Abstract
Temperature is an important environmental factor for ectotherms’ fitness and survival. The upper sublethal and lethal temperatures were compared between adults of three closely related destructive planthopper species, the small brown planthopper (Laodelphax striatellus, SBPH), the brown planthopper (Nilaparvata lugens, BPH), and the white-backed planthopper (Sogatella furcifera, WBPH) in the absence and presence of the host plant (Oryza sativa, var. Taichong1). Values of the critical thermal maxima (CTmax) were higher in SBPH than in both BPH and WBPH and higher in BPH than in WBPH, and values of the heat coma temperatures (HCT) were higher in both BPH and SBPH than in WBPH. CTmax and HCT values were higher in the presence than in the absence of plant material. Between sexes, females generally showed higher CTmax and HCT than males. The upper lethal temperatures (ULT50) measured in the absence of plant material were not significantly different among the planthopper species. The planthoppers also exhibited different behaviors in an increasing temperature regime, with fewer insects dropping-off from the plant in SBPH than in BPH and WBPH. These results indicate that SBPH and BPH are more heat tolerant than WBPH. The findings highlight the biological divergence in closely related planthopper species and the importance of performing the heat tolerance measurement in an ecologically relevant setting, which serves to predict seasonal and spatial occurrence patterns of the destructive planthopper species.
Similar content being viewed by others
Introduction
Insects are ectotherms and are of limited ability to regulate body temperature above and below optimum. Consequently, temperature can play a central role in insect development1, behavior2, and seasonality and distribution3,4,5. Therefore, thermal tolerance traits are usually examined in the laboratory to forecast ectotherms’ field success and pest outbreaks.
In thermal tolerance studies of insects, sub-lethal and lethal traits are usually characterized. Critical thermal limit, coma temperature and coma recovery times are the sub-lethal traits that are measured before the death of the organism. In contrast, lethal temperature and lethal time are parameters used to indicate lethal traits, where mortality is induced in the exposed organism6. When an insect is exposed to increasing temperatures for a certain duration, different observable and measurable events occur7: when the exposed organism shows uncoordinated movement until motionless, it reaches the critical thermal maximum (CTmax); when the appendages (legs and antennae) are incapable of movement and the organism enters a ‘heat coma’ state, it comes to the heat coma temperature (HCT)8. At even higher temperatures, the insect dies, and the temperature is the upper lethal temperature (ULT)7. ULT50 is the upper high temperature that results in 50% mortality in a sample population. Estimating these parameters offers a physiological insight into the events that are ecologically important8. For instance, insects at CTmax are incapable of moving move and are therefore unable to find food sources or escape from natural enemies1; and to a wider extent, this can affect insects’ distribution and potential range expansion3,9.
Rice is the staple food for one third of the world’s population and is attacked by three closely related destructive planthopper species: the small brown planthopper (SBPH) Laodelphax striatellus (Fallén), the brown planthopper (BPH) Nilaparvata lugens (Stål), and the white-backed planthopper (WBPH) Sogatella furcifera (Horvath) (Hemiptera: Delphacidae). The three species are similar in morphology and exhibit wing-dimorphism in the adult stage. They cause damage to rice plants directly by sucking phloem sap and indirectly by transmitting plant viruses10. However, the three planthopper species exhibit different tolerance to low temperatures; WBPH and BPH cannot survive 0 °C for 5 days10 while SBPH can overwinter locally at sub-zero temperatures11. WBPH and BPH can overwinter only in areas to the south of 21–25°N and occurrence in the northern areas depends on migration from the south, principally the Indo-China Peninsula. The planthopper species also differ in spatial distribution. WBPH and BPH can occur from the tropics to 42–44°N in Asia through migration, while SBPH occurs primarily in the temperate regions10. The temperatures of July and August in the subtropics during 1985–2010 average 27.3 °C and 27.0 °C, respectively, while in the temperate regions they are 28.8 °C and 28.0 °C, respectively, in China (data calculated from the Chinese Meteorological Data Sharing Service System, http://cdc.cma.gov.cn). Further, the three planthopper species also differ in host range. The three species all flourish on rice, but BPH is monophagous and WBPH and SBPH are polyphagous on rice, wheat, corn and other plants10. The planthopper species also show varying performance at high temperatures. An analysis of time-temperature-mortality models at constant temperatures between 35 and 40 °C revealed longer lethal time in SBPH than in WBPH and BPH12. Further tests employing varying high temperatures showed reduced nymphal duration, adult longevity and fecundity in WBPH and BPH in comparison with SBPH13. Piyaphongkul et al.8 measured CTmax, HCT and ULT of BPH and found that thermal tolerance differed between developmental stages. Furthermore, they indicated that acclimation increases heat tolerance in BPH14. Using transcriptional analysis, Huang et al.15 found temperature modulation of genes related to thermal tolerance in the three planthopper species and suggested that the differentially expressed genes of heat shock proteins in the three planthopper species at high temperature might contribute to their different capacities for heat tolerance. However, the differences in upper sublethal and lethal temperatures between the three planthopper species remain unaddressed, especially considering that the mid-latitude species seem most prone to experience heat stress4.
