Abstract
We recently identified the CDC25A phosphatase as a key actor in proliferation and differentiation in acute myeloid leukemia expressing the FLT3-ITD mutation. In this paper we demonstrate that CDC25A level is controlled by a complex STAT5/miR-16 transcription and translation pathway working downstream of this receptor. First, we established by CHIP analysis that STAT5 is directly involved in FLT3-ITD-dependent CDC25A gene transcription. In addition, we determined that miR-16 expression is repressed by FLT3-ITD activity, and that STAT5 participates in this repression. In accordance with these results, miR-16 expression was significantly reduced in a panel of AML primary samples carrying the FLT3-ITD mutation when compared with FLT3wt cells. The expression of a miR-16 mimic reduced CDC25A protein and mRNA levels, and RNA interference-mediated down modulation of miR-16 restored CDC25A expression in response to FLT3-ITD inhibition. Finally, decreasing miR-16 expression partially restored the proliferation of cells treated with the FLT3 inhibitor AC220, while the expression of miR-16 mimic stopped this proliferation and induced monocytic differentiation of AML cells. In summary, we identified a FLT3-ITD/STAT5/miR-16/CDC25A axis essential for AML cell proliferation and differentiation.
Similar content being viewed by others
Introduction
Acute myeloid leukemia are myeloid malignancies characterized by the accumulation of undifferentiated myeloid progenitors in the bone marrow. Although conventional chemotherapies usually induce complete remission process, frequent relapse lead to overall negative prognosis for the patients1. These pathologies originate from different categories of mutations accumulating in stem or progenitor hematopoietic cells, one of the most common concerning the tyrosine kinase receptor Fms-Like Tyrosine Kinase 3 (FLT3)2. Point mutations occuring in the activation loop (FLT3-TKD), and Internal Tandem Duplications (ITD) occuring on the juxtamembrane domain (FLT3-ITD) have been described, both leading to constitutive activation of the receptor. FLT3-ITD mutation is the most frequent and has been associated with a poor outcome3. In consequence, several tyrosine kinase inhibitors targeting FLT3 have been developped, and recently, an improvement in overall survival was shown for the first time through a randomized phase-3 controlled trial that combined first-generation multi-targeted type II FLT3-TKI midostaurin (PKC412) with standard chemotherapy in both newly diagnosed FLT3-ITD and FLT3-TKD AML, and which led to the approval of midostaurin in this indication in 2017 and its common use since then4. Among the next generation of highly specific FLT3 TKIs that have been developed, quizartinib (AC220) compares favorably in monotherapy to standard treatment in refractory or relapsed AML (QuANTUM-R trial (NCT02039726)5), and is currently being evaluated in first line treatment in combination with chemotherapy (QuANTUM-First trial (NCT02668653)). However, resistance to FLT3 TKI treatment still commonly occurs due to the persistence of downstream signaling pathways (e.g. MAPK), point mutations (e.g. FLT3-TKD), or micro-environmental factors3. Although significant efforts are being made to develop more efficient tyrosine kinase inhibitors (e.g. potent type II FLT3 TKI, namely gilteritinib, crenolanib or FF-10101) or to combine FLT3 inhibitors with other targeted therapies (e.g., with Bcl2 inhibitor venetoclax6, NCT03625505), specifically targeting FLT3-ITD leukemic cells remains a challenge for the next years.
Although early oncogenic pathways downstream of FLT3-ITD are well identifeid, cell cycle proteins that may account for deregulated proliferation of these cells remain poorly documented. In a recent work, we found that CDC25A protein level is higly dependent of FLT3-ITD activity, and that CDC25A governs proliferation and differentiation arrest of FLT3-ITD cells7.
CDC25 phosphatases (CDC25A, B and C) are master regulators of cyclin dependent kinase (CDK) activity, and CDC25A has been involved in CDK activation all along the cell cycle, by contrast with CDC25B and C which are more specialized in the G2 and M phases. CDC25A is overexpressed in several types of cancers, this overexpression beingoften correlated with a poor outcome8. We previously reported that CDC25A drives integrin-dependent proliferation of AML cells9, and is up-regulated downstream of oncogenic mutated tyrosine kinases NPM-ALK, BCR-ABL and JAK2V617F in different lymphoid and myeloid malignancies10,11. There are several pathways regulating CDC25A gene and protein12, and it is well established that discrete variations of the protein level affect genomic stability and cell transformation induced by various oncogenes13.
Consequently, better understanding of CDC25A regulation pathways in pathophysiological models appears to be of highest interest in order to identify new potential therapeutic targets.
We recently identified the STAT5 transcription factor as an intermediate between FLT3-ITD and CDC25A regulation in AML. However, the precise mechanism of this regulation remained undefined. In this paper we demonstrate that miR-16 regulates CDC25A protein levels downstream of FLT3-ITD in a STAT5-dependent manner, and that manipulating miR-16 has a high impact on the proliferation and differentiation of AML cells.
Results
STAT5 regulates CDC25A mRNA and protein levels
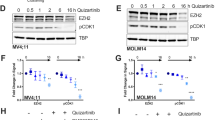
In a previous work7, we showed that FLT3-ITD inhibition induced rapid (2 hours) down-regulation of CDC25A mRNA and protein in the FLT3-ITD AML cell line MOLM-14, and we identified STAT5 as a regulator of CDC25A protein level in this model. Based on these data, we investigated the mechanisms by which CDC25A is regulated downstream of FLT3-ITD. Because STAT5 is a transcription factor, we first examined whether RNA interference-mediated STAT5 down-regulation could impact CDC25A mRNA level. As shown in Fig. 1A, 24 h after transfecting MV4-11 cells with STAT5 siRNA, the level of CDC25A mRNA was decreased by 50%, and CDC25A protein level was dramatically reduced at the same time (Fig. 1A). Similarly, a pharmacological inhibitor of STAT5 reduced both CDC25A mRNA and protein levels as early as 4 hours after treatment (Fig. 1B).
