Abstract
The misuse of pharmaceutical opioids is a major public health issue. In Australia, codeine was re-scheduled on 1 February 2018 to restrict access; it is now only available on prescription. The aim of this study was to measure the change in dental opioid prescriptions, one year before and after the codeine re-scheduling in Australia and to assess dental prescribing rates of opioids for 2018 by population and by clinician. Data was extracted for dental opioids for the year immediately prior and after the codeine up-schedule (1 February 2017-31 January 2019) from the publicly-available national prescription database (Pharmaceutical Benefits Scheme). Descriptive statistics, T-tests and odds ratios were used to identify significant prescribing differences. Codeine, codeine/paracetamol, oxycodone and tramadol use increased significantly the year after the codeine restriction than the previous year (13.8–101.1%). Australian dentists prescribed 8.6 prescriptions/1,000 population in 2018, with codeine/paracetamol accounting for most prescriptions (96%). The significant increase in opioid prescribing highlights that Australian dentists may be contributing to the misuse of pharmaceutical opioids. Educational efforts should be targeted at the appropriate use of opioids and patient selection. Dentists should be added to the prescription monitoring system SafeScript so they can make informed decisions for patients who are potentially misusing opioids.
Similar content being viewed by others
Introduction
Australia’s opioid crisis has been well documented; hospitalisations for pharmaceutical opioid poisoning surpassed those of heroin use and the misuse of pharmaceutical drugs as the most common reason for people to enter drug and alcohol treatment programs in Australia1. Pharmaceutical opioid deaths are now increasing at a greater rate than that due to heroin use as shown in a retrospective analysis from the National Coronial Information Systems between 2001 to 20122, and three people die every day from drug-related events involving pharmaceutical opioids3. Forty percent of deaths due to prescription opioid use were associated with a valid prescription of oxycodone for non-cancer pain (2001 to 2011)1.
The main source of opioids for non-medical use is leftover tablets of legitimate prescriptions sourced through social networks such as family and friends4. In Australia, codeine products, both over the counter (OTC) and prescription, comprised the most commonly misused pharmaceutical products, followed by oxycodone and tramadol5. Codeine is the most frequently used opioid analgesic in Australia6, and ranks fourth behind methadone, oxycodone, and morphine in being implicated in pharmaceutical opioid overdose deaths2. Nearly three in five (56%) codeine-containing analgesic products were purchased OTC in 20137. Prescription-only codeine utilisation was not accounted for in 21% of claims on Australia’s national medicines subsidy, the Pharmaceutical Benefits Scheme (PBS)7. A case series study reported that two in five (39%) patients with an opioid addiction were dependent on codeine alone8. The rate of codeine-related deaths increased several fold over ten years (3.5/million in 2000; 8.7/million in 2009)9. In response to the misuse, risks and toxicity, the Therapeutic Goods Administration (TGA) up-scheduled codeine from OTC and prescription to prescription only on 1 February 201810.
Dentists in Australia have increasingly been prescribing opioids; the most common product is codeine with paracetamol (Panadeine Forte)11. This pattern continues despite non-steroidal anti-inflammatory drugs (NSAID) being more appropriate for dental pain12,13. NSAIDs, alone or in combination with paracetamol, were found to be the most effective for dental pain while minimising adverse effects in an overview of systematic reviews14. Irrespective, dental opioid prescribing increased 30% between 2013 and 201611.
The legislative change to codeine scheduling appears to have reduced codeine misuse as highlighted in a recent analysis of monthly opioid exposure calls to the New South Wales Poisons Information Centre15. Additionally, studies of medical opioid prescribing indicate that there is no change in rates of dispensed opioid prescriptions after the legislative change compared to before (from January 2016 to December 2018)16. However, dental prescribing of opioids is increasing and some dentists are inappropriately preferencing the use of opioids over NSAIDs as recommended in the current Australian therapeutic guidelines17. The effect of the codeine rescheduling change on dental opioid prescribing is unknown. Thus, there were two aims of this study: 1) to assess prescribing rates of dental opioids in 2018 and 2) to analyse the dispensed use of dental opioids, one year before and one year after the up-scheduling of codeine.
Method
This study involved an analysis of dispensed prescriptions to the Australian population from outpatient pharmacies from a nationally-representative database. The data used in this study was obtained from Medicare Australia administered by the Australian Government Department of Health. As the data was publicly available and aggregated, no ethical approval was required. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting guideline was followed.
