Abstract
Coronary artery lesion (CAL) caused by Kawasaki disease (KD) is currently the most common acquired heart disease in children in many countries. Nevertheless, there is no single useful marker existing for predicting CAL of KD. Recently, many reports have noted that N-terminal pro-brain natriuretic peptide (NT-proBNP) can be utilized as a biomarker to predict CAL. Thus, we perform a meta-analysis to ascertain the diagnostic value of NT-proBNP in detecting CAL of KD in the acute phase. PubMed, the Cochrane Central Register of Controlled Trials, EMBASE, and China National Knowledge Infrastructure were searched to detect relevant publications. Finally, eight eligible studies were included. The overall diagnostic sensitivity and specificity were 0.84 (95% confidence interval [CI]: 0.78–0.89) and 0.71 (95% CI: 0.68–0.75), respectively. The area under the summary receiver operating characteristic curves value (SROC) curve was 0.8582 ± 0.0531. Moreover, the overall sensitivity and specificity across five studies adopted the threshold of approximately 900 ng/L were 0.82 (95% CI: 0.73–0.89) and 0.72 (95% CI: 0.68–0.76), respectively. SROC was 0.8868 ± 0.0486. This meta-analysis would be the first one to describe the role of NT-proBNP in detecting CAL of KD. We register this study with PROSPERO (CRD42019130083).
Similar content being viewed by others
Introduction
Kawasaki disease (KD) is a systemic, self-limited vasculitis. However, its etiology is still unknown. The major adverse impacts of this disease are mainly dependent on the occurrence of coronary artery lesions (CALs) among children paitents1. At present, KD ranks the top prevalence in pediatric acquired heart diseases around the world, including developed and rapidly industrializing countries2,3,4. Intravenous immunoglobulin (IVIG) has been identified to help reduce the prevalence of CAL to about 4%5. As stratified initial treatment in patients with high predictive risk could reduce this risk, lots of studies have focused on exploring potential biomarkers to diagnosis KD or predict the rate of CAL in KD patients, for example, erythrocyte sedimentation rate (ESR) and C reactive protein (CRP). But the predictive values of recognizing biomarkers are partly limited6.
As a biomarker for the diagnosis and monitoring disease progression in heart failure, serum N-terminal pro-brain natriuretic peptide (NT-proBNP) has been globally endorsed in clinical guidelines7,8. Moreover, NT-proBNP has been considered as a potential diagnostic biomarker for KD9,10. Since Kaneko et al.11 first reported NT-proBNP levels may associate with the development of CAL in KD, and it can be considered as a valuable biomarker to predict the risk of CAL in acute KD before initial IVIG treatment. Although a series of studies12,13,14 on the diagnostic value of NT-proBNP to detect CAL of KD have been carried out in recent years, but they could not achieve a convincing result. Thus, we carried out this meta-analysis to ascertain the diagnostic accuracy of NT-proBNP in detecting CAL of KD in the acute period before initial IVIG treatment.
Results
Search results
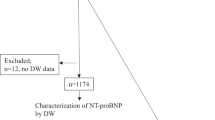
A total of 291 potentially relevant reports were retrieved. Among them, 27 papers were picked up by review their titles and abstracts with great topic interests. Then 5 studies were excluded due to the following reasons, including inappropriate research design without a diagnostic test (n = 8), lacking available data to generate a 2 × 2 table (n = 7), and failed to provide control data (n = 4). Finally, 8 studies11,14,15,16,17,18,19,20 with 197 CAL patients and 664 non-CAL patients in acute KD were included in this analysis. The flow chart of the study selection was illustrated in Fig. 1. Among the enrolled studies, the study design of four were retrospective trials, and the remaining four articles were prospective trials. The basic characteristics of all enrolled studies were presented in Table 1.
Study quality
We assessed the quality assessment of the included reports following the questions in the Quality Assessment of Diagnostic Accuracy Studies (QUADAS) list, and the results were illustrated in Table 2. Besides, all the eligible reports did not indicate follow-up time for CAL, which may be transient or permanent, which may increase bias the disease bias.
