Abstract
Connexins (Cxs) are membrane-spanning proteins which enable flow of information important for kidney homeostasis. Changes in their spatiotemporal patterning characterize blood vessel abnormalities and chronic kidney diseases (CKD). We analysed spatiotemporal expression of Cx37, Cx40, Cx43 and Cx45 in nephron and glomerular cells of developing, postnatal kidneys, and nephrotic syndrome of the Finnish type (CNF) by using immunohistochemistry, statistical methods and electron microscopy. During kidney development, strong Cx45 expression in proximal tubules and decreasing expression in glomeruli was observed. In developing distal nephron, Cx37 and Cx40 showed moderate-to-strong expression, while weak Cx43 expression gradually increased. Cx45/Cx40 co-localized in mesangial and granular cells. Cx43 /Cx45 co-localized in podocytes, mesangial and parietal epithelial cells, and with podocyte markers (synaptopodin, nephrin). Different Cxs co-expressed with endothelial (CD31) and VSMC (α –SMA) markers in vascular walls. Peak signalling of Cx37, Cx43 and Cx40 accompanied kidney nephrogenesis, while strongest Cx45 signalling paralleled nephron maturation. Spatiotemporal Cxs patterning indicate participation of Cx45 in differentiation of proximal tubules, and Cx43, Cx37 and Cx40 in distal tubules differentiation. CNF characterized disorganized Cx45 expression in proximal tubules, increased Cx43 expression in distal tubules and overall elevation of Cx40 and Cx37, while Cx40 co-localized with increased number of interstitial myofibroblasts.
Similar content being viewed by others
Introduction
During normal human kidney development, the uretheric bud induces the nearby metanephric mesenchyme to transform into metanephric cup cells, which than undergo through several developmental stages, including the renal vesicle stage, S-body and capillary loop stages, subsequently leading to mature nephron and glomeruli formation. Nephrogenesis is characterized by the mesenchymal to epithelial transition (MET) of future nephron cells, while podocytes also display a reverse process of epithelial to mesenchymal transition (EMT) at later developmental stages1. Podocytes are the key cells in formation of glomerular basement membrane (GBM), which increasingly expresses nephrin2 and synaptopodyn, cytoskeletal proteins critical for formation of the podocyte foot processes3. During normal kidney development, α –smooth actin (α –SMA) is increasingly expressed in smooth muscle cells in the walls of blood vessels (VSMC), pericytes and podocytes1, while the endothelium of blood vessels strongly expresses CD31 in the nephrogenic zone of the mouse4 and human developing kidneys1. Experimental animal models associate CD31 expression with development of atherosclerosis5. In chronic kidney disease (CKD), stronger expression of α –SMA actin is associated with the increased number of myofibroblasts in the interstitium of sclerotic kidneys, and with revascularization and regeneration of the affected kidneys4,6. CKD caused by the nephrotic syndrome of the Finnish type CNF) also shows changes in cytoskeletal filaments1. Recent studies indicate that in advanced stages of kidney disease, the amount of synaptopodyn in the urine can predict the extent of glomerular damage7.
Intercellular signaling via connexins (Cxs) has a major role both during normal kidney development, as well as in postnatal period and in progression of pathological changes. Cxs are membrane-spanning proteins, which by forming cell-to-cell channels and cell-to-extracellular space hemi-channels8 control cell proliferation and cell death9,10 as well as flow of information important for functional homeostasis within tissues and organs11. Cxs are widely spread in different organs, particularly in the vasculature, where they regulate vasomotion12. Therefore, alterations in normal Cx patterning are primarily observed in structural and functional changes of blood vessels13 and in severe vascular abnormalities14. Thus, acceleration of atherosclerosis was associated with Cx37 null mice15. While deletion of Cx45 was lethal in mouse development because of interrupted vessels formation16,17, its elevation led to development of hypertension18. Renal vasculature and the juxtaglomerular apparatus (JGA) have been shown to express Cx43, Cx40, Cx45 and Cx37, thus pointing to the role of those Cxs in several regulatory mechanisms within kidneys, including blood pressure control19,20.
Expression of different Cxs in tubular nephron cells and glomerular cells has been mostly analysed in rat and mice postnatal kidneys, but rarely during kidney development. Expression of various subtypes of Cxs was found along the kidney nephrons20,21, but clear morphological evidence for the presence of gap junctions has been reported only for their proximal tubules20. Expression of Cx37 was found in distal nephron20, in ascending limb and distal convoluted kidney tubules, while proximal tubules characterized its weaker expression22. Cx45 expression was observed in developing distal tubules of mice kidneys as well23.
Kidney glomeruli are structures which harbor several cells population and glomerular blood vessels, which display expression of different Cxs. Studies on Cxs expression in glomerular endothelial cells showed contradicting results24,25,26. Investigations on experimental animals reported that mesangial cells and podocytes can express Cx4326,27,28,29,30 and possibly Cx4531. In addition, mesangial cells expressed Cx37 and Cx4024.
