Abstract
This study investigated the effectiveness of Ai Chi compared to conventional water-based exercise on balance performance in individuals with chronic stroke. A total of 20 individuals with chronic stroke were randomly allocated to receive either Ai Chi or conventional water-based exercise for 60 min/time, 3 times/week, and a total of 6 weeks. Balance performance assessed by limit of stability (LOS) test and Berg balance scale (BBS). Fugl-Meyer assessment (FMA) and gait performance were documented for lower extremity movement control and walking ability, respectively. Excursion and movement velocity in LOS test was significantly increased in anteroposterior axis after receiving Ai Chi (p = 0.005 for excursion, p = 0.013 for velocity) but not conventional water-based exercise. In particular, the improvement of endpoint excursion in the Ai Chi group has significant inter-group difference (p = 0.001). Both groups showed significant improvement in BBS and FMA yet the Ai Chi group demonstrated significantly better results than control group (p = 0.025). Ai Chi is feasible for balance training in stroke, and is able to improve weight shifting in anteroposterior axis, functional balance, and lower extremity control as compared to conventional water-based exercise.
Similar content being viewed by others
Introduction
Stroke is a cerebral vascular disease caused by the interruption of the blood supply to the brain, cutting off the supply of oxygen and nutrients1. Damage to the brain tissue leads to sensory, motor, cognitive, and emotional deficits. With impaired motor and sensory functions, stroke patients suffer from deficits in balance control which plays crucial role in ambulatory function and thus as an important clinical indicator2,3,4,5. Balance is defined as the ability to maintain center of mass (COM) within the stability limits, the boundaries of the base of support (BOS)6. Balance control can be quantified by limit of stability (LOS) test, expressed by movement velocity, displacement excursion, and directional control7,8. Individuals with stroke usually show decline in the abovementioned balance performance9,10,11,12. Bohannon13 noted the correlation between static standing ability and independent mobility in stroke patients (r = 0.62). Lee et al.14 found that walking velocity is associated with maximal displacement excursion in LOS test (r = 0.68, p < 0.01) and Berg balance scale (r = 0.66, p < 0.01) in patients with stroke. In addition, the balance-related fall risks should also be addressed in people with chronic stroke15,16. Therefore, it is crucial to improve balance control in order to improve the balance-related activities for individuals with stroke.
Several elements, such as strengthening, postural control, weight shifting, and agility exercise, are necessary to be incorporated during balance training17. It has also been noted that increased somatosensory inputs and visual deprivation might exert positive effects on top of balance training, as well as enriched environment4,5,18,19. Water-based exercise, by utilizing the properties of water, including buoyancy, viscosity, turbulence, and hydrostatic pressure, has been suggested to improve balance control20,21. Two reviews summarized that the water-based exercise for neurological disorder covers a wide variety, including resistance training, movement facilitation, motor control training, balance training, coordination training and other specific techniques21,22. They indicated that stroke patients improved significantly more in weight shifting ability, dynamic balance, and functional mobility as compared with the land-based intervention21,22.
Ai Chi, first developed by Jun Konno in 1990s23, is one kind of water-based exercise emphasizing characteristics of balance training24. It resembles Tai Chi on land, complemented by Zen shiatzu and Watsu concepts25. Ai Chi is composed of 16 katas (movements), including breathing, upper extremity movements, lower extremity movements, trunk control, and coordinated movements23. With the properties and advantages of water, less weight bearing is required and larger displacement can be achieved. Currently, some studies have mentioned the benefits of Ai Chi for neurological involved patients21,22. Bayraktar et al. showed positive effects of 8 weeks of Ai Chi training on muscle strength, muscle endurance, functional mobility, and fatigue severity in patients with multiple sclerosis26. Noh et al. found that the balance performance and knee flexors strength improved more in the Ai Chi combining Halliwick therapy group than the conventional physiotherapy group in patients with stroke27. Pérez-de la Cruz et al. also showed the feasibility of Ai Chi on balance and functional capacity for people with Parkinson’s disease28.
