ‒ potential for sequestration
The mineral sequestration of CO2 using FBC fly ashes from hard coal is a particularly interesting option for southern Poland, where the potential for geological storage is limited20.
FBC fly ashes from hard coal combustion have been used in Poland for many years, mainly in mining, construction materials, and road construction4.
According to estimates from 2013, the amount of CO2 that can be disposed of using a mixture of fly ashes and solid waste from calcium flue gas desulfurization ‒ FBC fly ashes, assuming the use of waste already used in the mining industry and unused waste, is 11.3 Gg of CO2 per year21.
In 2016, 1,142,733 Mg of by-products of combustion were used in coal mines, of which 1,129,795 Mg were fly ashes (99.87%). It is estimated that 1,122,000 tons of by-products of combustion will be used in 202022. Assuming that the FBC fly ashes will be half of the by-products of combustion used in underground mining, i.e. 561,000 tons, the average potential amount of bound CO2 for the maximum binding capacity of CO2 will constitute 6,844.2 tons (12.2%), while the average maximum binding capacity of CO2 will amount to 5,329.5 tons (9.5%).
Taking into account the degree of carbonation, a maximum of 1,683 tons of CO2 can be bound for the degree of binding of 0.3 and 6,171 tons of CO2 for the degree of binding of 1.1.
Conclusions
Fluidized bed combustion (FBC) fly ashes have high content of CaO and free CaO, which makes them a good candidate for CO2 binding through mineral carbonation.
The studies confirmed the occurrence of the carbonation process. The main product of the mineral carbonation process is calcite. An additional effect is lowering the pH in the course of the process. It has been confirmed that the carbonation process affects the leachability of pollutant. The highest degree of binding was shown by PF 2 fly ashes. In all cases, the presence of hydrated silicates, aluminates, and calcium sulfoaluminates has been confirmed.
Fluidized bed combustion (FBC) fly ashes are also interesting material for CO2 binding because they are used in mines (suspension technology). The large-scale use of the suspension technology in the Polish underground mining may make it feasible to use it for CO2 binding, because of mines’ enormous experience5,6. This solves the problem of utilization of carbonation products. This way the waste will be managed and CO2 emission reduced.
References
IPCC. Climate Change 2014. Climate Change 2014: Synthesis Report https://doi.org/10.1017/CBO9781107415324 (2014).
Back, M., Kuehn, M., Stanjek, H. & Peiffer, S. Reactivity of alkaline lignite fly ashes towards CO2 in water. Environ. Sci. Technol. 42, 4520–4526 (2008).
Emitor 1997–2018. The emission of environmental pollution in power plants and in combined heat and power plants. (2018).
Uliasz-Bocheńczyk, A., Mazurkiewicz, M. & Mokrzycki, E. Fly ash from energy production – A waste, byproduct and raw material. Gospod. Surowcami Miner./Miner. Resour. Manag. 31, (2015).
Uliasz-Bocheńczyk, A., Mokrzycki, E., Mazurkiewicz, M., Piotrowski, Z. & Pomykala, R. CO2 Utilization within Ash–Water Mixtures Deposited in Underground Coal Mines. in Carbon Dioxide Sequestration in Geological Media – State of the Science. AAPG Studies in Geology No. 59 655–663 (2009).
Uliasz-Bochenczyk, A., Mokrzycki, E., Mazurkiewicz, M. & Piotrowski, Z. Utilization of carbon dioxide in fly ash and water mixtures. Chem. Eng. Res. Des. 84 (2006).
Uliasz-Bocheńczyk, A., Mokrzycki, E., Piotrowski, Z. & Pomykala, R. The underground storage of CO 2 with ash-water suspension. (Publishing House of Mineral and Energy Economy Research Institute of the Polish Academy of Sciences. (2007).
Report on the CO 2 market. No 90. (2019).
Fernández Bertos, M., Simons, S. J. R., Hills, C. D. & Carey, P. J. A review of accelerated carbonation technology in the treatment of cement-based materials and sequestration of CO2. J. Hazard. Mater. https://doi.org/10.1016/j.jhazmat.2004.04.019 (2004).
