Abstract
The extent to which commercially important Nephrops norvegicus lobsters feed on particulates in the wild is unknown, even though this could be an important way for burrow-dwelling females to avoid starvation during the long breeding season. This was investigated using δ13C and δ15N isotopic signatures in tissues with long and short turnover rates to provide diet discrimination and compare this between males and females. Secondary objectives examined size-related differences and calculated the trophic position based on the new results. Almost half the diet (47%) was made up of suspended particulate organic matter (POMsusp) alone. Fish was another important item in the diet, with plankton and invertebrate sources coming much lower down in dietary importance. Significantly more suspension feeding was observed in small or medium sized individuals than large ones in both sexes. However, there were no sex-related patterns, despite females being restricted to burrows for part of the analysis period. Female diet was almost identical to males and POMsusp comprised a large component of the diet in both sexes. The trophic position was estimated at 2.94 ± 0.16 (mean ± SD), which was at the lower end of the range reported in previous studies (2.60 to 4.32).
Similar content being viewed by others
Introduction
Dublin Bay prawn, Nephrops norvegicus, is a decapod crustacean and an important economic resource in Europe: global production of this fishery was 59,033 tons in 2016 of which the United Kingdom and Republic of Ireland were the main producers, capturing up to 32,708 and 10,379 tons per annum respectively during 2012–20161. Nephrops populations are distributed on semi-isolated mud patches which are assessed by ICES as separate Functional Units (FU), however this resource is not ‘managed’ via fishing quotas, and some FUs periodically display signs of over-exploitation2,3. The Marine Strategy Framework Directive and reformed Common Fisheries Policy (CFP) require ecosystem-based fisheries management which observes interactions among all components of the ecosystem, including trophic interactions4,5,6. Although not currently managed under the CFP, key gaps and ambiguities exist in knowledge of Nephrops’ diet and feeding ecology which should be addressed, given their economic importance and occasional over-exploitation.
Nephrops individuals are known to be opportunistic predators and scavengers, which seem to have a diet driven by prey abundance rather than prey preference7,8,9,10. Diet from stomach contents analyses seems to be similar across a wide geographical range in the north-eastern Atlantic and Mediterranean, composed mainly of crustaceans, polychaetes, molluscs and echinoderms8,9. A considerable contribution to the diet is also made by fish in southern Atlantic and Mediterranean samples11,12. However, some mystery surrounds the extent of feeding on particulates in Nephrops. In the absence of alternative food sources, such as in the aquarium, Nephrops was demonstrated to feed on planktonic items larger than 300–500 µm, which were later recovered from the stomach and intestine of the animal13. But, although some studies use particulate organic matter (POM) as the baseline for Nephrops trophic position estimation14,15, no studies have directly measured the importance of particulate food items in the diet of Nephrops in its natural habitat.
The lifestyle of male and female Nephrops differs significantly as females are restricted to burrows while brooding embryos over a long breeding period, from six to ten months, depending on latitude16. Therefore, it is logical to ask whether there are sex-related differences in diet arising from these lifestyle differences. Restriction to burrows for most of the year is evidenced by a lower percentage of females in fisheries catches during the breeding season, which is elongated in Irish and Scottish grounds over Autumn, Winter and early Spring17. For this reason, a seasonal decrease in nutritional status, i.e. ‘starvation’, has been predicted for females in Winter, compared with late Spring and Summer when they are observed to be present in the catch and actively feeding, after releasing their broods, moulting and mating5,10,16,18,19. A biochemical index for estimation of nutritional status from the Clyde Sea in Scotland suggested that, although females had reduced nutritional status in the Winter, this was not sufficiently low to indicate starvation10. At the same time, growth in females is also much lower than in males20 and the respective diets of males and females are still not fully understood.
Suspension feeding has been identified as a possible strategy for females to survive starvation while they are restricted to burrows during the long breeding period13. Since suspended food in the form of plankton biomass is seasonally lower during the Winter female burrow-dwelling period21,22, we propose that females instead feed on suspended POM (i.e. POMsusp). POMsusp represents a complex microscopic mixture of living and non-living organisms including phytoplankton, fecal pellets, detritus, bacteria and heterotrophs, but with distinct isotopic signatures from phyto- and zooplankton, particularly in coastal areas23,24, that can be an important food source to many organisms. The main aim of this study was therefore to investigate the relative importance of POMsusp in Nephrops’ diet (all sexes) as well as to examine sex-related differences in diet during the Spring-Summer period. Secondary objectives were to compare the diet composition between adult size classes as we might expect some dietary differences between the smaller and larger sizes due to differing abilities to compete over prey or to handle different prey items.
Stable isotopes analysis (SIA) was chosen to complement information from previous stomach contents analyses8,9,11,12. SIA can more fully represent POMsusp and soft-bodied prey items in the diet as well as providing a time-integrated view of feeding compared with a ‘snapshot’ provided by stomach contents analysis25. SIA analysis on tissues with different turnover rates can also demonstrate diet compositions over distinct periods26. For example, in the present study, 13C and 15N isotopic signatures in long and short-term storage tissues were used to compare signals in Nephrops’ diet between males and females, both in the period when females were in burrows during the lead-up to spawning and the period after females had spawned and were actively feeding, maturing new gonads and mating. A final aim was to determine Nephrops’ trophic position based on new SIA results from the present study. The SIA data were analysed within a Bayesian framework, an approach which is increasingly used to address ecological problems27,28,29,30,31,32,33,34.