Data sources
The Department of Health provides a national database on prescribed medicines in Australia that are recorded under the Pharmaceutical Benefits Scheme (PBS)18. We analysed opioids subsidised by the Australian Government for dental prescribers listed on the PBS including codeine, codeine 30 mg/paracetamol 500 mg, hydrocodone, morphine, oxycodone, and tramadol. For the first part of the study, we assessed opioid use one year prior immediately prior to and one year after the change in codeine re-scheduling to prescription only (1 February 2018) by extracting monthly data of PBS dispensed use of dental opioids from 1 February 2017 to 31 January 2019. In Australia, the government subsidises the cost of medicines listed on the PBS. Some medicines (and for specific indications) are not subsidised and so the prescriber will issue a ‘private’ prescription where the consumer funds the full cost of the medicine. These are not recorded under PBS data and therefore are not accounted for in the analysis. PBS prescriptions account for approximately 75% of dispensed medicine use in Australia, where prescriptions are dispensed in hospital settings and outpatient pharmacies19. However, most dental prescriptions would be from community pharmacies, of which PBS subsidised prescriptions represent around 93% of community prescriptions20. Therefore, the majority of dental prescriptions (closer to 93%) would be accounted for in this analysis. The ‘date of supply’ dataset was accessed as this is recommended when analysing medicine use19. Given the variations in processing, the dataset was accessed six months after the end study date (i.e. 1 August 2019)19. Dentists are urged to follow the Australian therapeutic guidelines17 but they are free to prescribe whatever medicines they choose. Despite the PBS being a national formulary, the laws governing the supply and prescribing of medicines varies among the states of Australia. The relevant difference is that dentists in Queensland are prohibited from prescribing oxycodone21.
For the second part of the study, we assessed the dental prescribing rates by population and clinician from 1 January to 31 December 2018. The number of registered dental practitioners were obtained from the Australian Health Practitioner Regulation Agency22 and the population value from the Australian Bureau of Statistics23.
Outcome measures
Three outcomes were measured: 1) dental opioid use for 2018 using the standardised defined daily dose (DDD)/1,000 inhabitants/day metric (see below); 2) prescribing rates for 2018 by population and by dentist; 3) the mean percentage difference for each individual opioid for the year before and after the rescheduling change.
Statistical analysis
We extracted and analysed data using Microsoft Excel. Descriptive statistics were used to determine dental opioid use for 2018 by population and by clinician. Medicine use was calculated using the accepted and standardised metric of the DDD per 1,000 populations per day18,24. The DDD value for each medicine was obtained from the World Health Organization Collaborating Centre for Drug Statistics Methodology; it is the average maintenance dose per day for the main indication in adults24. The DDD metric is acceptable to reflect utilisation for the opioids listed except oxycodone, as the WHO DDD has been found to be two to seven times higher than actual doses used for oxycodone25. Therefore, for oxycodone only, doses were converted to oral morphine equivalents and the morphine DDD used to calculate the DDD/1000 population/day metric to indicate utilisation26. In Australia, the number of tablets per pack is standardised and most products are prescribed in PBS-specified packs.
T-tests (two-tailed) were used to identify significant differences between the number of opioid prescriptions one year before and after the re-scheduling change; p < 0.05 was considered significant. Odds ratios were also used to quantify the proportionate change for each opioid for the year after the codeine restriction compared to the previous year, relative to the increase the population. The mid-year population was used, with the data obtained from the ABS.
Results
Dental opioid dispensed use in Australia
There were 213,933 dispensed dental opioid prescriptions in 2018 in Australia. Australian dentists prescribed 8.6 opioid prescriptions/1,000 population, 12.1 prescriptions/dentist and 0.1 total DDD/1000 inhabitants/day. Australian dentists preferentially prescribed the codeine 30 mg/paracetamol 500 mg product (accounted for 205,189 prescriptions, 96% of opioid prescriptions), followed by oxycodone (6,050 prescriptions, 2.8% of opioid prescriptions) and tramadol (2,490 prescriptions, 1.2% of opioid prescriptions) (Table 1).