Thermal tolerance has mostly been measured in the absence of the host plant, which may be problematic for phloem-feeding insects16. Host plant contact is reported to enhance aphids’ cold tolerance, i.e., lethal temperature17, lethal time18 and CTmin16. These results question the validity of measuring heat tolerance in the absence of the host plant for predictions and decisions concerning pest species (e.g., timing and severity of pest outbreaks, efficiency and establishment potential of invasive species, implications of climate change)16.
In this study, we hypothesize that the three closely related planthopper species differ in their heat tolerance and that contact with host plant can increase their ability to withstand heat stress. Heat tolerance was measured in terms of CTmax, HCT and ULT in both the absence and presence of their common host plant, which are expected to obtain results of ecological relevance to the field conditions. The current results will further our understanding of the spatio-temporal occurrence patterns of the three planthopper species in relation to their heat tolerance.
Results
CTmax
In the absence of plant material, the CTmax ranged between 37.5–39.5 °C, while in the presence of plant material, between 40.5–42.0 °C (Fig. 1A). Significant influence on CTmax was observed for planthopper species (F = 182.3, df = 2,480, P < 0.001), planthopper sex (F = 7.3, df = 1,480,, P = 0.014), plant material (F = 773.1, df = 1,480, P < 0.001), and the interaction between planthopper species and plant material (F = 8.1, df = 2,480, P = 0.038). Between the planthopper species, mean CTmax values were higher in SBPH than in both BPH and WBPH (Tukey test, P < 0.001), and higher in BPH than in WBPH (Tukey test, P = 0.038; Fig. 1B). CTmax values were significantly higher in the presence than in the absence of plant material and higher in the females than in the males.
When subjected to an increasing temperature regime, the insects exhibited different behaviors, i.e., dropping off or remaining on the tube wall or rice stem. Irrespective of species, proportions of insect dropping (Fig. 2) were found to be higher in the absence than in the presence of plant material at the 11 tested temperatures except 34 and 44 °C (χ2 ≥ 7.104, P ≤ 0.008). Between the species, proportions of insects dropping were different at seven out of the 11 tested temperatures (χ2 ≥ 7.120, P ≤ 0.028), with fewer insects dropping in SBPH. Regression of the cumulative proportions of insects dropping to temperatures (Table 1) showed that the temperatures with 50% insects dropping were 39.0, 40.4 and 38.9 °C for BPH, SBPH and WBPH, respectively, in the absence of plant material and 42.0, 43.0 and 41.3 °C for BPH, SBPH and WBPH, respectively, in the presence of plant material (Fig. 2).
Cumulative percentage of planthopper adults of three species dropping off in the absence (AHP, in solid bullet) and in the presence (PHP, in hollow bullet) of host plant when subjected to an increasing temperature regime from 26 °C to 35 °C at 0.5 °C/min and then from 35 °C to 50 °C at 0.1 °C/min. n = 80.