In developing rat kidneys, Cx45 was detected in glomeruli and distal tubules23, while Cx37, Cx45 and Cx46 characterized early stages of rat development32. In human mid-term fetuses, Cx43 expression was reported in tubules of the renal cortex33, while recent study on human fetuses reported Cx36 expression in proximal tubules and collecting ducts, and weakly in Henle’s loop and distal tubules, while Cx 43 expression characterized proximal tubules and glomeruli34. During normal EMT of human kidney stem cell cultures Cx43 expression gradually decreased35.
Different types of CKD share common pathophysiological mechanism, which includes inflammation and pathological accumulation of extracellular matrix leading to scar formation36. Decrease of Cx37 was observed in CKD kidneys, while increase in Cx43 characterised inflammatory cells, tubular, interstitial cells and in the endothelium of capillaries in hypertensive nephropathy37. However, data on the role of Cx43 in kidney pathology remained contradicting21: while Cx43 upregulation in podocytes characterized rat experimental glomerulonephritis and type 2 diabetes, its downregulation was seen in overt diabetic nephropathy. Therefore, Cx43 could be considered as marker for podocyte injury28,29.
Despite numerous investigations on the role of connexins in kidney homeostasis, there are still evident discrepancies about kidney regions specifically expressing different Cx isoforms. There is only one experimental study comparing expression of Cx in foetal and adult kidney tissue38, while data on the Cxs expression outside the vascular wall are still largely unknown. This specially refers small number of studies on human developing kidneys, which cover relatively short developmental period33,34. The aim of the present study was to describe changes in spatiotemporal expression of Cxs in developing and postnatal human kidneys, as well as their precise localization within different kidney cells by using co-expression studies with synaptopodyn, nephrin, CD31 and α –SMA. In addition, we compared intensity of signal between healthy kidneys and nephrotic syndrome of the Finnish type (CNF) in the light of possible faulty intercellular signalling associated with end- stage renal disease.
Materials and methods
Human tissue processing
In our study, 11 human embryonic and foetal tissues were acquired from the Department of Gynaecology and Obstetrics after spontaneous abortions, or after tubal pregnancies from the Department of Pathology. Tissues were processed with permission of the Ethical and Drug Committee of the University Hospital in Split in accordance with Helsinki Declaration. The age of conceptuses was evaluated between 8th and 38th developmental week from menstrual data and corresponding to the external measurements (crown–rump length) and the Carnegie staging system. All tissues were without sings of macerations and morphologically regular. In our research we also included postnatal tissue taken during autopsy of healthy 1,5- year old boy and nephrotic kidney tissue from 3 nephrectomised CNF patients (homozygous missense mutation c.1096 A > C; pSer366Arg in NPHS1 gene was detected in all three patients). Informed parental consent was obtained for the research purposes2. Tissues were furtherly processed and glass slides were prepared as previously described39. Tissue was fixed in 4% paraformaldehyde in phosphate buffer saline (PBS), dehydrated in ethanol dilutions, paraffin-embedded and serially cut as 5 µm thick sections. Appropriate tissue preservation was confirmed by Haematoxylin and Eosin staining of every 10th section. Tissue was analyzed using Olympus BX51 light microscope (Olympus, Tokyo, Japan).
Immunohistochemistry and immunofluorescence staining
Tissue sections were deparaffinized and then rehydrated in ethanol following standard protocol40 and treated with sodium citrate buffer. After washing with PBS, blocking buffer (Protein Block, Abcam, UK) was administered on the tissue covered area. Primary antibodies were applied (Table 1) overnight and rinsed in PBS. Suitable secondary antibodies were applied (Table 1) and incubated in humidity chamber for one hour. Considering the double staining with lectins, after the mentioned procedure of administering primary antibodies, sections were incubated with FITC-conjugated lectins (Dolichos Biforus Agglutinin - DBA or Lotus Tetragonolobus Lectin - LTL, Table 1) in dark. They were washed in PBS following the 2 hr incubation period at room temperature41. Slides were afterwards washed in PBS and treated with DAPI nuclear staining. Control for specificity was excluding primary antibody from the staining procedure. For imaging, Olympus fluorescence microscope (BX61; Tokyo, Japan) with a digital camera (DP71) was used. Images were captured by using the Olympus CellA software and assembled in the plates by Adobe Photoshop. For Fig. 5 exclusively, we used DS-Ri2 digital camera for imaging, and NIS-Elements F software for image capture.
Semi thin and ultra-thin sections and electron microscopy
In preparation for electron microscopy we used tissue samples of 10th and 22nd week-old human kidneys, 1.5 years healthy and 3-year CNF kidney tissue. After the 24- hour fixation in 4% paraformaldehyde, specimens were post-fixed in 1% osmium tetroxide for an hour, dehydrated in courses of ethanol and embedded in LX 112 resin. Toluidine blue was used for staining of previously cut semi thin sections (1 µm). Semi thin sections were additionally cut into ultrathin sections (0.05 µm thick) which were examined by transmission electron microscope (Zeiss 902 A, Germany) after being treated with uranyl acetate and lead citrate staining2.
Semi-quantification
The staining intensity of chosen antibodies was semi-quantitatively assessed by four categories: with (−) indicating the absence of any reactivity; (+) as a mild reactivity; (++) as moderate reactivity; and (+++) as strong reactivity (Table 2). Three researchers semi-quantitatively analyzed the staining intensity separately by image analysis software ImageJ (National Institutes of Health, Bethesda, MD, USA)1.