Diagnostic accuracy of NT-proBNP in detecting KD with CAL
Figure 2 summarizes the overall diagnostic value to predict KD with CAL by the level of NT-proBNP, demonstrating an overall sensitivity of 0.84 (95%CI, 0.78 to 0.89) with no significant heterogeneity (P = 0.5068, x2 = 6.29, I2 = 0.0%) (Fig. 2A), a specificity of 0.71 (95%CI, 0.68–0.75) combining a noticeable heterogeneity (P = 0.0009, x2 = 24.47, I2 = 71.4%) (Fig. 2B), and a diagnostic odds ratio (DOR) of 13.52 (95% CI, 6.98–26.21) with significant heterogeneity (P = 0.0474, Cochran-Q = 14.22, I2 = 50.8%) (Fig. 2C). Its area under the summary receiver operating characteristic curves (SROC) value (AUC) was counting as 0.8582 ± 0.0531 (Fig. 2D). The potential threshold effect is failed to be detected. Meta-regression has been used to measure the factors in inducing the heterogeneities, such as population nationalities, diagnostic criteria of CAL, study design, and total sample size. The meta-regression revealed the population nationalities made a significant contribution to the homogeneity, P = 0.037, t = −2.66, 95%CI (0.15, 0.92) (Fig. 3A). Besides, the diagnostic criteria of CAL was not a dramatic impact factor, P = 0.080, t = −2.11, 95%CI (0.13, 1.16) (Fig. 3B). Meanwhile, the meta-regression also did not detect the study design has a dramatic impact on the homogeneity of the enrolled studies too, P = 0.056, t = −2.37, 95%CI (0.07, 1.05) (Fig. 3C). Also, the total sample size is not a dramatic impact factor, P = 0.581, t = −0.58, 95%CI (0.11, 3.93) (Fig. 3D). Therefore, the differences among the study countries were the source of existing heterogeneity. Additionally, it’s interesting that we found there were five studies11,15,16,17,18 all had thresholds at approximately 900 ng/L. Therefore, we evaluated those studies and found the summary sensitivity was 0.82 (95%CI, 0.73–0.89), and the pooled estimation showed no significant heterogeneity (P = 0.6640, x2 = 2.39, I2 = 0.0%) (Fig. 4A). Meanwhile, the summary specificity was 0.72 (95%CI, 0.68–0.76), and the pooled estimation showed significant heterogeneity (P = 0.0047, x2 = 15, I2 = 73.3%) (Fig. 4B). The pooled DOR was 13.18 (95% CI, 5.40–32.21) with significant heterogeneity (P = 0.0678, Cochran-Q = 8.75, I2 = 54.3%) (Fig. 4C). The calculated AUC value was 0.8868 ± 0.0486 (Fig. 4D).
Sensitivity analysis and subgroup analysis
We systematically and qualitatively analyzed the sensitivity of the included studies to determine the influence of individual trials on the results of overall NT-proBNP and NT-proBNP (thresholds ≈ 900 ng/L), using STATA 15.1 for meta-analysis random-effects estimates. Finally, none of the studies was detected to incur undue weight in the analysis (Fig. 5). Besides, Meta-Disc 1.4 was utilized to detect whether there is any threshold effect in studies, and the result suggested that the Spearman correlation coefficient was −0.286 and P = 0.955, which was indicating no threshold effect related to heterogeneity existed. After then, we conducted two subgroup analyses by the study design and the total sample size. The pooled DOR of the prospective group was 32.52 (95% CI, 14.12–74.89) with low heterogeneity (P = 0.2094, Cochran-Q = 4.53, I2 = 33.8%), and the calculated AUC value was 0.9065 ± 0.0577. Besides, the pooled DOR of the retrospective group was 8.41 (95% CI, 5.19–13.62) with no significant heterogeneity (P = 0.4407, Cochran-Q = 2.70, I2 = 0.0%), and the calculated AUC value was 0.7357 ± 0.0692. Meanwhile, the pooled DOR of the total sample size (n ≤ 100) group was 18.37 (95% CI, 8.76–38.54) with no significant heterogeneity (P = 0.3729, Cochran-Q = 3.12, I2 = 4%), and the calculated AUC value was 0.8064 ± 0.1173. Additionally, the pooled DOR of the total sample size (n > 100) group was 11.73 (95% CI, 4.36–31.54) with moderate heterogeneity (P = 0.0184, Cochran-Q = 10.02, I2 = 70.1%), and the calculated AUC value was 0.8976 ± 0.0522. These results were shown in Table 3.