This study examined the importance of POMsusp as a food source in the diet of wild Nephrops, comparing this with other food sources. The specific hypotheses tested were: (i) suspension feeding is higher among the smaller (more vulnerable) adults, either because they remain in burrows to avoid enemies, or because they are too small to handle larger more mobile prey; and (ii) feeding patterns are sex-related, specifically there is higher suspension feeding in females than males during the period when females are brooding embryos and restricted to burrows compared with the period post-spawning when they are actively moulting, mating and feeding outside of the burrow.
Results
Importance of suspended particulate organic matter to Nephrops’ diet
Consumer isotopic values fell within the range of food source isotopic values for all four time periods (Fig. 1), indicating that all major Nephrops’ dietary sources were included in the analysis and no important dietary sources were missing. This arrangement between consumer and source values is a precondition for the SIMMR Bayesian mixing model to work adequately35. The SIMMR model output showed that POMsusp and fish were the main food sources for all consumer groups and time periods in the analysis. The estimated means of POMsusp contribution to the diet ranged between 12.0–47.4% but was generally high, >20% (Supplementary Table S1). Meanwhile, apart from fish, contributions from the other sources (phyto- and zooplankton, filter feeders, polychaetes and crustaceans) were much lower, with means ranging from 2.3–15.6% (Fig. 2). The estimated means for fish contribution to the diet ranged from 18.2–60.9%. Therefore, the main question about the importance of POMsusp in Nephrops’ diet was accepted to be the case in this study.
Contributions of putative sources to Nephrops’ diet. The figure shows posterior probability distributions of the contributions of all putative sources to Nephrops’ diet in different periods during Spring and Summer, 2014: ‘Spring long’ (8th March–29th May), ‘Spring short’ (10th–29th May), ‘Summer long’ (4th May–25th July) and ‘Summer short’ (6th–25th July).
After combining sources, the estimated means of ‘active feeding’ (i.e. filter feeders, polychaete, crustaceans and fish) ranged from 42.5–76.2%, while that of ‘suspension feeding’ (i.e. POMsusp, phytoplankton and zooplankton) ranged from 26.5–57.5%. More details on average contributions from all food sources and probability distributions of ‘active feeding’ and ‘suspension feeding’ to Nephrops’ diet can be seen respectively in Supplementary Table S1 and Fig. S1.
Size-related differences in Nephrops’ diet
There were some significant Nephrops size-related differences in suspension feeding (i.e. POMsusp, phytoplankton and zooplankton). The results showed that suspension feeding took place significantly more in small or medium sized individuals than large ones. Surprisingly, this occurred more frequently in male comparisons than in females. Suspension feeding was significantly higher in the small and medium sized males compared to larger males in the early time period, i.e. ‘Spring long’: 8th March–29th May (Fig. 3). In the late time period, i.e. ‘Summer long’ (4th May–25th July), it was significantly higher only in the small males compared to large males (Fig. 3). For females, the small size class was significantly more likely to suspension feed than larger ones in the ‘Spring short’ period (10th–29th May) (Fig. 3). No significant differences could be detected between the size groups in either sex in the ‘Summer short’ time period (6th–25th July) (Fig. 3).
Size-related differences in Nephrops’ diet from suspension feeding. The figure shows posterior probability distributions comparing contribution to the diet of suspension feeding (i.e. phytoplankton + zooplankton + POMsusp) in Nephrops of different sizes during Spring and Summer 2014:‘Spring long’ (8th March–29th May), ‘Spring short’ (10th–29th May), ‘Summer long’ (4th May–25th July) and ‘Summer short’ (6th–25th July). An asterix indicates a significant size-related difference in suspension feeding in the diet i.e. pBIC > 0.95 (provided by the SIMMR package according the Bayesian paradigm).
Sex-related differences in Nephrops’ diet
The comparison of suspension feeding between males and females produced no significant results across time periods including: 8th March–29th May (‘Spring long’), which includes part of the long breeding season of females, and 4th May–25th July (‘Summer long’) when both sexes are non-burrow dwelling and capable of feeding outside of burrows (Fig. 4). The contribution of active feeding and suspension feeding to the diet of males and females was also equivalent, with imperceptible differences observed between the sexes (Supplementary Fig. S1). Thus, the hypothesis of differences between male and female’s diet, related to the reproductive cycle of females is not upheld at Clew Bay.
Sex-related differences in Nephrops’ diet from suspension feeding. The figure shows posterior probability distributions comparing contribution to the diet of suspension feeding (i.e. phytoplankton + zooplankton + POMsusp) in Nephrops of different sexes, including periods when females were burrow-dwelling i.e. 8th March–29th May 2014 (‘Spring long’) and non-burrow dwelling periods 4th May–25th July 2014 (‘Summer long’). An asterix denotes a significant sex-related difference in suspension feeding in the diet i.e. pBIC > 0.95 (provided by the SIMMR package according the Bayesian paradigm), however no such differences were observed.
Trophic position of Nephrops norvegicus
The overall trophic position of N. norvegicus in Clew Bay, based on isotopic signatures in muscle and hepatopancreas (considered together) was estimated to be 2.94 ± 0.16 (mean ± SD), which represents an average over all time periods. The trophic position across different periods varied from 2.79 ± 0.12 (‘Spring short’) to 3.06 ± 0.10 (‘Spring long’/‘Summer long’ – with these two periods being identical; Table 1).
Discussion
The primary aim of this research was to investigate feeding on suspended particulate organic matter (POMsusp) in wild Nephrops as this may be a mechanism of avoiding seasonal starvation. Before looking at its contribution to the diet, we could establish that we had captured all the important dietary sources by the arrangement of isotopic values in the δ13C/δ15N bi-plots (Fig. 1). The position of consumer tissues within the polygon of sampled food sources for each of the four periods indicated that no important food source(s) were missing in the analysis.