Statistical analysis
Florescence intensity histograms were acquired for red and green fluorescence channels in ImageJ software (NIH, Bethesda, MD, USA). Pixels that had fluorescence intensity between 0 and 15 were considered background. Expression of different connexins was quantified as the area under the curve (AUC) of florescence intensity histograms, and we refer to this as florescence intensity unit (FIU) in the rest of the article. AUCs and their interval estimates were calculated by using AUC analysis routine in GraphPad Prism 8.0 software (Graph Pad, La Jolla, CA, USA). Statistical significance and effect sizes as well as respective 95%CI were calculated by ANOVA followed by Tukey post hoc test in GraphPad Prism 8.0 software (Graph Pad, La Jolla, CA, USA). Level of significance was set at p = 0.05. Expression of connexins during embryonic development was analyzed by finding peaks or nadirs in time series using the single sample t test.
Results
During early human development, the ureteric bud induces metanephric cup mesenchymal cells to gradually differentiate into renal vesicle epithelial cells, with centrally positioned lumen. The described process is known as mesenchymal-to epithelial transition1. During further development renal vesicle transforms into S-shaped body, which gives rise to immature glomeruli and different parts of nephron tubules.
Co-expression of Cx43 and Cx45 in the developing and postnatal kidney tissue
Double immunofluorescence method reveals co-expression of Cx45/Cx43 in the metanephric cup and collecting tubules at the earliest stages of kidney development (Fig. 1a–d). In immature glomeruli, Cx43 characterizes endothelial cells of vascular wall, while Cx45 is positioned in more peripheral part of the vascular wall (VSMC), thus Cx43/Cx45 expression mostly does not overlap. Cx45 is strongly expressed in some glomerular cells (probably corresponding to mesangial cells) (Fig. 1e–h).
Co-expression of Cx43 and Cx45 in the developing and post-natal human kidneys. (a–d) Kidneys of 8th week human embryo; (e–h) Kidneys of the 10th week human foetus; (i–l) Kidneys of 38th week human foetus; (m–p) Postnatal kidney tissue (1,5 years). expression of Cx43and Cx45 (arrows), metanephric cup (mc), collecting tubules (ct), blood vessels (bv), immature glomeruli (g), parietal epithelial cells (ec), proximal convoluted (pt) and distal convoluted tubules (dt). DAPI nuclear staining (c,g,k,o). Merged microphotographs show co-expression of Cx45/Cx43 (arrowheads) in the same cells but in different cellular compartments (d,h,l,p). Inset in (h) shows Cx43/Cx45 co-localization in mesangial cell, while inset in (p) shows their co-localization in podocytes. Double immunofluorescence staining to Cx43, Cx45 and DAPI, x100.
In the second half of intrauterine life, strong Cx45 expression characterizes all kidney structures, while weak Cx43 expression is observed in glomeruli, proximal and distal tubules. Cx43 expression is observed in the afferent and efferent arterioles and parietal epithelial cells. Again, Cx43/Cx45 co-expression is observed in in different cellular compartments of the same structures (Table 2, Fig. 1i–l).
In postnatal kidneys, Cx43/ Cx45 co-express in different cellular or membranous compartments of the same kidney structures including podocytes, walls of blood vessels and parietal epithelial cells, Cx43 is expressed moderately, while Cx45 strongly (Fig. 1m–p, inset Fig. 1p).
Co-expression of different Cxs with CD31 and α –SMA in developing and postnatal kidney tissue
Between 8th and 10th developmental week, human kidneys contain all described stages of nephron formation, with immature glomeruli positioned closest to the future medulla. Metanephric cup cells strongly express Cx45, moderately Cx40 and Cx37, and weakly Cx43. In immature glomeruli and collecting ducts, Cx43 is expressed weakly, Cx45 strongly, while Cx40 is expressed moderately-to-strongly in the form of dense granules. Cx37 is moderately expressed in some metanephric cup cells and immature glomeruli, but strongly in collecting tubules in the form of coarse grains (Table 2, see Fig. 2. a,e and i).
Co-expression of different Cxs (Cx43, Cx45, Cx40, Cx37) with CD31 and α –SMA in developing and post-natal human kidneys. (a–d,e–h) Kidneys of the 8th week human embryo; (i–l) Kidneys of the 10th week human fetus; (m–p) Kidneys of the 22nd week human fetus; (q–t) Kidneys of the 38th week human fetus; (u–x) Kidneys of 1,5 years old child. Cx37 and Cx40 expression (arrows), and CD31 and α –SMA expression (arrows), metanephric cup (mc), collecting tubules (ct), immature glomeruli (g), blood vessels (bv), interstitium (i), proximal convoluted (pt) and distal convoluted tubules (dt). DAPI nuclear staining (c,g,k,o,s,w). Merged microphotographs of different Cxs with CD31 and α –SMA show their co-expression (arrowheads) in the walls of blood vessels (d,h,l,p,t,x). Double immunofluorescence staining of DAPI nuclear stain with Cx37/CD31 (a–d) and with Cx40/CD31 (e–h) x20, and DAPI with Cx45/CD31 (q–t) x100. Double immunofluorescence staining of DAPI with Cx43/α –SMA (i–l), and Cx45/α –SMA (m–p), x100, and DAPI with Cx40/ α –SMA x20. (y) Florescence intensity histograms of different connexins during development and in postnatal period.