STAT5 regulates CDC25A protein and mRNA level in FLT3-ITD AML cells. (A) MV411 cells were transfected for 24 h (hours) with siRNA against STAT5A/B, and the CDC25A protein and mRNA levels were analyzed by western blot (left panel) and RT-qPCR (right panel) respectively. Western blot analysis of STAT5 protein and active phospho-STAT5 is also shown as a control of RNA interference efficiency (left panel). (B) MOLM14 and MV411 cells were treated for 4 h with STAT5 inhibitor II (1 µM), and the CDC25A protein and mRNA levels were analyzed by western blot (left panel) and RT-qPCR (right panel) respectively. These results are representative of at least 3 independent experiments. Error bars represent the standard error of the mean (SEM). Actin was used as a loading control in the western blot experiments. Full length blots are presented in Supplementary Fig. 3.
These data suggest that STAT5 regulates CDC25A, either directly through gene transcription, or indirectly through post-transcriptional mechanisms that affect its mRNA level, or both.
FLT3-ITD and STAT5 are involved in CDC25A transcription
To establish whether FLT3-ITD activity and STAT5 modulate CDC25A transcription, we performed chromatin-immunoprecipitation (ChIP) experiments on MOLM-14 or MV4-11 AML cells treated with an FLT3 inhibitor. The experimental design is shown in Fig. 2A. First, we performed RNA Polymerase II immune-precipitation and found that the association of RNA polymerase II with the CDC25A promoter was disrupted in the presence of the FLT3 inhibitor (Fig. 2B), indicating that FLT3-ITD activity indeed controls CDC25A transcription. Similar experiments performed with a STAT5 inhibitor suggest that STAT5 is involved in this transcriptional regulation (Fig. 2C). To obtain direct evidence of CDC25A transcriptional regulation by STAT5, we then performed ChIP experiments with a STAT5 antibody in MOLM-14 cells treated with the FLT3 inhibitor. These experiments revealed (i) that the STAT5 factor is indeed present on the CDC25A promoter in these leukemic cells, and (ii) that this association is dependent on FLT3-ITD activity (Fig. 2D). To our knowledge these data represent the first direct evidence of a transcriptional regulation of CDC25A by STAT5, and they demonstrate that FLT3-ITD activity controls this process.
STAT5-dependent transcriptional regulation of CDC25A downstream of FLT3-ITD. (A) Experimental design and position of the Q-PCR primer in the CDC25A promoter used for the CHIP experiments performed on MOLM-14 and MV4-11 cells. (B) MOLM14 and MV411 cells were treated for 4 h with the FLT3 inhibitor AC220 (2 nM). ChIP experiments were then performed with RNA Pol II antibody and mouse IgG as a control. The CDC25A TATA Box region was amplified by qPCR as described in A. (C) MOLM14 and MV411 cells were treated for 4 h with STAT5 inhibitor (1 μM). ChIP experiments were then performed with RNA Pol II antibody and mouse IgG as a control. The CDC25A TATA Box region was amplified by qPCR. (D) MOLM14 cells were treated for 4 h with AC220 (2 nM). ChIP experiments were then performed with STAT5 A/B antibody and rabbit IgG as a control. The CDC25A proximal promoter region was amplified by qPCR. These results are representative of at least 3 independent experiments. Error bars represent the SEM.
FLT3-ITD and STAT5 induce miR-16 down-regulation in AML cells
Since CDC25A is extensively regulated at transcriptional and post-transcriptional levels by several pathways12, we examined whether other mechanisms besides transcription could account for modifications in CDC25A level in AML cells. We specifically focused our attention on the micro RNA miR-16, a known tumor suppressor that has been reported to regulate CDC25A in response to UV-induced DNA damage14, and which was found to be down-regulated in murine FDCP-1 cells that express FLT3-ITD15. First, we tested the impact of FLT3 inhibition on miR-16 level in human MOLM-14 and MV4-11 cells. As shown in Fig. 3A, miR-16 was significantly increased after two hours of FLT3-ITD inhibition. In order to investigate the physiological meaning of these data, we established the status of miR-16 expression in primary cells from AML patients. As shown in Fig. 3B, miR-16 level was significantly lower in AML cells that express FLT3-ITD, further confirming that FLT3-ITD activity negatively regulates miR-16 expression. We then examined whether STAT5 could also regulate miR-16 expression in these cell lines. RNA interference-mediated down-regulation of STAT5 indeed increased miR-16 level to the same extent as FLT3 inhibition (Fig. 3C). In contrast, inhibiting either ERK or Akt, two other important pathways constitutively activated downstream of FLT3-ITD, did not modify miR-16 levels (Fig. 3D). From these experiments, we concluded that miR-16 level is negatively controlled by FLT3-ITD in a STAT5-dependent way in AML. Since miR-16 is part of two complex loci also expressing either miR-15-a, or miR-15-b, we also tested whether the level of these 2 micro-RNAs was dependent of FLT3 activity. As shown in Supplementary Fig. 1, FLT3 inhibition did not impact the levels of miR-15-a, or b, which indicates that the regulation of miR-16 expression by FLT3-ITD is not a regulation of either of the miR-16 cluster genes, as this would also impact miR-15-a or b expression.