Trends in dispensed use of dental opioids
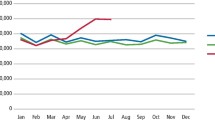
When comparing the year after the up-scheduling of codeine to the previous year, there was a significant increase in the mean difference for the most commonly dispensed four opioids. The use of codeine, codeine/paracetamol, oxycodone and tramadol increased significantly over the two-year period (codeine: 101.1% mean increase from 8.7 to 17.5 prescriptions, p = 0.002; codeine 30 mg/paracetamol 500 mg: 20.6% mean increase from 14,337.0 to 17,284.5 prescriptions., p < 0.0001; oxycodone: 23.5% mean increase from 416.1 to 514.0 prescriptions, p < 0.001; tramadol: 13.8% mean increase from 183.1 to 208.3 prescriptions, p = 0.03). This occurred despite the increase in the population and number of clinicians over the same period being much lower (1.6% and 3.1% respectively). (Table 2, Figs. 1 and 2). The use of hydromorphone and morphine had low use. When comparing the year before and after the re-scheduling change, hydromorphone use stayed the same with one prescription dispensed both the year before and after the change; morphine use decreased from 15 prescriptions to 3 prescriptions respectively.
When assessing the relative proportions of dispensed opioids, there was an increase in the relative proportions of opioids prescribed the year after the codeine restriction compared to the previous year, adjusted for the population increase. (codeine: OR: 2.0; codeine/paracetamol: OR: 1.2; oxycodone OR: 1.2; tramadol OR: 1.1).
Discussion
This is the first study to assess the effect of the codeine up-schedule on dental opioid prescribing in Australia. There was a substantial increase in dispensed dental opioid use after the up-scheduling of codeine with relatively smaller increases in the population and clinician numbers. As expected, this was significant for codeine and codeine with paracetamol, but also evident for oxycodone and tramadol. This increase occurred despite the Australian therapeutic guidelines indicating that opioids are not to be used first line to treat dental pain17, and tramadol is also not recommended17.
Multiple studies have confirmed that NSAIDs alone or in combination with paracetamol are more effective than opioid combinations for dental pain27 and as such opioids are not first line treatment in the current Australian guidelines17. Moreover, analgesic doses of codeine provide no extra pain relief in the surgical extraction of third molars compared to paracetamol and ibuprofen alone28. Furthermore, patients undergoing third molar extractions who received the opioid combination products had significantly more adverse effects compared to the patients who received the NSAID combination products27. Given the limited role of opioids in dental pain and that the vast majority of dental conditions require active treatment alone where medicines would only be adjunctive17, theoretically, the dental prescribing of opioids should not be increasing at a rate greater than the population change. Previous longitudinal studies of dental opioid prescribing in Australia showed increases in the use of opioids11 but this does not explain the significant increase after the codeine up-schedule. About one in four dentists in Australia (16-27%) would preference the use of analgesics only (paracetamol, paracetamol/codeine, tramadol or oxycodone) for dental pain, instead of NSAIDs29. Australian dentists tend to recommend tramadol if their patient is allergic or finds codeine ineffective30, despite both medicines requiring transformation by cytochrome P450 2D6 to the active metabolites. The pharmacogenomic variability of cytochrome P450 2D6 is well established and therefore patients who have inherited two non-functional alleles of this enzyme will likely find both medicines ineffective. In addition, other non-clinical factors have been identified for prescribing by Australian dentists, such as patient pressure, fear of litigation, and the desire by dentists to make patients feel well managed30.
While the increase in the use of paracetamol/codeine may be partly accounted for by the up-scheduling of codeine, the large increase suggests that dentists may also be inappropriately prescribing opioids, or some people may be acquiring opioids from dentists for misuse31. A case series analysis of people who sought help for opioid dependence showed that many initiated codeine for the treatment of headaches and dental pain8. A previous study in Boston, USA showed that one in eight (12%) people who presented to the emergency department with backache, dental pain or headache were seeking to acquire an opioid drug31. A pre-filled opioid prescription, given prior to the extraction of wisdom teeth, is an independent risk factor for persistent opioid use32. Dental opioid prescriptions may be associated with subsequent opioid abuse in adolescents and young adults33.