Taking together, water-based exercise is beneficial for balance performance in patients with stroke. Ai Chi is a specific water-based exercise which emphasizes the characteristics of balance control. However, whether Ai Chi can exert better effect on balance performance than conventional water-based exercise in people with stroke is not known. The aim of this study was to compare the effects of Ai Chi training with conventional water-based exercise on balance performance in people with stroke. We hypothesized that Ai Chi can result in superior effects on balance control than conventional water-based exercise people with stroke.
Results
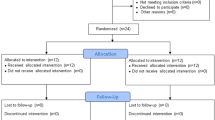
Flow of participants through the study
Recruitment occurred between April 2016 and September 2017 according to the Institutional Review Board (IRB) approval period. There were 20 participants (14 males and 6 females with mean age of 54.6 years old) provided informed consent and were randomly assigned to the experimental or the control group (n = 10 for each group) (Fig. 1). There were no significant differences in baseline demographic information between the experimental and the control groups (Tables 1 and 2).
The results of dynamic balance indicated by LOS are shown in Table 3 and Fig. 2. Within-group comparisons revealed significant improvements in endpoint excursion (EPE, p = 0.005), maximal excursion (MXE, p = 0.007), and movement velocity (MVL, p = 0.013) in anteroposterior direction after Ai Chi training (Fig. 2a,b,d). Ai Chi training also significantly improved the EPE in anteroposterior direction as compared to the control training (p = 0.001). There were no significant changes in LOS test after control training.
Regarding the functional balance performance, both groups improved significantly in Berg Balance Scale (BBS) scores after training (p = 0.005, p = 0.043, for Ai Chi and control group respectively) with no significant group difference (Table 4). However, 7 out of 10 subjects improved at least 4 points (minimal detectable change, MDC29) after Ai Chi training, while 2 out of 10 improved beyond the MDC after control training (p = 0.025 for group comparison).
The results of gait performance are shown in Table 5. Participants demonstrated significant improvement in speed (p = 0.047) and stride length (p = 0.028) after Ai Chi training while participants showed significant improvement in stride length (p = 0.009) after control training. However, there were no significant inter-group differences.
Both groups also improved significantly in lower extremity motor control as indicated by Fugl-Meyer assessment (FMA) after training (p = 0.001, p = 0.009, for Ai Chi and control group respectively), and Ai Chi group improved more than the control group (p = 0.030) (Table 6).
Discussion
Our results showed that the Ai Chi exerted superior effects on anteroposterior dynamic balance control and lower extremity motor control than conventional water-based exercise in individual with chronic stroke. The gait speed improved only after Ai Chi training. However, both Ai Chi and conventional water-based exercise improved the lower extremity motor control and BBS. We also noted that no drop-out or adverse event reported during the 6-week study period for both groups.
Ai Chi is one of the special of aquatic exercise resembling Tai Chi on land, however, its effects on balance has not yet been established in people with stroke. In this study, we demonstrated that Ai Chi exerted significant effects on center of gravity (COG) displacement, especially in anteroposterior direction, and such significant effects cannot be achieved by conventional aquatic exercise. Two displacements of COG were recorded during LOS test, EPE and MXE. As EPE is the distance travelled at the first attempt which might indicate the confident excursion of the subjects. MXE, on the other hand, is the maximal result after several attempts. Therefore, the difference between EPE and MXE may imply the strategy and confidence of the subjects during the COG displacement. In current study, improvement of EPE was greater than MXE in anteroposterior direction after Ai Chi training, which may indicate that the confidence for further anteroposterior displacement has been established. According to Eng et al., improvement in COG control along the anteroposterior axis was more difficult than in lateral axis for individuals with chronic stroke30. In addition, the anteroposterior margin of stability is critical when the post-stroke patients walk or encounter destabilizing environment31. Taken together, the increased anteroposterior excursion after Ai Chi training may result in faster walking speed as demonstrated in the present study.
Though BBS could be improved by both water-based programs, only Ai Chi group has reached MDC significantly. Since BBS is a clinical test, reaching MDC suggested “true” change after intervention32. Balance impairment is one of the most important internal factor associated with fall33, and BBS is a sensitive measure for identifying not only balance deficits but also fall risk34. We further analyzed the decrease of fall risk in both groups. The latest cutoff score of increased fall risk in stroke was set at 49 (score ≦ 49)35. Changing from BBS ≦ 49 to BBS > 49 indicated the decreased fall risk. At baseline, 8 people in Ai Chi group had higher fall risk and 5 (62%) of them decreased the fall risk. On the other hand, 5 people had higher fall risk in control group, but only 1 (20%) has decreased the fall risk.
