Abstract
Recent studies including the SPRINT trial have shown beneficial effects of intensive systolic blood pressure reduction over the standard approach. The awareness of the J-curve for diastolic blood pressure (DBP) causes some uncertainty regarding the net clinical effects of blood pressure reduction. The current analysis was performed to investigate effects of low on-treatment DBP on cardiovascular risk in the SPRINT population. The primary composite outcome was the occurrence of myocardial infarction, acute coronary syndrome not resulting in myocardial infarction, stroke, acute decompensated heart failure or death from cardiovascular causes. The prevalence of primary outcomes was significantly higher in subjects within low DBP in both standard (44–67 mmHg [10.8%] vs 67–73 mmHg [6.7%] vs 73–78 mmHg [5.1%] vs 78–83 mmHg [4.4%] vs 83–113 mmHg [4.3%], p < 0.001) and intensive treatment (38–61 mmHg [6.7%] vs 61–66 mmHg [4.1%] vs 66–70 mmHg [4.5%] vs 70–74 mmHg [2.7%] vs 74–113 mmHg [3.4%], p < 0.001) arms. After adjusting for covariates, low DBP showed no significant effects on cardiovascular risk. Therefore, while reaching blood pressure targets, low DBP should not be a matter of concern.
Similar content being viewed by others
Introduction
The American College of Cardiology, American Heart Association and the European Society of Cardiology, recently adopted new goals for antihypertensive therapy following the release of results from new clinical studies including the Systolic Blood Pressure Intervention Trial (SPRINT)1,2,3. SPRINT showed that achieving systolic blood pressure (SBP) of less than 120 mmHg, rather than 140 mmHg, contributed to reduced risks of fatal and nonfatal major cardiovascular events and overall mortality. The new recommendations supporting intensive blood pressure reduction meet the fact that a significant percentage of patients receiving antihypertensive therapy still do not achieve their target BP values4. While lower BP reduced the risk of cardiovascular events and improved survival, it also created doubts regarding the J-curve hypothesis. In 1979 it was shown that lower diastolic blood pressure (DBP) may increase the risk of myocardial infarction among hypertensive patients5,6. The results of other studies addressing this issue confirmed that intensive DBP reduction may increase the risk of coronary artery disease (CAD)7,8. There are few studies published before and after SPRINT which established an optimal target DBP. Randomised trials showed inconclusive results or considered out-dated DBP targets9,10. Other investigations comprised post-hoc analyses of randomised trial participants, including the SPRINT population11,12,13,14,15. Overall, the results of these studies were incoherent, applied methodologies differed between studies and investigators pooled data from different studies, such as one report which combined data from SPRINT and the Action to Control Cardiovascular Risk in Type 2 Diabetes (ACCORD). The International Verapamil–Trandolapril Study (INVEST) study focused on hypertensive subjects with stable CAD and J-curve relationships between both systolic and diastolic BP and primary outcomes were found16. However, the J- shape of the curve was more pronounced for DBP than for SBP, and the risk of myocardial infarction and death increased when the DBP was <75 mmHg16. Considering these findings, it is still unclear whether a substantial BP reduction is beneficial and safe among all subgroups of hypertensive patients. In the SPRINT study, intensive control of SBP reduced the risk of cardiovascular disease among high-risk patients1. In contrast, the ACCORD study, which included diabetic patients, demonstrated that a similar reduction in SBP did not affect cardiovascular morbidity and mortality17. In this study, we evaluated the effects of low on-treatment DBP on primary composite outcomes and investigated whether other clinical factors influenced these outcomes in hypertensive subjects.
Results
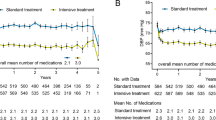
A total of 8890 participants (64.7% men, 35.3% women) were included in this study. Among these participants, 4438 (49.9%) of them were allocated to the standard (<140 mmHg) treatment arm while 4452 (50.1%) patients were placed in the intensive treatment arm (<120 mmHg). The primary composite outcome occurred a total of 461 (5.2%) subjects, including 272 (6.1%) from the standard and 189 (4.2%) from the intensive treatment arm. The overall mean SBP/DBP was 128.1/71.4 mmHg, while mean SBP/DBP in standard and intensive treatment arm were 135.3/75.1 mmHg and 120.9/67.7 mmHg.
