Abstract
Traditionally, the study of anatomy in insects has been based on dissection techniques. Micro-computed tomography (micro-CT) is an X-ray based technique that allows visualization of the internal anatomy of insects in situ and does not require dissections. We report on the use of micro-CT scans to study, in detail, the internal structures and organs of the coffee berry borer (Hypothenemus hampei), the most damaging insect pest of coffee worldwide. Detailed images and videos allowed us to make the first description of the aedeagus and the first report of differences between the sexes based on internal anatomy (flight musculature, midgut shape, hindgut convolutions, brain shape and size) and external morphology (lateral outline of the pronotum and number of abdominal tergites). This study is the first complete micro-CT reconstruction of the anatomy of an insect and is also the smallest insect to have been evaluated in this way. High quality rendered images, and additional supplementary videos and 3D models are suitable for use with mobile devices and are useful tools for future research and as teaching aids.
Similar content being viewed by others
Introduction
Ever since the introduction of coffee from Africa to various continents, it has become an important commodity not only to producing countries, but also to billions of consumers that take delight on its flavour, aroma, and stimulatory effects1. Amongst the many agronomic problems faced by coffee producers, insect pests and plant pathogens are of paramount importance2. Climate change is also posing an enormous challenge to the continuity of commercial coffee production and to the survival of wild coffee species3,4.
The coffee berry borer, Hypothenemus hampei (Ferrari) (Coleoptera: Curculionidae: Scolytinae: Cryphalini), is the most economically important insect pest of coffee due to its cryptic life cycle inside the coffee berry, which makes it quite hard to manage5. The insect is currently present in most coffee producing countries5 and worldwide losses are likely over US$500 million, with yearly losses in Brazil alone estimated at US$215–358 million6. The insect has also been reported to have infested vast areas, e.g., over 715,000 ha in Colombia by 19987 and more than 800,000 ha by 20028. Even though > 1,800 papers have been published on the coffee berry borer9, knowledge about its internal anatomy is restricted to a handful of papers that use dissection techniques10,11,12,13,14.
Insect dissection techniques were first used more than 400 years ago by Aldrovandi15 and Malpighi16. Even though dissections have most definitely been useful, resulting in thousands of papers on the internal anatomy of insects, they result in an inevitable distortion of the internal structures and organs. A relatively new technique based on X-rays, known as micro-computed tomography (micro-CT), allows visualization of the insect’s internal anatomy in situ, without the need for dissection; results have been validated by comparing them with classical destructive methodologies17,18.
We present a detailed study of all the anatomical structures and organs in the coffee berry borer using micro-CT, with the exception of the respiratory system, which is presented separately19. This is in contrast to other insect-related micro-CT papers that focus on specific aspects such as cephalic anatomy20, metamorphosis21,22, tracheal systems23,24,25,26,27,28, mycetangia29,30,31, mandibular muscles32, the alimentary canal33, respiratory volume34, genitalia35 and the brain36. Together with the study on the respiratory system19, this paper represents the first holistic view of the entire anatomy of the coffee berry borer, and to the best of our knowledge, is the smallest insect to have been subjected to micro-CT.
Results
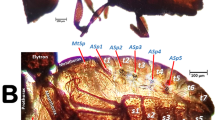
Previously unreported external differences between the sexes were found: males had eight abdominal tergites (Fig. 1A) and, in lateral view, the outline of the pronotal disc appeared uniformly curved (Fig. 1B); the females had seven abdominal tergites (Fig. 1C) and the outline of the pronotal disc had a slight concave depression (Fig. 1D).
Left dorso-lateral views of a male (A) and a female (C) coffee berry borer. Elytra have been removed using software thereby revealing the dorsal part of the thoracic and abdominal segments. Abdominal tergites (t) and the position of the abdominal respiratory spiracles (ASp) are labelled sequentially (terminology after Hopkins39). Insets (B,D) show a uniformly curved pronotal disc in males (B) in contrast to a concave depression on the posterior pronotal third in females (D; blue arrows). Note different scales were used for each sex. The meso- and metanotum were removed by software to observe the reduced flight muscles in the male (E) in contrast to conspicuous flight muscles in the female (F).
The general arrangement of the internal structures and organs of both sexes can be seen in Figs. 2,3, and Supplementary Videos S1, S9. The digestive system, including the excretory system, are shown in Figs. 4, 5, and Supplementary Videos S1–S3, S9. Differences between the sexes in the shape of the midgut and the trajectories of the convolutions that make up the midgut and hindgut are shown in Fig. 4G,H, and Supplementary Video S2. Detailed views of the external and internal structures of the proventriculus, including the musculature are shown in Figs. 6, 7. Light microscopy views are included (Fig. 6G,H) for comparison with the micro-CT reconstructions (Fig. 6A–F; Supplementary Videos S3, S9).
Alimentary canal of a male (A,B,C) and a female coffee berry borer (D,E,F): right-lateral (A,D), ventral (B,E), and dorsal (C,F) views. Red arrows indicate the rectal ampulla. Lateral views of the male (G) and female (H) alimentary canals with the trajectories and convolutions of the midgut and hindgut shown in yellow. Each time the digestive tracts began a new trajectory curve, the new tract is marked with an incremental number. Anterior-transversal (I) and sagittal (J) cuts close to the stomodaeal valve of the midgut reveal the internal papillae.
Right-sagittal view of the internal anatomy of the female, showing the main musculature, dorsal vessel and nervous system (A), and a dorsal view of the digestive and nervous systems (B). Dorsal (C) and right-lateral (D) views of a male focusing on the positioning of the nervous system in relation to the digestive system.
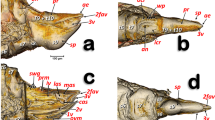
Details of the proventriculus: micro-CT volume-rendered images (A–F) and light microscopy photographs (G,H). Right-lateral view (A,G); posterior view showing the stomodaeal valve (=cardiac sphincter) (B). Progressive transverse slices (thickness ca. 20 µm) from the basal to the apical zone, each marked with arrows numbered 1 to 3 (A,C,D,E); sagittal mid-slice (F); postero-lateral view (H). Most of the nomenclature after Eaton50.