Publication bias
We used funnel plots and the Deeks’ test to assess publication bias in the included studies. Each dot plots in these graphs represented a study. The distance between each dot and the vertical line indicated bias in each study. Symmetric distribution indicated no publication bias existed. Funnel plots in Fig. 6A–F present a degree of symmetry, suggesting that there is no potential for publication bias among the included articles.
Funnel plots for the assessment of potential publication bias. The funnel graphs plot the Bsquare root of the effective sample size (1/ESS1/2) against the DOR. Each circle represents each study in the meta-analysis. Asymmetry of the circle distribution between regression lines indicates potential publication bias. (A) Total pooled result, (B) Thresholds ≈ 900 ng/L pooled result, (C) Prospective pooled result, (D) Retrospective pooled result, (E) Total sample size (n ≤ 100) pooled result, (F) Total sample size (n > 100) pooled result.
Discussion
The diagnostic accuracy of NT-proBNP in detecting KD with CAL in the acute phase was systematically evaluated in our study. Finally, we found that NT-proBNP can be a valuable biomarker for predicting CAL of KD. The previous meta-analysis10 showed that NT-proBNP level was mildly higher than in KD patients of the acute phase compared with the febrile control patients. In our study, we found that the mean NT-proBNP level in KD patients with CAL was much higher than in KD patients without CAL (approximately 2500 ng/L vs. 800 ng/L) (Table 1). However, the precise mechanism of elevated NT-proBNP in KD patients is still unclear21. Nevertheless, factors known to affect the NT-proBNP levels, such as cardiac function22 or inflammatory cytokines23, were not evaluated in all the included studies, which could cause biased results. Therefore, further well-designed studies are needed to evaluate the value of NT-proBNP in predicting CAL of KD.
The present study suggested that the overall diagnostic sensitivity and specificity of NT-proBNP for diagnosis KD with CAL in the acute phase were 0.84 and 0.71, AUC of SROC was 0.8582. Then, we evaluated five studies, which all had thresholds at approximately 900 ng/L. Finally, we found the overall diagnostic sensitivity and specificity of NT-proBNP (threshold ≈ 900 ng/L) for diagnosis KD with CAL were 0.82 and 0.72, AUC of SROC was 0.8868, which was slightly higher than the overall diagnostic accuracy of NT-proBNP. In general, those found suggested that NT-proBNP can be used as a biomarker for detecting CAL of KD. Additionally, the diagnostic accuracy in the threshold of about 900 ng/L shows a little higher than the overall diagnostic accuracy, which still only indicates that NT-proBNP has diagnostic value, but cannot indicate that 900 ng/L is the recommended threshold. The specific threshold range needs to be determined by more well-designed clinical studies with larger sample size. Besides, the meta-regression showed that the differences among the study countries were the source of heterogeneity.
Furthermore, we conducted two subgroups analysis by the study design and the total sample size, and these results suggested that both the type of research and the total sample size might be the sources of heterogeneity. Besides, the calculated AUC of value was 0.9065 ± 0.0577 for the prospective group and 0.8976 ± 0.0522 for the total sample size (n > 100) group, which showed prospective studies with large sample size (n > 100) were superior designed studies to evaluate the value of NT-proBNP in predicting CAL of KD.
