Abstract
Event-free survival (EFS) is considered the most reliable surrogate endpoint for overall survival (OS) in randomised controlled trials (RCTs) of adjuvant therapies for malignant tumours. However, the surrogacy of intermediate endpoints such as EFS for OS in trials of patients with osteosarcoma has not been investigated to date. In this study, we investigated the correlation between OS and intermediate endpoints in RCTs of localised osteosarcoma. A systematic search identified 20 relevant RCTs. The correlations between the surrogate endpoints and OS were evaluated using weighted linear regression analyses and by calculating the Spearman rank correlation coefficients (ρ). The strength of the correlation was determined by calculating the coefficient of determination (R2). A total of 5,620 patients were randomly assigned to 45 treatment arms in the eligible 20 RCTs. The correlation between the hazard ratios for EFS and OS was moderate (R2 = 0.456, ρ = 0.440); this correlation tended to be weaker for patients with localised osteosarcoma excluding the patients with metastases. Overall, the trial-level correlation between the surrogate endpoints and OS was not robust in RCTs of osteosarcoma published to date. Hence, the suitability of the intermediate endpoints as surrogates for OS could not be confirmed.
Similar content being viewed by others
Introduction
Osteosarcoma is the most frequently diagnosed primary malignant bone tumour, with an annual incidence of approximately 800 patients in the United States1, and 200 in Japan2. The prognosis of patients with osteosarcoma has dramatically improved following the introduction of multi-drug combination chemotherapy regimens. The current standard chemotherapeutic agents for osteosarcoma include methotrexate, doxorubicin (Adriamycin), and cisplatin (the MAP regimen) as well as ifosfamide (IFM)3.
Histological response to preoperative chemotherapy is a known prognostic factor for patients with osteosarcoma. Patients who are good responders (i.e., those who exhibit ≥90% necrosis in the resected tumour specimen) have been shown to have better prognoses than those who are poor responders (i.e., <90% necrosis)4, although trials aimed at determining the optimal combination chemotherapy regimen for the latter group have been ongoing. The EURAMOS1 trial, which is the largest randomised controlled trial (RCT) for osteosarcoma to date, found no significant difference in the outcomes of patients treated with MAP alone versus those treated with MAP plus IFM and etoposide. As such, the investigators recommended MAP alone as the standard regimen for osteosarcoma, including in the poor responders5. In Japan, however, IFM is combined with MAP as postoperative chemotherapy for patients who respond poorly to MAP preoperatively6; randomised phase 3 trials are currently underway to verify the effectiveness of this combination regimen7.
Improvement of overall survival (OS) is the most important goal when treating patients with malignant tumours. OS can be clearly defined, as it is the final, immutable endpoint, and is therefore preferred in clinical trials investigating most malignant tumours. However, there are limitations when using OS as a primary endpoint, as it requires more number of patients and additional follow-up involving more time and effort as well as higher costs. Moreover, the emergence of effective new drugs might prolong the post-progression survival and OS, and the effects of the protocol and post-protocol treatments may overlap, thereby influencing OS, making the interpretation of RCT results difficult. Hence, a number of RCTs of patients with localised osteosarcoma have used the intermediate measure of event-free survival (EFS) as their primary endpoint, as this avoids the abovementioned limitations. However, adopting EFS as the primary endpoint requires strong evidence of its correlation with OS; yet, the suitability of EFS as a surrogate endpoint for OS in RCTs of localised osteosarcoma has not been verified. To that end, we conducted a meta-analysis of all published RCTs of localised osteosarcoma to investigate the suitability of the intermediate endpoints including EFS and pathological response rate (RR) as surrogates for OS.
Methods
Study selection and data extraction
A systematic search of PubMed, Scopus, EBSCOhost MEDLINE, and the Cochrane Central Register of Controlled Trials was conducted according to the ‘Preferred Reporting Items for Systematic Reviews and Meta-Analyses’ guidelines8. The search was conducted for all English-language RCTs related to chemotherapy for localised osteosarcoma that were published between January 1974 and July 2019. Eligible studies were RCTs of chemotherapy for newly diagnosed localised osteosarcoma. Non-randomised clinical trials, reviews, and meta-analyses were excluded. The clinical trials extracted by the search were screened and cross-checked independently by two authors.