Micro-CT volume rendered images of the internal structures of the proventriculus from different perspective views: right-lateral (A,B,C), right-antero-posterior (D), anterior (E), posterior (F), left antero-posterior (G) and right-postero-anterior (H). Different perspectives of the grinding sclerotized complex isolated (E–H). A: a superficial window was opened, and the grinding sclerotized complex removed; B,D: as A but including the grinding complex; C: as B but the grinding complex has been superficially sagittally cut, eliminating one side of the octagonal structure.
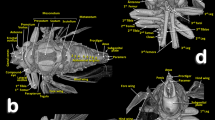
The general internal anatomy of an adult female (with organs and muscles) is shown in Fig. 5A, with the dorsal vessel, aorta, and the nervous system highlighted in different colours. The nervous system and its positional relationship with the digestive system is shown in Fig. 5, and Supplementary Videos S4, S9. Detailed views of the nervous system in both sexes are shown in Figs. 8, 9. The female and male reproductive systems are shown in Fig. 10 and Supplementary Video S5. A study of the aedeagus is shown in Fig. 11 and Supplementary Videos S6, S7.
Nervous systems of a female (A,B) and a male (C,D) coffee berry borer, showing the main ganglia and nerves (A,C: dorsal; B,D: ventral). The abdominal nerves are not shown in the male. The nerve insertions in the thoracic-abdominal-complex ganglion are indicated with blue arrows. Terminology after Atkins and Chapman75 and Snodgrass45.
Lateral views of the nervous system of a female (A) and a male (B) coffee berry borer allowing comparison of the relative size of the central nervous system in relation to the head. Details in frontal (C,E) and latero-frontal (D,F) views of the central nervous system of a female (C,D) and a male (E,F) focusing on the differences between sexes.
Female (A–E) and male (F–I) coffee berry borer reproductive systems, in latero-dorsal (A,C), lateral (E,G) and dorsal (B,D,F,H) views. Details of the distal part of the spermatheca, with the spermathecal muscles and the spermatic gland (D). Figures (C,E,H and I) show the positions of the insect corresponding to figures (A,B,F and G), respectively.
Rendered images of different views of the aedeagus: left-lateral (A,E); left-latero-dorsal (B,F); dorsal (C,G); and ventral (D,H). Images at the bottom have been rendered transparent by software to enhance the view of internal structures. Abbreviations: ad = apical denticles (titillators); lpa = left parameres; rpa = right parameres; ml = median lobe (‘penis’); pa = paramere apophysys; rpa = right paramere; sd = subapical dorsal denticles; ti = titillators. Terminology after Lindroth and Palmen70.
Discussion
The presence of eight tergites in the male has been described previously7,8 (Fig. 1A) but the presence of only seven in the female is new (Fig. 1B). Nüsslin37 stated that Hypothenemus (Cryphalini) females had eight tergites while Wood38 stated in reference to the 8th tergite that “In all other scolytids and most curculionids it is of reduced size, lacks pubescence and is telescoped beneath and hidden by tergum 7”; this has been reported for various bark beetle species38,39,40,41. As part of this study, we dissected ten females reared in artificial diet and did not find a covered/obscured 8th tergite. Therefore, it is possible that, either there is variation in the number of tergites amongst the 181 species of Hypothenemus5, or the 7th and 8th tergites are fused, as was reported in Conophthorus (Scolytini)41. Interestingly, there has been one report of a male coffee berry borer reared on artificial diet that had only seven tergites (Fig. 2C in Vega et al.42). However, in our study, we dissected ten males from the same colony and all of them had eight tergites.
A previously unrecorded external characteristic that varied between the sexes in our study occurred in lateral view, i.e., the outline of the pronotal disc in males was uniformly curved (Fig. 1B) while in females it had a slight concave depression (Fig. 1D). As has been previously described, coffee berry borer males are smaller than females, have vestigial wings5,42 and rudimentary compound eyes43 (Figs. 1, 8, 9E,F; Supplementary Videos S1, S4). Other characteristics that vary between sexes in bark and ambrosia beetles are discussed by Kirkendall et al.44 and, for Hypothenemus in particular, by Wood38 and Vega et al.5.
The alimentary canal, from mouth to anus, consisted of three main parts: foregut (stomodaeum), midgut (mesenteron) and hindgut (proctodeum). In contrast to the midgut, the foregut and the hindgut were ectodermic derivates and chitinized. The foregut was comprised of the preoral cavity, the mouthparts (where food is masticated), the pharynx, and the esophagus, which was enlarged posteriorly into the crop (where food is temporarily stored before entering the proventriculus, a gizzard-like organ located under the prothorax and whose main function is to grind and filter food particles). From there, food enters the anterior midgut (also referred to as stomach and ventriculus) and eventually the hindgut. Waste is excreted through the posterior opening of the alimentary canal (i.e., the anus). The insertion of Malpighian tubules was coincident with the limit between the mesodeum (midgut) and the proctodeum (hindgut) separation, as described by others45,46,47,48.
The pharynx had chitinized ridges, where the pharynx dilator muscles were inserted, and opened into the preoral cavity. The preoral cavity ran across the head, below the brain (Figs. 2, 3, 5; Supplementary Videos S1, S2), through the nervous system (Fig. 5; Supplementary Videos S4, S9), connecting with the crop via a short esophagus. The crop was comprised of two contiguous cavities, one small anterior one and another more voluminous one connected to the proventriculus (Figs. 2, 3, 4, 5, 6A; Supplementary Videos S2, S9).