Our results of both group were in line with previous studies stating that aquatic therapy can improve lower extremity function and balance performance27,36. However, our results further indicated that Ai Chi was more effective than conventional water-based exercise in these two outcome measures. Such superior effect of Ai Chi may be due to that Ai Chi incorporated closed kinetic chain (CKC) exercise while conventional exercise often used open kinetic chain (OKC) movements. CKC exercise involved multiple joints and coordinated muscles while OCK usually isolated one joint. Lower extremity CKC exercise facilitated ankle neuromuscular control superiorly and appeared to be more effective in weight translation37. Integrating feedback from the entire lower extremity and further simulating the mechanoreceptors were documented during CKC exercise38. With foot sole fixed on the ground, CKC exercise required much eccentric control of knee extensor, as well as calf muscle stretching. Consequently, tibialis anterior could generate higher force and work more effectively39. The above mentioned benefits of CKC exercise may explain the superior effects of Ai Chi to conventional water-based exercise in present study.
Another noticeable difference was the movement composition. Conventional water-based exercise was impairment-emphasizing using single-joint movement to focus on exercising the affected limbs, while Ai Chi involved coordinated movements. In particular, the second half movements of Ai Chi (movement #7 to #16) were classified as total coordinated body movements23. Four of which (movement #10 to #13) highlighted weight shifting in the anteroposterior axis, and only two, sideways (movement #9, #14). The rest of the movements (movement #8, #9, #15) stressed axial rotation of the trunk and whole body. The last movement (movement #16) required more advanced control of the trunk and lower extremity. Albeit these movements, a diversity of positions were embedded during Ai Chi exercise, including static wide stance, lunge, one-leg stance, and ultimately dynamic crossing steps and jump-landing. Transition of positions was accounted for effective balance training17. It was consistent with our results where weight translation has improved significantly in Ai Chi group, particularly in anteroposterior axis, and the result was transferred to the improved BBS.
In this study, we further noted that the directional control (DCL) did not improve as the MXE and EPE did after Ai Chi training. According to motor learning principles, a movement consolidation includes kinematic accuracy, which was acquainted from a large scale of trials and errors, and the later-coming dynamic effects of force40. We thus speculated that the increased displacement (indicated by MXE and EPE) but not the kinematic accuracy (indicated by DCL) may reflect the movement consolidation has not yet been established.
The small sample size of our study is one of the limitations. A larger randomized controlled clinical trial is needed to validate the reported benefits of the Ai Chi intervention for people with chronic stroke. Despite the small sample size, the effect size is relatively strong for our outcomes (LOS: 0.57; BBS: 0.44)41. In addition, the therapist was not blinded to the exercise group and, although unavoidable, this limitation may introduce bias. Also, the decrease in fall risk was reported according to the BBS in present study, however, the reduction of fall incidence after training warrants further follow-up. Furthermore, it should be noted that both Ai Chi and conventional water-based exercise included gait training to enhance the effects of exercise training. However, this may dilute the distinction between the Ai Chi and conventional water-based exercise.
This is the first study comparing the effects of Ai Chi with conventional water-based exercise in individuals with stroke. With a total of 18 sessions in 6 weeks, Ai Chi resulted in better improvement in anteroposterior balance control and lower extremity motor function than conventional water-based exercise. Both water-based exercise improved BBS score, however, only Ai Chi training decreased the possible fall risks.
Methods
Design
This was a single-blinded (assessor blinded) randomized controlled trial with pre- and post-test. The randomization was block randomization with the block of 4 and was conducted via sealed envelope drawn by a person who was not involved in the study. Participants with chronic stroke were recruited and referred by doctors in a general hospital. Stroke diagnosis, age, gender, stroke type, lesion side, and duration of hemiparesis were obtained from patient interviews and medical chart. Subjects who met the selection criteria and consented to participate were randomly allocated to the experimental group or the control group. All participants were assessed before (pre) and after completing the 6 weeks of intervention (post) by a physical therapist who was blinded to the group allocation. Assessment included balance performance, gait performance, and motor control of lower extremity. Participants in experimental group received Ai Chi intervention and control group received conventional water-based exercise for 60 min each session, 3 sessions per week for a total of 6 weeks.