Although CAL in KD may be transient or permanent24, all the included studies did not provide follow-up time for CAL, which may bias the diagnostic value of NT-proBNP. Further rigorous studies, with unified inclusion and exclusion criteria and a consecutive enrolment design, needed to evaluate the diagnostic value of NT-proBNP in KD with CAL diagnosis.
Our meta-analysis has several limitations. First, the number of included studies was small (n = 8), and all of them were conducted in Asian populations, which means these results may not generalize to other areas. Second, no included articles combine NT-proBNP with other laboratory tests, such as ESR, CRP, to identify the diagnostic accuracy of KD with CAL, which could work as a better method for detection. Third, all of the follow-up time of the included articles were unclear, which may lead to the deviation of CAL diagnosis, which could further affect the accuracy of NT-proBNP in predicting CAL of KD.
In conclusion, despite these limitations, this is the first meta-analysis that showed NT-proBNP could be used as a biomarker for detecting CAL of KD. Besides, further well-designed studies with a large sample size are needed to strictly evaluate the value of NT-proBNP in predicting CAL of KD.
Materials and Methods
Study protocol and ethics statement
We performed this analysis following a predetermined protocol according to the recommendations of Deeks25. The data collection and reporting were by the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) Statement26 (S Table 1). Due to it is a systematic literature study, ethical approval was not necessary. The protocol for this analysis was registered with PROSPERO (CRD42019130083).
Search strategy
PubMed, the Cochrane Central Register of Controlled Trials (CENTRAL), EMBASE, and China National Knowledge Infrastructure (CNKI) through Mar 13th, 2019 were searched to detect relevant studies. Search terms of PubMed database were (Mucocutaneous Lymph Node Syndrome[MeSH Terms] OR Kawasaki disease OR Kawasaki syndrome) AND (pro-brain natriuretic peptide[MeSH Terms] OR NT-proBNP OR N-terminal pro-BNP OR NTproBNP OR NT-BNP). Search terms for the CENTRAL, EMBASE, and CNKI with the corresponding search results are listed in S Appendix 1. English and Chinese were the language limits for retrieval.
Study selection
Articles were screened preliminarily by title and abstract after citations selected by the systematic search. And then, potentially relevant articles were retrieved by full text, while assessed for compliance to inclusion and exclusion criteria.
Inclusion criteria: (1) all cases must meet the KD diagnostic criteria; (2) randomized or non- randomized controlled, cohort studies, clinical trials evaluating NT-proBNP in blood samples; (3) contained the data that can calculate true positive (TP), false negative (FN), false positive (FP), and true negative (TN), such as specificity, sensitivity and sample size; (4) all studies had KD with non-CAL subjects as the control group; (5) The samples were taken from patients with acute KD before initial IVIG treatment.
Exclusion criteria: (1) reviews, editorials, abstracts, letters, expert opinions, conferences articles, or case reports without controls; (2) unable to construct 2 × 2 table; (3) duplicated publications.
Data collection and assessment of study quality
The eligibility of studies was assessed by two investigators (Xiaolan Zheng, Yi Zhang) independently by the title and abstract. At the same time, the divergences and the quality of reports were determined by a third reviewer (Yifei Li) according to inclusion or exclusion criteria. The quality assessment of all included reports was evaluated by the two investigators (Xiaolan Zheng, Lei Liu) independently following the QUADAS list27. As well-conducted research might score lower in the absence of relevant parts of the methodology and results, the assessments were reported in descriptive form only. Finally, the data from which can calculate TP, FP, FN, and TN were extracted by two investigators (Xiaolan Zheng, Peng Yue).