The quintiles of DBP were calculated for the standard (1st quintile 44–67 mmHg, 2nd 67–73 mmHg, 3rd 73–78 mmHg, 4th 78–83 mmHg, 5th 83–113 mmHg) and intensive treatment arm (1st quintile 38–61 mmHg, 2nd 61–66 mmHg, 3rd 66–70 mmHg, 4th 70–74 mmHg, 5th 74–113 mmHg). There were significant differences in the occurrence of primary endpoints within quintiles of both treatment arms (standard: 10.8% vs 6.7% vs 5.1% vs 4.4% vs 4.3%, p < 0.001; intensive: 6.7% vs 4.1% vs 4.5% vs 2.7% vs 3.4%, p < 0.001). Kaplan–Meier curves showing survival rates among the arms are presented in Figs 1 and 2. Log-rank test confirmed significant differences between curves among the DBP quintiles in the standard and intensive treatment arms (p < 0.0001). The clinical characteristics of the quintiles in each treatment arm are shown in Tables 1 and 2.
The highest rates cardiovascular and renal disease and older age were observed among the quintiles with the highest incidence of primary endpoints. Despite having lower SBP, worse prognoses were observed in the quintiles with lower DBP. The patients in quintiles with lower DBP received more antihypertensive drugs. The difference in pulse pressure was also observed between the quintiles in both treatment arms. The patients with lower on-treatment DBP had also lower baseline DBP. Higher use of statins and aspirin was associated with better lipid profile, but this result was observed in the quintiles with worse outcomes. Similar findings were observed in both treatment arms.
The primary outcome rates were compared between the corresponding DBP quintiles in standard and intensive treatment arm. When the 1st and 2nd quintiles were compared, there was a higher incidence of primary outcomes in the standard treatment arm compared to the intensive treatment arm (10.8% vs 6.7%, p = 0.003 and 6.7% vs 4.1%, p = 0.023). There was no significant difference in between the 3rd, 4th and 5th DBP quintiles in the two treatment arms (5.1% vs 4.5%, p = 0.649; 4.4% vs 2.7%, p = 0.064, 4.3% vs 3.4%, p = 0.342).
Among the standard and intensive treatment arms, on-treatment DBP was a poor predictor of primary endpoints. Receiver operating characteristic (ROC) analysis showed the following area under the curve (AUC) measurements: 0.605 (95% CI: 0.569–0.641) and 0.581, 95% CI: 0.536–0.626 (Fig. 3).
We used Cox proportional hazard risk model to assess the impact of on-treatment DBP and pulse pressure on primary endpoint event risks among the total analysed population of patients. After adjusting for age, sex, history of clinical cardiovascular disease or chronic kidney disease, treatment arm, current smoking status and body mass index, our analysis revealed no influence of on-treatment DBP on the occurrence of primary outcome (Table 3). However, on-treatment pulse pressure was shown to be an independent predictor of primary outcome event risks after adjusting for other cofactors in the multivariable Cox model (Table 3). The analysis comparing participants in the quintiles of total analysed population is shown in Table S1 and Fig. S1.
Discussion
This study shows that DBP is not an independent risk factor for cardiovascular events. Worse prognoses among participants with low on-treatment DBP may be explained by the fact that these patients were older, often had previous cardiovascular disease or chronic kidney disease and more often were smokers. The impact of those factors on the primary endpoints was high, what is expressed by hazard ratios in Table 3.
Three other studies based on SPRINT data investigated the J-curve hypothesis of DBP. Kalkman et al. re-analysed merged SPRINT and ACCORD data to verify the J-shaped curve hypothesis related to SBP and DBP13. The authors found an increased risk associated with lower on-treatment DBP with a nadir at 85 mmHg, which indicated that their results confirmed J-shaped curve hypothesis. However, the impact of potential interactions between low DBP and confounding variables was not assessed. In our study, the influence of low on-treatment DBP on primary outcomes disappeared after adjusting for confounding factors.