Double fluorescence of CD31 with Cx40 and Cx37 (Fig. 2a–d,e–h), and double fluorescence of α –SMA with Cx43 (Fig. 2i–l), shows their co-localization in the wall of glomerular blood vessels.
During further development (10th to 38th week), glomeruli became mature, while proximal convoluted tubules and distal convoluted tubules become morphologically distinguishable. During that period, Cx45 is moderately expressed in the glomeruli and distal tubules, while strongly in proximal tubules (Fig. 2m,q). Cx43 slightly increases to moderate in the distal tubules, while Cx40 display moderate-to-strong expression in all three structures. Cx37 expression is moderate in the whole kidney, while weakly in the proximal tubules (Table 2).
In the 22nd and 38th developmental week, moderate-to-strong co-expression of Cx45 co-expresses with strong expression of CD31 and α –SMA in the walls of blood vessels (Table 1, Fig. 2m–p,q–t).
In postnatal healthy kidneys, expression of Cx45 increases only in glomeruli, while Cx43 increases from weak to moderate in all kidney structures. While Cx37 shows no changes in its expression pattern, expression of Cx40 is postnatally mild in glomeruli, moderate in proximal tubules, but increases to strong in distal tubules, (Table 2., see Fig. 2u). Double staining of Cx40 and α –SMA shows their co-localization in intraglomerular and extraglomerular blood vessels (Fig. 2u–x)
Co-expression of different Cxs with CD31 and α –SMA in CNF
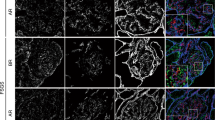
In CNF kidneys, glomeruli display different degrees of pathological changes, while some proximal tubules appear dilated thus forming small cysts. Compared to healthy postnatal kidneys, in CNF kidneys expression and distribution of different Cxs changes.: Cx45 and Cx43 expression slightly decreases in glomeruli, while Cx40 and Cx37expression increases in some distal tubules (Table 2). Cx43/Cx45 co-express in different cellular-compartments of the same kidney structures (Table 2, Fig. 3a–d). Cx37 shows irregular expression in the form of coarse granules within glomeruli, some parts of proximal and distal tubules. Double staining of Cx37 and CD31 shows their co-expression in the walls of blood vessels (Fig. 3e–h). Cx40 displays reduced expression in affected glomeruli, increased expression in distal tubules and moderate expression in interstitium, Double staining of Cx40 with α –SMA shows their co-localization in interstitial myofibroblasts (Fig. 3i–l).
Co-expression of different Cxs with CD31 and α –SMA in CNF kidneys. (a–l) CNF post-natal kidneys (3,5 years). Cx43, Cx40 and Cx37 expression (arrows), and CD31 and α –SMA is expression (arrows), glomeruli (g), proximal tubules (pt) and distal tubules (dt), cysts of proximal tubules (c), blood vessels (bv) and interstitium (i). DAPI nuclear staining (c,g,k). Merged microphotographs of different Cxs with CD31 and α –SMA show their co-expression (arrowhead) in the wall of blood vessels and population of interstitial myofibroblasts (mf) (d,h,l). Double immunofluorescence staining of DAPI with Cx43 /Cx45 (a–d) x100; DAPI with Cx37/CD31 (e–h) x20, and DAPI with Cx40/α –SMA x100. (m) Fluorescence intensity histograms comparing healthy and CNF postnatal kidneys.
Co-expression of nephrin and synaptopodin with different Cxs in developing and CNF kidneys
Up to the 10th developmental week, synaptopodin expression is absent in the kidney structures (Table 2.). In the 22nd foetal week, Cx43 is mildly expressed in the glomeruli, proximal and distal tubules, and strongly in the juxtaglomerular apparatus (jga), while synaptopodin expression is moderate in the glomeruli, and very mild in proximal and distal tubules (Table 2). Nephrin expression has been previously shown to increases during human development2
Synaptopodin only partly co-expresses with Cx43 in the glomerular cell population probably corresponding to podocytes (Fig. 4a–d), as well as does Cx45 with nephrin (Fig. 4e–h).
Co-expression of synaptopodin and nephrin with different Cx in developing and CNF kidneys. (a-d,e–h) Human kidneys in the 22nd developmental week. (i–l,m–p) Postnatal CNF kidneys. Cx43and Cx43 expression (arrows), synaptopodin and nephrin expression (arrows), glomeruli (g), proximal (pt) and distal tubules (dt), juxtaglomerular region (jga). DAPI nuclear staining (c,g,k,o). Merged view of Cx43 or Cx45 with synaptopodin shows co-expression of the two markers (arrowheads) in the glomeruli (podocytes) of healthy kidneys (d,h), and absence of their co-expression in CNF kidneys (l,p). Double immunofluorescence staining of DAPI with Cx43 or Cx45 with nephrin or synaptopodin x100.