HCT
Considered by sex and species of the planthopper adults, the average HCT ranged between 41.5–43.5 °C in the absence of plant material (Fig. 3A) and between 42.5–44.0 °C in the presence of plant material (Fig. 3B). Among the planthopper species, HCT differed significantly (χ2 ≥ 11.007, P ≤ 0.004), and was lower in WBPH than in both BPH (Z ≥ 2.396, P ≤ 0.017) and SBPH (Z ≥ 2.281, P ≤ 0.001), irrespective of sex and presence of plant material. The mean HCT values were also lower in BPH than SBPH in plant presence for both sexes (Z ≥ 2.31, P ≤ 0.021) but in plant absence, lower in females of SBPH than BPH (Z = 3.147, P = 0.002).
Significantly higher HCT values were observed in the presence than in the absence of plant material in each combination of planthopper sex and species (Z ≥ 3.474, P ≤ 0.001) except female BPH (Z = 1.275, P = 0.202). Between the sexes, BPH females in the absence of plant material and SBPH females in the presence of plant material showed higher HCT than their respective male counterparts (Z ≥ 4.027, P ≤ 0.001).
ULT
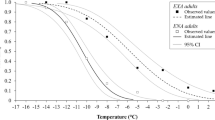
ULT50 was detected when the insects were exposed to a series of high temperatures ranging from 37.5 to 45.0 °C (Fig. 4). There were no significant differences in ULT50 between the planthopper species shown by the overlapping of the 95% confidence limits. Between the sexes, WBPH females (95% confidence limits 40.04–42.01 °C) showed significantly higher ULT50 than males (39.18–39.99 °C).
Discussion
Temperature is a pivotal abiotic component that significantly affects ectotherms. It influences the life history traits and physiology of insects19, and is an important factor determining the geographical distribution and richness of insects3,4,5, especially under climate change scenarios20.
Host plant contact can influence heat tolerance traits of herbivores. In the three planthopper species, CTmax and HCT measured in the presence of plant material were higher than in the absence of plant material. In a similar design, Alford et al.16 observed lower CTmin in three aphid species in the presence than in the absence of plant material and proposed that performing the measurement in the absence of plant material could result in underestimation of thermal tolerance. Host plant contact may contribute to herbivore tolerance to extreme temperatures in that: (1) an herbivore can evade thermal stress through movement in/on the host plant5; (2) feeding from the host plant by phloem feeders can add to their thermal tolerance. Alford et al.16 argued that phloem sap is rich in cryoprotectant carbohydrates and continuous feeding under low temperatures can enhance aphid supercooling ability and thus lower their CTmin. When exposed to high temperatures, the accumulated polyols21 and glucose, protein and glycerol22 can stabilize protein structure against thermal denaturation; (3) feeding during exposure to high temperatures can also help maintain a water balance against dehydration through ingestion of food and water23. Insect dropping-off at extreme temperatures is a result of loss of locomotion coordination24,25. Planthoppers dropped more in the absence than in the presence of plant material at high temperatures or dropped by 50% at a lower temperature in the absence than in the presence of plant material, which further indicates that host plant contact can enhance insect’s heat tolerance. Although lack of traction on the glass tube may contribute to the insect dropping-off, the insects do drop off only when they lose locomotion coordination.
CTmax also differ between insect species, as shown in three Cetoniidae species26. In the cicada Magicicada cassini, CTmax is 43 °C27. The three planthopper species differed significantly in the upper sublethal temperatures. The planthopper adults became immobilized at 37.5–39.5 °C in the absence of plant material; this corresponds to a previous report of CTmax of 38 °C for BPH adults8. Irrespective of sex and presence of plant material, the CTmax values were higher in SBPH than in both BPH and WBPH and the HCT values were higher in both BPH and SBPH than in WBPH, indicating that SBPH and BPH are more heat tolerant than WBPH. These results correspond to an early report of longer lethal time in SBPH than in WBPH and BPH12. Because the insects were reared for six generations in the laboratory, a direct effect of sampling locations on heat tolerance can be excluded; instead, the high heat tolerance in SBPH may be adaptive to the relatively hot summer in the temperate regions where SBPH principally occurs. Additionally, SBPH is small in size (with body length of 3.33 to 4 mm) in comparison with BPH and WBPH (3.7 to 5 mm)28, which can add to the high heat tolerance in SBPH8. Since CTmax represents the effective limits to coordinated movement7, it can have important ecological significance for insect behavior8.