Participants provided written informed consent of study procedures approved by ethical committees at Cheng Hsin General Hospital and National Yang-Ming University. This trial was registered in 10/06/2016 at http://www.anzctr.org.au/ (ACTRN12616000769482) and conformed to the CONSORT checklist. All experiments were performed in accordance with relevant guidelines and regulations.
Participants
The inclusion criteria of this study were: (1) age between 20 and 80 years old, (2) 6 months post first-ever stroke with unilateral motor deficits, (3) ability to walk independently for at least 15 meters with or without use of walking aids, and (4) a score of >24 on the mini-mental state examination (MMSE). The exclusion criteria were: (1) receiving water-based exercise on a regular basis, (2) unstable vital signs (resting heart rate >100 bpm, body temperature >38 °C, or respiration rate >20 breaths per minute), (3) history of major cardiovascular event (such as myocardial infarction, heart failure, or endocartditis), (4) any hydrotherapy contraindication, such as pregnancy, incontinence, open wound or intubation, and (5) history of other diseases known to possibly interfere with participating the study, such as diabetic neuropathy or uncontrolled hypertension.
Outcome measures
The primary outcome of this study was balance performance including dynamic balance and functional balance. Dynamic balance was assessed by LOS test using the SMART Balance system (NeuroCom International, Inc, USA). To assess the LOS, the subject stood on the force plate and shifted his/her center of pressure (COP) to reach maximal distance in the target direction as quickly and accurately as possible without moving the feet. Four directions (forward, backward, non-affected side, and affected side) were assessed in random order. MVL, EPE, MXE, and DCL were collected during the LOS test in this study7,8. MVL is defined as the average speed in a specific direction. EPE is the distance covered in the very first attempt towards the target, expressed in percentage. MXE is defined as the farthest distance traveled by the COP during the trial. DCL is defined as the amount of movement in the intended direction minus the amount of extraneous movement. A DCL score of 100% indicates that the participant does not deviate from a straight path during the test42. The anteroposterior direction and lateral direction were calculated and reported in this study. In anteroposterior direction, displacement percentage of EPE and MXE was the sum of forward and backward direction; MVL and DCL was the average. In lateral direction, displacement percentage of EPE and MXE was the sum of affected and non-affected direction; MVL and DCL was the average. Functional balance was assessed by BBS. BBS is a valid and reliable scale containing 14 items from a sitting position to standing on one foot. Each item is scored from 1 to 4 with a highest possible total score of 56. Higher score stands for better balance performance32.
Secondary outcomes included gait performance and motor control of lower extremity. Gait performance was assessed by GAITRite system (CIR system, Inc, USA). Participants were asked to walk 3 times on a GAITRite carpet with sensors attached underneath. The carpet was 3.66 m in length and 0.61 m in width. Spatio-temporal parameters were recorded and calculated. Gait parameters of interest in this study included speed, cadence, stride length, stride time, stride length variability, stride time variability, spatial asymmetry ratio, and temporal asymmetry ratio43. Variability was expressed by standard deviation/mean ×100% to indicate the consistency of gait pattern44. Asymmetry ratio was quantified by the percentage as follows: paretic side/non-paretic side ×100%, with step length indicating for spatial parameters and step time for temporal45. The averaged value of 3 trials were used for data analysis. The lower extremity motor control was evaluated by FMA scale which has been reported with good reliability for stroke patients46,47. The total score of lower extremity part is 34, and higher score indicates better control of lower extremity. All the data were collected in Neurological Physiotherapy Lab in National Yang-Ming University.
Intervention programs
Participants in experimental group and control group received exercise program in a 120 cm deep, 35 °C therapeutic pool with therapist and participant ratio 1:2. Both groups were instructed by an experienced aquatic physiotherapist with Ai Chi certificate.