The extracted data included the publication date, trial name, study phase, patient enrolment period, number of patients, sex, age, number of metastatic diseases, neo-adjuvant or adjuvant regimens, drugs and doses in the standard and experimental arms, primary and secondary endpoints, intention-to-treat (ITT) analysis, post-protocol treatment, pathological response to chemotherapy, survival, and adverse events. A phase 2/3 study was considered as phase 3 for the purposes of this analysis. The medians, hazard ratios (HRs), 95% confidence intervals (CIs), and P-values for both EFS and OS were extracted; disease-free survival was considered as the same as EFS in this study. The pathological RR was defined as the proportion of assessed patients with a complete or partial response based on the criteria of each study. Data on 1-year, 3-year, and 5-year EFS and OS rates were extracted based on Kaplan-Meier estimates; when these data were not described, Kaplan-Meier plots for EFS or OS were constructed for their estimation as binary proportions. The variances were calculated according to the methods guided by the Cochrane9,10. Data were extracted and crosschecked by two authors. In the event of discrepancies, a third author arbitrated to reach a consensus.
Statistical analysis
The EFS and OS values were obtained by meta-analyses using pooled HRs and their corresponding 95% CIs. The odds ratios (ORs) and corresponding 95% CIs for 1-, 3-, and 5-year EFS and OS were also calculated. The meta-analyses were performed using inverse variance and a Mantel-Haenszel random- or fixed-effects model11. If the P-value in the heterogeneity test was less than 0.1, the random-effects model was applied. Cochrane’s Q-test and I2 statistics were used to evaluate heterogeneity. Meta-analyses were completed using the Review Manager software (version 5.3; Nordic Cochrane Centre, Cochrane Collaboration, Copenhagen, Denmark).
The association of HR for OS and HRs or ORs for surrogate endpoints was assessed to evaluate the trial-level surrogacy. A weighted least-square regression for log (HR for OS) and log (HR for EFS) was used for the evaluation of the relationship between OS and EFS with weights equal to the sample size of the trial12,13. The coefficient of determination (R2) was used to evaluate the strength of the association. The bootstrapping was used to estimate the 95% CIs of the R2 surrogacy measures. The nonparametric Spearman’s rank correlation coefficients (ρ) were also used to evaluate the correlation between surrogate endpoints and OS.
Coefficient values were assessed as follows; >0.9, excellent; >0.75, very good; >0.5, good; >0.25, moderate; and ≤0.25, poor14. Sensitivity analyses were performed by excluding studies that comprised patients with metastatic disease or those whose treatment arms included radiation therapy and immuno-stimulants from the evaluation of surrogacy. The statistical analyses were completed using SAS (version 9.4; SAS institute, Cary, NC, USA). P-values reflected two-sided tests, with P < 0.05 indicating statistical significance.
Results
Characteristics of the eligible RCTs
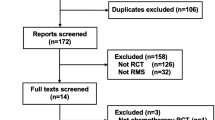
Our systematic search of the literature identified 1,798 articles; 83 duplicate publications were excluded and remaining 1,715 studies were screened further. The full texts of 45 studies were finally evaluated after excluding 1,670 publications. Among these, nine repeat publications, nine non-chemotherapeutic studies, four non-neo-adjuvant or non-adjuvant setting studies, and three publications describing incomplete studies were further excluded. The remaining 20 RCTs were incorporated into the present analysis (Fig. 1, Table S1)5,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33. The characteristics of the eligible RCTs are summarised in Table 1.