miR-16 level is dependent on FLT3-ITD activity and STAT5 in AML cells. (A) MOLM-14 and MV4-11 cells were treated for 2 h with the FLT3 inhibitor AC220 (2 nM) and the miR-16 level was analyzed by RT-qPCR. (B) Basal miR-16 levels were analyzed by RT-qPCR and normalized to a control (MOLM14). A break in the Y axis was added for easier presentation of the results. (C) MV4-11 cells were transfected for 24 h with STAT5A/B siRNA and the miR-16 level was analyzed by RT-qPCR. (D) MOLM-14 and MV4-11 cells were treated for 2 h with ERK or Akt inhibitors (50 nM and 5 µM respectively) and the miR-16 level was analyzed by RT-qPCR.(E) MOLM-14 and MV4-11 cells were transfected for 24 h with a premiR-16 and the CDC25A protein level was analyzed by western blot. (F) MOLM-14 and MV4-11 cells were transfected for 24 h with a miR-16 inhibitor and treated for 2 h with AC220 (2 nM), the CDC25A and Pim1 protein levels were analyzed by western blot. (G) MV4-11 cells were transfected for 24 h with a miR-16 inhibitor and STAT5A/B siRNA, and the CDC25A protein level was analyzed by western blot. These results are representative of at least 3 independent experiments. Error bars represent the SEM. Actin was used as a loading control in the western blot experiments. ns: non-specific. Full length blots are presented in Supplementary Fig. 4.
MiR-16 regulates CDC25A protein level
We then examined whether miR-16 regulates CDC25A protein level downstream of FLT3-ITD. Transfection of FLT3-ITD AML cell lines with miR-16 mimic indeed dramatically reduced CDC25A protein levels (Fig. 3E). The low level of CDC25A protein in these experiments is caused by the cell cycle arrest induced after cell transfection. To further specify this process, we then used an RNA interference strategy to repress miR-16 expression in MOLM-14 and MV4-11 cells treated with the FLT3i AC220 (Fig. 3G). As expected, down-regulating miR-16 in these conditions totally rescued CDC25A protein expression on FLT3 inhibition, demonstrating that miR-16 is the major regulator of CDC25A protein level downstream of FLT3-ITD. Interestingly, in opposition to results obtained by Kim el al.15, we observed no rescue of the Pim1 kinase level by miR-16 inhibition, suggesting that miR-16 does not regulate Pim1 expression downstream of FLT3-ITD in our cell model. We then tested whether miR-16 also regulates CDC25A protein expression downstream of STAT5. As expected, inhibiting miR-16 completely restored CDC25A protein expression in MOLM-14 cells transfected with STAT5 siRNA (Fig. 3F), which confirms the existence of a FLT3-ITD/STAT5/miR-16/CDC25A axis in these AML cells.
To further specify how miR-16 regulates CDC25A translation, we then measured CDC25A mRNA level by RT-QPCR. In contrast to our observations concerning CDC25A protein level, miR-16 inhibition only very partially restored CDC25A mRNA expression when FLT3-ITD is inhibited in MOLM-14 cells, and not at all when FLT3-ITD is inhibited or STAT5 is down-regulated in MV4-11 cells (Supplementary Fig. 2A,B). Moreover, when CDC25A mRNA stability was studied using an Actinomycin-D chase experiment only a low impact of FLT3 inhibition (Supplementary Fig. 2C) and no impact of STAT5 knockdown (Supplementary Fig. 2D) on CDC25A mRNA half-life were shown. Altogether these results imply that although miR-16 might somehow destabilize CDC25A mRNA, its major function resides in the inhibition of mRNA translation.
The STAT5/miR-16/CDC25A axis is specific to FLT3-ITD AML cells
We then examined whether these molecular pathways specifically work downstream of FLT3-ITD. In order to do this, we first used the HEL leukemic cell line, which expresses the mutant tyrosine kinase JAK2V617F but not the FLT3 receptor, and in which we previously described a STAT5-dependent regulation of CDC25A11. As shown in Fig. 4A, RNA interference-mediated down-regulation of STAT5 actually induced a dramatic decrease in CDC25A protein level, but inhibiting miR-16, which was modestly increased under these conditions (Fig. 4B), did not restore CDC25A protein level. This indicates that STAT5 regulation of CDC25A does not depend on miR-16 in this cell line. In the OCI-AML3 leukemic cell line, which expresses wild type FLT3, activating FLT3 with FLT3 ligand, or inhibiting the receptor with AC220, modified neither CDC25A protein nor the miR-16 level (Fig. 4C,D). Overall, these data obtained in human leukemic cells suggest that several different signaling pathways operate in different cell lines to regulate CDC25A, and that the STAT5/miR-16/CDC25A pathway specifically acts downstream of the FLT3-ITD mutant receptor in AML.
The STAT5/miR-16/CDC25A axis is specific to FLT3-ITD positive cells. (A) HEL cells were transfected for 24 h with a miR-16 inhibitor and STAT5A/B siRNA. CDC25A and STAT5 protein levels were analyzed by western blot. (B) HEL cells were transfected for 24 h with STAT5A/B siRNA and miR-16 level was analyzed by RT-qPCR. (C) OCI-AML3 cells were treated for 2 h with the FLT3 inhibitor AC220 (2 nM) and/or FLT3 ligand (6 nM), and the CDC25A protein level was analyzed by western blot. (D) OCI-AML3 cells were treated for 2 h with the FLT3 inhibitor AC220 (2 nM) and/or FLT3 ligand (6 nM) and the miR-16 level was analyzed by RT-qPCR. These results are representative of at least 3 independent experiments. Error bars represent the SEM. Actin was used as a loading control in the western blot experiments. ns: non-specific. Full length blots are presented in Supplementary Fig. 5.