Extreme high temperature can cause mortality in insects. Fifty percent of the planthopper adults did not survive exposures of only 3 min at 39.5–41.5 °C. Similarly, approximately 50% of BPH adults were killed during 2 to 6 min exposures to 42.5 °C8. The current ULT50 values in the planthoppers are comparable to those in insects of other orders. In the tsetse fly Glossina pallidipes, ULT50 ranged between 35–38 °C during exposures of 1 to 3 h19 and in the adult codling moth Cydia pomonella, it was 44 °C following a 2 h exposure29. Denaturizing of proteins, enzyme structure modification and desiccation are proposed as the causes for insect death at high temperatures9,30. However, ULT50 did not differ significantly between the three planthopper species.
Insect stages and sexes may show differential vulnerability to high temperatures. In the present study, females were generally more heat tolerant than males. This may be due to the fact that male planthoppers are smaller in size and volume than females. Previous reports show that the gain and loss of heat are proportionate to the ratio of surface area to volume8 that is usually larger for small insects than for large insects; therefore, the planthopper males may have gained more heat in exposure to increasing high temperature than the females and thus showed lower heat tolerance than the females. The sexual difference in heat tolerance may also result from their physiological difference in response to high temperatures, which may involve the accumulation of sugars21 and transcriptional variation31. While constant high temperature induces up-regulation of most heat shock protein-related genes in both sexes, the expression of Hsp25.4 and DnaJ5 are down-regulated in the male fat body of Bombyx mori31.
The heat tolerance determined in the three planthopper species is of significance in predicting their population dynamics. The heat tolerance measured in the presence of plant material is more ecologically relevant and thus can provide results of higher relevance to the natural conditions16. Although field temperatures rarely reach the thermal limits gained in this study, temperatures as high as 40 °C or more occur and usually last for several hours daily during heat waves7. Despite the measurement of heat tolerance with the presence of plant materials in this study, caution has to be exercised in applying the data in actual pest population prediction, because the microclimates experienced by the insects in the fields, especially pre-exposure to temperature extremes or fluctuating temperatures, can greatly affect insect responses to high temperatures. Nevertheless, our results of heat tolerance echo the population dynamics of the three planthopper species. The average peak densities of SBPH and BPH in the 21th century is almost double that of the 20th century10. Yield losses due to SBPH damage at heading stage reached 10–20%, which never occurred in the 20th century32. It can be expected that the heat-tolerant insect pest species will better adapt to temperature increases associated with global warming in the future.
Methods
Insect cultures
Stock culture of the planthoppers (SBPH, BPH and WBPH) were established from samples collected from Zhengzhou (N34°53′35.70″, E113°38′8.53″; SBPH) and Xing’An (N25°36′13.10″, E110°41′17.30″; BPH and WBPH), China and reared on potted rice seedlings (var. Taichong1) at tillering stage in insect-proof cages. Insect cultures were housed in an environment-controlled insectary at 26 °C and LD16:8 h photoperiod. Prior to use in the experiment, the three planthopper species had been reared for 6 generations.