At times, almost half (47%) of the diet of Nephrops was made up of POMsusp. These lobsters did show variety in their diet, however, and another important item in the diet was fish, while plankton and invertebrate sources came far below these items in dietary importance. Reliance on POMsusp and fish, rather than on invertebrates, appears initially surprising, considering predominance of invertebrates in stomach content analysis8,9. However, many crustaceans are predatory on fish, which is apparently independent of their size36. Capture of flatfish, for example, may present little difficulty to Nephrops, whose diet may also be subsidised from discards arising from inshore fishing activity in Clew Bay. The high level of feeding on particulate matter was more surprising, however another burrowing decapod crustacean, Neotrypaea californiensis, has recently been shown to be primarily reliant on POMsusp as a food source33.
Due to their burrowing lifestyle and long breeding season in females, much effort has gone into investigating seasonal starvation in Nephrops10,37,38,39. The ability to feed on particulate food sources would help to counteract starvation brought about by these lifestyle restrictions. When Nephrops individuals were maintained in unfiltered seawater in an aquarium, they showed an intermediate nutritional status between control animals with no access to food and those from the wild, which was suggestive of suspension feeding, at least in-extremis with no other food available13. The present results add to this by demonstrating the importance of suspension feeding to the diet of wild individuals, showing that they utilised this food source at a significant level. Previous work has theorised that 65–68% of daily energy intake was available for growth from suspension feeding at sufficient particulate densities8. Although our study does not address energy intake directly, our estimates of suspension feeding in the diet often reached 50% (particularly in short-term tissues). This likely represents a considerable amount of suspension feeding-derived energy that is available for growth (Supplementary Table S1).
In fact, it has long been acknowledged that POMsusp is an important seasonal source of food for benthic organisms in Winter40,41. Not all suspended food particles were equally important, however, for example phyto- and zooplankton were far less important than POMsusp in Nephrops (Fig. 2; Supplementary Table S1). Sediment organic matter (SOM) could be another important food source for benthic organisms like Nephrops. A practical difficulty is distinguishing SOM from POMsusp because the latter eventually falls to the seafloor and therefore forms one component of the SOM. Although we cannot discount the possibility that, as well or instead of feeding on POMsusp, Nephrops also picks POM up off the sediment while deposit feeding on a mix of SOM/POM, we do not have evidence to support this idea. POMsusp and SOM present distinct isotopic signatures42. For example, compared with POMsusp, SOM was shown to be enriched in δ13C and depleted in δ15N42. Had SOM with this42 profile been substituted for POMsusp in the present study, Nephrops samples would have fallen outside the isotopic polygon. Also, the δ13C/δ15N bi-plot in our study showed no missing dietary items, which might have been expected had SOM been important in the diet. Other studies have also shown distinct POM and SOM signatures28,43. Future studies combining fatty acid analysis and SIA may further disentangle the various sources of organic particulates and their relative importance, including sources found inside lobster burrows (e.g.33).
The hypothesis suggesting a size-related difference in Nephrops diet was accepted for several comparisons. Suspension feeding was higher in smaller compared to larger size classes for males in particular, e.g. during ‘Spring long’: 8th March–29th May (small and medium males compared to large males) and ‘Summer long’: 4th May–25th July (small males compared to large males). Males may suffer more competition for active food items than females44 which may force smaller individuals to rely on particulate food sources. The same size-related difference, i.e. a higher proportion of suspended food in the diet of small females compared with larger ones, was borne out in only one time period: the ‘Spring short’ feeding on 10th–29th May. This was not seen for the equivalent tissue later in the season ‘Summer short’ on 6th–25th July. Interestingly, the size-related differences we observed did not appear to be related to a limitation on predation capacity in smaller lobsters. Indeed, at times, the contribution to the diet by fish was even higher in smaller individuals than in the larger ones, e.g. 48.1% versus 44.0% and 44.7% versus 28.1% for small versus large males in two of the four periods analysed (Fig. 2, Supplementary Table S1). However, without further research it is difficult to interpret the reason for this, for example, it is possible that smaller individuals may feed on fisheries discard or on larger individual’s leftover prey.
Although the isotopic signal from long and short-term storage tissues varied substantially, there was no difference in ‘Spring’ and ‘Summer’ diets when similar storage tissues were compared (Supplementary Fig. S1). The difference between long and short-term storage tissues arises because these represent different time intervals, 19 and 81 days respectively. Active feeding was higher in long-term storage tissues, whereas suspension feeding was increased in short-term tissues (Supplementary Fig. S1). Without further experiments, the reasons for this are unclear, however.
The hypothesis related to sex-specific diets was rejected. Males and females were remarkably similar in diet, even in the Spring period (8th March–29th May), even though the tissues sampled from this time represented a period when females were mostly brooding. Larval release at Clew Bay begins around the second week of April45, until which point, the females stay inside burrows to brood their developing embryos. Our results demonstrate that this period of burrow dwelling does not prevent females from accessing the same food items as males. It has been suggested that feeding by females during the breeding season may simply take place closer to the burrow mouth10,46 or females may bury food within or adjacent to burrows15. Although the sexes have similar diets, as shown in the present study, the overall opportunity for feeding may be reduced in females. However, starvation and sex-specific reduction in nutritional status has been previously examined and found to be absent10, with corroborating evidence from biochemical markers that suggest good nutritional condition throughout the year in females38. Although Clew Bay is a particularly shallow site, the same major food groups (i.e. plankton and particulates, macroinvertebrates and fish) are available in deeper habitats7,8,9,10,11,12. Nephrops diurnal emergence does vary with depth47 but we can think of no plausible reason for this to interact with the availability of POMsusp or other food groups. Therefore, we believe the results are transferrable to other Nephrops populations, although other locations may have slightly different groups of macroinvertebrates (echinoderms, in particular, were not abundant in the sediments at Clew Bay).