The participants in the experimental group received 16-kata Ai Chi for warm-up and main exercise. Each kata and its movement characteristics were described in Supplementary Table S1. In each session, participants practiced the first 3 katas of Ai Chi as warm-up for 15 min. Those katas were mainly for breathing control and symmetrical arm movements in wide stance position. After the warm-up exercise, participants performed 3–4 katas with 10–15 repetition for each katas as the main exercise program for 30 min. Gait training for another 15 min was administered to conclude the intervention. The difficulties of the katas progressed every week until week 5. In the last week, participants were asked to practice a full Ai Chi program as the main exercise program23. The progression was shown in Supplementary Table S2.
Participants in the control group also started a session with 15 min warm-up by practicing active movement of the affected side. The 30-min main exercise included stretching exercise and resistance exercise for affected leg. For the stretching exercise, the participants were in a sitting position in water with 90-degree hip and knee flexion and feet on the floor for 30 seconds and then stood up for 5 times. The participants were allowed to lean back on the wall or to use the handrail for balance. Resistance training included four combinations of movements: hip flexion-extension, hip abduction-adduction, knee flexion-extension, ankle dorsiflexion-plantar flexion. In hip and knee movements, a flotation aid was placed and fixed around the ankle of the affected side. Ankle movement was floatation-free. Participants were asked to exercise the affected leg as fast as they can without losing standing balance. The therapist provided verbal feedback and manual guidance as needed during movement. Each movement was practiced 10 times as one set, and the participants needed to practice 3 sets for the resistance training. During the first set of practice, the participants were asked to hold the handrail with the non-affected hand. During the second and the third set of practice, the participants were encouraged to practice without holding the handrail. Gait training for 15 min was administered to conclude the intervention.
Statistical analysis
A sample of 14 participants was calculated to be sufficient for power of 0.80 and a two-tailed alpha level of 0.05 to detect a between-group difference in BBS (effect size 1.03)27. To account for some attrition, 20 participants were recruited.
All the data was analyzed by SPSS 24.0. Descriptive statistics were generated for all variables, and distributions of variables were expressed as mean ± standard deviation or frequency. Normal distribution of outcomes cannot be confirmed by Shapiro-Wilk test, and thus inter-group difference of baseline data was analyzed by Mann-Whitney U test for continuous variables or X2 test for nominal scales. Mann-Whitney U test was used to compare the percentage of changes of outcomes between groups, and Wilcoxon signed rank test for within group comparisons. Statistical significant level was set at 0.05.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Stroke, cerebrovascular accident, http://www.who.int/topics/cerebrovascular_accident/en/ (2015).
Yoo, J., Lim, K. B., Lee, H. J. & Kwon, Y. G. Cardiovascular response during submaximal underwater treadmill exercise in stroke patients. Annals of rehabilitation medicine 38, 628–636 (2014).
de Haart, M., Geurts, A. C., Huidekoper, S. C., Fasotti, L. & van Limbeek, J. Recovery of standing balance in postacute stroke patients: a rehabilitation cohort study. Archives of physical medicine and rehabilitation 85, 886–895 (2004).
Bonan, I. V. et al. Reliance on visual information after stroke. Part I: balance on dynamic posturography1. Archives of physical medicine and rehabilitation 85, 268–273 (2004).
Bonan, I. V. et al. Reliance on visual information after stroke. Part II: effectiveness of a balance rehabilitation program with visual cue deprivation after stroke: a randomized controlled trial1. Archives of physical medicine and rehabilitation 85, 274–278 (2004).
O’Sullivan, S. B., Schmitz, T. J. & Fulk, G. Physical rehabilitation. (FA Davis, 2013).
Wallmann, H. W. Comparison of elderly nonfallers and fallers on performance measures of functional reach, sensory organization, and limits of stability. The Journals of Gerontology Series A: Biological Sciences and Medical Sciences 56, M580–M583 (2001).
Alfeeli, A. K., Alghunaim, S. M., Baqer, A. B., Shehab, D. K. & Ahmed, M. M. Postural stability and balance training program in hemiparetic stroke patients. Macedonian Journal of Medical Sciences 6, 251–254 (2013).
Goldie, P., Matyas, T., Evans, O., Galea, M. & Bach, T. Maximum voluntary weight-bearing by the affected and unaffected legs in standing following stroke. Clinical Biomechanics 11, 333–342 (1996).