The proventriculus (Fig. 6, Supplementary Videos S3, S9) was an octagon-shaped organ and once thought to have taxonomic value in Scolytoidea48,49,50,51. A series of outer band muscles surrounded eight heavily sclerotized units (Figs. 6, 7), as previously described by Eaton50 for Ips mexicanus (=radiatae) (Hopkins); these muscles exert a peristaltic function (contraction and relaxation) that grinds and moves food particles through the proventriculus. Another band of inner muscles also have a peristaltic function on the grinding structures, formed by eight plates that appeared as a whole and in cross section with a starry arrangement (Figs. 6C–E, 7E,F). Each of these were formed in two parts, an anterior plate and a posterior plate. Internally these plates formed masticatory teeth, which triturated food particles. The presence of long ‘saber-like bristles’ and masticatory brush-teeth also assist in food grinding and filtering (Figs. 6C–H, 7; Supplementary Videos S2, S3). The general structure agreed with Calders’s52 ‘Type 7’ Scolytinae.
Eaton50 described the grinding-straining mechanism of food moving through the proventriculus of I. mexicanus as follows: “Its passage through the proventriculus is probably restricted to the side channels formed by the rows of lamellae, since the obstructing brushes extending into the lumen would prevent direct movement of the food material through the central opening” and indicated this probable passage of food in Fig. 8 (p. 46). However, in our study of the coffee berry borer, it could be clearly seen that the lumen was filled with food particles while the spaces marked by Eaton50 as possible passageways for food, were devoid of food particles (Fig. 6C–F).
From the proventriculus the food moves through the cardiac sphincter (also known as the cardiac or stomodaeal valve; Fig. 6B–F) to the anterior midgut. There were differences between the sexes in the anterior midgut. In the male coffee berry borer, the anterior midgut was much dilated anteriorly (Figs. 2B, 4A–C,G; Supplementary Video S2), which was not apparent in the female (Figs. 3B, 4D–F,H; Supplementary Videos S2, S9). After scanning eight females and two males, it was clear that males had a conspicuously dilated midgut that was not present in females and this was not dependent on how full the cavity was with food. Moreover, we observed that the female (but not in the male) had short papillae covering the entire surface of the anterior part of the midgut, close to the stomodaeal valve; these were also present on 2/3 of the ventral surface (Fig. 4I,J). This arrangement of papillae has not been described previously in curculionids52.
The midgut has no chitinous lining and is the primary site for secretion of digestive enzymes into the lumen and for absorption of nutrients53, although it should be noted that salivary enzymes (e.g., amylases) secreted in the foregut can also initiate digestive action54. There is considerable variation in the pH of the foregut, midgut and hindgut of Coleopteran species53, with the midgut and hindgut of the coffee berry borer having an acidic pH (4.5–5.2)55. Aggregation pheromones in Dendroctonus and Ips are also produced in the midgut56.
The transit of food through the alimentary canal in the coffee berry borer takes ca. 24h57. The anterior midgut of the coffee berry borer has a gradient in oxygen content, with a microaerophilic region close to the surface (0–300 µm depth) followed by an anaerobic region (400–600 µm depth)57. This gradient allows for the activity of oxygen-dependent and anaerobic enzymes. For example, N-demethylases (involved in caffeine metabolism in the coffee berry borer) are oxygen-dependent58, while the fermentation of polysaccharides such as mannan in the coffee bean5,59,60, is an anaerobic process61.
The posterior midgut had two diverticula (right and left), also known as gastric caeca (Figs. 2–4, 5B; Supplementary Videos S2, S9); these increase the surface area of the midgut and are involved in water absorption, digestive activity, and uptake of nutrients54.
Insertion of the Malpighian tubules is recognized as the beginning of the hindgut45,46,47,62. Insertion of six Malpighian tubules has been reported in other Scolytinae51,63,64,65,66,67 with the distal portion of the Malpighian tubules embedded in the tissues surrounding the rectum65,66,67. However, after exhaustive visualization of these tubules in both sexes of the coffee berry borer, we only observed four long Malpighian tubules, in contrast with the six reported previously11. They appeared to be folded several times, going forwards and backwards (Figs. 2, 3, 4A–F, 5C,D; Supplementary Videos S1, S2, S4, S9); the distal portions, which were situated at the level of the anterior midgut (stomach), did not surround the rectum. The distal portions are immersed in haemolymph and serve a urinary function (i.e., as excretory organs that remove nitrogenous wastes, mainly uric acid) and also balance water and salt levels in the haemolymph. The anterior part of the hindgut is also referred to as the ileum or intestinum, and the posterior part in some insects is divided into colon and rectum (these three parts are also known as pylorus, ileum and rectum47, and are responsible for absorption of solutes such as amino acids, salts and water46,47,68. The rectum is the most dorsal part of the digestive system, and was slightly displaced to the right side, running backwards to open externally at the anus (Figs. 2–4, 5B–D; Supplementary Videos S1, S2, S4, S9).
It has been reported that the length of the alimentary canal in Dendroctonus armandi is almost three times the length of the insect67. In the coffee berry borer, the length of the alimentary canal was 2.82 mm for the male and 3.48 mm for the female; the ratio of the length of the alimentary canal to the body of the insect was 2.9 and 2.3, for the male and female, respectively. Moreover, we observed additional differences between the sexes in the number of trajectories of the midgut and hindgut (based on the number of times the alimentary canal began a new trajectory curve). We observed nine different trajectories in the male compared with eight in the female (Fig. 4G,H; Supplementary Video S2). Males have a shorter body length than females, suggesting that additional folding of the tubes of mid- and hindgut was necessary to fit into the smaller space. In both sexes a rectal ampulla was connected to the final tract of the rectum (Figs. 2–3, 4A–F; Supplementary Videos S1, S2); in other Coleoptera this ampulla has a defensive function via secretions53.
The male reproductive system (Figs. 2, 10F–I, 11; Supplementary Videos S1, S5) was comprised of two testes touching medially and positioned above the midgut (containing lobular testis follicles where spermatozoa are formed46,47). Each testis had a vas deferens and a single elongated accessory gland, in contrast to three pairs of accessory glands per pair of testes in D. monticolae69. Accessory glands are involved in the production of seminal fluids that mix with sperm to form the ejaculate;45 the vas deferens dilates to form a small seminal vesicle for storage prior to ejaculation47. The vasa deferentia on both sides meet to form an ejaculatory duct that is connected to the aedeagus.