The importance of POMsusp as a dietary source in Nephrops has implications for its trophic position. Based on isotopic signatures in the present study, trophic level was calculated to be 2.94 ± 0.16 (mean ± SD) i.e. at the lower end of previous estimates for Nephrops (2.60–4.3214,15,43) that were also derived using SIA. SIA can reveal lower trophic status in consumers compared with stomach contents analysis, because the latter can underestimate soft-bodied prey25. In the case of Nephrops, it would be almost impossible to detect from stomach contents, the fact that up to half of the diet derived from POMsusp. Such disproportionate measurement of prey items acts to artificially inflate trophic position based on stomach contents alone.
As the present study shows, smaller and medium-sized males fed on significantly higher suspended food than larger ones, therefore the potential to suspension feed may be an important mechanism for avoiding aggressive encounters over food between males. This is potentially important because the growth (and hence biomass) of male individuals is strongly density-dependent at Clew Bay44 (- densities at Clew Bay vary between 0 and 15 individuals per pot fished45). Body size also varies across fishing grounds - smaller Nephrops are found at FUs with higher stock densities, most likely as a result of reduced growth potential due to intraspecific competition5,44. We suggest that feeding on POM is an important lifestyle adaptation in both males (counteracting competitive interactions) and females (counteracting burrow-dwelling) but that Nephrops diet is remarkably similar in the sexes. The knowledge that fish is also an important component of the diet in all groups examined at Clew Bay means that, in theory, reduced subsidy from fisheries discards to scavengers like Nephrops under the EU Landings Obligation could affect feeding opportunities for this species in the future.
Methods
Study area
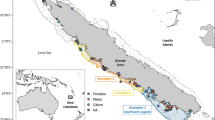
The research was conducted on an inshore population at Clew Bay in the west of Ireland. Samples were collected in Clew Bay (53.87°N and 9.64°W) on substrates which are dominated by mud and sand and in water depths ranging from 5–40 m (average 20 m). The tidal range is around 5 m and the residence time of water in the inner bay is likely to be short, ~2 days48,49. Pot fishing for Nephrops is seasonal in this area and runs from April – August, therefore the field sampling programme was limited to this period.
Sample collection
All samples including Nephrops and putative prey (benthic macrofauna, zooplankton, phytoplankton and suspended particulate organic matter), were collected from the study site on two occasions which were eight weeks apart, i.e. on the 29-May-2014 (nominated ‘Spring long’ or ‘Spring short’, depending on the tissue – see below and Table 2) and the 25-July-2014 (‘Summer long’ or ‘Summer short’ –Table 2). Nephrops were collected by baited creels on both dates. As Nephrops interacts with benthic communities both on and beneath the sediment, these potential prey items were obtained in two ways: (1) via five 15 min bottom trawl, using a standard 2 m beam trawl with a chain mat, a stretched mesh (bar length: 20 mm) and a codend liner with a knotless mesh (bar length: 4 mm) and (2) using a day grab Van Veen 12.110–250 cm²/3.14 L. A total of 17 different putative prey taxa were sampled from different groups: tunicates, polychaetes, bivalves, gastropods, crustaceans and fish on both dates (see Supplementary Table S2). Zooplankton and phytoplankton were sampled on both days using a 57 cm ring diameter and 250 µm mesh WP-2 plankton net towed behind the boat (15 min tows). The choice of plankton net assumed Nephrops can feed on plankton items larger than 300–500 µm13. Assuming that Nephrops consumers fell within the range of diet sources in an isotope bi-plot, we could be satisfied that no important food sources were missing from the analysis (indeed, this was the case - see Results section 2.1). To sample POMsusp, water samples were taken via a Niskin bottle triggered at around 1 m above the seafloor. All samples were held on ice during transit and then transferred to a −20 °C freezer until processing for stable isotopes analysis.
Stable isotope sample preparation
Potential Nephrops food ‘source’ tissues were processed as follows: phytoplankton and zooplankton samples were cleaned under the microscope. POMsusp was concentrated by filtering seawater (around 5 L was filtered for each sample) on precombusted glass filters and stored frozen (−20 °C). POMsusp samples were acid-washed to remove any carbonates, which consisted of adding 1 ml 0.1 M HCl, following the protocol developed by50. All macrofaunal items that were dominant in both abundance and biomass in grabs were sampled for SIA using various tissues, depending on the organism (see Supplementary Table S2 for details).
‘Consumer’ (Nephrops) tissues were subsampled from the fisheries catch by selecting n = 10 replicate individuals within each of three size classes (small, medium and large) for both sexes (see Supplementary Table S3). After thawing at room temperature, carapace length, weight without chelipeds (to avoid bias due to claw loss) and sex was recorded for all individuals. Nephrops tissue was sampled from muscle (tail) and hepatopancreas for both males and females, with hepatopancreas in this case representing a shorter-term storage tissue and muscle representing a longer-term storage tissue (see below).
All tissues sampled were oven dried in 2 ml tubes at 60 °C for at least 48 h. Each dried sample was then ground with a mortar and pestle to a fine homogenous powder. Varying amounts of lipids amongst species and tissue types can result in errors in δ13C isotope values if not removed from the tissue prior to measurement51. Therefore, all source and consumer samples underwent lipid correction of three 8 ml washes (or until the supernatant was clear) of 2:1 chloroform:methanol solvent according methodology developed by52. Samples were again dried in the oven at 60 °C for 48 h to remove any remaining solvent. Aliquots of lipid extracted tissue of 400–600 μg were weighed into tin capsules for stable isotope analysis.