Au-Yeung, S. S., Ng, J. T. & Lo, S. K. Does balance or motor impairment of limbs discriminate the ambulatory status of stroke survivors? American journal of physical medicine & rehabilitation 82, 279–283 (2003).
Dettmann, M. A., Linder, M. T. & Sepic, S. B. Relationships among walking performance, postural stability, and functional assessments of the hemiplegic patient. American journal of physical medicine 66, 77–90 (1987).
Mottet, D., Van Dokkum, L. E. H., Froger, J., Gouaïch, A. & Laffont, I. Trajectory formation principles are the same after mild or moderate stroke. PloS one 12, e0173674 (2017).
Bohannon R.W. Gait performance of hemiparetic stroke patients: selected variables. Archives of physical medincine and rehabilitation 68, 777–781 (1987).
Lee, C. C., Wang, R. Y. & Yang, Y. R. Correlations among balance and mobility measures for patients with stroke. Formosan Journal of Physical Therapy 28, 139–146 (2003).
Harris, J. E., Eng, J. J., Marigold, D. S., Tokuno, C. D. & Louis, C. L. Relationship of balance and mobility to fall incidence in people with chronic stroke. Physical therapy 85, 150–158 (2005).
Belgen, B., Beninato, M., Sullivan, P. E. & Narielwalla, K. The association of balance capacity and falls self-efficacy with history of falling in community-dwelling people with chronic stroke. Archives of physical medicine and rehabilitation 87, 554–561 (2006).
Au-Yeung, S. S., Hui-Chan, C. W. & Tang, J. C. Short-form Tai Chi improves standing balance of people with chronic stroke. Neurorehabilitation and neural repair 23, 515–522 (2009).
Bayouk, J. F., Boucher, J. P. & Leroux, A. Balance training following stroke: effects of task-oriented exercises with and without altered sensory input. International Journal of Rehabilitation Research 29, 51–59 (2006).
Pollock, A., Baer, G., Langhorne, P. & Pomeroy, V. Physiotherapy treatment approaches for the recovery of postural control and lower limb function following stroke: a systematic review. Clinical Rehabilitation 21, 395–410 (2007).
Becker, B. E. Aquatic therapy: scientific foundations and clinical rehabilitation applications. Pm&r 1, 859–872 (2009).
Iatridou, G. et al. The effectiveness of hydrokinesiotherapy on postural balance of hemiplegic patients after stroke: a systematic review and meta-analysis. Clinical rehabilitation 32, 583–593 (2018).
Marinho-Buzelli, A. R., Bonnyman, A. M. & Verrier, M. C. The effects of aquatic therapy on mobility of individuals with neurological diseases: a systematic review. Clinical rehabilitation 29, 741–751 (2015).
Sova, R. & Konno, J. Ai chi: Balance, harmony & healing. (DSL, 1999).
Covill, L. G., Utley, C. & Hochstein, C. Comparison of ai chi and impairment-based aquatic therapy for older adults with balance problems: a clinical study. Journal of geriatric physical therapy 40, 204–213 (2017).
Brody, L. T. & Geigle, P. R. Aquatic exercise for rehabilitation and training. (Human Kinetics, 2009).
Bayraktar, D. et al. Effects of Ai-Chi on balance, functional mobility, strength and fatigue in patients with multiple sclerosis: a pilot study. NeuroRehabilitation 33, 431–437 (2013).
Noh, D. K., Lim, J. Y., Shin, H. I. & Paik, N. J. The effect of aquatic therapy on postural balance and muscle strength in stroke survivors—a randomized controlled pilot trial. Clinical rehabilitation 22, 966–976 (2008).
Pérez-de la Cruz, S., Luengo, A. G. & Lambeck, J. Effects of an Ai Chi fall prevention programme for patients with Parkinson’s disease. Neurología (English Edition) 31, 176–182 (2016).
Hiengkaew, V., Jitaree, K. & Chaiyawat, P. Minimal detectable changes of the Berg Balance Scale, Fugl-Meyer Assessment Scale, Timed “Up & Go” Test, gait speeds, and 2-minute walk test in individuals with chronic stroke with different degrees of ankle plantarflexor tone. Archives of physical medicine and rehabilitation 93, 1201–1208 (2012).
