Abstract
Parkinson’s disease (PD) is a progressive, neurodegenerative movement disorder characterized by the loss of dopaminergic (DA) neurons. Limited understanding of the early molecular pathways associated with the demise of DA neurons, including those of inflammatory exacerbation of neurodegeneration, is a major impediment to therapeutic development. Recent studies have implicated gene-environment interactions in PD susceptibility. We used transcriptomic profiling in a Drosophila PD model in response to paraquat (PQ)-induced oxidative stress to identify pre-symptomatic signatures of impending neuron dysfunction. Our RNAseq data analysis revealed extensive regulation of innate immune response genes following PQ ingestion. We found that PQ exposure leads to the activation of the NF-κB transcription factor, Relish, and the stress signaling factor JNK, encoded by the gene basket in Drosophila. Relish knockdown in the dopaminergic neurons confers PQ resistance and rescues mobility defects and DA neuron loss. Furthermore, PQ-induced toxicity is mediated through the immune deficiency signaling pathway. Surprisingly, the expression of Relish-dependent anti-microbial peptide (AMPs) genes is suppressed upon PQ exposure causing increased sensitivity to Gram-negative bacterial infection. This work provides a novel link between PQ exposure and innate immune system modulation underlying environmental toxin-induced neurodegeneration, thereby underscoring the role of the innate immune system in PD pathogenesis.
Similar content being viewed by others
Introduction
Parkinson’s disease (PD) is an age-associated neurodegenerative disorder primarily caused by the progressive loss of dopaminergic (DA) neurons from the substantia nigra in the brain1. The clinical motor symptoms include rest tremor, rigidity, progressive bradykinesia and postural instability. Apart from the motor deficits, PD is also linked to several non-motor symptoms, including sleep disorder, depression, constipation, anxiety, impaired reaction time, that often manifest during the early pre-clinical stages of PD2. Although the precise molecular mechanisms involved in the neurodegenerative process remain unclear, there is increasing evidence that PD is a complex multifactorial disorder caused by a combination of genetic and environmental factors, which affect multiple key signaling pathways in different cell types leading to the loss of DA neurons3,4. One of the major challenges to therapeutic development is the lack of clear understanding of the pre-symptomatic molecular pathways that are either triggered or suppressed in PD pathogenesis.
Chronic neuroinflammation is a common emerging hallmark of several neurodegenerative diseases, including PD5. Post-mortem studies of brains indicate activation of innate immune glial cells and elevated levels of pro-inflammatory factors as common features of PD patients6,7. Activated glial cells induce inflammatory responses to promote defense against pathogen invasion or tissue damage8. However, improper resolution leads to uncontrolled persistent inflammation that contributes to neurotoxicity. Expression of several Toll-like receptors (TLRs), which are responsible for initiating the inflammatory response, has been observed in post-mortem PD brain samples9. Moreover, TLR4 has been implicated in the abnormal deposition of α-synuclein, a protein that accumulates in brain cells of PD patients10. Consistent with these findings, several recent studies demonstrate that anti-inflammatory compounds exhibit significant protective functions for DA neurons in PD models. Treatment with the synthetic anti-inflammatory steroid, dexamethasone, had a beneficial effect against neurodegeneration and reduced activation of glial cells in mouse PD models11. Administration of minocycline, a tetracycline derivative, effectively protects DA neurons in both mouse and Drosophila models by regulating the mitogen-activated protein kinase (MAPK) signaling pathways, which plays a critical role in controlling the expression of pro-inflammatory genes12,13. In mammalian PD models, activation of c-Jun N-terminal Kinase (JNK) has been implicated in PQ-induced oxidative stress and neurodegeneration14. Moreover, in transgenic mice models, activated JNK has been detected in a leucine-rich repeat kinase 2 (LRRK2) mutant, a gene linked to autosomal dominant familial PD15. These findings provide strong link between dysregulated inflammatory responses and PD pathogenesis.