Evaluation indicators
We measured the following indicators of NT-proBNP: sensitivity, specificity, DOR, and SROC. Sensitivity represented the proportion of patients in KD patients with CAL, which were correctly identified by the positive results of NT-proBNP. Besides, specificity expressed the KD cases with non-CAL that were correctly identified by the negative results of NT-proBNP. Also, the DOR more reliably defined a summary of test performance, rather than merely pooling specificity and sensitivity in individual reports. DOR was an independent index with a range of 0 ~ infinity. The higher the DOR, the better the discrimination28. The SROC was plotted by combining sensitivity and specificity. Furthermore, AUC was calculated as a global measurement of test performance29, and the closer the AUC was to 1, the better the test performance would be.
Publication bias
Funnel plots and the Deeks’ test were used to assess the publication bias. It indicated a potential publication bias when the asymmetric distribution of data dot in the funnel plot with a quantified result of P < 0.0530.
Heterogeneity and meta-regression
The heterogeneity of pooling sensitivity and specificity were examined by the x2 test, while the heterogeneity of pooling DOR was examined by the Cochran Q test. The I2 test in every pooling analysis to quantitatively was also conducted to assess the proportion of total variation in the study. I2 value would range from 0 to 100%, with values of 25, 50, and 75%, respectively, as evidence of low, moderate, and high heterogeneity31. The threshold effect was suggested by a curvilinear shape in the SROCs. Furthermore, the meta-regression was carried out to detect the potential factors that would cause heterogeneity. All the possible factors were extracted from the baseline measurement and original testing procedures, which were included in meta-regression. The meta-regression can determine the correlation between the potential factors and the existing heterogeneity. The factor should have a dramatic impact on the homogeneity of the enrolled studies with a P-value < 0.05 when a significant difference was discovered.
Sensitivity analysis
We performed the sensitivity analysis to determine the influence of individual studies on the results. Meta-Disc 1.432 was used for detecting threshold effects in reports.
Statistical analysis
Meta-Disc 1.4 was utilized to perform data analysis. Besides, STATA 15.1 (Stata Corporation, College Station, Texas, USA) was utilized to assess the publication bias and perform meta-regression analysis. Homogenous results utilized the random-effects model for statistical analysis, while the heterogeneous (I2 < 50%) results utilized a fixed-effects model, and the data were presented using a forest map.
Data availability
The authors confirm that all the data based findings are fully available without restriction. All relevant data are included in the paper and references.
References
Newburger, J. W. et al. Diagnosis, treatment, and long-term management of Kawasaki disease: a statement for health professionals from the Committee on Rheumatic Fever, Endocarditis and Kawasaki Disease, Council on Cardiovascular Disease in the Young, American Heart Association. Circulation 110, 2747–2771, https://doi.org/10.1161/01.cir.0000145143.19711.78 (2004).
Singh, S., Vignesh, P. & Burgner, D. The epidemiology of Kawasaki disease: a global update. Archives of disease in childhood 100, 1084–1088, https://doi.org/10.1136/archdischild-2014-307536 (2015).
Singh, S. et al. Mortality in children with Kawasaki disease: 20 years of experience from a tertiary care centre in North India. Clinical and experimental rheumatology 34, S129–133 (2016).
Dietz, S. M. et al. Dissecting Kawasaki disease: a state-of-the-art review. European journal of pediatrics 176, 995–1009, https://doi.org/10.1007/s00431-017-2937-5 (2017).
McCrindle, B. W. et al. Diagnosis, Treatment, and Long-Term Management of Kawasaki Disease: A Scientific Statement for Health Professionals From the American Heart Association. Circulation 135, e927–e999, https://doi.org/10.1161/cir.0000000000000484 (2017).
Singh, S., Jindal, A. K. & Pilania, R. K. Diagnosis of Kawasaki disease. International journal of rheumatic diseases 21, 36–44, https://doi.org/10.1111/1756-185x.13224 (2018).
Yancy, C. W. et al. ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. Journal of the American College of Cardiology 62, e147–239, https://doi.org/10.1016/j.jacc.2013.05.019 (2013).
Ponikowski, P. et al. ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC)Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. European heart journal 37, 2129–2200, https://doi.org/10.1093/eurheartj/ehw128 (2016).