CNF kidneys show decreased and only partial expression of synaptopodin in the glomeruli, and very weak expression in proximal and distal tubules (Table 2; Fig. 4i–l). Cx43 and synaptopodin do not co-express in the podocytes (Fig. 4i–l), while nephrin expression in CNF glomeruli is very mild (Fig. 4m–p).
Co-expression of different Cxs with markers of proximal tubules (DBA) and distal tubules (LTL) in developing, postnatal and CNF kidneys
In 8th to 10th developmental week, only small number of kidney tubules showed strong signal to LTL, while there was no signal to DBA at that developmental stage. Double staining of Cx37 with LDL shows absence of their co-expression in distal tubule, while positive signal to Cx37 was seen in collecting ducts (Table 2, Fig. 5a–d). In contrast to developing kidneys, postnatal kidneys showed strong reactivity to both DBA in proximal tubules and LDL in distal tubules. Double staining of Cx40 with DBA showed their co-expression in proximal tubules (Fig. 5e–h), while Cx43 co-expressed with LDL in distal tubules (Fig. 5i–l).
Co-expression of different Cxs with markers of proximal tubules (DBA) and distal tubules (LTL) in developing, postnatal and CNF kidneys. (a–d) Human kidneys in the 10th developmental week. (e–h,i–l) Human postnatal kidneys (1,5 years). (m–p) Postnatal CNF kidneys (3,5 years). Cx37, Cx40 and Cx45 expression (arrows), expression of markers for proximal (DBA) and distal (LTL) tubules (arrows), proximal tubules (pt), distal tubules (dt), collecting tubules (ct), interstitium (i), glomeruli (g), blood vessels (bv). Co-expression of Cx37 with LTL is missing in 10th developmental week kidneys (a–d), while co-expression of Cx40/DBA (arrowhead) (e–h) and Cx43/LTL (arrowhead) (i–l) is seen in proximal and distal tubules of postnatal kidneys. In CNF, Cx37 co-expresses with DBA (arrowhead) in dilated proximal tubules. Double immunofluorescence staining of DAPI with Cx37/LTL, Cx40/DBA, Cx43/LTL and Cx37/DBA x40.
In CNF kidneys, dilated proximal tubules strongly reacted with DBA. Double staining of Cx37 with DBA shows their co-expression in proximal tubules (Fig. 5m–p).
By using specific markers for proximal and distal tubules, we could prove that morphological criteria for detection of different parts of nephron tubules in our study accorded with their labelling with specific markers for proximal and distal tubules.
Electron microscopy
In the 6th developmental week, abundant gap junctions interconnect epithelial tubular cells (Fig. 6a). During progression of development gap junctions are found in different parts of kidney tissue, including glomerular cells (Fig. 6b). In the CNF kidneys, besides typical depletion of podocyte pedicles, we observed connexins in the whole kidney tissue, but most abundantly between the tubular cells (Fig. 6c).
Transmission electron microscopy (TEM) of the connexins in the developing kidney tissue and CNF kidneys. (a) Connexins (arrows) are observed between the tubular cells in the 6th developmental week. (b) Connexins (arow) interconnect glomerular cells in the kidney tissue of the 21st week human embryo. (c) Connexins (arrows) between the tubular cells in the CNF kidneys.
Quantification and semi-quantification of Cxs expression during development and postnatal period
Semi-quantification of Cxs expression in different parts of nephron is shown in Table 2.
Analysis of the mean fluorescence signal of different Cxs revealed significant difference of maximal signal (peak signalling) between Cxs: Cx37, Cx43 and Cx40 displayed very similar course of fluorescent intensity signalling, being strongest in the 21st developmental week, but overall intensity of Cx40 was stronger (p = 0.0003, p = 0.02, p < 0.0001, respectively). The strongest fluorescence signalling characterized Cx45, with peak in the 38th developmental week (p = 0.03). In postnatal period, intensity of florescence signal dropped, except for Cx40 which remained high (Fig. 2y). All Cxs signals are much stronger in CNF kidneys compared to healthy postnatal kidneys (Fig. 3m).
Discussion
Connexins are transmembrane proteins that form gap junctions, which in the kidney tissue primarily contribute to renal haemodynamics, as they mostly localize in the walls of blood vessels. However, they also seem to have essential role in tubular epithelial and glomerular function42, in prenatal development, morphogenesis and tissue differentiation. By now, numerous experimental studies resulted in controversial data about spatio-temporal expression of specific Cx sub-types in the kidney tissue.
In the present study, strong expression of Cx45 displayed decreasing course in glomeruli of developing kidneys, while its strong expression in proximal tubules continued into postnatal period. In contrast, during development of mouse kidneys, Cx45 expression paralleled appearance of renin producing cells, but postnatally disappeared38. Compared to healthy postnatal kidneys, CNF kidneys showed disorganized distribution of Cx45 in proximal tubules, what might be associated with faulty signalling. CNF has been previously shown to be histologically characterized by tubular primary cilia dysregulation and appearance of proximal tubules cysts43. We also found both Cx45 and Cx40 expression in glomerular mesangial and granular cells, thus confirming possibility of appearance of heterotopic channels between different Cxs, which has been shown in experimental animals19,38,44. However, use of different techniques in studies analysing Cxs expression led to conflicting conclusions. Thus, Cx45 was detected in mouse distal tubules by immunohistochemistry23 but its mRNA failed to be confirmed32.