MiR-16 regulates FLT3-ITD AML cell proliferation and differentiation
Considering that we identified CDC25A as a master regulator of FLT3-ITD AML proliferation and differentiation, we decided to investigate whether this is also the case for miR-16. Expressing miR-16 mimic in MOLM-14 or MV4-11 cells for 72 h significantly decreased their proliferation (Fig. 5A). Interestingly, this was not the case in the OCI-AML3 AML cells that express wild type FLT3. Down-regulating miR-16 partially restored proliferation of MOLM-14 or MV4-11 cells treated with the FLT3 inhibitor AC220 (Fig. 5B), suggesting that miR-16 regulation and functions could be central to the therapeutic effects of FLT3 inhibitors. We then tested the importance of miR-16 in a MOLM-14-TKD cell line that is resistant to FLT3-ITD inhibition7,16. Overexpressing miR-16 also reduced cell proliferation in this case, suggesting that targeting this pathway could overcome some of the resistance observed with FLT3-ITD kinase inhibitors (Fig. 5A). In accordance with these results, the clonogenic potential of primary cells from a FLT3-ITD-positive patient was reduced when miR-16 was overexpressed, while there was no impact in FLT3wt primary samples (Fig. 5C).
miR-16 inhibits cell proliferation in FLT3-ITD cell lines and primary cells, and triggers monocytic differentiation in FLT3-ITD AML cells. (A) MOLM-14 and MV4-11 (FLT3-ITD), OCI-AML3 (FLT3wt) and MOLM-14-TKD (FLT3-ITD inhibition resistant) cell lines were transfected with pre-miRcontrol or pre-miR-16 and live cells were counted at 72 h after trypan blue staining. (B) MOLM-14 and MV4-11 cells were transfected with pre-miRcontrol or pre-miR-16 as in A, and then treated with AC220 (2 nM) 24 h after transfection. 72 h after transfection, cells were counted after trypan blue staining. (C) Primary cells from three AML patients were transduced with a lentiviral vector expressing a scrambled shRNA (Ctrl, black bars) or the miR-16 (miR-16, white bars), and clonogenic potential was assessed by colony forming assay 7 days after transduction (left panel). Transduction efficiency was assessed by RT-qPCR after 3 days (right panel). (D)- F. MOLM-14, OCI-AML3, MOLM14-TKD or MV4-11 cells were transfected with pre-miRcontrol or pre-miR-16 as in A, and then labeled with anti CD11 (D) and CD14 (E) or anti CD15 (F) antibodies in order to monitor their differentiation state. Annexin V labeling was performed in parallel in order to exclude dying cells from the FACS analysis. These results are representative of at least 3 independent experiments. Error bars represent the SEM.
In addition to proliferation, we then examined whether miR-16 down-regulation by FLT3-ITD was involved in the differentiation block of these cells. As seen in Fig. 5D, miR-16 expression induced differentiation of MOLM-14 cells, as indicated by increased expression of the cell surface differentiation marker CD11b. Interestingly, this was also the case in MOLM-14-TKD cells that are resistant to FLT3 inhibition, but not in cells that express wild type FLT3 (OCI-AML3). Finally, we also observed re-expression of the CD14 but not the CD15 surface differentiation marker with miR-16 overexpression (Fig. 5E,F), suggesting that monocytic rather than granulocytic differentiation was induced by miR-16 in FLT3-ITD cells, which is highly consistent with our previous work on CDC25A7.
Discussion
In this work we investigated the molecular mechanisms that control CDC25A transcription and translation in acute myeloid leukemia cells that express the FLT3-ITD mutant tyrosine kinase receptor. This investigation picks up where our previous study leaves off, in which we identified this phosphatase as a master determinant of proliferation and differentiation arrest in this leukemic subtype7. CDC25A major function is the activation of CDK complexes, but its direct impact has been also reported on the tyrosine kinase receptor EGRF17. In consequence, we tested whether in our model, CDC25A could directly regulate FLT3-ITD activity. As can be seen in Supplementary Fig. 3, CDC25 inhibition did not affect tyrosine kinase phosphorylation of FLT3-ITD, nor STAT5 phosphorylation, suggesting that CDC25A most likely regulates AML cells proliferation through its canonical function of CDKs activator.
In general, CDC25A expression is tightly regulated in myeloid malignancies that express tyrosine kinase mutations7,11,18 in which STAT5 and STAT3 pathways are most often activated. Transcriptional regulations of CDC25A by E2F119, myc20 and STAT321 have been described previously in different cellular contexts. However, the question of STAT5-mediated transcriptional regulation of CDC25A remained an open one. In our study, STAT5 is identified at the CDC25A promoter for the first time, and we highlight the importance of STAT5 activity for proper CDC25A transcription downstream of the FLT3-ITD receptor. It has been reported that STAT3 associates with myc or Rb to respectively activate or inhibit CDC25A transcription21. Therefore, whether STAT5 associates with transcriptional partners to finely tune CDC25A transcription in AML cells remains an interesting question.
The importance of STAT factors in hematological models has been extensively documented22,23, and researchers have been taking interest in the clinical relevance of these factors as targets in various cancer models24,25. Recently, a study in MDS and AML models showed an impairment in leukemic growth in vivo and an increase in differentiation in primary progenitor cells with the use of a clinical antisense nucleotide that targets STAT326. Our current findings along with those of other studies27,28 highlight and specify the importance of STAT5 as part of an essential pathway for FLT3-ITD AML proliferation and survival, and efforts to target STAT5 in AML are in progress29.
In addition to this direct transcriptional regulation, we have highlighted another CDC25A regulation pathway downstream of FLT3-ITD involving STAT5 as well. In fact, we identified micro-RNA 16 as a key negative regulator of CDC25A protein expression downstream of FLT3-ITD and STAT5 in AML cells. First, our data support the argument that STAT5 is an actor in miR-16 regulation downstream of FLT3-ITD. STAT5 has been shown to regulate miR-16-2 and miR15-b expression in multiple myeloma30 by inhibiting the transcription of their cluster gene, SMC4. However, in AML cell lines, we found no effect of FLT3 inhibition on miR-15-a, or -b expression, which suggests a more complex regulation of miR-16 that may involve maturation rather than transcriptional regulation of one or both cluster genes (for a review see31).