Stable isotope ratios (δ13C and δ15N) of all samples were measured at the Stable Isotope Core Laboratory of Washington State University using an elemental analyser (ECS 4010, Costech Analytical, Valencia, CA) connected to a continuous flow isotope ratio mass spectrometer (Delta PlusXP, Thermofinnigan, Bremen) and expressed as parts per thousand (‰) (further details can be found in the Supplementary Methods).
Data analysis
The package SIMMR - Stable Isotopes Mixing Models in R53 was used to estimate the likely contribution of each putative food source to the diet of Nephrops by solving mixing equations for stable isotopic data within a Bayesian framework. SIMMR model outputs are posterior probability distributions representing the likelihood of a specific source being part of the diet of the consumer, with their respective credible intervals. SIMMR was run based on the following input data: 13C and 15N isotope signatures of consumers, mean 13C and 15N isotope signatures of sources i.e. putative prey groups and their standard deviations and estimates for 13C and 15N trophic enrichment factors (means and standard deviations – see below).
For the initial analysis, to show the importance of POMsusp in the diet and to ensure that all dietary sources were captured in the analysis, sources were divided into seven taxa/groupings: (i) Crustaceans; (ii) Filter feeders; (iii) Fish; (iv) Phytoplankton; (v) Polychaeta; (vi) POMsusp and (vii) Zooplankton. Meanwhile, consumers were grouped in all possible combinations of size (small, medium, large), sex (male and female) for long/short-term storage tissues (respectively, muscle and hepatopancreas), providing 12 different combinations overall. Comparison of diet between these consumer groups formed the basis of further hypothesis testing, i.e. statistical comparisons of ‘active feeding’ versus ‘suspension feeding’, as described in ‘Statistical design’, below. The SIMMR model was run twice based on isotopic signatures (for both consumers and food sources) collected in each of the first and second sampling days. Next, four experimental time ‘Periods’ were defined based on the combination of the two sampling dates and two different tissues representing a long (muscle) or short (hepatopancreas) residence times (rt) (Table 2). Residence time for muscle tissue was 81.1 days, obtained from isotopic incorporation rates and discrimination factors in Neogonodactylus bredini (mantis shrimp)54, while rt for hepatopancreas was estimated as 19.3 days. This was calculated from the 13C half-life for hepatopancreas tissues in Callinectes sapidus55 (further details of these calculations can be found in the Supplementary Methods).
Trophic enrichment (or ‘fractionation’) factors (TEFs) of 3.0 ± 0.6‰ for δ13C and 0.9 ± 0.3‰ for δ15N were chosen, based on estimates from mantis shrimp muscle54, which is the best taxon-specific information available. These values contrast with widely-used values from previous meta-analysis56 that present averages from 61 different species of aquatic and terrestrial vertebrates and invertebrates in a variety of taxa: arthropods, molluscs, nematodes, birds, fish and mammals (for information, values in56 were 0.5 ± 0.13‰ for δ13C and 2.3 ± 0.18‰ for δ15N). Nevertheless, we chose the mantis shrimp values54, firstly, on the basis that these fractionation values were calculated from a decapod crustacean: taxonomic relatedness is important due to evidence that TEFs are taxon-specific due to shared physiological processes at taxon level57,58,59,60,61. Secondly, the values in54 represented lipid-corrected stable isotope ratios for consumers and prey, as also used in our study, and were from a diet shift controlled laboratory experiment.
Statistical design
Each group of consumers subjected to hypothesis testing included 10 replicate consumer samples (n = 10). This sample size seems adequate in bootstrapped simulations, which have shown an absence of large biases in statistical inference of stable isotope data with >8 replicate consumer samples62. For hypothesis testing, sources were combined by the function ‘combine_sources’ of SIMMR package into ‘active feeding’ (i.e. filter feeders, polychaete, crustaceans and fish) and ‘suspension feeding’ (i.e. POMsusp, phytoplankton and zooplankton). In order to test our hypotheses, posterior distributions of ‘suspension feeding’ by consumers were compared in several ways. Size-related differences in consumers were examined across all 8 possible ‘Sex’ x ‘Period’ combinations (i.e. all combinations of males and females in 4 time periods). Sex-related differences in consumers were examined across 6 combinations of 3 ‘Size’ groups in 2 periods, ‘Spring long’ and ‘Summer long’. These periods represent an equivalent number of feeding days but with the key difference that ‘Spring long’ included part of the period where females were brooding embryos in Clew Bay, i.e. up until ~10th April45, whereas ‘Summer long’ was a non-brooding period. Any dietary differences associated with female brooding could be judged against males using this comparison. Please note that, as they had just completed their reproductive cycle and had spawned, none of the females sampled actually contained embryo masses, however we could assume that 84–92% of our sample (n = 60) of females had bred, based on previous work17,63,64,65. The suspension feeding contribution was compared across each of the above groups using the function ‘compare_ groups’ from the SIMMR package. This function gives the probability pBIC of ‘any diet source’s proportion in one treatment being greater than the proportion of the same source in another treatment’ with pBIC > 0.95 considered to indicate significant differences66.