Lin, K. H. et al. Usefulness of natriuretic peptide for the diagnosis of Kawasaki disease: a systematic review and meta-analysis. BMJ Open 5, e006703, https://doi.org/10.1136/bmjopen-2014-006703 (2015).
Yu, J., Li, H. H. & Dong, L. Meta-analysis: Diagnostic Value of N-Terminal Pro-brain Natriuretic Peptide for Kawasaki Disease. Clin Lab 62, 1903–1910, https://doi.org/10.7754/Clin.Lab.2016.160126 (2016).
Kaneko, K. et al. Prediction of the risk of coronary arterial lesions in Kawasaki disease by brain natriuretic peptide. Pediatr Cardiol 32, 1106–1109, https://doi.org/10.1007/s00246-011-9986-8 (2011).
Chen, T. T. et al. Relationship between heart rate variability and coronary artery lesion in children with Kawasaki disease. Zhongguo dang dai er ke za zhi = Chinese journal of contemporary pediatrics 17, 607–612 (2015).
Kim, M. K., Song, M. S. & Kim, G. B. Factors Predicting Resistance to Intravenous Immunoglobulin Treatment and Coronary Artery Lesion in Patients with Kawasaki Disease: Analysis of the Korean Nationwide Multicenter Survey from 2012 to 2014. Korean Circ J 48, 71–79, https://doi.org/10.4070/kcj.2017.0136 (2018).
Yoshimura, K. et al. N-terminal pro-brain natriuretic peptide and risk of coronary artery lesions and resistance to intravenous immunoglobulin in Kawasaki disease. J Pediatr 162, 1205–1209, https://doi.org/10.1016/j.jpeds.2012.11.026 (2013).
Qiu Huixian, R. M., Qi, C., Yuanhai, Z., Rongzhou, W. & Rulian, X. Prediction of the Risk of Coronary Arterial Lesions in Kawasaki Disease by N – terminal Brain Natriuretic Peptide. J Med Res 41, 166–168, https://doi.org/10.3969/j.issn.1673-548X.2012.04.052 (2012).
Chen Yanli, D. X. The value of N-terminal pro-brain natriuretic peptide in predicting coronary arterial lesions in kawasaki disease. Guangdong Medical Journal 35, 3075–3077, https://doi.org/10.13820/j.cnki.gdyx.2014.19.041 (2014).
Huiling, L., Yaping, L. & Xiufen, H. Prediction of the risk of coronary arterial lesions in Kawasaki disease by N-terminal pro-brain natriuretic peptide. Zhonghua er ke za zhi = Chinese journal of pediatrics 53, 300–303 (2015).
Lee, H. Y. & Song, M. S. Predictive factors of resistance to intravenous immunoglobulin and coronary artery lesions in Kawasaki disease. Korean J Pediatr 59, 477–482, https://doi.org/10.3345/kjp.2016.59.12.477 (2016).
Fan Jianghua, D. W., Haiyan, L., Longgui, Y. & Yan, T. Prediction of Plasma NT-proBNP and PCT for Early Kawasaki Disease Coronary Artery Lesions. 24(9), 1–5, https://doi.org/10.13407/j.cnki.jpp.1672-108X.2018.09.001 (2018).
Jung, J. Y. et al. N-terminal pro-brain natriuretic peptide and prediction of coronary artery dilatation in hyperacute phase of Kawasaki disease. Am J Emerg Med 37, 468–471, https://doi.org/10.1016/j.ajem.2018.06.021 (2019).
Shiraishi, M. et al. N-terminal pro-brain natriuretic Peptide as a useful diagnostic marker of acute Kawasaki disease in children. Circulation journal: official journal of the Japanese Circulation Society 77, 2097–2101 (2013).
Nasser, N., Perles, Z., Rein, A. & Nir, A. NT-proBNP as a marker for persistent cardiac disease in children with history of dilated cardiomyopathy and myocarditis. Pediatr Cardiol 27, 87–90, https://doi.org/10.1007/s00246-005-1027-z (2006).