Baciocchi, R., Costa, G., Polettini, A., Pomi, R. & Prigiobbe, V. Comparison of different reaction routes for carbonation of APC residues. in Energy Procedia 1, 4851–4858 (2009).
Polettini, A. & Pomi, R. Leaching behaviour of incineration bottom ash by accelerated ageing. J. Hazard. Mater. B113, 209–215 (2004).
Rendek, E., Ducom, G. & Germain, P. Carbon dioxide sequestration in municipal solid waste incinerator (MSWI) bottom ash. J. Hazard. Mater. https://doi.org/10.1016/j.jhazmat.2005.07.033 (2006).
Deja, J. Immobilization of Cr6+, Cd2+, Zn2+ and Pb2+ in alkali-actived slag binders. Cem. Concr. Res. 32, 1971–1979 (2002).
Giergiczny, Z. & Król, A. Immobilization of heavy metals (Pb, Cu, Cr, Zn, Cd, Mn) in the mineral additions containing concrete composites. Journal of Hazardous Materials https://doi.org/10.1016/j.jhazmat.2008.03.007 (2008).
Todorovic, J. & Ecke, H. Demobilisation of critical contaminants in four typical waste-to-energy ashes by carbonation. in Waste Management. https://doi.org/10.1016/j.wasman.2005.11.011 (2006).
Román-Ross, G., Cuello, G. J., Turrillas, X., Fernández-Martínez, A. & Charlet, L. Arsenite sorption and co-precipitation with calcite. Chem. Geol. https://doi.org/10.1016/j.chemgeo.2006.04.007 (2006).
Costa, G. et al. Current status and perspectives of accelerated carbonation processes on municipal waste combustion residues. Environmental Monitoring and Assessment https://doi.org/10.1007/s10661-007-9704-4 (2007).
Reeder, R. J. Interaction of divalent cobalt, zinc, cadmium, and barium with the calcite surface during layer growth. Geochim. Cosmochim. Acta https://doi.org/10.1016/0016-7037(96)00034-8 (1996).
Tesoriero, A. J. & Pankow, J. F. Solid solution partitioning of Sr2+, Ba2+, and Cd2+ to calcite. Geochim. Cosmochim. Acta https://doi.org/10.1016/0016-7037(95)00449-1 (1996).
Uliasz-Misiak, B. & Przybycin, A. Present and future status of the underground space use in Poland. Environ. Earth Sci. 22, 1–15 (2016).
Uliasz-Bocheńczyk, A. & Mokrzycki, E. Mineral sequestration of CO2 with the use of energy waste – An attempt to estimate the Polish potential. Gospod. Surowcami Miner./Miner. Resour. Manag. 29 (2013).
Drobek, L., Kanafek, J. & Pierzyna, P. Utilisation of ccps in hard-coal mines – current state, prospects for years 2016-20, technical and environmental aspects. in XXIII International Conference – Ashes From Power Generation (ed. Kornacki, A.) 225–237 (2016).
Ebrahimi, A. et al. Sustainable transformation of fly ash industrial waste into a construction cement blend via CO2 carbonation. J. Clean. Prod. https://doi.org/10.1016/j.jclepro.2017.04.037 (2017).
Ji, L. et al. Insights into Carbonation Kinetics of Fly Ash from Victorian Lignite for CO2 Sequestration. Energy and Fuels. https://doi.org/10.1021/acs.energyfuels.7b03137 (2018).
Noack, C. W. et al. Comparison of alkaline industrial wastes for aqueous mineral carbon sequestration through a parallel reactivity study. Waste Manag. https://doi.org/10.1016/j.wasman.2014.03.009 (2014).
Montes-Hernandez, G., Pérez-López, R., Renard, F., Nieto, J. M. & Charlet, L. Mineral sequestration of CO2 by aqueous carbonation of coal combustion fly-ash. J. Hazard. Mater. https://doi.org/10.1016/j.jhazmat.2008.04.104 (2009).