The trophic position of Nephrops was determined based on the isotopic signatures of consumers and prey according to a modified version of the following equation67:
where \({\delta }^{15}{N}_{c}\) is the isotopic signature of the consumer Nephrops, Nbase is that of the food base (herein phytoplankton), λ is the trophic position of the base (λ = 1 for primary producers) and Δn is an estimate of the average increase in Δ15N per trophic position/level, herein set at 3.4‰ based on estimates for aquatic food webs67,68. However, because the TEF for δ15N, is significantly lower for decapods54 than for many other taxa56,67,68, we modified the Eq. (1) to incorporate this and prevent an erroneous underestimation of Nephrops’ trophic position, as follows:
where 0.9 is the TEF for δ15N of mantis shrimp54 corresponding to the average increase in Δ15N per trophic position/level; this was subtracted from the isotopic signatures of the consumers in Eq. (1) to facilitate a more accurate trophic position calculation for a decapod, as is the case in the present study. Because this manipulation of the equation underestimates the trophic position in one level, a correction was required by adding one trophic position/level at the end of the calculation, as seen in Eq. (2).
For estimating Nephrops’ overall trophic position, the isotopic signatures in the tissues (muscle and hepatopancreas together) of all individuals sampled in both sampling days (n = 120) were used. In this case, the trophic position of each individual was estimated, and the average of these values was considered to represent the trophic position of Nephrops in Clew Bay in 2014. The average of the nitrogen isotopic signatures of the phytoplankton collected in each of the 2 sampling days was used as \(\,{\delta }^{15}{N}_{base}\).
Data availability
Supplementary Information accompanies this paper. The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
FAO. FishStatJ: software for fishery statistical season series. Available at, http://www.fao.org/fishery/statistics/en (Accessed: 01/09/2019) (2016).
Anon. The Stock Book -Annual Review for Fish Stocks in 2011 with Management Advice for 2012. The Marine Institute, 494pp (2011).
Lordan, C. et al. Porcupine Bank Nephrops Grounds (FU16) 2013 UWTV Survey Report and catch options for 2014 Available at, http://hdl.handle.net/10793/912 (Accessed: 01/09/2019) (2013).
Hobday, A. J. et al. Ecological risk assessment for the effects of fishing. Fish. Res. 108, 372–384 (2011).
Johnson, M. P., Lordan, C. & Power, A. M. Habitat and ecology of N. norvegicus. Adv. Mar. Biol. 64, 27–63 (2013).
Möllmann, C. et al. Implementing ecosystem-based fisheries management: from single-species to integrated ecosystem assessment and advice for Baltic Sea fish stocks. ICES J. Mar. Sci. 71, 1187–1197 (2014).
Chapman, C. J. & Rice, A. L. Some direct observations on the ecology and behaviour of the Norway lobster Nephrops norvegicus. Mar. Biol. 10, 321–329 (1971).
Parslow-Williams, P., Goodheir, C., Atkinson, R. J. A. & Taylor, A. C. Feeding energetics of the Norway lobster, Nephrops norvegicus in the Firth of Clyde, Scotland. Ophelia 56, 101–120 (2002).
Bell, T., Tuck, I. & Dobby, H. Nephrops Species. In Lobsters: Biology, Management, Aquaculture and Fisheries (ed. Phillips, B. F.) 357–413 (John Wiley & Sons Ltd, 2013).
Watts, A. J. R., Albalat, A., Smith, I. P., Atkinson, R. J. A. & Neil, D. M. Seasonal nutritional status in Norway lobsters, Nephrops norvegicus (L.): are females nutritionally compromised over the winter? Mar. Biol. Res. 12, 563–572 (2016).
Gual-Frau, A. & Gallardo-Cabello, M. Analisis de la frecuencia y habitos alimenticios de la ‘Cigala’; Nephrops norvegicus (Linneo, 1758) en el mediterraneo occidental (Crustacea: Nephropsidae). An. Inst. Cienc. del Mar y Limnol. Univ. Nal. Autón. México 15(1), 151–165 (1988).
Cristo, M. & Cartes, J. E. A comparative study of the feeding ecology of Nephrops norvegicus (L.), (Decapoda: Nephropidae) in the bathyal Mediterranean and the adjacent Atlantic. Sci. Mar. 62, 81–90 (1998).
Loo, L. O., Baden, S. P. & Ulmestrand, M. Suspension feeding in adult Nephrops norvegicus (L.) and Homarus gammarus (L.) (Decapoda). Neth. J. Sea Res. 31, 291–297 (1993).
Loc’h, F. L. & Hily, C. Stable carbon and nitrogen isotope analysis of Nephrops norvegicus/Merluccius merluccius fishing grounds in the Bay of Biscay (Northeast Atlantic). Can. J. Fish. Aquat. Sci. 62, 123–132 (2005).
Watts, A. J. R. Nutritional status and trophic dynamics of the Norway lobster Nephrops norvegicus (L.) (PhD Thesis). University of Glasgow Available at, http://theses.gla.ac.uk/id/eprint/3335 (Accessed: 01/09/2019) (2012).
Powell, A. & Eriksson, S. P. Reproduction: Life Cycle, Larvae and Larviculture. Adv. Mar. Biol. 64, 201–245 (2013).
de Figueiredo, M. J. & Thomas, H. J. On the biology of the Norway Lobster, Nephrops norvegicus (L.). J. Cons. Perm. Int. Explor. Mer. 31, 89–101 (1967).
Sardà, F. Reproduction and Moult Synchronism in Nephrops norvegicus (L.) (Decapoda, Nephropidae) in the Western Mediterranean: Is Spawning Annual or Biennial? Crustaceana 60, 186–199 (1991).
Sardà, F. A review (1967–1990) of some aspects of the life history of Nephrops norvegicus. ICES Mar. Sci. Symp. 199, 78–88 (1995).
Haynes, P. S. et al. Growth in Nephrops norvegicus from a tag-recapture experiment. Sci. Rep. 6, 1–11 (2016).