In another paper re-analysing the SPRINT data, Stensrud et al. assumed, according to the results of previous studies, that mean DBP < 60 mmHg may worsen the effects of lowering BP14,18. After adjustment for potential confounding variables, the authors did not observe unfavourable effects of DBP < 60 mmHg. It is important to note that the authors used a cut-off value of DBP < 60 mmHg while in other studies, DBP < 70 mmHg was used as the lower limit for DBP19,20,21. These results may imply that DBP < 60 mmHg did not increase patients’ risk when cofactors were included in the analysis. Also, recent European guidelines selected DBP = 70 mmHg as a lower limit for recommended on-treatment DBP3. The role of pulse pressure should be also emphasised here. We found that on-treatment pulse pressure is an independent risk factor for primary endpoint events, even after adjusting for confounding factors. In the study examining SPRINT data, Pareek et al. showed that the excess risk associated with higher pulse pressure at baseline appeared to be related to age, sex and other major cardiovascular risk factors, thus limiting its clinical utility among high-risk SPRINT patients22. Authors concluded that baseline pulse pressure should not limit the selection of appropriate candidates for intensive BP reduction. However, some studies found that baseline pulse pressure might be an independent risk factor for acute coronary events23.
The SPRINT data analysis was also performed by Lee et al.15. The authors determined that DBP < 55 mmHg at during a single visit was associated with adverse clinical effects. The adopted methodology evaluated the effect of single episode of hypotension rather than examining the effect of permanently low on-treatment DBP on adverse events. In contrast to the results of a study by Stensrud et al., DBP < 55 mm at a single visit significantly increased the risk of complications according to the results of a multivariable Cox proportional analysis.
According to the results of previous SPRINT re-analyses, our study involved the comparison within the quintiles of DBP in both treatment arms. This methodology helped reveal the key differences within subpopulations across on-treatment DBP levels and the necessity of assessing the impact of confounding factors.
The results of other studies investigating the DBP-related J-curve hypothesis were ambiguous. The differences between analysed endpoints, DBP thresholds and study populations (baseline prevalence of CAD, diabetes mellitus or stroke) made it difficult to draw clear conclusions and compare results with our results.
The results of HOT trial (Hypertension Optimal Treatment) did not reveal differences in the incidence of stroke risk, overall and cardiovascular-related mortality and myocardial infarction between the subgroups with DBP ≤90, ≤85 and ≤80 mmHg. The incidence of all myocardial infarctions was almost significant, and the authors concluded that optimal DBP is 82.6 mmHg10. In the Hypertension Objective Treatment Based on Measurement by Electrical Devices study (HOMED-BP), the impact of usual (125–134/80–84 mmHg) vs. tight (<125/80 mmHg) BP control was investigated24. The authors did not evidence regarding a J-shaped curve relationship between BP reduction and clinical endpoints24. According to the J-shaped curve hypothesis, low compliance among the tight control arm, on-treatment DBP may be higher than unsafe BP levels. Post-hoc analyses of The Valsartan Antihypertensive Long-Term Use Evaluation and INVEST trials did not provide data on the detrimental effects of low on-treatment DBP11,25. Other re-analyses confirmed the J-shaped hypothesis for on-treatment DBP in subjects of Ongoing Telmisartan Alone and in combination with Ramipril Global Endpoint Trial (ONTARGET) and merged ONTARGET and Telmisartan Randomised Assessment Study in ACE Intolerant Participants with Cardiovascular Disease (TRANSCEND) trials12,19,26. The J-shaped curve relationship between DBP and cardiovascular mortality was also observed after long-term follow-up in the analysis of the National Health and Nutrition Examination Survey III and Diabetes Heart Study performed by Gomadam et al.27 Our multivariable Cox model showed that DBP was not a significant cofactor. Based on the data obtained from this study and previous studies, the evidence for a J-shaped relationship between DBP and cardiovascular risk is inconclusive. Despite the weakness of evidence regarding the J-shaped curve hypothesis, this phenomenon is more pronounced in the subjects with CAD28,29. Furthermore, CAD is more frequent in the subjects with lower DBP, which suggests that this effect may be cumulative20,30,31,32. In our analysis of individuals with a high risk of cardiovascular disease, including current CAD patients, we found no evidence that a J-shaped curve existed for on-treatment DBP when some cofactors were included into the analysis.