Determination of critical thermal maximum (CTmax)
CTmax of the three planthopper species was measured using a double-walled glass column (Weber column, height 20 cm and diameter 16 cm) connected to a programmed alcohol bath (Ministat 230-cc-NR, Huber Ltd., Germany). The double-walled glass column was similar to that used by Powell and Bale33. A group of 10 male or female adult planthoppers (3 day old) collected directly from the insectary into a glass tube (10 cm high, 2.5 cm diameter) covered with gauze was transferred into the Weber column preset at 26 °C. The column was subsequently closed with a plastic lid to minimize airflow and maintain a stable thermal environment and to prevent condensation. Uniformity of temperature in the column has previously been reported16. Before the programmed temperature increase, the planthoppers were allowed to recover at 26 °C for 30 min from handling. The observation was repeated in 4 replicates separately for males and females of each of the three planthopper species.
The temperature within the column was programmed to increase from 26 °C to 35 °C at 0.5 °C/min and then from 35 °C to 50 °C at 0.1 °C/min8. Temperature measured by a thermocouple within the glass tube was recorded by software (JULABO easy temp. professional) and insect movement at 11 temperatures, by a high efficiency video camera (Panasonic, HDC-HS700 DV). The video was played back for determination of CTmax: the temperature at which an insect exhibits uncoordinated movement and drops to the bottom of the glass tube and becomes immobile7. It was observed that most insects were on the glass tube wall after recovery from handling and when temperature increased to 34 °C, the insects were all clinging to the glass tube. The temperatures that the insects experienced in the glass tube were the designated temperatures.
CTmax was also determined in the presence of plant material to provide conditions that are relevant to the field conditions as above. The difference was that the 10 adult planthoppers were aspirated onto three segments of rice stem (about 8.5 cm in height) secured with roll paper in the glass tube (10 cm high, 2.5 cm diameter). After a 30 min recovery from handling, the insects were monitored by the video camera.
Determination of heat coma temperature (HCT)
HCT was determined using the same Weber column as for CTmax. The temperature within the column was also initially set at 26 °C. The procedures were largely the same as that for CTmax measurement except that the insects were individually placed in cellular holes (diameter 1.2 cm) of a transparent plastic plate (height 10 cm, width 12 cm) within the column and allowed to settle for 30 min before temperature increase. After the temperature treatment as above, the video was watched to record the temperatures at which a planthopper last walked and moved its legs and antennae7.
To provide field relevant conditions, HCT was also determined for the insects in the presence of plant material as above, where the planthoppers were individually aspirated onto three segments of rice stem (about 8.5 cm in height) in a glass tube. Four replicates were exercised separately for males and females of each of the three planthopper species.
Determination of upper lethal temperature (ULT)
ULT50 of the three planthopper species was determined for males and females separately. Preliminary experiments were conducted to find out the temperature range that could result in zero to 100 percent mortality. To determine ULT50, a group of 15 insects within a 0.9-ml Eppendorf tube was exposed to the temperatures (37.5, 39.0, 40.5, 42.0, 43.5, and 45.0 °C) generated with the programmable alcohol bath. The temperature in the bath was increased from 26 °C to a target temperature at 0.5 °C min−1 as above and maintained there for 3 min. After the heat stress exposure, the bath temperature was decreased back to the rearing temperature (26 °C) at the same rate as for warming. Thereafter, the planthoppers were transferred to a recovery glass tube (4 cm × 40 cm) containing rice plants (25 days old) and allowed to recover at 26 °C. Survival was assessed at 72 h after exposure. Four replicates were performed for each combination of species and sex at each of the high temperatures.
Data analysis
Data from each replicate were averaged and SEs were calculated. The CTmax data were subjected to General Linear Model for significant influence of planthopper species, planthopper sex and presence of plant material, and mean difference between the planthopper species were separated by Tukey test. HCT data were subjected to Kruskal Wallis Test for significant effects of planthopper species, planthopper sex and presence of plant material individually due to equal variance could not be assumed in homogeneity analysis. When there was a significant effect, the differences were tested by Mann-Whitney U Test. Data of planthopper behaviors (dropping-off or remaining on the glass tube wall or the rice stem) were analyzed using a Pearson Chi-Square test in Crosstabs to determine if the proportion of dropping-off individuals differed between the test arena or the species. In the multiple comparisons, the tests were adjusted using the Bonferroni correction. Further, the cumulative proportions of insects dropping were regressed with temperatures using logistic estimation and the temperatures at which 50% insects dropping were calculated. The temperature resulting in 50% of mortality (ULT50) was determined by probit analysis using the binomial distribution. Significant difference in ULT50 was based on non-overlap between the 95% confidence limits34. All the data analysis was performed using SPSS 16.0 (SPSS Inc., Chicago, USA).