In our study, different kidney structures showed weak Cx43 expression, which during development gradually increased to mild only in distal tubules. While Cx43 expression increased in glomeruli and proximal tubules of postnatal healthy kidneys, it became irregular and strong in distal tubules of CNF kidneys. In contrast to our results, previous study on human foetal kidneys missed to show early Cx43 expression in the metanephric cup cells34, what might be explained by higher sensitivity of immunofluorescence antibodies used in the present study. Discrepancies in the reported expression of CX43 and Cx45 might also reflect interspecies differences in Cxs expression20. In comparison to Cx45, Cx43 expression was confined only to apical cytoplasmic compartment of tubular nephron cells, while Cx43 / Cx45 co-expression characterized different cellular compartments in mesangial cells, podocytes, and parietal epithelial cells. Our study also showed that both Cx43 and Cx45 co-localized with markers for podocyte pedicles, thus pointing to importance of those two Cxs subtypes in podocyte signalling. Dedifferentiation of human podocytes in CNF kidneys has been previously shown to be associated with changes in cytoskeletal arrangement, presence of primary cilia and re-expression of nestin1,2. Additionally, interactions between connexins were suggested to be crucial in regulation of EMT of developing kidney cells45, which in CNF seems to be disturbed.
Studies on animals showed that Cx43 might be primarily involved in vasomotion, but also in gene transcription, ATP and vesicle release, cytoskeletal dynamics and cell stress11. In CKD, deletion of Cx43 caused anti-inflammatory effect and reduced interstitial fibrosis, while similar to our study - its disruption led to depletion of podocyte pedicles46. In contrast, Cx43 upregulation promoted pro-inflammatory environment21 thus suggesting that alterations in expression of Cxs might be associated with development of CKD37.
During development, moderate-to-strong expression of Cx37 in human kidney structures continued into postnatal period. Similar to our study, previous animal studies have also shown expression of Cx37 primarily in distal nephron20. Importance of early expression of Cx37 and Cx43 was implicated in kidney development, while their elevation was referred in neonatal unilateral ureteral obstruction32. We also noticed overall elevation of Cx40 and Cx37 expression in CNF kidneys, particularly in distal tubules.
Moderate-to strong expression of Cx40 during human kidney development decreased in glomeruli and increased in distal tubules in postnatal period. Opposite to our study, Cx40 expression in rats was observed in intra and extraglomerular mesangial cells and non-glomerular endothelial cells29.
While use of different techniques in analysing Cxs expression in kidney tissue often led to conflicting results, application of immunohistochemical method in kidney tissues of different animal species and humans revealed a certain degree of conservation in their spatio-temporal expression.
In conclusion, in developing human kidneys co-expression of different Cxs with markers for endothelial cells (CD31) and VSMC (α –SMA) indicated importance of communications between those two cell populations for vascular wall integrity. Overall signalling intensity of Cx37, Cx43 and Cx40 had its peak in the 22nd developmental week and accompanied a process of nephrogenesis, while peak of Cx45 expression in the 38th week corresponded to period of functional nephron maturation47,48. Based on the Cxs spatiotemporal expression in glomerular and tubular human kidney cells, we suggested importance of Cx45 in differentiation of proximal tubules, and Cx43, Cx37 and Cx40 in differentiation of distal tubules. Additionally, fine cytoplasmic balance between Cx43 and Cx45 co-expression might be crucial for differentiation of glomerular cell population, including podocytes, mesangial and parietal epithelial cells. The observed changes in Cxs expression characterized all structures in developing and CNF kidneys, thus pointing to Cxs as possible target in treatment of kidney diseases and repair processes29. Alterations in gap junction activity have been shown to cause structural and functional damage in several kidney diseases21,29,37. We also showed increased number of myfibroblasts co-localizing with Cx40 in the interstitium of CNF kidneys, thus implying possible involvement of Cx40 in regeneration processes49. Namely, expansion of myofibroblast population, which acquired some functional and structural characteristics of smooth muscle cells50 lead to extensive deposition of interstitial extracellular matrix, which is the key characteristic of renal fibrosis in CKD. In addition, some dedifferentiated renal fibroblasts re-expressed mesenchymal markers, what might be considered as a sign of EMT6. Therefore, treatment that would re-establish mature fibroblast phenotype could be another option for treating renal fibrosis6,51.
References
Filipovic, N. et al. Immunohistochemical and electronmicroscopic features of mesenchymal-to-epithelial transition in human developing, postnatal and nephrotic podocytes. Histochem Cell Biol 147, 481–495 (2017).
Vukojevic, K. et al. Glomeruli from patients with nephrin mutations show increased number of ciliated and poorly differentiated podocytes. Acta Histochem. 120, 748–756 (2018).
Kato, T. & Mizuno, S. Nephron, Wilms’ tumor-1 (WT1), and synaptopodin expression in developing podocytes of mice. Exp Anim. 66, 183–189 (2017).