Despite the fact that coronary blood flow primarily occurs during diastole and depends on DBP, it should be emphasised that the clinical effects of DBP cannot be interpreted separately from on-treatment SBP. It is difficult to decide on a patient-by-patient basis whether the benefit of intensive SBP reduction outweighs the harms associated with low DBP. Beddhu et al. evaluated the interaction between low baseline DBP and intensive SBP reduction among SPRINT participants and found adverse effects associated with intensive treatment to lower SBP among patients with low baseline DBP33. Our comparison of corresponding quintiles of DBP in both treatment arms suggests better outcomes are associated with intensive rather than standard treatment, despite lower on-treatment DBP. According to these results, on-treatment DBP should be only interpreted as the consequence of SBP lowering and SBP < 130 mmHg should be considered a treatment goal.
The most important limitation to our study is the fact that despite the large sample size, post-hoc reasoning could be affected by potential bias. Secondly, there were essential differences identified between automated and clinical BP measurements. Therefore, it was difficult to compare our results to those of the SPRINT trial due to differences in methods34,35. The studies performed by Kalkman, Stensrud and Lee had the same disadvantage; therefore, their choice of corresponding harmful DBP threshold may be also questioned. Furthermore, our findings do not apply to diabetic subjects or these with the history of stroke. However, the ACCORD and SPS3 trials already investigated these subgroups17,18. Considering the results of Di Nora et al., who showed that the SPRINT results were not reproducible within a community-based cohort of Caucasian participants, our results could also vary based on the characteristics of the investigated population36.
In conclusion, our data showed that low on treatment DBP does not independently influence cardiovascular risk in patients allocated to intensive and standard blood pressure lowering strategies. These results are in line with the conclusions of similar studies11,12,13,14,27. In that context, low on-treatment DBP should be considered as a marker of high cardiovascular risk rather than an independent risk factor26.
Methods
Study population
SPRINT was a randomised and multi-centre study that determined whether patients benefitted from intensive efforts to decrease SBP1. Participants were randomised to intensive (target SBP < 120 mmHg) and standard treatment (target SBP < 140 mmHg) arms. Although a target of DBP was not established, patients were treated to achieve DBP of <90 mmHg after meeting their goal SBP measurement37. Nearly 10,000 subjects who presented high risks of cardiovascular diseases and adverse cardiovascular events (older age; SBP, 130–180 mmHg and the history of cardiovascular or chronic kidney disease or Framingham Risk Score for 10-year cardiovascular risk >15%) were enrolled in the study. Eligibility and exclusion criteria are described previously38.
BP measurements and other clinical data
SBP and DBP were measured three times per visit using an automated system (Model 907, Omron Healthcare). According to the study protocol, we decided to analyse the interval from the 6-month visit until the end of the study due to the stability of BP values among the participants. We used the mean of BP measurements on each visit during the analysed period to describe on-treatment DBP and SBP. The pulse pressure was calculated as the difference between SBP and DBP. Other data including age, body mass index, height, lipid profile, blood glucose levels and overall medical history were considered.
Outcome
The primary composite outcomes of the SPRINT trial included myocardial infarction, acute coronary syndrome not resulting in myocardial infarction, stroke, acute decompensated heart failure or death from cardiovascular complications. SPRINT proved that allocation to the intensive treatment arm improved cardiovascular outcomes among hypertensive patients with a high risk of cardiovascular disease. We decided to investigate the association between DBP and primary outcomes, as defined by the original trial.