Pinet, P. R. Invitation to Oceanography (Third ed.) 323–360 (Jones and Bartlett Publishers Inc., 2003).
O’Boyle, S. & Silke, J. A review of phytoplankton ecology in estuarine and coastal waters around Ireland. J. Plankton Res. 32, 99–118 (2010).
Stowasser, G. et al. Food web dynamics in the Scotia Sea in summer: A stable isotope study. Deep-Sea. Research II. 59–60, 208–221 (2012).
Harmelin-Vivien, M. Comparison of C and N stable isotope ratios between surface particulate organic matter and microphytoplankton in the Gulf of Lions (NW Mediterranean). Cont. Shelf Res. 28(15), 1911–1919 (2008).
Wieczorek, A. M., Power, A. M. & Browne, P. C. T. Stable-isotope analysis reveals the importance of soft-bodied prey in the diet of lesser spotted dogfish Scyliorhinus canicula. J. Fish Biol. 93, 685–693 (2018).
Fry, B. Stable Isotope Ecology. 173–176 (Springer Science+Business Media, 2006).
Ben-David, M., Flynn, R. W. & Schell, D. M. Annual and seasonal changes in diets of martens: evidence from stable isotope analysis. Oecologia 111, 280–291 (1997).
Carrasco, N. K. & Perissinotto, R. Spatial and temporal variations in the diet of the mysid Mesopodopsis africana in the St. Lucia Estuary (South Africa). MEPS 417, 127–138 (2010).
Fanelli, E. et al. Food partitioning and diet temporal variation in two coexisting sparids, Pagellus erythrinus and Pagellus acarne. J. Fish Biol. 78, 869–900 (2011).
Negrete, P. et al. Temporal variation in isotopic composition of Pygoscelis penguins at Ardley Island, Antarctic: Are foraging habits impacted by environmental change? Polar Biol. 40, 903–916 (2016).
Segura-García, I., Briones-Fourzán, P., de Lestang, S. & Lozano-Álvarez, E. Dietary partitioning between sympatric species of spiny lobster in a coral reef system. Bull Mar Sci. 92, 355–369 (2016).
Villegas, M., Newsome, S. D. & Blake, J. G. Seasonal patterns in δ2H values of multiple tissues from Andean birds provide insights into elevational migration. Ecol. Appl. 26, 2383–2389 (2016).
Bosley, K. M., Copeman, L. A., Dumbauld, B. R. & Bosley, K. L. Identification of burrowing shrimp food sources along an estuarine gradient using fatty acid analysis and stable isotope ratios. Estuar. Coast. 40, 1113–1130 (2017).
Herman, R. W. et al. Seasonal consistency and individual variation in foraging strategies differ among and within Pygoscelis penguin species in the Antarctic Peninsula region. Mar. Biol. 164, 115 (2017).
Phillips, D. L. et al. Best practices for use of stable isotope mixing models in food-web studies. Can. J. Zool. 92, 823–835 (2014).
Hickman, C. P. Jr., Roberts, L. S. & Larson, A. Integrated Principles of Zoology (Ninth ed.) 489–512 (Wm. C. Brown Communications Inc., 1995).
Mente, E., Carter, C. G., Barnes, R. S. & Karapanagiotidis, I. T. Protein synthesis in wild-caught Norway lobster (Nephrops norvegicus L.). J. Exp. Mar. Biol. Ecol. 409, 208–214 (2011).
Rotllant, G. et al. The effects of seasonal variation on the nutritional condition of Nephrops norvegicus (Astacidea: Nephropidae) from wild populations in the western Mediterranean. J. Mar. Biol. Assoc. UK. 94, 763–773 (2014).
Watts, A. J. R., McGill, R. A. R., Albalat, A. & Neil, D. M. Biophysical and biochemical changes occur in Nephrops norvegicus during starvation. J. Exp. Mar. Biol. Ecol. 457, 81–89 (2014).
Riley, G. A. Particulate Organic Matter in Sea Water. Adv. Mar. Biol. 8, 1–118 (1971).
Darnaude, A. M. Fish ecology and terrestrial carbon use in coastal areas: implications for marine fish production. J. Anim. Ecol. 74, 864–876 (2005).
Ziolkowska, M., Sokołowski, A. & Pierre, R. Spatial and temporal variability of organic matter sources and food web structure across benthic habitats in a low diversity system (southern Baltic Sea). J. Sea Res. 141, 47–60 (2018).
Hill, J. M. Structure and flow of carbon and nitrogen to the western Irish Sea Nephrops norvegicus fishery: a stable isotope approach (PhD Thesis). Queen Mary University of London Available at, http://qmro.qmul.ac.uk/xmlui/handle/123456789/1483 (Accessed: 01/09/2019) (2007).
Merder, J. et al. Density dependent growth in ‘catch-and-wait’ fisheries explains body size differences in Nephrops norvegicus. Ambio. https://doi.org/10.1007/s13280-019-01158-1 (2019).
Power, A.M. et al. Field recorded data on habitat, density, growth and movement of Nephrops norvegicus. Scientific Data, https://doi.org/10.1038/s41597-019-0013-x (2019).
Katoh, E., Sbragaglia, V., Aguzzi, J. & Breithaupt, T. Sensory Biology and Behaviour of Nephrops norvegicus. Adv. Mar. Biol. 64, 65–106 (2013).
Lauria, V., Power, A. M., Lordan, C., Weetman, A. & Johnson, M. P. Spatial Transferability of Habitat Suitability Models of Nephrops norvegicus among Fished Areas in the Northeast Atlantic: Sufficiently Stable for Marine Resource Conservation? Plos One 10(2), 1–19 (2015).