Our data also point to the fact that miR-16 is an important determinant of CDC25A protein level downstream of FLT3-ITD. MiR-16-dependent regulation of CDC25A has been previously reported in response to UV radiation-induced DNA damage, participating in the checkpoint response and proliferation arrest observed in these conditions14. In this study, we demonstrate that miR-16-dependent regulation of CDC25A is not restricted to DNA damage response, and could participate in the response of leukemic cells to tyrosine kinase inhibitors. There are 2 seed sequences of miR-16 in the 3’UTR region of CDC25A mRNA. This strongly suggests a direct interaction between miR-16 and CDC25A mRNA, as was suggested with a luciferase reporter system in Hela cells in response to UV radiation14. However, in this study we did not detect a significant impact of miR-16 expression on CDC25A mRNA level in AML cells, which suggests that depending on the cell and the environmental conditions, the functional miR-16 molecular mechanisms may differ.
In a previous work, Kim et al. described a down-regulation of miR-16 by FLT3-ITD15. Pim1 regulation by miR-16 has been described in a murine cell line model overexpressing FLT3-ITD15. However our data do not support the argument that Pim1 is a miR-16 target in the human FLT3-ITD AML cell lines MOLM-14 and MV4-11 (see Fig. 3E). It remains to be established whether this discrepancy is due to the distinct AML cell models used in both studies or to other unknown parameters. However, considering that transcriptional regulation of Pim1 by STAT5 is a well-established event downstream of FLT3-ITD32,33, this may suggest the existence of a complex oncogenic signaling network including STAT5, miR-16, Pim1, and CDC25A that still needs to be clarified.
MiR-16 is considered to be a tumor suppressor because it negatively regulates the expression of pro-oncogenic proteins involved in cell proliferation and cell death. Therefore, some studies aimed at expressing miR-16 as a therapeutic approach have been developed in a murine model of CLL34,35. However the use of micro RNAs as therapeutic tools remains rather unsuccessful as they have never progressed beyond a phase I trial36. This emphasizes the need for a clearer understanding of the networks of proteins regulated downstream of these oligonucleotides. Although we describe CDC25A as an important target of miR-16 in this study, it is unlikely that all the effects of miR-16 expression in FLT3-ITD cells are only due to CDC25A regulation. For instance, in terms of the effects of miR-16 expression on the differentiation process, it should be noted that this RNA is involved in retinoic acid induced differentiation37, and that PU.1, an important transcriptional regulator of myeloid differentiation, has been described as a miR-16 target38. Whether or not the miR-16-dependent differentiation that we observed in FLT3-ITD AML cells is linked with these pathways remains to be investigated. With regards to its cancer relevance, another interesting miR-16 target that has been established is the anti-apoptotic protein Bcl239. Bcl2 inhibitors are currently being introduced as potential therapeutic tools in AML40, and interestingly, a synergy between venetoclax and the FLT3-inihibitor quizartinib has been described41. It would be interesting to establish whether miR-16-dependent regulation of Bcl2 is involved in this process.
In conclusion, our data point to the following model, in which FLT3-ITD activity leads to miR16 repression through a STAT5-dependent mechanism that remains to be precised. In consequence, STAT5 activation downstream of FLT3-ITD contributes to CDC25A protein expression by two different ways: STAT5 represses miR16, which is a negative regulator of CDC25A translation in this model, and directly participates to CDC25A gene transcription. This pathways participates to proliferation and differentiation arrest of FLT3-ITD AML cells. This model is summarized in Fig. 6.
A model of CDC25A regulation by STAT5 and miR16 downstream of FLT3-ITD. FLT3-ITD activated STAT5 positively regulates the transcription of the CDC25A gene, and negatively regulates miR16 expression. miR16 is a negative regulator of CDC25A translation in these cells. Direct transcriptional activity of STAT5 together with miR16 down regulation are key events leading to CDC25A protein expression, and consequent proliferation in these AML cells.
Methods
Cell lines and reagents
Human acute myeloid leukemia cell lines MOLM-14, MV4-11, OCI-AML3, HEL (DSMZ, Braunschweig, Germany), and MOLM-14-TKD (provided by Dr. J Tamburini, Paris) were cultured in RPMI 1640 medium (Gibco, Life Technologies, Carlsbad, CA, USA), supplemented with 10% Fetal Bovine Serum (Gibco, Life Technologies, Carlsbad, CA, USA). All cells were cultured at 37 °C and 5% CO2 in a humidified incubator, and were regularly tested for mycoplasma contamination. The Akt inhibitor VIII and STAT5 inhibitors were purchased from Calbiochem (San Diego, CA, USA). The FLT3 inhibitor quizartinib AC220 and the MEK inhibitor PD0325901 were purchased from Selleck Chemicals (Houston, TX, USA). FLT3 ligand was purchased from R&D Systems (Minneapolis, MN, USA), and D Actinomycin was purchased from Sigma (Saint-Louis, MO, USA).