Dental prescribing of opioids varies substantially across countries. Underscoring the findings from this analysis of dental opioid prescribing rates, a recent study by Suda et al. compared opioid prescribing between US and English dentists in 201634. Australian dentists prescribe around four times less than those in the US (35.4 opioid prescriptions/1,000 US population versus 8.6 opioid prescriptions/1,000 Australian population) and almost five times less than those in the US when adjusting by clinician (58.2 opioid prescriptions/US dentist versus 12.1 opioid prescriptions/Australian dentist)34. In addition, US dentists prescribe a larger range of opioids and long-acting opioids than Australian dentists34. Notwithstanding this substantial difference to US dentists, Australian dentists prescribe 21 times more opioids than English dentists when adjusted for population (0.5 prescriptions/1,000 English population), and about ten times the rate when adjusting by clinician34. This vast difference in prescribing rates occurs despite all three countries displaying similar levels of oral health as measured by such as decayed, missing and filled teeth indexes, and levels of edentulousness35. Suda et al. noted that one major difference among prescribing practises is that English dentists are restricted to prescribe from a medicine formulary; they can only prescribe dihydrocodeine on the NHS34. Nevertheless, such a striking contrast must be questioned whether this is due to prescribing practices alone, or potentially opioid seeking behaviour across these different countries. Further, it may well be that there are much higher levels of opioid use in England as these medications are readily available over the counter and thus their use merely not measurable.
The limitations of this study are that the Australian PBS data only captures prescribing on the PBS and does not include prescriptions that are privately funded. Furthermore, no prescription would be generated where the dentist both prescribed and supplied the medicine directly to patients. It is expected that these sources of prescriptions will be relatively low; dental dispensing is rare in Australia. Prescription details such as dose, frequency and duration of treatment were not available therefore it was not possible to assess the appropriateness of the prescriptions. These data are not linked to morbidity or mortality data so it is not possible to ascertain any link between dental opioid prescribing and opioid misuse. Nevertheless, these data allowed for international comparisons and demonstrated prescribing trends in most of the population.
This study has substantial public health implications. It should initiate a call to action for dentists regarding their role in responsible opioid prescribing, as well as to re-evaluate the recommendations of opioids in dental practice. Given the established misuse of pharmaceutical opioids in Australia5 and other countries, and the associated public health burden, dentists should only prescribe opioids if NSAIDs and paracetamol have not been effective or cannot be tolerated; they should ensure a true therapeutic need exists. Continuing education efforts could be directed towards the appropriate use and limitations of opioids in general dental practice, with future research aimed at developing strategies to improve opioid prescribing among dentists at an individual level.
Strategies could be implemented for the early identification of people who are at higher risk of developing opioid dependence. Opioid-dependent individuals tend to also have co-morbidities of pre-existing chronic pain and psychiatric conditions8. High rates of concomitant prior mental health, substance use and chronic pain issues were recorded for these deaths due to codeine toxicity in a prospective study from the National Coronial Information System9. Given that the predominant source of drugs for misuse is mostly through leftover pills from legitimate prescriptions sourced through social networks4, manufacturers could consider the formulation of opioids in smaller pack sizes to reduce the number of leftover pills from a prescription.
One other measure to assist with the prescribing of medicines prone to misuse is the Safe Script program36, which is obligatory in the Australian states of Victoria and Tasmania from April 2020. This initiative allows prescribers to access real-time prescription monitoring for all patients who acquire medicines prone to misuse from each prescriber and pharmacist to assist with ‘doctor shopping’ and to prevent the increased acquisition of these drugs from multiple clinicians. The initiative is only implemented in two Australian states and includes medical doctors, nurses, and pharmacists only. Given the significant increase in the use of codeine since the scheduling change, including dentists in the program will assist them to make more informed decisions when prescribing drugs of dependence to people who are seeking opioids for non-medical use.
Conclusion
Underscoring the opioid crisis in Australia, this study illustrates how the increased prescribing of opioids by dentists, especially since the codeine restriction, may be contributing to the epidemic and how dental prescriptions can be a source of misuse. While guidelines are in place, further restrictions from the PBS such as prescribing authority prescriptions for selected indications and restricted quantities are some ways to curtail prescribing and limit the number of tablets prescribed. The prescription monitoring system - Safescript - should include dentists to allow them to be aware when prescribing opioids for people who acquire them for non-medical use. Consideration should be given when prescribing opioids for patients who have chronic pain, and procedures established for the early identification of people who may be prone to opioid dependence. Further research is needed to investigate the appropriateness of prescribing practices, and both undergraduate and continuing education of dentists should be targeted about the role of opioids in dental pain management.