The 5,620 patients whose data were extracted from the 20 eligible RCTs were randomly assigned to 45 treatment arms. All arms comprised combination chemotherapy regimens with two to eight cytotoxic drugs except one arm whose subjects underwent radiation alone and another whose patients underwent combination chemotherapy and bilateral lung irradiation18. No study involved administering a molecular targeted therapy, while two studies involved immuno-stimulants as protocol treatments27,31. Chemotherapy was administered in a neo-adjuvant setting in 37 treatment arms in 17 studies, and in an adjuvant setting in 7 treatment arms in 4 studies. Nine and three studies defined their primary endpoints as EFS and RR, respectively; only two RCTs used OS. Among the 20 RCTs, 5 involved patients with metastatic disease (n = 253 in total). The median EFS and OS were not reached in 14 (of 20) and 10 (of 13) RCTs, respectively; therefore, median survival analyses were not conducted in our study. Secondary malignant neoplasms were considered as events for EFS in 8 RCTs, and the total number of the patients that developed secondary malignancies was 36 (1.1%) out of 3,202.
The HRs for EFS and OS were 1.01 (95% CI 0.92, 1.11, P = 0.78) and 1.02 (95% CI 0.92, 1.14, P = 0.66) (Fig. 2 and S1). There were no significant differences between the standard and experimental arms in terms of time-to-event endpoints, RRs, and severe adverse events (Table 2).
Association between intermediate endpoints and OS
Neither of the investigated intermediate endpoints showed excellent or very good association with the HR of OS. To assess the correlation between surrogate endpoints and OS, a weighted least-square regression model was fitted for the trial-level HRs, weighted with the sample size of the trial. For example, the linear regression equation of HR for EFS was as follows;
The trial-level correlation between the HRs of EFS and OS was moderate (R2 = 0.456, 95% CI 0.112, 0.799) (Fig. 3A and Table 3). The Spearman’s rank correlation coefficient (ρ) between the HRs of EFS and OS was 0.440 (95% CI −0.147, 0.797, P = 0.13). The association with the HR of OS was assessed as good (i.e., the coefficient of determination was >0.5) for the 5-year EFS (R2 = 0.530, 95% CI 0.210, 0.850; ρ = 0.564, 95% CI 0.019, 0.851, P = 0.0447), 3-year OS (R2 = 0.647, 95% CI 0.328, 0.912; ρ = 0.758, 95% CI 0.355, 0.923, P = 0.0027), and 5-year OS (R2 = 0.745, 95% CI 0.540, 0.951; ρ = 0.841, 95% CI 0.540, 0.951, P = 0.0003) (Fig. 3B–D and Table 3). On the other hand, the R2 for the association between the HR of OS and pathological RR was poor (R2 = 0.242, 95% CI 0.00, 0.709; ρ = −0.464, 95% CI −0.927, 0.558, P = 0.3542).
Next, we conducted sensitivity analyses to evaluate the surrogacy after excluding the five studies that involved patients with metastatic disease. The coefficients of determination for EFS and OS in studies of localised osteosarcoma tended to be lower than those in all studies together. The correlation between the HRs of EFS and OS was poor (R2 = 0.156, 95% CI 0.00, 0.511; ρ = 0.083, 95% CI −0.615, 0.708, P = 0.8312) (Table 4). The association between the HR of OS and RR in studies of localised osteosarcoma alone was good (R2 = 0.598, 95% CI 0.075, 1.00); however, the correlation was inverse and not significant (ρ = −0.800, 95% CI −0.996, 0.697, P = 0.20). Further sensitivity analyses to evaluate the surrogacy after excluding the three studies that involved radiation therapy and immuno-stimulants in the treatment arms were performed. The coefficients of determination for EFS and OS in the chemotherapy-based studies also showed similar tendency as those in the localised osteosarcoma studies (Table 5).
Discussion
One of the advantages of using a surrogate endpoint instead of OS in clinical trials is that the required number of events can be observed more quickly; therefore, the study period can be shortened and the cost reduced. However, the concern remains whether the surrogate endpoint correctly reflects the OS, which is the true endpoint. The correlation between intermediate endpoints and OS has never been investigated in RCTs for osteosarcoma; ours is the first such investigation to date.