Western Blot
1 × 106 cells were lysed in 60 μl of NuPAGE® LDS Sample Buffer (Novex, Life Technologies, Carslbad, CA, USA), sonicated for 4 × 7 seconds, and boiled for 3 minutes. Proteins were then resolved on NuPAGE® 4–12% Bis-Tris Gels and transferred to nitrocellulose membrane. The membrane was saturated for 1 hour in Tris Buffer Saline with Tween 0.1% (TBS-T) containing 5% non-fat milk. Membranes were blotted with suitable antibodies overnight at 4 °C, washed thrice with TBS-T and incubated for 1 hour with HRP-conjugated secondary antibody (Promega, Madison, WI, USA). After four additional washes, detection was achieved with ECLTM or ECLTM prime western blotting detection reagents (GE Healthcare, Chicago, IL, USA). The antibodies used were: monoclonal anti-CDC25A (F-6, sc-7389) and anti-Pim1 (12H8, sc-13513) from Santa Cruz; polyclonal anti-phospho-STAT5 A/B (Tyr 694, 9351), anti-STAT5 A/B (9363), from Cell Signaling Technology (Beverly, MA, USA) and anti-α-actin (MAB1501) from Millipore (Burlington, MA, USA).
RT-qPCR
Cells were lysed in TRIzol (Invitrogen, Life Technologies, Carlsbad, CA, USA) and total RNA was then extracted according to the manufacturer’s instructions. For gene expression analysis, cDNA was generated with the SuperScript III First-Strand Synthesis System for RT-PCR (Invitrogen, Life Technologies, Carlsbad, CA, USA) according to the manufacturer’s instructions. The PCR was performed with TaqMan® Gene Expression Master Mix (Applied Biosystems, Life Technologies, Carlsbad, CA, USA) with 10 ng of cDNA, The primer used was Hs00947994_m1 (Applied Biosystem, Life Technologies, Carlsbad, CA, USA) for CDC25A. GUSB (Hs00939627_m1) and B2M (Hs00984230_m1) were used as housekeeping genes. For mature micro-RNA expression analysis, cDNA was generated using the TaqMan® micro-RNA reverse transcription kit (Applied Biosystems, Life Technologies, Carlsbad, CA, USA), and PCR was performed using the TaqMan® Universal Master Mix with UNG (Applied Biosystems, Life Technologies, Carlsbad, CA, USA), and the TaqMan microRNA assays (Applied Biosystems, Life Technologies, Carlsbad, CA, USA) according to the manufacturer’s instructions. The primers used were miR-16 (assay 000391) and U6 snRNA (assay 001973) as a control. Results were analyzed with the StepOneTM software v2.2.2 (Applied Biosystems, Life Technologies, Carlsbad, CA, USA) using the conventional ΔΔCt method. PCR was carried out on a StepOneTM (Applied Biosystems, Life Technologies, Carlsbad, CA, USA).
RNA transfection
Leukemic cell lines were transfected with the Neon transfection system (Life Technologies, Carlsbad, CA, USA). Cells (2 × 106) were re-suspended in 100 μl of resuspension buffer, and various doses of RNA were added. RNA sequences were: specific STAT5A and STAT5B siRNA (ON-TARGETplus SMARTpool, human STAT5A and STAT5B, Dharmacon, Lafayette, CO, USA) or siRNA control (sigenome control pool non targeting #2, or ON-TARGETplus control pool, Dharmacon, Lafayette, CO, USA), anti-miR control or anti-miR-16 (Life Technologies, Carlsbad, CA, USA), and pre-microRNA negative control or pre-microRNA-16 (Life Technologies, Carlsbad, CA, USA). Cells were then transfected with the nucleofector device (program 5 for MOLM14, MV4-11 and OCI-AML3 cell lines, custom program for the HEL cell line), and subsequently re-suspended in normal culture medium at a concentration of 1 × 106 cells/ml.
Chromatin immuno-precipitation (ChIP)
Cells were crosslinked, lysed and sonicated according to42, then STAT5 and RNA polymerase II immunoprecipitation was performed according to43. The antibodies used for immunoprecipitation were STAT5 A (sc-1081, Santa Cruz Biotechnology, Santa Cruz, CA, USA) and STAT5 B (sc-835, Santa Cruz Biotechnology), 1.2 µg each, and RNA Polymerase II CTD (MABI0601, Clinisciences, Nanterre, France), 2 µg. DNA from ChIP was isolated with a phenol/chloroform extraction, and used for qPCR with the primers listed in Supplementary Table 1. Results were then normalized using the percent input method.
Patient samples
Patient AML samples used in this work were obtained after informed consent in accordance with the Declaration of Helsinki and were stored in the HIMIP collection. In compliance with French law, the HIMIP collection was declared to the Ministry of Higher Education and Research (DC 2008-307 collection 1) and a transfer agreement was obtained (AC 2008-129) after approval by the ethics committees (Comité de Protection des Personnes Sud-Ouest et Outremer II and the APHP ethics committee). Clinical biological annotations of the samples were declared to the CNIL (French Data Protection Authority). All were diagnosed at the Department of Hematology of Toulouse University Hospital. The patients’ characteristics are listed in Supplementary Table 2.
Patient samples and clonogenic assays
Patient samples’ origins and characteristics are described in the Online Supplementary Methods and Supplementary Table 2.
Frozen patient cells were thawed in IMDM medium with 20% FBS and immediately processed for treatment. For clonogenic assays, cells were grown in duplicate in H4230 methylcellulose medium (Stem Cell Technologies, Vancouver, BC, Canada) supplemented with 10% 5637-conditionned medium in 35 mm petri dishes for 7 days in a humidified CO2 incubator. Leukemic colonies were then scored under an inverted microscope.
Transduction of primary cells
To generate lentiviral vectors that express the micro-RNA 16, the miR-16 hairpin sequence was cloned into the pLKO.1 lentiviral vector. We used 293T packaging cells co-transfected with lentiviral protein (GAG, POL, and REV) that encode plasmids and our control (a scrambled shRNA hairpin sequence) or miR-16 plasmids. Supernatants containing lentivirus were collected for the next two day. Patient cells were thawed, plated at 1.5 million cells/ml and spinoculated for an hour with the concentrated lentiviral particles, before being inoculated for clonogenic assays.