The structure of the aedeagus in insects was studied in-depth by Snodgrass45 and updated for Coleoptera by Lindroth and Palmen70. The aedeagus is an important taxonomical characteristic used for species identification of insects in general and is of particular relevance in studies of Coleoptera71 including bark beetles72. However, to the best of our knowledge, there has been no previous detailed description of the aedeagus in the coffee berry borer. There are only draft schematic drawings and not detailed photographs10,11. The micro-CT rendered images of the aedeagus of the coffee berry borer (Figs. 2A,B, 10F–I, 11; Supplementary Videos S6, S7) show two lateral lobes (parameres) and a medial lobe (the intromitting structure, the penis) between them, with a rounded distal part opening called the gonopore. The distal extremity of the aedeagus is armed with apical spines and subapical-dorsal denticles that, as a whole, form anchoring structures (known as titillators45) helping ensure the sexes remain connected during copulation. The general structure of the aedeagus corresponds to the vaginate type described by Lindroth and Palmen70 where the parameres are elongated, forming a dorsal channel through which the penis moves.
The female reproductive system (Figs. 3, 10A–E; Supplementary Videos S1, S5) is comprised of two ovaries (located at each side of the body, slightly beneath the hindgut), each containing two ovarioles. Each ovariole had a terminal filament forming the suspensory apparatus of the mature ovary;45 a germarium in which oocytes are produced from oogonia; a vitellarium in which yolk is deposited into the oocytes through a membranous tube;47 and the ovariole pedicel that enlarges to form the calyx for the reception of eggs45. From each calyx emerged a short lateral oviduct; both lateral oviducts joined to form the common oviduct leading to the female gonopore. Close to the end of the common oviduct, on the right side, in a dorsal position was an ampulla-shaped accessory gland; between the accessory glands (very close to each other) was a hook-shaped spermatheca. The micro-CT rendered images obtained here were totally coincident with light microscopic images obtained following dissection10,11,14. Details of the hooked distal part of the spermatheca, spermatic gland, and the spermathecal muscles described recently14 were enhanced using micro-CT (Fig. 10D; Supplementary Video S8). Román-Ruiz et al.14 first photographed and described the spermatic gland as a “cell agglomeration on the curved distal end of the spermatheca”, but they added that “a further study will be necessary to confirm this finding”. The micro-CT reconstructions allowed us to visualize the structure previously identified as the spermatic gland (Fig. 10A,B,D; Supplementary Video S8) thus confirming its existence and structure.
The circulatory system in insects is responsible for the movement of haemolymph into the haemocoel spaces where organs are immersed. The haemolymph enters various contractile chambers of a dorsal vessel (heart) and is pumped forwards through an anterior aorta. This is what it is considered as an open circulatory system. The circulatory system does not have a key role in the transport of gases as this is achieved by an aero-vascular system formed by a complex of tubes (tracheal system)45,46,47,73. In the coffee berry borer, as in other insects, the circulatory system is quite simple in contrast with the complexity of the tracheal respiratory system19. The dorsal vessel was segmented in the female; it was in a medial position within the abdomen, just below the tergites. The dorsal vessel had three conspicuous dilated chambers beneath tergites 4–6 which were prolonged anteriorly and posteriorly by aortic vessels.
When observing the musculature (Figs. 1E,F, 2, 3, 5A), it is noteworthy that the dorso-longitudinal and dorso-ventral flight muscles were clearly visible in the female (Figs. 1F, 3A, 5A) and absent in the male (Figs. 1E, 2A). This is likely to be because males have lost their ability to fly as they have vestigial wings, spending their entire life inside the coffee berry5,42. The micro-CT rendered images of the flight muscles are completely comparable to, and actually clearer, than those previously published from dissected insects13. Various muscles connected to the pharynx and mouthparts were clearly visible inside the head (mandibular, pharyngeal, maxillary and labial muscles; Figs. 2A, 5A). Head flexor and extensor muscles (Fig. 5A) were clearly visible in the pronotum. While we do not describe here every muscle visible in the micro-CT rendered images and supplementary videos, it will be the subject of a future paper.
The anatomy of the nervous system of insects was described extensively by Snodgrass45 and summarized and updated by Grassé74. There have been several studies on the anatomy of the nervous system in different families and species of Coleoptera53,74. To our knowledge, despite the extensive revision by Calder52, the most detailed anatomical studies of the nervous system of bark beetles is that of D. pseudotsugae by Atkins and Chapman75, and our own study of the central nervous system of the coffee berry borer (Figs. 2A, 3A, 5,8,9; Supplementary Videos S1, S4, S9); both showed similar general patterns of organisation.
In the coffee berry borer, the brain (Figs. 2A, 3A, 5), located above the anterior end of the foregut, was connected to a ventral cord by two circumpharyngeal connectives (Figs. 5, 8, 9A,B). Most of the ganglions in the ventral cord were fused, so it was only possible to distinguish a subesophageal ganglion (located beneath the esophagus in a ventral posterior cephalic position; Figs. 5, 8, 9A,B,D,F) and a large thoracic-abdominal complex ganglion (in a ventral thoracic position just below the proventriculus; Figs. 2A, 3A, 5, 8, 9A,B,D–F). Other Scolytinae species have a prothoracic separated ganglion52,75. In other species, the connectives linking the brain to the ventral cord are known as circumesophageal connectives45,46,47,75. However, it is clear that, in the coffee berry borer, these connectives surround the pharynx and not the esophagus (Figs. 2A, 3A, 5; Supplementary Videos S4, S9). For this reason, we describe them as circumpharyngeal connectives.