EFS is often selected as the primary endpoint in RCTs of localised osteosarcoma. Among the 20 RCTs analysed in our study, EFS was adopted as the primary endpoint in nine, including all of the latest four. Our results indicated that EFS might only have a moderate correlation with OS. In terms of other time-to-event endpoints, 3-year OS and 5-year OS had stronger correlations with OS than did EFS, although neither of the intermediate endpoints we investigated indicated a ‘very good’ or ‘excellent’ association with OS assessed by R2. The suitability of EFS and RR as intermediate endpoints for OS was not confirmed in the present study; however, if there is a need to select intermediate endpoints in RCT for osteosarcoma, 3-year OS and 5-year OS could be recommended as surrogates for OS.
Because some of the RCTs analysed in this study involved patients with metastatic disease, not all RCTs investigated localised disease only. Although the same treatment strategy is often pursued for patients with localised and metastatic osteosarcomas, their prognoses are largely different; this may result in biases when these two groups are analysed together. Therefore, a sensitivity analysis was performed using only the RCTs that investigated patients with localised osteosarcoma. This analysis revealed that the correlation between EFS and OS was inferior to that observed in the overall analysis. The same trend was observed at other time-to-event endpoints except for the 1-year EFS. The RR showed a good correlation, with an R2 value of 0.598; however, it was an inverse correlation (ρ = −0.800), and no surrogacy was validated. Our data showed that, even when analysing RCTs of patients with localised osteosarcoma alone, the intermediate endpoints were not found to be suitable surrogates for OS.
The fastest-obtainable surrogate endpoint is the RR. In many RCTs of patients with osteosarcoma, postoperative chemotherapy regimens were switched according to the histological responses to preoperative chemotherapy; in three of these RCTs, RR was selected as the primary endpoint25,29,30. Although poor responders with unfavourable RRs are known to have worse prognoses4, our results showed that RR was not a suitable surrogate endpoint for OS. A possible reason for this is that the outcome of osteosarcoma treatments might be influenced not only by the effect of preoperative chemotherapy but also the quality of surgery and the effect of postoperative chemotherapy. In our meta-analysis, there were six RCTs that showed significant differences in RR between treatment groups; however, none showed significant differences in OS. Even if the pathological RR is improved by increasing the intensity of chemotherapy or the number of drugs, it has not been linked to the improvement of survival34. This might be because the effectiveness of preoperative chemotherapy alone does not fully reflect the outcome of the overall treatment.
In the field of sarcoma, three analyses of the endpoint surrogacy were performed in patients with advanced soft tissue sarcoma (STS)35,36,37. The first found a high correlation between progression-free survival (PFS) and OS; however, only the simple correlation coefficient was calculated, whereas the coefficient of determination was not35. In the second study, 14 RCTs encompassing 2,846 patients with advanced STS were compiled to compare the surrogacy of intermediate endpoints for OS. Although this was the first study to collect and analyse data from individual patients with advanced STS, PFS, time-to-progression, and time-to-treatment failure were found not to be surrogate endpoints for OS in both the individual and trial levels36. The third study was a trial-level surrogacy validation investigation using published data; however, all RCTs used in that analysis were trials of first-line treatments for advanced STS, and the standard treatment in all RCTs was the same (doxorubicin monotherapy). The correlation between PFS and OS in that study was moderate, while the correlations of other short-term endpoints with OS were relatively poor37. As our analysis of RCTs of patients with localised osteosarcoma who were treated with multimodal regimens did not show strong surrogacy, and since meta-analyses of RCTs investigating patients with advanced STS treated with chemotherapy alone also failed to identify a reliable surrogate endpoint, caution should be taken when planning to use such intermediate markers as primary endpoints.
There were several limitations in our study. First of all, the analyses were based on published data and not on individual patient data; therefore, only trial-level surrogacy was analysed while individual-level surrogacy was not. Second, there were variations in the definitions of time-to-event endpoints between trials, indicating heterogeneity among included RCTs. Third, the study phases and ITT analyses were clearly described in only 6 and 7 of the 20 RCTs, respectively, while none described post-protocol treatments; this indicated that the qualities of the RCTs used in our study were not high. Finally, some trials included patients with metastatic disease (comprising 4.5% of all patients analysed); therefore, the analyses were not exclusively of patients with localised osteosarcoma.