References
Weekes, L. M. Multidisciplinary approach to reducing pharmaceutical misuse. Med. J. Aust. 204, 96 (2016).
Roxburgh, A. et al. Trends in heroin and pharmaceutical opioid overdose deaths in Australia. Drug. Alcohol. Depend. 179, 291–298, https://doi.org/10.1016/j.drugalcdep.2017.07.018 (2017).
Welfare, A. I. o. H. a. Opioid harm in Australia and comparison between Australia and Canada., (AIHW, Canberra, 2018).
Hulme, S., Bright, D. & Nielsen, S. The source and diversion of pharmaceutical drugs for non-medical use: A systematic review and meta-analysis. Drug. Alcohol. Depend. 186, 242–256, https://doi.org/10.1016/j.drugalcdep.2018.02.010 (2018).
Australian Institute of Health and Welfare. National Drug Strategy Household Survey 2016 Detailed findings, 2016).
Degenhardt, L. et al. The extent and correlates of community-based pharmaceutical opioid utilisation in Australia. Pharmacoepidemiol. Drug. Saf. 25, 521–538, https://doi.org/10.1002/pds.3931 (2016).
Gisev, N. et al. To what extent do data from pharmaceutical claims under-estimate opioid analgesic utilisation in Australia? Pharmacoepidemiol. Drug. Saf. 27, 550–555, https://doi.org/10.1002/pds.4329 (2018).
Nielsen, S. et al. Comparing treatment-seeking codeine users and strong opioid users: Findings from a novel case series. Drug. Alcohol. Rev. 34, 304–311, https://doi.org/10.1111/dar.12224 (2015).
Roxburgh, A. et al. Trends and characteristics of accidental and intentional codeine overdose deaths in Australia. Med. J. Aust. 203, 299, https://doi.org/10.5694/mja15.00183 (2015).
Australian Government Department of Health, T. G. A. Codeine information hub; Changes to patient access for medicines containing codeine, https://www.tga.gov.au/codeine-info-hub
Teoh, L., Marino, S. K., McCullough, R. & Part, M. 2. Current prescribing trends of dental non-antibacterial medicines in Australia from 2013 to 2016. Aust. Dent. J. 63, 338–346, https://doi.org/10.1111/adj.12613 (2018).
Becker, D. E. & Phero, J. C. Drug therapy in dental practice: nonopioid and opioid analgesics. Anesth Prog 52, 140–149, https://doi.org/10.2344/0003-3006(2005)52[140:DTD]2.0.CO;2 (2005).
Aminoshariae, A., Kulild, J. C., Donaldson, M. & Hersh, E. V. Evidence-based recommendations for analgesic efficacy to treat pain of endodontic origin: A systematic review of randomized controlled trials. J. Am. Dental Assoc. 147, 826–839, https://doi.org/10.1016/j.adaj.2016.05.010 (2016).
Moore, P. A. et al. Benefits and harms associated with analgesic medications used in the management of acute dental pain: An overview of systematic reviews. J. Am. Dental Assoc. 149, 256–265 e253, https://doi.org/10.1016/j.adaj.2018.02.012 (2018).
Cairns, R., Schaffer, A. L., Brown, J. A., Pearson, S. A. & Buckley, N. A. Codeine use and harms in Australia: evaluating the effects of re-scheduling. Addiction, https://doi.org/10.1111/add.14798 (2019).
Middleton, M. & Nielsen, S. Changes in Australian prescription opioid use following codeine rescheduling: A retrospective study using pharmaceutical benefits data. Int. J. Drug. Policy 74, 170–173, https://doi.org/10.1016/j.drugpo.2019.08.008 (2019).
Group, O. a. D. E. Therapeutic Guidelines: Oral and Dental Version 3. (Therapeutic Guidelines Ltd, 2019).
Australian Government Department ofHealth. About the PBS, http://www.pbs.gov.au/info/about-the-pbs (2017).
Mellish, L. et al. The Australian Pharmaceutical Benefits Scheme data collection: a practical guide for researchers. BMC Res. Notes 8, 634, https://doi.org/10.1186/s13104-015-1616-8 (2015).
Australian Government Department of Health. Australian Statistics on Medicines 2011, http://www.pbs.gov.au/info/statistics/asm/asm-2011#info_on_asm (2011).
Queensland Health Act 1937. (1 October 2017).