Anon. A Survey of Selected Littoral and Sublittoral Sites in Clew Bay, Co.Mayo. A report prepared by Aqua-Fact International Ltd for Dúchas, Department of Arts Heritage and the Gaeltacht Available at, https://www.npws.ie/sites/default/files/publications/pdf/Aquafact_1999_Clew_Bay.pdf (Accessed: 01/09/2019) (1999).
Anon. CLAMS Co-ordinated Local Aquaculture Management Systems Group, Clew Bay Co. Mayo (2001) Available at http://www.gesaq.org/p2clew/documents/clams_clew_bay_2001.pdf. (Accessed: 01/09/2019).
Jacob, U., Mintenbeck, K., Brey, T., Knust, R. & Beyer, K. Stable isotope food web studies: a case for standardized sample treatment. MEPS 287, 251–253 (2005).
Post, D. M. et al. Getting to the fat of the matter: models, methods and assumptions for dealing with lipids in stable isotope analyses. Oecologia 152, 179–189 (2007).
Bligh, E. G. & Dyer, W. J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Phys. 37, 911–917 (1959).
Parnell, A. simmr: A Stable Isotope Mixing Model. R package version 0.3 (2016). Available at, https://CRAN.R-project.org/package=simmr (Accessed: 01/09/2019).
deVries, M. S., del Rio, C. M., Tunstall, T. S. & Dawson, T. E. Isotopic incorporation rates and discrimination factors in mantis shrimp crustaceans. PLoS One 10(4), 1–16 (2015).
Vedral, A. J. Blue crab residency and migration in the Mobile Bay estuary: a stable isotope study investigating connectivity (PhD Thesis). University of Alabama Available at, http://acumen.lib.ua.edu/content/u0015/0000001/0001038/u0015_0000001_0001038.pdf (Accessed: 01/09/2019) (2012).
McCutchan, J. H., Lewis, W. M. Jr., Kendall, C. & McGrath, C. C. Variation in trophic shift for stable isotope ratios of carbon, nitrogen, and sulfur. Oikos 102, 378–390 (2003).
Vanderklift, M. A. & Ponsard, S. Sources of variation in consumer-diet δ15N enrichment: a meta-analysis. Oecologia 136, 169–192 (2003).
Caut, S., Angulo, E. & Courchamp, F. Variation in discrimination factors (Δ15N and Δ13C): the effect of diet isotopic values and applications for diet reconstruction. J. Appl. Ecol. 46, 443–453 (2009).
Suring, E. & Wing, S. R. Isotopic turnover rate and fractionation in multiple tissues of red rock lobster (Jasus edwardsii) and blue cod (Parapercis colias): Consequences for ecological studies. J. Exp. Mar. Biol. Ecol. 370, 56–63 (2009).
del Rio, C. M. & Carleton, S. A. How fast and how faithful: the dynamics of isotopic incorporation into animal tissues. J. Mammal. 93, 353–359 (2012).
Remy, F., Darchambeau, F., Melchior, A. & Lepoint, G. Impact of food type on respiration, fractionation and turnover of carbon and nitrogen stable isotopes in the marine amphipod Gammarus aequicauda (Martynov, 1931). J. Exp. Mar. Biol. Ecol. 486, 358–367 (2017).
Pearson, J. & Grove, M. Counting sheep: sample size and statistical inference in stable isotope analysis and palaeodietary reconstruction. World Archaeol. 45(3), 373–384 (2013).
Thomas, H. J. The Spawning and Fecundity of the Norway Lobsters (Nephrops norvegicus L.) around the Scottish Coast. J. Cons. perm. int. Explor. Mer. 29, 221–229 (1964).
Thomas, H. J. & Figueiredo, M. J. Seasonal Variations in the Catch Composition of the Norway Lobster, Nephrops norvegicus (L.) around Scotland. J. Cons. perm.int. Explor. Mer. 30, 75–85 (1965).
Farmer, A. S. D. Reproduction in Nephrops norvegicus (Decapoda: Nephropidae). J. Zool. 174, 161–183 (1974).
Masson, M. E. J. A tutorial on a practical Bayesian alternative to null-hypothesis significance testing. Behav. Res. Methods 43, 679–690 (2011).
Vander Zanden, M. J. & Fetzer, W. W. Global patterns of aquatic food chain length. Oikos 116, 1378–1388 (2007).
Post, D. M. Using stable isotopes to estimate trophic position: models, methods, and assumptions. Ecology 83, 703–718 (2002).
Acknowledgements
This work was supported by the Irish Research Council Government of Ireland Postgraduate Scholarship GOIPG/2017/1465 and a College of Science Fellowship from the National University of Ireland Galway. We are grateful to Dylan Barrett, Liam Fullbrook, John Galvin, Albert Lawless and Eoin MacLoughlin for help with fieldwork and to Mark P. Johnson and Colm Lordan for helpful discussions.
Author information
Authors and Affiliations
Contributions
A.M.P., C.A.S.S. and C.T.G. designed the study and carried out the statistical analysis; A.M.P. and C.A.S.S. wrote the manuscript, prepared the figures and reviewed the manuscript. C.T.G., A.M.W. and P.B. carried out the fieldwork, prepared the samples and reviewed the manuscript. A.M.P. and C.A.S.S. obtained the funding.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Santana, C.A.d.S., Wieczorek, A.M., Browne, P. et al. Importance of suspended particulate organic matter in the diet of Nephrops norvegicus (Linnaeus, 1758). Sci Rep 10, 3387 (2020). https://doi.org/10.1038/s41598-020-60367-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-60367-x
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.