Munro, D. A. D., Hohenstein, P. & Davies, J. A. Cycles of vascular plexus formation within the nephrogenic zone of the developing mouse kidney. Sci Rep. 7, 3273, https://doi.org/10.1038/s41598-017-03808-4 (2017).
Liu, L. & Shi, G. P. CD31: beyond a marker for endothelial cells. Cardiovasc Res. 94, 3–5 (2012).
Boor, P. & Floege, J. The renal (myo-)fibroblast: a heterogeneous group of cells. Nephrol Dial Transplant 27, 3027–3036 (2012).
Kwon, S. K., Kim, S. J. & Kim, H. Y. Urine synaptopodin excretion is an important marker of glomerular disease progression. Korean J Intern Med 31, 938–943 (2016).
Sala, G., Badalamenti, S. & Ponticelli, C. The Renal Connexome and Possible Roles of Connexins in Kidney Diseases. Am J Kidney Dis 67, 677–687 (2016).
Moorby, C. D. & Gherardi, E. Expression of a Cx43 deletion mutant in 3T3 A31 fibroblasts prevents PDGF-induced inhibition of cell communication and suppresses cell growth. Exp Cell Res 249, 367–376 (1999).
Seul, K. H., Kang, K. Y., Lee, K. S., Kim, S. H. & Beyer, E. C. Adenoviral delivery of human connexin37 induces endothelial cell death through apoptosis. Biochem Biophys Res Commun 319, 1144–1151 (2004).
Ribeiro-Rodrigues, T. M., Martins-Marques, T., Morel, S., Kwak, B. R. & Girao, H. Role of connexin 43 in different forms of intercellular communication - gap junctions, extracellular vesicles and tunnelling nanotubes. J Cell Sci 130, 3619–3630 (2017).
Schuster, A., Oishi, H., Beny, J. L., Stergiopulos, N. & Meister, J. J. Simultaneous arterial calcium dynamics and diameter measurements: application to myoendothelial communication. Am J Physiol Heart Circ Physiol 280, H1088–1096 (2001).
Haefliger, J. A., Nicod, P. & Meda, P. Contribution of connexins to the function of the vascular wall. Cardiovasc Res. 62, 345–356 (2004).
Simon, A. M. & McWhorter, A. R. Vascular abnormalities in mice lacking the endothelial gap junction proteins connexin37 and connexin40. Dev Biol. 251, 206–220 (2002).
Wong, C. W. et al. Connexin37 protects against atherosclerosis by regulating monocyte adhesion. Nat Med 12, 950–954 (2006).
Kruger, O. et al. Defective vascular development in connexin 45-deficient mice. Development. 127, 4179–4193 (2000).
Kumai, M. et al. Loss of connexin45 causes a cushion defect in early cardiogenesis. Development. 127, 3501–3512 (2000).
Li, L. et al. The enhancement of Cx45 expression and function in renal interlobar artery of spontaneously hypertensive rats at different age. Kidney Blood Press Res 40, 52–65 (2015).
Hanner, F. et al. Connexin45 is expressed in the juxtaglomerular apparatus and is involved in the regulation of renin secretion and blood pressure. Am J Physiol Regul Integr Comp Physiol 295, R371–380 (2008).
Hanner, F., Sorensen, C. M., Holstein-Rathlou, N. H. & Peti-Peterdi, J. Connexins and the kidney. Am J Physiol Regul Integr Comp Physiol 298, R1143–1155 (2010).
Abed, A. et al. Targeting connexin 43 protects against the progression of experimental chronic kidney disease in mice. Kidney Int. 86, 768–779 (2014).
Stoessel, A., Himmerkus, N., Bleich, M., Bachmann, S. & Theilig, F. Connexin 37 is localized in renal epithelia and responds to changes in dietary salt intake. Am J Physiol Renal Physiol 298, F216–223 (2010).
Butterweck, A., Gergs, U., Elfgang, C., Willecke, K. & Traub, O. Immunochemical characterization of the gap junction protein connexin45 in mouse kidney and transfected human HeLa cells. J Membr Biol 141, 247–256 (1994).
Haefliger, J. A. et al. Connexins 40 and 43 are differentially regulated within the kidneys of rats with renovascular hypertension. Kidney Int. 60, 190–201 (2001).
Takenaka, T. et al. Expression and role of connexins in the rat renal vasculature. Kidney Int. 73, 415–422 (2008).
Zhang, J. & Hill, C. E. Differential connexin expression in preglomerular and postglomerular vasculature: accentuation during diabetes. Kidney Int. 68, 1171–1185 (2005).
Hillis, G. S. et al. The expression of connexin 43 in human kidney and cultured renal cells. Nephron. 75, 458–463 (1997).
Sawai, K. et al. Redistribution of connexin43 expression in glomerular podocytes predicts poor renal prognosis in patients with type 2 diabetes and overt nephropathy. Nephrol Dial Transplant 21, 2472–2477 (2006).
Morioka, T. et al. Glomerular expression of connexin 40 and connexin 43 in rat experimental glomerulonephritis. Clin Exp Nephrol 17, 191–204 (2013).