Australian Health Practitioner Regulation Agency. Dental Board of Australia - Registrant Data, (September 2018).
Australian Government. Australian Bureau of Statistics, http://www.abs.gov.au/.
World Health Organisation for Drug Statistics Methodology Centre. ATC/DDD Index 2017, https://www.whocc.no/atc_ddd_index/
Nielsen, S. et al. Defined daily doses (DDD) do not accurately reflect opioid doses used in contemporary chronic pain treatment. Pharmacoepidemiol. Drug. Saf. 26, 587–591, https://doi.org/10.1002/pds.4168 (2017).
Nielsen, S., Degenhardt, L., Hoban, B. & Gisev, N. A synthesis of oral morphine equivalents (OME) for opioid utilisation studies. Pharmacoepidemiol. Drug. Saf. 25, 733–737, https://doi.org/10.1002/pds.3945 (2016).
Daniels, S. E., Goulder, M. A., Aspley, S. & Reader, S. A randomised, five-parallel-group, placebo-controlled trial comparing the efficacy and tolerability of analgesic combinations including a novel single-tablet combination of ibuprofen/paracetamol for postoperative dental pain. Pain 152, 632–642, https://doi.org/10.1016/j.pain.2010.12.012 (2011).
Best, A. D. et al. Efficacy of Codeine When Added to Paracetamol (Acetaminophen) and Ibuprofen for Relief of Postoperative Pain After Surgical Removal of Impacted Third Molars: A Double-Blinded Randomized Control Trial. J. Oral. Maxillofac. Surg. 75, 2063–2069, https://doi.org/10.1016/j.joms.2017.04.045 (2017).
Teoh, L., Marino, S. K. & McCullough, R. M. A survey of prescribing choices of general dentists in Australia. BMC Oral. Health 19, 193, https://doi.org/10.1186/s12903-019-0882-6. (2019).
Teoh, L., Marino, S. K. & McCullough, R. M. Perceptions, attitudes and factors that influence prescribing by general dentists in Australia: a qualitative study. J. Oral. Pathol. Med. 48, 647–654, https://doi.org/10.1111/jop.12909 (2018).
Weiner, S. G. et al. Characteristics of emergency department “doctor shoppers”. J. Emerg. Med. 48, 424–431 e421, https://doi.org/10.1016/j.jemermed.2014.11.008 (2015).
Harbaugh, C. M. et al. Persistent Opioid Use After Wisdom Tooth Extraction. JAMA 320, 504–506, https://doi.org/10.1001/jama.2018.9023 (2018).
Schroeder, A. R., Dehghan, M., Newman, T. B., Bentley, J. P. & Park, K. T. Association of Opioid Prescriptions From Dental Clinicians for US Adolescents and Young Adults With Subsequent Opioid Use and Abuse. JAMA Intern. Med. 179, 145–152, https://doi.org/10.1001/jamainternmed.2018.5419 (2019).
Suda, K. J. et al. Comparison of Opioid Prescribing by Dentists in the United States and England. JAMA Netw. Open. 2, e194303, https://doi.org/10.1001/jamanetworkopen.2019.4303 (2019).
Blinkhorn, A. S. Comparison of the dental health of adults and children living in NSW with their counterparts in the US and UK. N. S W Public. Health Bull. 20, 52–55, https://doi.org/10.1071/NB08067 (2009).
Department of Health and Human Services, V. S. G. SafeScript, https://www2.health.vic.gov.au/public-health/drugs-and-poisons/safescript.
Acknowledgements
This research was supported by the Australian Government Research Training Program Scholarship (no. 241616) (LT).
Author information
Authors and Affiliations
Contributions
L.T. conceived the methodology, contributed to acquisition of the data, complied the results, analysed and interpreted the data, drafted the manuscript and gave final approval. S.H. conceived the methodology, assisted with the analysis and interpretation of the results, drafting of the manuscript and gave final approval. R.M. assisted with the analysis and interpretation of the results and gave final approval. M.Mc. contributed to the analysis and interpretation of the data, presentation of the results, drafted the manuscript and gave final approval.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Teoh, L., Hollingworth, S., Marino, R. et al. Dental opioid prescribing rates after the up-scheduling of codeine in Australia. Sci Rep 10, 8463 (2020). https://doi.org/10.1038/s41598-020-65390-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-65390-6
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.