References
Tewksbury, J. J., Huey, R. B. & Deutsch, C. A. Putting the heat on tropical animals. Sci. 320, 1296–1297 (2008).
Long, Y., Hu, C., Shi, B., Yang, X. & Hou, M. Effects of temperature on mate-location in the brown planthopper, Nilaparvata lugens (Homoptera: Delphacidae). Environ. Entomol. 41, 1231–1238 (2012).
Bale, J. S. et al. Herbivory in global climate change research: direct effects of rising temperature on insect herbivores. Global Change Biol. 8, 1–16 (2002).
Hoffmann, A. A., Chown, S. L. & Clusella-Trullas, S. Upper thermal limits in terrestrial ectotherms: how constrained are they? Funct. Ecol. 27, 934–949 (2013).
Ma, G., Bai, C.-M., Wang, X.-J., Majeed, M. Z. & Ma, C.-S. Behavioural thermoregulation alters microhabitat utilization and demographic rates in ectothermic invertebrates. Anim. Behav. 142, 49–57 (2018).
Bürgi, L. P. & Mills, N. J. Ecologically relevant measures of the physiological tolerance of light brown apple moth, Epiphyas postvittana, to high temperature extremes. J. Insect Physiol. 58, 1184–1191 (2012).
Hazell, S. P. et al. Hyperthermic aphids: insights into behaviour and mortality. J. Insect Physiol. 56, 123–131 (2010).
Piyaphongkul, J., Pritchard, J. & Bale, J. Can tropical insects stand the heat? A case study with the brown planthopper Nilaparvata lugens (Stål). PloS ONE 7, e29409 (2012).
Hance, T., van Baaren, J. & Boivin, G. Impact of extreme temperatures on parasitoids in a climate change perspective. Ann. Rev. Entomol. 52, 107–126 (2007).
Heong, K. L., Cheng J. & Escalada M. M. Rice planthoppers: ecology, management, socio economics and policy. Springer, Dordrecht, the Netherland (2015).
An, Z.-F., Zhang, A.-M. & Liu, X.-D. Effect of low temperature shock on the development and fecundity of Laodelphax striatellus (Fallen). Chin. Bull. Entomol. 5, 1309–1313 (2011).
Ma, J., Hu, G. & Cheng, J. Survival analysis of three planthopper species at extreme high temperatures: a time temperature-mortality model. Entomol. J. East China 7, 85–90 (1998a).
Ma, J., Hu, G. & Cheng, J. Ecological performances of three planthopper species at simulated fluctuating temperature. Entomol. J. East China 7, 85–90 (1998b).
Piyaphongkul, J., Pritchard, J. & Bale, J. Effects of acclimation on the thermal tolerance of the brown planthopper Nilaparvata lugens (Stål). Agri. Forest. Entomol. 16, 174–183 (2014).
Huang, H.-J. et al. Comparative analysis of the transcriptional responses to low and high temperatures in three rice planthopper species. Mol. Ecol. 26, 2726–2737 (2017).
Alford, L., Burel, F. & Baaren, J. Improving methods to measure critical thermal limits in phloem-feeding pest insects. Ent. Exp. Appl. 159, 61–69 (2016).
Griffiths, E. & Wratten, S. D. Intra- and interspecific differences in cereal aphid low-temperature tolerance. Ent. Exp. Appl. 26, 161–167 (1979).
Butts, R. A., Howling, G. G., Bone, W., Bale, J. S. & Harrington, R. Contact with the host plant enhances aphid survival at low temperatures. Ecol. Entomol. 22, 26–31 (1997).