Micro-CT also allowed us to visualize different pairs of nerves including optic (to innervate eyes) and antennal (to innervate antennae) nerves in the brain; and mandibular nerves originating in the subesophageal ganglion (to innervate the mandibles) (Figs. 5A, 8A,B,D,9A,B,D). Nerve insertions into the thoracic-abdominal-complex ganglion and long abdominal nerves were also clearly visible (Figs. 8A–C, 9E); together, these innervate the wings, legs and abdominal muscles. Note that the abdominal nerves in the male were not segmented and have not been included in figures. Details of the different ganglia, such as the frontal ganglion (situated ventral to the brain) (Fig. 8D) were also visualized (Figs. 8, 9).
Sexual differences in the nervous system of insects has been reported, both in morphology, size and in the structure of the neural connections, e.g., in social insects76 and flies77. In the coffee berry borer, there were conspicuous differences both in shape, size and relationship to the head of the nervous system, between the female and the male. The female had a larger, more laterally extended brain lobes, and thicker optic lobes (Fig. 9C,D) than the male (Fig. 9E,F). In relation to the size of the head, the brain of the female is smaller (Figs. 3A, 9A) than the male brain (Figs. 2A, 9B). The smaller brain and thinner optic nerves observed in the male is related to the biology of the species; males remain inside the coffee berry and have rudimentary compound eyes, vestigial wings, and flight muscles, as discussed above.
Conclusions
The use of micro-CT to elucidate the anatomical structures and organs of the coffee berry borer has facilitated a complete reconstruction of the anatomy of the insect, revealing the actual position of internal structures and organs without the distortions encountered using dissection methods. The technique has also allowed us to obtain detailed rendered images and videos of the aedeagus, which we used to describe it for the first time. This study is the first complete micro-CT reconstruction of the anatomy of an insect and is also the smallest insect to have been evaluated in this way. Moreover, we report previously unreported differences between the sexes with respect to both external morphology (lateral outline of the pronotum and number of abdominal tergites), and internal anatomy (flight musculature, midgut shape, hindgut convolutions and the shape and size of the brain). We have included supplementary videos and a 3D model that is suitable for use with mobile devices and could be a useful tool for scientists working on other insects as well as a teaching aid.
Methods
Insects
Coffee berry borers were reared in artificial diet at the USDA, ARS, Beltsville laboratory78.
Micro-CT scans
The specimens were killed by submerging them in 30% ethanol for 25 minutes. They were then dehydrated in an ethanol series (50%, 70%, 80%, 90%; 25 minutes in each concentration), and stained in 1% iodine in absolute ethanol for three days. Stained insects were placed in a well and submerged in hexamethyldisilazane for 4 h and air-dried overnight. For the scans, specimens were glued with cyanoacrylate to the tip of a nylon fishing line 200 μm in diameter as described by Alba-Tercedor79 and scanned using a Bruker SkyScan 1172 microtomograph (Bruker-micro CT, Kontich, Belgium) with a Hamamatsu L702 X-ray source and a Ximea 11 megapixels camera. The setting parameters were as follows: voltage = 49 kV; current = 64 µA; isotropic voxel size = 0.52 µm; image rotation step = 0.5° for the male and 0.4° for the female; 360° of rotation scan, and no filter, resulting in a scan duration of 2 h: 47 min: 55 s and 722 × -ray images for the male, and 3 h: 29 min: 39 s and 902 × -ray images for the female.
Image reconstruction, measurements and supplementary videos
Bruker micro-CT’s Skyscan software (NRecon, DataViewer, CTAnalyser) was used for primary reconstructions and the ‘cleaning’ process to obtain datasets for ‘slices’ as described previously by Alba-Tercedor79. Volume-rendered images and Supplementary Videos S1–S8 were obtained using Amira’s software, v. 6.7.0 (Thermo Fisher Scientific, Waltham, MA)80,81. The built-in ‘volrenRed.col’ colour filter was selected to obtain volume-rendered reconstructions in Figs. 1A,C, 6A–F, 7, 10. Different anatomical parts were independently segmented to finally obtain the rendered colorized images of Figs. 2–10. To be able to obtain the actual texture of structures in desired colours, after segmentation, each structure was subjected to the following arithmetic operation: A*(B > 0), where A represents the whole animal and B the segmented structure. The total length of the alimentary canal was measured manually from the oral opening to the anus using Amira’s measuring tool. Figs. 4I,J, Supplementary Video S9 and Supplementary 3D models S10 and S11 for both sexes were obtained using CTVox (a Bruker micro-CT’s Skyscan software). Colours were obtained by adjusting the transfer function curves in accordance with the transparency of the structures to X-ray, as described by Alba-Tercedor79.
Light microscopy study of the proventriculus
A female was cleared by submersion in 10% aqueous KOH for 48 h at room temperature, followed by dissection and subsequent mounting on a slide in a variation of Hoyer’s liquid82. Figure 6G,H were obtained using a Samsung Note 8 smartphone connected to the ocular of an Olympus CH-2 binocular microscope.
Data availability
The datasets generated and analyzed during the course of the study are available from J.A.T. upon reasonable request.
Change history
24 December 2019
An amendment to this paper has been published and can be accessed via a link at the top of the paper.
28 January 2021
An amendment to this paper has been published and can be accessed via a link at the top of the paper.
References
Vega, F. E. The rise of coffee. Am. Sci. 96, 138–145 (2008).
Wintgens, J. N. (Ed.). Coffee: Growing, Processing, Sustainable Production.Weinheim: Wiley-VCH (2004).
Ziska, L. H., et al. Climate change, carbon dioxide, and pest biology, managing the future: Coffee as a case study. Agronomy 8, 152 (2018).
Davis, A. P. et al. High extinction risk for wild coffee species and implications for coffee sector sustainability. Sci. Adv. 5, eaav3473 (2019).
Vega, F. E., Infante, F. & Johnson, A. J. The genus Hypothenemus, with emphasis on H. hampei, the coffee berry borer. In: Bark Beetles: Biology and Ecology of Native and Invasive Species (F. E., Vega and R. W., Hofstetter, Eds), pp. 427–494. San Diego: Academic Press (2015).