Data source
Anonymised data (SPRINT_ POP Research Materials) for the current analysis were obtained from the National Heart, Lung and Blood Institute (NHLBI) Biologic Specimen and Data Repository Information Coordinating Centre. This manuscript does not necessarily reflect the opinions or views of the SPRINT_POP or the NHLBI. Our study received the approval of the Ethics Committee at Medical University of Warsaw. The SPRINT study was approved by the institutional review board at each participating study site. Informed consent was obtained from all participants. All methods were carried out in accordance with relevant guidelines and regulations.
Statistical analysis
This was a retrospective study. The investigations were performed within the subgroups selected by DBP quintiles separately for the subjects assigned to intensive and standard treatment arms. Continuous data were presented as mean and standard deviation. Discrete values were presented as number and percentage. The analysis of variance and Chi Square (χ2) tests were used for between-arm comparisons. Survival was analysed using Kaplan–Meier curves, and log-rank test was applied to assess difference in survival curves. ROC curve analysis was used to assess the predictive value of DBP. A Cox proportional hazard risk model was used to explain the impact of the independent variable (DBP) and other predictors on the occurrence of primary composite outcomes. A p-value < 0.05 was considered significant. All employed tests were two-tailed. All calculations were made using R 3.4.0 software (R Foundation for Statistical Computing, Vienna, Austria) for statistical computations. Standard, ‘survival’, ‘survminer’ and ‘pROC’ packages were used39,40,41,42.
Data Availability
Data that support the findings of this study are available from the NHLBI, but restrictions apply regarding the availability of these data. Since these data are under the licence for the current study, they are not publicly available. The data are available from NHLBI upon reasonable request. The authors have no right to share the data.
References
Wright, J. T. et al. A Randomized Trial of Intensive versus Standard Blood-Pressure Control. NEJM 373, 2103–2116, https://doi.org/10.1056/NEJMoa1511939 (2015).
Whelton, P. K. et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. JACC, https://doi.org/10.1016/j.jacc.2017.11.006 (2017).
Williams, B. et al. 2018 ESC/ESH Guidelines for the management of arterial hypertension: The Task Force for the management of arterial hypertension of the European Society of Cardiology and the European Society of Hypertension: The Task Force for the management of arterial hypertension of the European Society of Cardiology and the European Society of Hypertension. J Hypertens 36, 1953–2041, https://doi.org/10.1097/hjh.0000000000001940 (2018).
Muntner, P. et al. Potential U.S. Population Impact of the 2017 ACC/AHA High Blood Pressure Guideline. JACC 71, 109–118, https://doi.org/10.1016/j.jacc.2017.10.073 (2018).
Stewart, I. M. Relation of reduction in pressure to first myocardial infarction in patients receiving treatment for severe hypertension. Lancet 1, 861–865 (1979).
Stewart, I. M. Lowering blood-pressure. Lancet 2, 422 (1979).
Cruickshank, J. M., Thorp, J. M. & Zacharias, F. J. Benefits and potential harm of lowering high blood pressure. Lancet 1, 581–584 (1987).
Alderman, M. H., Ooi, W. L., Madhavan, S. & Cohen, H. Treatment-induced blood pressure reduction and the risk of myocardial infarction. JAMA 262, 920–924 (1989).
Hannson, L. The BBB Study: the effect of intensified antihypertensive treatment on the level of blood pressure, side-effects, morbidity and mortality in “well-treated” hypertensive patients. Behandla Blodtryck Battre. Blood Press 3, 248–254 (1994).
Hansson, L. et al. Effects of intensive blood-pressure lowering and low-dose aspirin in patients with hypertension: principal results of the Hypertension Optimal Treatment (HOT) randomised trial. Lancet 351, 1755–1762, https://doi.org/10.1016/s0140-6736(98)04311-6 (1998).
Kjeldsen, S. E. et al. No evidence for a J-shaped curve in treated hypertensive patients with increased cardiovascular risk: The VALUE trial. Blood Press 25, 83–92, https://doi.org/10.3109/08037051.2015.1106750 (2016).