In conclusion, our first-of-its-kind trial-level investigation of the suitability of intermediate endpoints as surrogates for OS in RCTs of osteosarcoma did not confirm their suitability, even though the rareness of the disease and the difficulty inherent in conducting large-scale RCTs were taken into account. As such, there is a need to develop novel and effective intermediate endpoints when performing RCTs for patients with osteosarcoma.
Data availability
The datasets generated and/or analysed in the current study are available from the corresponding author on reasonable request.
References
American Cancer Society. Cancer facts and figures 2020. American Cancer Society https://www.cancer.org/research/cancer-facts-statistics/all-cancer-facts-figures/cancer-facts-figures-2020.html. (Accessed 2 May 2020).
Japanese Orthopaedic Association Musculoskeletal Tumor Committee. Bone Tumor Registry in Japan 2015. Tokyo: National Cancer Centre (2015)
Casali, P. G. et al. Bone sarcomas: ESMO-PaedCan-EURACAN Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 29(Suppl 4), iv79–95, https://doi.org/10.1093/annonc/mdy310 (2018).
Bacci, G. et al. Grade of chemotherapy-induced necrosis as a predictor of local and systemic control in 881 patients with non-metastatic osteosarcoma of the extremities treated with neoadjuvant chemotherapy in a single institution. Eur. J. Cancer 41, 2079–2085, https://doi.org/10.1016/j.ejca.2005.03.036 (2005).
Marina, N. M. et al. Comparison of MAPIE versus MAP in patients with a poor response to preoperative chemotherapy for newly diagnosed high-grade osteosarcoma (EURAMOS-1): an open-label, international, randomised controlled trial. Lancet Oncol. 17, 1396–1408, https://doi.org/10.1016/S1470-2045(16)30214-5 (2016).
Iwamoto, Y. et al. Multiinstitutional phase II study of neoadjuvant chemotherapy for osteosarcoma (NECO study) in Japan: NECO-93J and NECO-95J. J. Orthop. Sci. 14, 397–404, https://doi.org/10.1007/s00776-009-1347-6 (2009).
Iwamoto, Y. & Tanaka, K. The activity of the Bone and Soft Tissue Tumor Study Group of the Japan Clinical Oncology Group. Jpn. J. Clin. Oncol. 42, 467–470, https://doi.org/10.1093/jjco/hys059 (2012).
Moher, D., Liberati, A., Tetzlaff, J. & Altman, D. G., PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ. 339, b2535, https://doi.org/10.1136/bmj.b2535 (2009).
Smith, C. T. Meta-analysis of time-to-event data. Cochrane Training https://training.cochrane.org/resource/meta-analysis-time-event-data. Accessed 2 May 2020 (2018).
Tierney, J. F. et al. Practical methods for incorporating summary time-to-event data into meta-analysis. Trials. 8, 16, https://doi.org/10.1186/1745-6215-8-16 (2007).
Mantel, N. & Haenszel, E. Statistical aspects of the analysis of data from retrospective studies of disease. J. Natl. Cancer Inst. 22, 719–748 (1959).
Buyse, M. et al. Progression-free survival is a surrogate for survival in advanced colorectal cancer. J. Clin. Oncol. 25, 5218–5224, https://doi.org/10.1200/JCO.2007.11.8836 (2007).
Burzykowski, T. et al. Evaluation of tumor response, disease control, progression-free survival, and time to progression as potential surrogate end points in metastatic breast cancer. J. Clin. Oncol. 26, 1987–1992, https://doi.org/10.1200/JCO.2007.10.8407 (2008).
Lassere, M. N. The biomarker-surrogacy evaluation schema: a review of the biomarker-surrogate literature and a proposal for a criterion-based, quantitative, multidimensional hierarchical levels of evidence schema for evaluating the status of biomarkers as surrogate endpoints. Stat. Methods Med. Res. 17, 303–340, https://doi.org/10.1177/0962280207082719 (2008).
Winkler, K. et al. Neoadjuvant chemotherapy for osteogenic sarcoma: results of a Cooperative German/Austrian study. J. Clin. Oncol. 2, 617–624, https://doi.org/10.1200/JCO.1984.2.6.617 (1984).