Oliveira, C. M., Auad, A. M., Mendes, S. M. & Frizzas, M. R. Economic impact of exotic insect pests in Brazilian agriculture. J. Appl. Entomol. 137, 1–15 (2013).
Duque O., H. Economics of coffee berry borer (Hypothenemus hampei) in Colombia. In “Workshop, Coffee Berry Borer: New Approaches to Integrated Pest Management”, Mississippi State University, Starkville, Mississippi, May 1–5, 2000,14 pp. (2000).
Duque-Orrego, H., Márquez-Q., A. & Hernández-S., M. (2002) Estudios de caso sobre costos de manejo integrado de la broca del café en el Departamento de Risaralda. Cenicafé 53, 106–118 (2002).
Pérez, J., Infante, F. & Vega, F. E. A coffee berry borer (Coleoptera: Curculionidae: Scolytinae) bibliography. J. Insect Sci. 15, 83 (2015).
Rubio-Gómez, J. D., Bustillo-Pardey, A. E., Vallejo-Espinosa, L. F., Benavides Machado, P. & Acuña-Zornosa, J. R. Morfología del sistema reproductor femenino y masculino de Hypothenemus hampei. Cenicafé 58, 75–82 (2007).
Rubio G., J. D., Bustillo P., A. E., Vallejo E., L. F., Acuña Z., J. R. & Benavides, M. P. Alimentary canal and reproductive tract of Hypothenemus hampei (Ferrari) (Coleoptera: Curculionidae, Scolytinae). Neotrop. Entomol. 37, 143–151 (2008).
Constantino, L. M. et al. Aspectos biológicos, morfológicos y genéticos de Hypothenemus obscurus e Hypothenemus hampei (Coleoptera: Curculionidae: Scolytinae). Rev. Colomb. Entomol. 37, 173–182 (2011).
López-Guillén, G. J. et al. Morphology and structural changes in flight muscles of Hypothenemus hampei (Coleoptera: Curculionidae) females. Environ. Entomol. 40, 441–448 (2011).
Román-Ruiz, A. K. et al. Description of the sperm and spermatheca of Hypothenemus hampei (Coleoptera: Curculionidae: Scolytinae) for the differentiation of mated and unmated females. Ann. Entomol. Soc. Amer. 110, 353–359 (2017).
Aldrovandi, U. De animalibus insectis libri septem: cum singlulorum iconibus ad viuum expressis. Bonon: apud Joan Bapt. Bellagamban (1602).
Malpighi, M. Dissertatio Epistolica de Bombyce. London: J. Martyn & J. Allestry, regiæ societatis typographos (1669).
Wipfler, B., Pohl, H., Yavorskaya, M. I. & Beutel, R. G. A review of methods for analysing insect structures – the role of morphology in the age of phylogenomics. Curr. Opin. Insect Sci. 18, 60–68 (2016).
Alba-Tercedor, J. & Alba-Alejandre, I. Comparing micro-CT results of insects with classical anatomical studies: The European honey bee (Apis mellifera Linnaeus, 1758) as a benchmark (Insecta: Hymenoptera, Apidae). Microscopy & Analysis 3, 12–15 (2019).
Alba-Tercedor, J., Alba-Alejandre, I. & Vega, F. E. Revealing the respiratory system of the coffee berry borer (Hypothenemus hampei, Coleoptera: Curculionidae: Scolytinae) using micro-computed tomography. Sci. Rep. in press.
Richter, A. et al. The cephalic anatomy of workers of the ant species Wasmannia affinis (Formicidae, Hymenoptera, Insecta) and its evolutionary implications. Arthropod Struct. Dev. 49, 26–49 (2019).
Lowe, T., Garwood, R. J., Simonsen, T. J., Bradley, R. S. & Withers, P. J. Metamorphosis revealed: Time-lapse three-dimensional imaging inside a living chrysalis. J. Roy. Soc. Interface 10, 20130304 (2013).
Helm, B. R. et al. Micro-computed tomography of pupal metamorphosis in the solitary bee Megachile rotundata. Arthropod Struct. Dev. 47, 521–28 (2018).
Greenlee, K. J. et al. Synchrotron imaging of the grasshopper tracheal system: Morphological and physiological components of tracheal hypermetry. Am. J. Physiol. Regul. Integr. Comp. Physiol. 297, R1343–1350 (2009).
Greco, M. et al. 3-D visualisation, printing, and volume determination of the tracheal respiratory system in the adult desert locust, Schistocerca gregaria. Entomol. Exp. Appl. 152, 42–51 (2014).
Iwan, D., Kamiński, M. J. & Raś, M. The last breath: A ΜCT-based method for investigating the tracheal system in Hexapoda. Arthropod Struct. Dev. 44, 218–27 (2015).
Ha, Y. R., Yeom, E., Ryu, J. & Lee, S. J. Three-dimensional structures of the tracheal systems of Anopheles sinensis and Aedes togoi pupae. Scientific Reps. 7, 44490 (2017).
Raś, M., Iwan, D. & Kamiński, M. J. The tracheal system in post-embryonic development of holometabolous insects: A case study using the mealworm beetle. J. Anat. 232, 997–1015 (2018).
Wasserthal, L. T., Cloetens, P., Fink, R. H. & Wasserthal, L. K. X-ray computed tomography study of the flight-adapted tracheal system in the blowfly Calliphora vicina, analysing the ventilation mechanism and flow-directing valves. J. Exp. Biol. 221, jeb176024 (2018).
Li, Y. et al. Structure of the ambrosia beetle (Coleoptera: Curculionidae) mycangia revealed through micro-computed tomography. J. Insect Sci. 18(5), 13 (2018).
Li, Y., Ruan, Y.-Y., Stanley, E. L., Skelton, J. & Hulcr, J. Plasticity of mycangia in Xylosandrus ambrosia beetles. Insect Sci. 26, 737–742 (2018).