Sato, N., Sagawa, K., Sasaguri, Y., Inoue, O. & Kato, H. Immunopathology and cytokine detection in the skin lesions of patients with Kawasaki disease. J Pediatr 122, 198–203, https://doi.org/10.1016/s0022-3476(06)80113-7 (1993).
Kuo, H. C. Preventing coronary artery lesions in Kawasaki disease. Biomedical journal 40, 141–146, https://doi.org/10.1016/j.bj.2017.04.002 (2017).
Deeks, J. J. Systematic reviews in health care: Systematic reviews of evaluations of diagnostic and screening tests. BMJ (Clinical research ed.) 323, 157–162 (2001).
Moher, D., Liberati, A., Tetzlaff, J. & Altman, D. G. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS medicine 6, e1000097, https://doi.org/10.1371/journal.pmed.1000097 (2009).
Whiting, P., Rutjes, A. W., Reitsma, J. B., Bossuyt, P. M. & Kleijnen, J. The development of QUADAS: a tool for the quality assessment of studies of diagnostic accuracy included in systematic reviews. BMC medical research methodology 3, 25, https://doi.org/10.1186/1471-2288-3-25 (2003).
Glas, A. S., Lijmer, J. G., Prins, M. H., Bonsel, G. J. & Bossuyt, P. M. The diagnostic odds ratio: a single indicator of test performance. Journal of clinical epidemiology 56, 1129–1135 (2003).
Moses, L. E., Shapiro, D. & Littenberg, B. Combining independent studies of a diagnostic test into a summary ROC curve: data-analytic approaches and some additional considerations. Statistics in medicine 12, 1293–1316 (1993).
Deeks, J. J., Macaskill, P. & Irwig, L. The performance of tests of publication bias and other sample size effects in systematic reviews of diagnostic test accuracy was assessed. Journal of clinical epidemiology 58, 882–893, https://doi.org/10.1016/j.jclinepi.2005.01.016 (2005).
Higgins, J. P. & Thompson, S. G. Quantifying heterogeneity in a meta-analysis. Statistics in medicine 21, 1539–1558, https://doi.org/10.1002/sim.1186 (2002).
Zamora, J., Abraira, V., Muriel, A., Khan, K. & Coomarasamy, A. Meta-DiSc: a software for meta-analysis of test accuracy data. BMC medical research methodology 6, 31, https://doi.org/10.1186/1471-2288-6-31 (2006).
Acknowledgements
This work was supported by grants from the National Natural Science Foundation of China (No. 81700360, 81741025, 81570369 and 81571515) and the Technologic Program from Sichuan Province Government Foundation, China (No. 2018JY0603).
Author information
Authors and Affiliations
Contributions
Kaiyu Zhou, Yimin Hua, Gang Wu and Yifei Li participated in research design. And Xiaolan Zheng, Yi zhang, Lei Liu, Peng Yue, and Yifei Li collectively contributed to the data collection and analysis. Xiaolan Zheng, Yifei Li, and Chuan Wang helped to interpret the results from the analysis. Finally, Xiaolan Zheng wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zheng, X., Zhang, Y., Liu, L. et al. N-terminal pro-brain natriuretic peptide as a biomarker for predicting coronary artery lesion of Kawasaki disease. Sci Rep 10, 5130 (2020). https://doi.org/10.1038/s41598-020-62043-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-62043-6
This article is cited by
-
Development of a nomogram prediction model for early identification of persistent coronary artery aneurysms in kawasaki disease
BMC Pediatrics (2023)
-
N-Terminal Pro-Brain Natriuretic Peptide Levels in Kawasaki Disease, Sepsis and Other Febrile Illnesses
Indian Pediatrics (2023)
-
Cardiac Biomarkers Aid in Differentiation of Kawasaki Disease from Multisystem Inflammatory Syndrome in Children Associated with COVID-19
Pediatric Cardiology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.