Working Party on Bone Sarcoma. A trial of chemotherapy in patients with osteosarcoma. Br. J. Cancer 53, 513–518, https://doi.org/10.1038/bjc.1986.81 (1986).
Bacci, G. et al. Adriamycin-methotrexate high dose versus adriamycin-methotrexate moderate dose as adjuvant chemotherapy for osteosarcoma of the extremities: a randomized study. Eur. J. Cancer Clin. Oncol. 22, 1337–1345 (1986).
Krailo, M. et al. A randomized study comparing high-dose methotrexate with moderate-dose methotrexate as components of adjuvant chemotherapy in childhood nonmetastatic osteosarcoma: a report from the Childrens Cancer Study Group. Med. Pediatr. Oncol. 15, 69–77 (1987).
Winkler, K. et al. Neoadjuvant chemotherapy of osteosarcoma: results of a randomized cooperative trial (COSS-82) with salvage chemotherapy based on histological tumor response. J. Clin. Oncol. 6, 329–337, https://doi.org/10.1200/JCO.1988.6.2.329 (1988).
Burgers, J. M. et al. Osteosarcoma of the limbs. Report of the EORTC-SIOP 03 trial 20781 investigating the value of adjuvant treatment with chemotherapy and/or prophylactic lung irradiation. Cancer 61, 1024–1031 10.1002/1097-0142(19880301)61:5%3C1024::aid-cncr2820610528%3E3.0.co;2-p (1988).
Bacci, G. et al. Primary chemotherapy and delayed surgery (neoadjuvant chemotherapy) for osteosarcoma of the extremities. The Istituto Rizzoli Experience in 127 patients treated preoperatively with intravenous methotrexate (high versus moderate doses) and intraarterial cisplatin. Cancer 65, 2539–2553 10.1002/1097-0142(19900601)65:11%3C2539::aid-cncr2820651125%3E3.0.co;2-m (1990).
Bramwell, V. H. et al. A comparison of two short intensive adjuvant chemotherapy regimens in operable osteosarcoma of limbs in children and young adults: the first study of the European Osteosarcoma Intergroup. J. Clin. Oncol. 10, 1579–1591, https://doi.org/10.1200/JCO.1992.10.10.1579 (1992).
Souhami, R. L. et al. Randomised trial of two regimens of chemotherapy in operable osteosarcoma: a study of the European Osteosarcoma Intergroup. Lancet 350, 911–917, https://doi.org/10.1016/S0140-6736(97)02307-6 (1997).
Meyers, P. A. et al. Intensification of preoperative chemotherapy for osteogenic sarcoma: results of the Memorial Sloan-Kettering (T12) protocol. J. Clin. Oncol. 16, 2452–2458, https://doi.org/10.1200/JCO.1998.16.7.2452 (1998).
Bacci, G. et al. A comparison of methods of loco-regional chemotherapy combined with systemic chemotherapyas neo-adjuvant treatment of osteosarcoma of the extremity. Eur. J. Surg. Oncol. 27, 98–104, https://doi.org/10.1053/ejso.2000.1056 (2001).
Goorin, A. M. et al. Presurgical chemotherapy compared with immediate surgery and adjuvant chemotherapy for nonmetastatic osteosarcoma: Pediatric Oncology Group Study POG-8651. J. Clin. Oncol. 21, 1574–1580, https://doi.org/10.1200/JCO.2003.08.165 (2003).
Meyers, P. A. et al. Osteosarcoma: a randomized, prospective trial of the addition of ifosfamide and/or muramyl tripeptide to cisplatin, doxorubicin, and high-dose methotrexate. J. Clin. Oncol. 23, 2004–2011, https://doi.org/10.1200/JCO.2005.06.031 (2005).
Lewis, I. J. et al. Improvement in histologic response but not survival in osteosarcoma patients treated with intensified chemotherapy: a randomized phase III trial of the European Osteosarcoma Intergroup. J. Natl. Cancer Inst. 99, 112–128, https://doi.org/10.1093/jnci/djk015 (2007).