Hwan Seul, K. & Beyer, E. C. Heterogeneous localization of connexin40 in the renal vasculature. Microvasc Res. 59, 140–148 (2000).
Just, A. et al. Connexin 40 mediates the tubuloglomerular feedback contribution to renal blood flow autoregulation. J Am Soc Nephrol 20, 1577–1585 (2009).
Silverstein, D. M. et al. Expression of connexins in the normal and obstructed developing kidney. Pediatr Nephrol. 18, 216–224 (2003).
Hieda, K. et al. Spatial relationship between expression of cytokeratin-19 and that of connexin-43 in human foetal kidney. Anat Cell Biol 46, 32–38 (2013).
Raduly, G. et al. The immunoexpression of aquaporin 1, PAX2, PAX8, connexin 36, connexin 43 in human foetal kidney. Rom J Morphol Embryol 60, 437–444 (2019).
Ullmann, U. et al. Epithelial-mesenchymal transition process in human embryonic stem cells cultured in feeder-free conditions. Mol Hum Reprod 13, 21–32 (2007).
Prakoura, N., Kavvadas, P. & Chadjichristos, C. E. Connexin 43: a New Therapeutic Target Against Chronic Kidney Disease. Cell Physiol Biochem 49, 985 (2018).
Toubas, J. et al. Alteration of connexin expression is an early signal for chronic kidney disease. Am J Physiol Renal Physiol 301, F24–32 (2011).
Kurtz, L., Janssen-Bienhold, U., Kurtz, A. & Wagner, C. Connexin expression in renin-producing cells. J Am Soc Nephrol 20, 506–512 (2009).
Becic, T., Kero, D., Vukojevic, K., Mardesic, S. & Saraga-Babic, M. Growth factors FGF8 and FGF2 and their receptor FGFR1, transcriptional factors Msx-1 and MSX-2, and apoptotic factors p19 and RIP5 participate in the early human limb development. Acta Histochem. 120, 205–214 (2018).
Vukojevic, K., Carev, D., Sapunar, D., Petrovic, D. & Saraga-Babic, M. Developmental patterns of caspase-3, bax and bcl-2 proteins expression in the human spinal ganglia. J Mol Histol 39, 339–349 (2008).
Luetic, M. et al. PUFAs supplementation affects the renal expression of pannexin 1 and connexins in diabetic kidney of rats. Histochem Cell Biol 153, 165–175 (2020).
Abed, A. B., Kavvadas, P. & Chadjichristos, C. E. Functional roles of connexins and pannexins in the kidney. Cell Mol Life Sci 72, 2869–2877 (2015).
Saraga, M. et al. Mechanism of cystogenesis in nephrotic kidneys: a histopathological study. BMC Nephrol. 15, 3, https://doi.org/10.1186/1471-2369-15-3 (2014).
Rackauskas, M. et al. Gating properties of heterotypic gap junction channels formed of connexins 40, 43, and 45. Biophys J. 92, 1952–1965 (2007).
Jose, P. A., Chen, S. & Armando, I. Connections in chronic kidney disease: connexin 43 and connexin 37 interaction. Am J Physiol Renal Physiol 301, F21–23 (2011).
Matsuuchi, L. & Naus, C. C. Gap junction proteins on the move: connexins, the cytoskeleton and migration. Biochim Biophys Acta 1828, 94–108 (2013).
Faa, G. et al. Marked interindividual variability in renal maturation of preterm infants: lessons from autopsy. J Matern Foetal Neonatal Med. 23(Suppl 3), 129–133 (2010).
Hinchliffe, S. A., Sargent, P. H., Howard, C. V., Chan, Y. F. & van Velzen, D. Human intrauterine renal growth expressed in absolute number of glomeruli assessed by the disector method and Cavalieri principle. Lab Invest. 64, 777–784 (1991).
Gustafsson, F. et al. Expression of connexin 37, 40 and 43 in rat mesenteric arterioles and resistance arteries. Histochem Cell Biol 119, 139–148 (2003).
Eyden, B. The myofibroblast: phenotypic characterization as a prerequisite to understanding its functions in translational medicine. J Cell Mol Med 12, 22–37 (2008).
Asada, N. et al. Dysfunction of fibroblasts of extrarenal origin underlies renal fibrosis and renal anemia in mice. J Clin Invest 121, 3981–3990 (2011).
Acknowledgements
This work was supported by the University of Split School of Medicine Institutional grant for excellence 2016; Croatian Science Foundation (grant no. IP-06-2016-2575); Ministry of Science, Education and Sports of the Republic of Croatia (grant no. 021-2160528-0507).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Additionally, material preparation, data collection and analysis were performed by Ivona Kosovic, Natalija Filipovic, Benjamin Benzon Merica Glavina Durdov and Ivana Bocina, while the first draft of the manuscript was written by Mirna Saraga-Babic, Katarina Vukojevic and Marijan Saraga.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kosovic, I., Filipovic, N., Benzon, B. et al. Spatio-temporal patterning of different connexins in developing and postnatal human kidneys and in nephrotic syndrome of the Finnish type (CNF). Sci Rep 10, 8756 (2020). https://doi.org/10.1038/s41598-020-65777-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-65777-5
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.