Sleight, P. et al. Prognostic value of blood pressure in patients with high vascular risk in the Ongoing Telmisartan Alone and in combination with Ramipril Global Endpoint Trial study. J Hypertens 27, 1360–1369, https://doi.org/10.1097/HJH.0b013e32832d7370 (2009).
Kalkman, D. N. et al. J Curve in Patients Randomly Assigned to Different Systolic Blood Pressure Targets: An Experimental Approach to an Observational Paradigm. Circulation 136, 2220–2229, https://doi.org/10.1161/circulationaha.117.030342 (2017).
Stensrud, M. J. & Strohmaier, S. Diastolic hypotension due to intensive blood pressure therapy: Is it harmful? Atherosclerosis 265, 29–34, https://doi.org/10.1016/j.atherosclerosis.2017.07.019 (2017).
Lee, T. C., Cavalcanti, R. B., McDonald, E. G., Pilote, L. & Brophy, J. M. Diastolic hypotension may attenuate benefits from intensive systolic targets: secondary analysis of a randomized controlled trial. AJM, https://doi.org/10.1016/j.amjmed.2018.05.022 (2018).
Pepine, C. J. et al. A calcium antagonist vs a non-calcium antagonist hypertension treatment strategy for patients with coronary artery disease. The International Verapamil-Trandolapril Study (INVEST): a randomized controlled trial. JAMA 290, 2805–2816, https://doi.org/10.1001/jama.290.21.2805 (2003).
Cushman, W. C. et al. Effects of intensive blood-pressure control in type 2 diabetes mellitus. NEJM e 362, 1575–1585, https://doi.org/10.1056/NEJMoa1001286 (2010).
McEvoy, J. W. et al. Diastolic Blood Pressure, Subclinical Myocardial Damage, and Cardiac Events: Implications for Blood Pressure Control. JACC 68, 1713–1722, https://doi.org/10.1016/j.jacc.2016.07.754 (2016).
Bohm, M. et al. Achieved blood pressure and cardiovascular outcomes in high-risk patients: results from ONTARGET and TRANSCEND trials. Lancet 389, 2226–2237, https://doi.org/10.1016/s0140-6736(17)30754-7 (2017).
Vidal-Petiot, E. et al. Cardiovascular event rates and mortality according to achieved systolic and diastolic blood pressure in patients with stable coronary artery disease: an international cohort study. Lancet 388, 2142–2152, https://doi.org/10.1016/S0140-6736(16)31326-5 (2016).
Somes, G. W., Pahor, M., Shorr, R. I., Cushman, W. C. & Applegate, W. B. The role of diastolic blood pressure when treating isolated systolic hypertension. Arch Intern Med 159, 2004–2009 (1999).
Pareek, M. et al. Pulse Pressure, Cardiovascular Events, and Intensive Blood-Pressure Lowering in the Systolic Blood Pressure Intervention Trial (SPRINT). Am J Med. 2019 Jan 16, https://doi.org/10.1016/j.amjmed.2019.01.001, [Epub ahead of print].
Glasser, S. P et al. Is pulse pressure an independent risk factor for incident acute coronary heart disease events? The REGARDS study. American Journal of Hypertension, 27(4), 555–563, https://doi.org/10.1093/ajh/hpt168.
Asayama, K. et al. Cardiovascular outcomes in the first trial of antihypertensive therapy guided by self-measured home blood pressure. Hypertens Res 35, 1102–1110, https://doi.org/10.1038/hr.2012.125 (2012).
Messerli, F. H. et al. Dogma disputed: can aggressively lowering blood pressure in hypertensive patients with coronary artery disease be dangerous? Ann Intern Med 144, 884–893 (2006).
Bohm, M. et al. Achieved diastolic blood pressure and pulse pressure at target systolic blood pressure (120–140 mmHg) and cardiovascular outcomes in high-risk patients: results from ONTARGET and TRANSCEND trials. Eur Heart J 39, 3105–3114, https://doi.org/10.1093/eurheartj/ehy287 (2018).