Jiang, Z.-R., Kinoshita, S., Sasaki, O., Cognato, A. I. & Kajimura, H. Non-destructive observation of the mycangia of Euwallacea interjectus (Blandford) (Coleoptera: Curculionidae: Scolytinae) using X-ray computed tomography. Entomol. Sci. 22, 173–181 (2019).
Li, D., Zhang, K., Zhu, P., Wu, Z. & Zhou, H. 3D configuration of mandibles and controlling muscles in rove beetles based on micro-CT technique. Analyt. Bioanalyt. Chem. 401, 817–25 (2011).
Martín-Vega, D. et al. Micro-computed tomography visualization of the vestigial alimentary canal in adult oestrid flies. Med. Vet. Entomol. 32, 378–382 (2018).
Shaha, R. K., Vogt, J. R., Han, C. S. & Dillon, M. E. A micro-CT approach for determination of insect respiratory volume. Arthropod Struct. Dev. 42, 437–442 (2013).
Simonsen, T. J. & Kitching, I. J. Virtual dissections through micro-CT scanning: a method for non-destructive genitalia ‘dissections’ of valuable Lepidoptera material. Syst. Entomol. 39, 606–618 (2014).
Smith, D. B. et al. Exploring miniature insect brains using micro-CT scanning techniques. Sci. Rep. 6, 21768 (2016).
Nüsslin, O. Phylogenie und system der borkenkäfer. Zeitschrift fürwissenschaftliche Insektenbiologie 8, 1–5 (1911).
Wood, S. L. A revision of North American Cryphalini (Scolytidae: Coleoptera). Univ. Kansas Sci. Bull 36, 959–1089 (1954).
Hopkins, A. D. Contributions toward a monograph of the scolytid beetles. The genus Dendroctonus. U. S. Department of Agriculture, Bureau of Entomology, Technical Series, No. 17, Part I, pp. 1–164 (1909).
Furniss, M. M. Biology of Trypophloeus striatulus (Coleoptera: Scolytidae) in felt-leaf willow in interior Alaska. Environ. Entomol. 33, 21–27 (2004).
Santiago-Blay, J. A. & Young, T. L. Reliable sexing of adult Conophthorus (Coleoptera: Scolytidae) beetles. Can. Entomol. 127, 605–7 (1995).
Vega, F. E. et al. A mysterious wing spine in male coffee berry borers (Coleoptera: Curculionidae: Scolytinae). Fla. Entomol. 98, 352–353 (2015).
Vega, F. E. et al. On the eyes of male coffee berry borers as rudimentary organs. PLoS ONE 9, e85860 (2014).
Kirkendall, L. R., Biedermann, P. H. W. & Jordal, B. H. Evolution and diversity of bark and ambrosia beetles. In: Bark Beetles: Biology and Ecology of Native and Invasive Species (F. E., Vega and R. W., Hofstetter, Eds), pp. 85–156. San Diego: Academic Press (2015).
Snodgrass, R. E. Principles of Insect Morphology. New York: McGraw-Hill Book Company, Inc. (1935).
Gillott, C. Entomology, 3rd ed. Dordrecht: Springer (2005).
Chapman, R. F. The Insects: Structure and Function, 5th ed. (S. J., Simpson and A. E., Douglas, Eds). Cambridge: Cambridge University Press (2013).
Swaine, J. M. Canadian bark-beetles. Part II. A preliminary classification with an account of the habits and means of control. Dominion of Canada, Department of Agriculture, Entomological Branch, Bulletin No. 14 (Technical Bulletin), 1–143 (1918).
Beal, J. A. The development of the proventriculus of Pityogenes hopkinsi Swaine. Ann. Entomol. Soc. Am. 20, 522–539 (1927).
Eaton, C. B. The anatomy and histology of the proventriculus of Ips radiatae Hopkins. Ann. Entomol. Soc. Am. 35, 41–49 (1942).
Nobuchi, A. A comparative morphological study of the proventriculus in the adult of the Superfamily Scolytoidea (Coleoptera). Bull. Gov. For. Exp. Stn. (Jpn.) 224, 40–58 (1969).
Calder, A. A. The alimentary canal and nervous system of Curculionoidea (Coleoptera): gross morphology and systematic significance. J. Nat. Hist. 23, 1205–1265 (1989).
Crowson, R. A. The Biology of the Coleoptera. (Academic Press, London, 1981).
Klowden, M. J. Physiological Systems in Insects. (Elsevier/AcademicPress, Amsterdam, 2007).
Valencia-Jiménez, A. Amylase and protease inhibitors as alternative against herbivore insect. In: Coffee Biotechnology and Quality (T., Sera, C. R., Soccol, A., Pandey, and S., Roussos, Eds), pp. 287–296. Proceedings, 3rd International Seminar on Biotechnology in the Coffee Agro-Industry, Londrina, Brazil. Dordrecht: Kluwer Academic Publishers (2000).
Blomquist, G. J. et al. Pheromone production in bark beetles. Insect Biochem. Mol. Biol. 40, 699–712 (2010).
Ceja-Navarro, J. A. et al. Gut microbiota mediate caffeine detoxification in the primary insect pest of coffee. Nature Comms. 6, 7618 (2015).
Summers, R. M. et al. Novel, highly specific N-demethylases enable bacteria to live on caffeine and related purine alkaloids. J. Bacteriol. 194, 2041–2049 (2012).
Redgwell, R. & Fischer, M. Coffee carbohydrates. Braz. J. Plant Physiol. 18, 165–174 (2006).
Aguilera-Gálvez, C., Vásquez-Ospina, J. J., Gutiérrez-Sanchez, P. & Acuña-Zornosa, R. Cloning and biochemical characterization of an endo-1,4-ß-mannanase from the coffee berry borer, Hypothenemus hampei. BMC Res. Notes 6, 333 (2013).
Balaman, S. Y. Decision-Making for Biomass-Based Production Chains. The Basic Concepts and Methodologies. (Academic Press, San Diego, 2018).