Le Deley, M. C. et al. SFOP OS94: a randomised trial comparing preoperative high-dose methotrexate plus doxorubicin to high-dose methotrexate plus etoposide and ifosfamide in osteosarcoma patients. Eur. J. Cancer 43, 752–761, https://doi.org/10.1016/j.ejca.2006.10.023 (2007).
Ferrari, S. et al. Neoadjuvant chemotherapy with methotrexate, cisplatin, and doxorubicin with or without ifosfamide in nonmetastatic osteosarcoma of the extremity: an Italian sarcoma group trial ISG/OS-1. J. Clin. Oncol. 30, 2112–2118, https://doi.org/10.1200/JCO.2011.38.4420 (2012).
Bielack, S. S. et al. Methotrexate, doxorubicin, and cisplatin (MAP) plus maintenance pegylated interferon alfa-2b versus MAP alone in patients with resectable high-grade osteosarcoma and good histologic response to preoperative MAP: first results of the EURAMOS-1 good response randomized controlled trial. J. Clin. Oncol. 33, 2279–2287, https://doi.org/10.1200/JCO.2014.60.0734 (2015).
Piperno-Neumann, S. et al. Zoledronate in combination with chemotherapy and surgery to treat osteosarcoma (OS2006): a randomised, multicentre, open-label, phase 3 trial. Lancet Oncol. 17, 1070–1080, https://doi.org/10.1016/S1470-2045(16)30096-1 (2016).
Senerchia, A. A. et al. Results of a randomized, prospective clinical trial evaluating metronomic chemotherapy in nonmetastatic patients with high-grade, operable osteosarcomas of the extremities: A report from the Latin American Group of Osteosarcoma Treatment. Cancer 123, 1003–1010, https://doi.org/10.1002/cncr.30411 (2017).
Bramer, J. A., van Linge, J. H., Grimer, R. J. & Scholten, R. J. Prognostic factors in localized extremity osteosarcoma: a systematic review. Eur. J. Surg. Oncol. 35, 1030–1036, https://doi.org/10.1016/j.ejso.2009.01.011 (2009).
Zer, A., Prince, R. M., Amir, E. & Abdul Razak, A. Evolution of randomized trials in advanced/metastatic soft tissue sarcoma: End point selection, surrogacy, and quality of reporting. J. Clin. Oncol. 34, 1469–1475, https://doi.org/10.1200/JCO.2015.64.3437 (2016).
Savina, M. et al. Surrogate endpoints in advanced sarcoma trials: a meta-analysis. Oncotarget 9, 34617–34627, https://doi.org/10.18632/oncotarget.26166 (2018).
Tanaka, K., Kawano, M., Iwasaki, T., Itonaga, I. & Tsumura, H. Surrogacy of intermediate endpoints for overall survival in randomized controlled trials of first-line treatment for advanced soft tissue sarcoma in the pre- and post-pazopanib era: a meta-analytic evaluation. BMC Cancer 19, 56, https://doi.org/10.1186/s12885-019-5268-2 (2019).
Acknowledgements
This work was supported in part by the National Cancer Centre Research and Development Fund [grant number 29-A-3] and AMED [grant number JP17ck0106336]. We would like to acknowledge Kureha Special Laboratory Co., Ltd. (Tokyo, Japan) for its assistance in performing the statistical analysis. We would like to thank Editage (www.editage.jp) for English language editing.
Author information
Authors and Affiliations
Contributions
K.T. and H.T. conceived the study concept and design. K.T., M.K., T.I., and I.I. performed the systematic search of the literature and extraction of data. K.T, M.K., T.I., I.I., S.M. and H.T. performed statistical analysis, interpretation of data, and drafting and revision of the manuscript. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tanaka, K., Kawano, M., Iwasaki, T. et al. Surrogate endpoints for overall survival in randomised controlled trials of localised osteosarcoma: A meta-analytic evaluation. Sci Rep 10, 8573 (2020). https://doi.org/10.1038/s41598-020-65591-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-65591-z
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.