Eng, J. J. et al. A community-based group exercise program for persons with chronic stroke. Medicine and science in sports and exercise 35, 1271 (2003).
Hak, L. et al. Stepping strategies used by post-stroke individuals to maintain margins of stability during walking. Clinical biomechanics 28, 1041–1048 (2013).
Blum, L. & Korner-Bitensky, N. Usefulness of the Berg Balance Scale in stroke rehabilitation: a systematic review. Physical therapy 88, 559–566 (2008).
Saverino, A., Benevolo, E., Ottonello, M., Zsirai, E. & Sessarego, P. Falls in a rehabilitation setting: functional independence and fall risk. Europa medicophysica 42, 179–184 (2006).
Maeda, N., Kato, J. & Shimada, T. Predicting the probability for fall incidence in stroke patients using the Berg Balance Scale. Journal of International Medical Research 37, 697–704 (2009).
Patterson, K. K., Inness, E., McIlroy, W. E. & Mansfield, A. A retrospective analysis of post-stroke Berg Balance Scale scores: how should normal and at-risk scores be interpreted? Physiotherapy Canada 69, 142–149 (2017).
Chu, K. S. et al. Water-based exercise for cardiovascular fitness in people with chronic stroke: a randomized controlled trial1. Archives of physical medicine and rehabilitation 85, 870–874 (2004).
Kwon, Y. J., Park, S. J., Jefferson, J. & Kim, K. The effect of open and closed kinetic chain exercises on dynamic balance ability of normal healthy adults. Journal of physical therapy science 25, 671–674 (2013).
Wilk, K. E. et al. A comparison of tibiofemoral joint forces and electromyographic activit during open and closed kinetic chain exercises. The American journal of sports medicine 24, 518–527 (1996).
Lee, N. K. et al. The effects of closed and open kinetic chain exercises on lower limb muscle activity and balance in stroke survivors. NeuroRehabilitation 33, 177–183 (2013).
Krakauer, J. W., Ghilardi, M. F. & Ghez, C. Independent learning of internal models for kinematic and dynamic control of reaching. Nature neuroscience 2, 1026 (1999).
Coolican, H. Research methods and statistics in psychology. (Psychology Press, 2017).
Liston, R. A. & Brouwer, B. J. Reliability and validity of measures obtained from stroke patients using the Balance Master. Archives of physical medicine and rehabilitation 77, 425–430 (1996).
Hsu, A. L., Tang, P. F. & Jan, M. H. Analysis of impairments influencing gait velocity and asymmetry of hemiplegic patients after mild to moderate stroke1. Archives of physical medicine and rehabilitation 84, 1185–1193 (2003).
Wang, R. Y. et al. Effects of combined exercise on gait variability in community-dwelling older adults. Age 37, 40 (2015).
Wonsetler, E. C. & Bowden, M. G. A systematic review of mechanisms of gait speed change post-stroke. Part 1: spatiotemporal parameters and asymmetry ratios. Topics in stroke rehabilitation 24, 435–446 (2017).
Sanford, J., Moreland, J., Swanson, L. R., Stratford, P. W. & Gowland, C. Reliability of the Fugl-Meyer assessment for testing motor performance in patients following stroke. Physical therapy 73, 447–454 (1993).
Pandian, S., Arya, K. N. & Kumar, D. Minimal clinically important difference of the lower-extremity fugl–meyer assessment in chronic-stroke. Topics in stroke rehabilitation 23, 233–239 (2016).
Acknowledgements
This work was supported by grants from the Ministry of Science and Technology (MOST 106-2314-B-010-040-MY3) and Cheng Hsin General Hospital (Grant No. CY10702).
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments: P.H.K., R.Y.W. Performed the experiments: P.H.K. Analyzed the data: P.H.K. Wrote the paper: P.H.K., R.Y.W., S.F.C., Y.R.Y., T.C.L. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ku, PH., Chen, SF., Yang, YR. et al. The effects of Ai Chi for balance in individuals with chronic stroke: a randomized controlled trial. Sci Rep 10, 1201 (2020). https://doi.org/10.1038/s41598-020-58098-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-58098-0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.