Flow cytometry
Cells were stained for 30 min (minutes) with the anti-human monoclonal antibody CD11b-PE (IM2581U, Beckman Coulter, Brea, CA, USA), CD14-FITC (555397, BD Biosciences, San Jose, CA, USA), or CD15-APC (551376, BD Biosciences, San Jose, CA, USA). Annexin V-FITC or APC (BD Biosciences, San Jose, CA, USA) and 7-AAD (Cell viability assay, BD Biosciences, San Jose, CA, USA) were used to assess cell viability. Data were collected on a MacsQuant cytometer (BD Biosciences, San Jose, CA, USA), and analyzed using the FlowJo software (BD Biosciences, San Jose, CA, USA).
Statistics
Experiments in cell lines were performed at least 3 times. Results are expressed as mean value +/− SEM. Statistical analysis of the data was performed by the Mann-Whitney U test using the GraphPad Prism software, version 5.0 (GraphPad Software Inc., La Jolla, CA, USA). Differences were considered as significant for p values < 0.05 (*), p < 0.01 (**), p < 0.001 (***).
Data availability
All data generated or analysed during this study are included in this published article (and its Supplementary Information files).
References
Döhner, H., Weisdorf, D. J. & Bloomfield, C. D. Acute myeloid leukemia. N. Engl. J. Med. 373, 1136–52 (2015).
Papaemmanuil, E. et al. Genomic Classification and Prognosis in Acute Myeloid Leukemia. N. Engl. J. Med. 374, 2209–2221 (2016).
Daver, N., Schlenk, R. F., Russell, N. H. & Levis, M. J. Targeting FLT3 mutations in AML: review of current knowledge and evidence. Leukemia 33, 299–312 (2019).
Stone, R. M. et al. Midostaurin plus Chemotherapy for Acute Myeloid Leukemia with a FLT3 Mutation. N. Engl. J. Med. 377, 454–464 (2017).
Cortes, J. E. et al. Efficacy and Safety of Single-Agent Quizartinib (Q), a Potent and Selective FLT3 Inhibitor (FLT3i), in Patients (pts) with FLT3-Internal Tandem Duplication (FLT3-ITD)-Mutated Relapsed/Refractory (R/R) Acute Myeloid Leukemia (AML) Enrolled in the Global, Phase 3, Randomized Controlled Quantum-R Trial. Blood 132, 563 (2018).
Grundy, M. et al. Predicting effective pro-apoptotic anti-leukaemic drug combinations using co-operative dynamic BH3 profiling. PLoS One 13, e019068 (2018).
Bertoli, S. et al. CDC25A governs proliferation and differentiation of FLT3-ITD acute myeloid leukemia. Oncotarget 6, 38061–38078 (2015).
Boutros, R., Lobjois, V. & Ducommun, B. CDC25 phosphatases in cancer cells: key players? Good targets? Nat. Rev. Cancer 7, 495–507 (2007).
Fernandez-Vidal, A. et al. Cell adhesion regulates CDC25A expression and proliferation in acute myeloid leukemia. Cancer Res. 66, 7128–35 (2006).
Fernandez-Vidal, A. et al. Upregulation of the CDC25A phosphatase down-stream of the NPM/ALK oncogene participates to anaplastic large cell lymphoma enhanced proliferation. Cell Cycle 8, 1373–9 (2009).
Gautier, E. F. et al. The cell cycle regulator CDC25A is a target for JAK2V617F oncogene. Blood 119, 1190–1199 (2012).
Fernandez-Vidal, A., Mazars, A. & Manenti, S. CDC25A: a rebel within the CDC25 phosphatases family? Anticancer Agents Med. Chem. 8, 825–831 (2008).
Ray, D. & Kiyokawa, H. CDC25A phosphatase: a rate-limiting oncogene that determines genomic stability. Cancer Res. 68, 1251–3 (2008).
Pothof, J. et al. MicroRNA-mediated gene silencing modulates the UV-induced DNA-damage response. EMBO J. 28(14), 2090–2099 (2009).
Kim, K. T. et al. MicroRNA-16 is down-regulated in mutated FLT3 expressing murine myeloid FDC-P1 cells and interacts with Pim-1. Plos One 7, e44546 (2012).
Larrue, C. et al. Antileukemic Activity of 2-Deoxy-d-Glucose through Inhibition of N-Linked Glycosylation in Acute Myeloid Leukemia with FLT3-ITD or c-KIT Mutations. Mol. Cancer Ther. 14, 2364–2373 (2015).
Wang, Z., Wang, M., Lazo, J. S. & Carr, B. I. Identification of epidermal growth factor receptor as a target of CDC25A protein phosphatase. J. Biol. Chem. 277, 19470–19475 (2002).
Seedhouse, C. et al. Impaired S-phase arrest in acute myeloid leukemia cells with a FLT3 internal tandem duplication treated with clofarabine. Clin. Cancer Res. 15, 7291–7298 (2009).
Vigo, E. et al. CDC25A phosphatase is a target of E2F and is required for efficient E2F-induced S phase. Mol. Cell Biol. 19, 6379–6395 (1999).
Galaktionov, K., Chen, X. & Beach, D. Cdc25 cell-cycle phosphatase as a target of c-myc. Nature. 382, 511–517 (1996).
Barré, B., Vigneron, A. & Coqueret, O. The STAT3 transcription factor is a target for the Myc and riboblastoma proteins on the Cdc25Apromoter. J. Biol. Chem. 280, 15673–15681 (2005).