Reynolds, B., Reddy, K. & Argyle, M. Field Application of Accelerated Mineral Carbonation. Minerals. https://doi.org/10.3390/min4020191 (2014).
Siriruang, C., Toochinda, P., Julnipitawong, P. & Tangtermsirikul, S. CO2 capture using fly ash from coal fired power plant and applications of CO2-captured fly ash as a mineral admixture for concrete. J. Environ. Manage. https://doi.org/10.1016/j.jenvman.2016.01.010 (2016).
Mazzella, A., Errico, M. & Spiga, D. CO2 uptake capacity of coal fly ash: Influence of pressure and temperature on direct gas-solid carbonation. J. Environ. Chem. Eng. https://doi.org/10.1016/j.jece.2016.09.020 (2016).
Ukwattage, N. L., Ranjith, P. G., Yellishetty, M., Bui, H. H. & Xu, T. A laboratory-scale study of the aqueous mineral carbonation of coal fly ash for CO2 sequestration. J. Clean. Prod. https://doi.org/10.1016/j.jclepro.2014.03.005 (2015).
Uliasz-Bochenczyk, A. Application of Fly Ash From Hard Coal Combustion in Water Boilers for CO2 Sequestration via Mineral Carbonation. Rocz. Ochr. Sr. (2008).
Uliasz-Bocheńczyk, A. CO 2 mineral sequestration in selected wastes. (Publishing House of Mineral and Energy Economy Research Institute of the Polish Academy of Sciences. (2009).
Nyambura, M. G., Mugera, G. W., Felicia, P. L. & Gathura, N. P. Carbonation of brine impacted fractionated coal fly ash: Implications for CO2 sequestration. J. Environ. Manage. https://doi.org/10.1016/j.jenvman.2010.10.008 (2011).
Muriithi, G. N. et al. Comparison of CO2 capture by ex-situ accelerated carbonation and in in-situ naturally weathered coal fly ash. J. Environ. Manage. https://doi.org/10.1016/j.jenvman.2013.05.027 (2013).
Jo, H. Y., Kim, J. H., Lee, Y. J., Lee, M. & Choh, S. J. Evaluation of factors affecting mineral carbonation of CO2 using coal fly ash in aqueous solutions under ambient conditions. Chem. Eng. J. https://doi.org/10.1016/j.cej.2011.12.023 (2012).
Han, S. J., Im, H. J. & Wee, J. H. Leaching and indirect mineral carbonation performance of coal fly ash-water solution system. Appl. Energy https://doi.org/10.1016/j.apenergy.2014.12.074 (2015).
Liu, W. et al. CO2 sequestration by direct gas–solid carbonation of fly ash with steam addition. J. Clean. Prod. https://doi.org/10.1016/j.jclepro.2017.12.281 (2018).
Siriwardena, D. P. & Peethamparan, S. Quantification of CO2 sequestration capacity and carbonation rate of alkaline industrial byproducts. Constr. Build. Mater. https://doi.org/10.1016/j.conbuildmat.2015.05.035 (2015).
Rao, A., Anthony, E. J., Jia, L. & Macchi, A. Carbonation of FBC ash by sonochemical treatment. Fuel https://doi.org/10.1016/j.fuel.2007.02.004 (2007).
Acknowledgements
The phase composition, leachability of pollutants and the microstructural analysis were determined at the Faculty of Materials Science and Ceramics at the AGH University of Science and Technology. The work was supported by the AGH University of Science and Technology [subvention research programs no. 16.16.100.215] and the Mineral and Energy Economy Research Institute of the Polish Academy of Sciences [statutory research].
Author information
Authors and Affiliations
Contributions
Alicja Uliasz-Bochenczyk and Eugeniusz Mokrzycki wrote the main manuscript text, prepared figures and reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Uliasz-Bocheńczyk, A., Mokrzycki, E. The potential of FBC fly ashes to reduce CO2 emissions. Sci Rep 10, 9469 (2020). https://doi.org/10.1038/s41598-020-66297-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-66297-y
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.