Terblanche, J. S., Clusella-Trullas, S., Deere, J. A. & Chown, S. L. Thermal tolerance in a south-east African population of the tsetse fly Glossina pallidipes (Diptera, Glossinidae): implications for forecasting climate change impacts. J. Insect Physiol. 54, 114–127 (2008).
Hu, C., Hou, M., Wei, G., Shi, B. & Huang, J. Potential overwintering boundary and voltinism changes in the brown planthopper, Nilaparvata lugens, in China in response to global warming. Climatic Change 132, 337–352 (2015).
Hendrix, D. L. & Salvucci, M. E. Polyol metabolism in homopterans at high temperatures: accumulation of mannitol in aphids (Aphididae: Homoptera) and sorbitol in whiteflies (Aleyrodidae: Homoptera). Comp. Biochem. Physiol. A 120, 487–494 (1998).
Ghaedi, B. & Andrew, N. R. The physiological consequences of varied heat exposure events in adult Myzus persicae: a single prolonged exposure compared to repeated shorter exposures. PeerJ 4, e2290 (2016).
Danks, H. V. Dehydration in dormant insects. J. Insect Physiol. 46, 837–852 (2000).
Ma, G. & Ma, C.-S. Climate warming may increase aphids’ dropping probabilities in response to high temperatures. J. Insect Physiol. 58, 1456–1462 (2012).
Alford, L., Andrade, T. O., Georges, R., Burel, F. & van Baaren, J. Could behaviour and not physiological thermal tolerance determine winter survival of aphids in cereal fields? PloS ONE 9, e114982 (2014).
Renault, D., Vernon, P. & Vannier, G. Critical thermal maximum and body water loss in first instar larvae of three Cetoniidae species (Coleoptera). J. Thermal Biol. 30, 611–617 (2005).
Heath, J. E., Hanegan, J. L., Wilkin, P. J. & Heath, M. S. Adaptation of the thermal responses of insects. Am. Zool. 11, 147–158 (1971).
Heong, K. L. & Hardy, B. Planthoppers: new threats to the sustainability of intensive rice production systems in Asia. Los Baños (Philippines): International Rice Research Institute (2009).
Chidawanyika, F. & Terblanche, J. S. Rapid thermal responses and thermal tolerance in adult codling moth Cydia pomonella (Lepidoptera: Tortricidae). J. Insect Physiol. 57, 108–117 (2011).
Dillon, M. E., Wang, G. & Huey, R. B. Global metabolic impacts of recent climate warming. Nat. 467, 704–706 (2010).
Wang, H. et al. Transcriptome analysis of the Bombyx mori fat body after constant high temperature treatment shows differences between the sexes. Mol. Biol. Rep. 41, 6039–6049 (2014).
Wang, H. D., Zhu, J. L., Zhu, L. M., Lu, Q. & Wang, J. L. Study on assessment of yield loss caused by small brown planthopper and control threshold at heading stage. Plant Prot. 27, 17–20 (2007).
Powell, S. J. & Bale, J. Effect of long-term and rapid cold hardening on the cold torpor temperature of an aphid. Physiol. Entomol. 31, 348–352 (2006).
Shen, J. & Wu, Y. Insecticide resistance in cotton bollworm and its management. China Agricultural Press, Beijing, China (1995).
Acknowledgements
We thank Chunsen Ma for allowing us using the programmed alcohol bath and Fei Li, Xiaoqin Sun, and Huan Liu for technical assistance with the experiments. This research was financially supported by the National Key R&D Program of China (2016YFD0300701, 2018YFD0200306). The granting agency had no role in the design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
M.H. conceived and together with S.A. designed the experiments. S.A. and P.L. performed the experiments. M.H. contributed reagents/materials. S.A. and M.H. analyzed the results. S.A., P.L. A.A. and M.H. joined writing and reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ali, S., Li, P., Ali, A. et al. Comparison of upper sublethal and lethal temperatures in three species of rice planthoppers. Sci Rep 9, 16191 (2019). https://doi.org/10.1038/s41598-019-52034-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-52034-7
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.