Coffer, P. J., Koenderman, L. & de Groot, R. P. The role of STATs in myeloid differentiation and leukemia. Oncogene 19, 2511–2522 (2000).
Bunting, K. D. STAT5 signaling in normal and pathologic hematopoiesis. Front. Biosci. 12, 2807–2820 (2007).
Rani, A. & Murphy, J. J. STAT5 in Cancer and Immunity. J. Interferon Cytokine Res. 36, 226–37 (2016).
Groner, B. & von Manstein, V. Jak Stat signaling and cancer: Opportunities, benefits and side effects of targeted inhibition. Mol. Cell Endocrinol. 451, 1–14 (2017).
Shastri, A. et al. Antisense STAT3 inhibitor decreases viability of myelodysplastic and leukemic stem cells. J. Clin. Invest. 128, 5479–5488 (2018).
Gerloff, D. et al. NF-κB/STAT5/miR-155 network targets PU.1 in FLT3-ITD-driven acute myeloid leukemia. Leukemia 29, 535–547 (2015).
Nogami, A. et al. Inhibition of the STAT5/Pim Kinase Axis Enhances Cytotoxic Effects of Proteasome Inhibitors on FLT3-ITD-Positive AML Cells by Cooperatively Inhibiting the mTORC1/4EBP1/S6K/Mcl-1 Pathway. Transl. Oncol. 12, 336–349 (2019).
Wingelhofer, B. et al. Pharmacologic inhibition of STAT5 in acute myeloid leukemia. Leukemia. 32, 1135–1146 (2018).
Li, G. et al. STAT5 requires the N-domain for suppression of miR15/16, induction of bcl-2, and survival signaling in myeloproliferative disease. Blood 115, 1416–1424 (2010).
Liu, T., Xu, Z., Ou, D., Liu, J. & Zhang, J. The miR-15a/16 gene cluster in human cancer: A systematic review. J. Cell Physiol. 234, 5496–5506 (2019).
Nosaka, T. et al. STAT5 as a molecular regulator of proliferation, differentiation and apoptosis in hematopoietic cells. EMBO J. 18, 4754–4765 (1999).
Mizuki, M. et al. Suppression of myeloid transcription factors and induction of STAT response genes by AML-specific Flt3 mutations. Blood 101, 3164–3173 (2003).
Kasar, S. et al. Systemic in vivo lentiviral delivery of miR-15a/16 reduces malignancy in the NZB de novo mouse model of chronic lymphocytic leukemia. Genes Immun. 13, 109–119 (2012).
Cutrona, G. et al. Effects of miRNA-15 and miRNA-16 expression replacement in chronic lymphocytic leukemia: implication for therapy. Leukemia 31, 1894–1904 (2017).
Beg, M. S. et al. Phase I study of MRX34, a liposomal miR-34a mimic, administered twice weekly in patients with advanced solid tumors. Invest. New Drugs. 35, 180–188 (2017).
Gao, S. M. et al. miR-15a/16-1 enhances retinoic acid-mediated differentiation of leukemic cells and is up-regulated by retinoic acid. Leuk. Lymphoma 52, 2365–2371 (2011).
Moon, H. G., Yang, J., Zheng, Y. & Jin, Y. miR-15a/16 regulates macrophage phagocytosis after bacterial infection. J. Immunol. 193, 4558–4567 (2014).
Cimmino, A. et al. miR-15 and miR-16 induce apoptosis by targeting BCL2. Proc. Natl. Acad. Sci. USA 102, 13944–13949 (2005).
Wei, A. H. et al. Venetoclax Combined With Low-Dose Cytarabine for Previously Untreated Patients With Acute Myeloid Leukemia: Results From a Phase Ib/II Study. J. Clin. Oncol. JCO1801600 [Epub ahead of print] (2019).
Singh Mali, R. et al. FLT3-ITD Activation Mediates Resistance to the BCL-2 Selective Antagonist, Venetoclax, in FLT3-ITD Mutant AML Models. Blood 130, 1348 (2017).
Pelish, H. E. et al. Mediator kinase inhibition further activates super-enhancer-associated genes in AML. Nature 526, 273–276 (2015).
Rascle, A., Johnston, J. A. & Amati, B. Deacetylase activity is required for recruitment of the basal transcription machinery and transactivation by STAT5. Mol. Cell Biol. 23, 4162–4173 (2003).
Acknowledgements
We would like to sincerely thank Laure Guitton-Sert for her technical input in designing ChIP experiments. We thank Carine Valle at the CRCT molecular biology facility and Manon Farcé at the CRCT cytometry facility for their helpful discussions of the analyses. We would also like to thank Véronique De Mas, Cécile Demur and Eric Delabesse for the management of the BRC-HIMIP Biobank (Biological Resources Centres-INSERM Midi-Pyrénées “Cytothèque des hémopathies malignes”). Gabrielle Sueur is a recipient of the Ligue Nationale contre le Cancer.
Author information
Authors and Affiliations
Contributions
G.S., S.M., and S.B. concepted and designed the study; G.S., A.Bo., and M.G. performed experiments; S.B. and V.MDM. contributed clinical samples; G.S., S.M., and S.B. wrote the manuscript; A.Be., and S.M. insured administrative, technical and material support; S.M. and S.B. supervised the project.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sueur, G., Boutet, A., Gotanègre, M. et al. STAT5-dependent regulation of CDC25A by miR-16 controls proliferation and differentiation in FLT3-ITD acute myeloid leukemia. Sci Rep 10, 1906 (2020). https://doi.org/10.1038/s41598-020-58651-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-58651-x
This article is cited by
-
JAK/STAT in leukemia: a clinical update
Molecular Cancer (2024)
-
Regulation of proton partitioning in kinase-activating acute myeloid leukemia and its therapeutic implication
Leukemia (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.