Gomadam, P. et al. Blood pressure indices and cardiovascular disease mortality in persons with or without diabetes mellitus. J Hypertens 36, 85–92, https://doi.org/10.1097/hjh.0000000000001509 (2018).
Mancia, G. & Grassi, G. Aggressive blood pressure lowering is dangerous: the J-curve: pro side of the arguement. Hypertension 63, 29–36, https://doi.org/10.1161/01.hyp.0000441190.09494.e9 (2014).
Verdecchia, P., Angeli, F., Mazzotta, G., Garofoli, M. & Reboldi, G. Aggressive blood pressure lowering is dangerous: the J-curve: con side of the arguement. Hypertension 63, 37–40, https://doi.org/10.1161/01.hyp.0000439102.43479.43 (2014).
Mancia, G. Should blood pressure reduction be aggressive in patients with hypertension and coronary artery disease? Lancet 388, 2061–2062, https://doi.org/10.1016/s0140-6736(16)31398-8 (2016).
Vidal-Petiot, E. et al. Relationships Between Components of Blood Pressure and Cardiovascular Events in Patients with Stable Coronary Artery Disease and Hypertension. Hypertension 71, 168–176, https://doi.org/10.1161/hypertensionaha.117.10204 (2018).
Unni, G. J-Curve Phenomenon - Current Understanding and Clinical Implications. Hypertension J 2, 16–20, https://doi.org/10.5005/jp-journals-10043-0024 (2016).
Beddhu, S. et al. Influence of baseline diastolic blood pressure on effects of intensive compared with standard blood pressure control. Circulation 137, 134–143, https://doi.org/10.1161/CIRCULATIONAHA.117.030848 (2018).
Agarwal, R. Implications of Blood Pressure Measurement Technique for Implementation of Systolic Blood Pressure Intervention Trial (SPRINT). JAHA 6, https://doi.org/10.1161/jaha.116.004536 (2017).
Filipovsky, J. et al. Automated compared to manual office blood pressure and to home blood pressure in hypertensive patients. Blood Press 25, 228–234, https://doi.org/10.3109/08037051.2015.1134086 (2016).
Di Nora, C. et al. Systolic blood pressure target in systemic arterial hypertension: Is lower ever better? Results from a community-based Caucasian cohort. Eur J Intern Med. 48, 57–63, https://doi.org/10.1016/j.ejim.2017.08.029 (2018).
Systolic Blood Pressure Intervention Trial (SPRINT) protocol, https://www.sprinttrial.org/public/Protocol_Current.pdf.
Ambrosius, W. T. et al. The design and rationale of a multicenter clinical trial comparing two strategies for control of systolic blood pressure: the Systolic Blood Pressure Intervention Trial (SPRINT). Clin Trials 11, 532–546, https://doi.org/10.1177/1740774514537404 (2014).
R Core Team. R: A language and environment for statistical computing. R Foundation for Statistical Computing, https://www.R-project.org/.
Therneau, T. A Package for Survival Analysis in S. Version 2.38, https://CRAN.R-project.org/package=survival.
Kassambara, A. Drawing survival curves using ‘ggplot2’, https://CRAN.R-project.org/package=survminer.
Robin, X. et al. pROC: an open-source package for R and S+ to analyze and compare ROC curves. BMC bioinformatics 12, 77, https://doi.org/10.1186/1471-2105-12-77 (2011).
Author information
Authors and Affiliations
Contributions
All the authors contributed to the conception of the study, interpretation of the results, prepared, revised and approved the manuscript. P.S. performed all statistical analyses. P.S. and J.L. performed the literature search.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sobieraj, P., Lewandowski, J., Siński, M. et al. Low on-treatment diastolic blood pressure and cardiovascular outcome: A post-hoc analysis using NHLBI SPRINT Research Materials. Sci Rep 9, 13070 (2019). https://doi.org/10.1038/s41598-019-49557-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-49557-4
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.