Romoser, W. S. The Science of Entomology. (Macmillan Publishing Co., Inc, New York, 1973).
Aslam, N. A. An assessment of some internal characters in the higher classification of the Curculionidae s.l. (Coleoptera). Trans. R. Ent. Soc. London 113, 417–480 (1961).
Baker, W. V. & Estrin, C. L. The alimentary canal of Scolytus multistriatus (Coleoptera: Scolytidae): a histological study. Can. Entomol. 106, 673–686 (1974).
Díaz, E., Cisneros, R. & Zuñiga, G. Comparative anatomical and histological study of the alimentary canal of the Dendroctonus frontalis (Coleoptera: Scolytidae) complex. Ann. Entomol. Soc. Am. 93, 303–311 (2000).
Díaz, E., Arciniega, O., Sánchez, L., Cisneros, R. & Zuñiga, G. Anatomical and histological comparison of the alimentary canal of Dendroctonus micans, D. ponderosae, D. pseudotsugae pseudotsugae, D. rufipennis, and D. terebrans (Coleoptera: Scolytidae). Ann. Entomol. Soc. Am. 96, 144–152 (2003).
Bu, S.-H. & Chen, H. The alimentary canal of Dendroctonus armandi Tsai and Li (Coleoptera: Curculionidae: Scolytinae). Coleopterists Bull. 63, 485–496 (2009).
Wigglesworth, V. B. The Principles of Insect Physiology. (Methuen & Co., Ltd, London, 1942).
Cerezke, H. F. The morphology and functions of the reproductive systems of Dendroctonus monticolae Hopk. (Coleoptera: Scolytidae). Can. Entomol. 96, 477–500 (1964).
Lindroth, C. H. & Palmen, E. Coleoptera. In: Taxonomist’s Glossary of Genitalia in Insects, 2nd ed. (S. L., Tuxen, Ed.), pp. 80–88. Copenhagen: Ejnar Munksgaard. (1970).
Zunino, M. 2012. Cuarenta años de anatomía de las piezas genitales en la taxonomía de los escarabajos (Coleoptera: Scarabaeoidea): el estado del arte. Dugesiana 18, 197–206 (2012).
Fuchs, G. Morphologische Studien über Borkenkäfer. II. Die europäischen Hylesinen. E. Reinhardt, München, 50 pp. (1912).
Wigglesworth, V. B. A theory of tracheal respiration in insects. Proc. R. Soc. B, Biol. Sci. 106, 229–250 (1930).
Grassé, P.-P. Le Système Nerveux Des Insectes. pp. 321–510 In Traité de Zoologie. Anatomi, Systematique, Biologie. T. VIII, fasc. III. Paris: Masson et Cie. (1975).
Atkins, M. D. & Chapman, J. A. Studies on the nervous system anatomy of the Douglas fir beetle, Dendroctonus pseudotsugae Hopk. (Scolytidae). Can. Entomol. 89, 80–86 (1957).
Beani, L., Dessì-Fulgheri, F., Cappa, F. & Toth, A. The trap of sex in social insects: From the female to the male perspective. Neurosci. & Biobehav. Revs. 46, 519–33 (2014).
Cachero, S. A., Ostrovsky, A. D., Yu, J. Y., Dickson, B. J. & Jefferis, G. S. X. E. Sexual dimorphism in the fly brain. Curr. Biol. 20, 1589–1601 (2010).
Vega, F. E., Kramer, M. & Jaramillo, J. Increasing coffee berry borer (Coleoptera: Curculionidae: Scolytinae) female density in artificial diet decreases fecundity. J. Econ. Entomol. 104, 87–93 (2011).
Alba-Tercedor, J. From the sample preparation to the volume rendering images of small animals: A step by step example of a procedure to carry out the micro-CT study of the leafhopper insect Homalodisca vitripennis (Hemiptera: Cicadellidae). Bruker Micro-CT Users Meeting, May 5–8, 2014, Ostend, Belgium, pp. 260–288 (2014).
Stalling, D., Westerhoff, M. & Hege, H.-C. Amira: A highly interactive system for visual data analysis. Vis. Handb. 749–767 (2005).
Thermo Fisher Scientific. Amira 3D Visualization and Analysis Software. Available at: http://www.thermofisher.com/amira-avizo (2017).
Alba-Tercedor, J. Efemerópteros. In: Curso Práctico de Entomología (J. A., Barrientos, Ed.), pp. 511–522. Barcelona, Alicante: Asociación Española de Entomología, Centro Iberoamericano de la Biodiversidad (2004).
Acknowledgements
We appreciate the help provided by the Skyscan’s staff at Bruker Micro-CT: Alexander Sasov, Stephan Boons, Xuan Liu, Vladimir Kharitonov, Phil Salmon, Kjell Laperre and Wesley De Boever. Also, to Kathy Tinoco, Clark Shaunte and Jean-Luc Garnier at Thermo Fisher Scientific for assistance with the Amira software, and to Alejandra Sánchez-Erostegui. This paper benefitted from sub-award agreement S15192.01 between Kansas State University (KSU) and the University of Granada, as part of the USDA-NIFA Award 2014-70016-23028 to Susan J. Brown (KSU), “Developing an Infrastructure and Product Test Pipeline to Deliver Novel Therapies for Citrus Greening Disease” (2015–2020).
Author information
Authors and Affiliations
Contributions
J.A.-T., I.A.A. and F.E.V. conceived and designed the study. I.A.A. and J.A.-T. conducted the microtomographic study and recorded the images and videos. F.E.V., I.A.A., and J.A.-T. interpreted the results and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Alba-Alejandre, I., Alba-Tercedor, J. & Vega, F.E. Anatomical study of the coffee berry borer (Hypothenemus hampei) using micro-computed tomography. Sci Rep 9, 17150 (2019). https://doi.org/10.1038/s41598-019-53537-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-53537-z
This article is cited by
-
Anatomical changes of Tenebrio molitor and Tribolium castaneum during complete metamorphosis
Cell and Tissue Research (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.