Over the last 15 years, investigations of neurodegenerative disease have incorporated invertebrate models such as Drosophila melanogaster and Caenorhabditis elegans on the principle that cellular and molecular mechanisms of neurodegeneration, metabolism, stress response and neuronal function are highly conserved16,17,18. These models offer powerful genetic tools and short generation times to provide an entrée into genetic screens for identifying potentially important network components and cellular responses that may then be validated in cell culture or in vivo mammalian models. Significant effort has been devoted to identifying early signatures of neurodegenerative disease onset, with the goal of intervention at a point presumably more amenable to modulation or cure. RNA sequencing (RNAseq) transcriptome analysis has emerged as a powerful strategy to investigate differential gene regulation and to identify early and predictive molecular signatures of neurodegenerative disease19,20. However, most of the reported transcriptomic studies have mainly focused on genetic PD models, and it is not clear whether genetic mutation and environmental insult individually trigger the same molecular responses or distinct, interacting pathways.
There is accumulating evidence for increased innate immune activation in PD21. In Drosophila, the innate immune response to bacterial and fungal pathogens is mainly controlled by two signaling cascades, the Toll and the immune deficiency (IMD) pathways, both of which activate members of the Nuclear Factor kappa B (NF-κB) family of transcription factors22. The Toll pathway primarily responds to infections with fungi and Gram-positive bacteria, while the IMD pathway is predominantly activated by infection with Gram-negative bacteria. Peptidoglycans derived from the Gram-negative bacterial cell wall bind to the transmembrane peptidoglycan recognition protein (PGRP)-LC or the intracellular PGRP-LE receptors. Following receptor dimerization, the adaptor protein IMD initiates intracellular signaling by associating with Fas-associated death domain protein (FADD), which then recruits the caspase-8 homolog death related ced-3/Nedd2-like caspase (DREDD). DREDD cleaves off the amino-terminal portion of IMD and stabilizes through IMD ubiquitination. Activated DREDD also cleaves the inhibitory C-terminal domain of Relish, a homologue of mammalian NF-κB23. Relish, the key transcription factor activated by stimulation of the IMD signaling pathway, undergoes nuclear translocation upon activation, leading to the transcription and synthesis of antimicrobial peptides (AMPs)24. In mammalian models, NF-κB also plays an essential role in the inflammatory response through regulation of genes encoding pro-inflammatory mediators25. Increased NF-κB activation has been detected in the brains of PD patients, emphasizing the link between immune activation and neurodegenerative disease26. Similarly, increased NF-κB activation has been linked to age-related neurodegeneration in Drosophila27. Therefore, tight regulation of the IMD pathway is crucial to avoid uncontrolled chronic inflammation. The immune pathways are regulated by several negative feedback loops to avoid prolonged inflammatory responses28. Earlier studies have shown that flies with mutations in a gene that encodes a repressor of the IMD pathway, defense repressor 1 (dnr1) display neurodegeneration due to increased expression of Relish target genes encoding AMPs29. In addition, flies with mutations in ATM, a gene associated with the neurodegenerative disease ataxia-telangiectasia (A-T) in humans, show elevated levels of AMPs in glial cells demonstrating that Relish is involved in the development of the degenerative disease pathology30,31. A recent study in humans links the immune system to PD by demonstrating a heightened immune response to α-synuclein32. We have previously reported that PQ treatment induces an inflammatory response that parallels mammalian neuroinflammation through the induction of nitric oxide synthase (NOS), a critical component of the Drosophila innate immune response33. Increasing evidence suggests a critical role of chronic intestinal inflammation to PD pathogenesis34. It has also been hypothesized that activation of innate immune genes could play a neuroprotective role35. Altogether, these findings demonstrate that dysregulation of immune pathways in Drosophila also can contribute to neurodegeneration, as in mammals. Given the striking parallels of Drosophila and mammalian inflammatory responses during neurodegeneration, we postulated that controlled induction of neurodegeneration in Drosophila would be an entrée into the earliest cellular responses to neuron insult and that many of these responses will be conserved.
Epidemiological studies have identified the herbicide, paraquat (PQ), as a potential environmental risk factor in the onset of PD36,37,38,39. Previously, we found that exposure of adult flies to low doses of PQ (3–20 mM) resulted in a recapitulation of parkinsonism movement disorders, including tremors, loss of balance and slowed locomotion, accompanied by loss of dopaminergic neurons, with regionally specific differences in neuron susceptibility3,13. In the present study, we employed transcriptomic profiling of gene expression responses to transient PQ exposure using Drosophila heads to build a temporal landscape of pre-symptomatic gene responses to neuron damage – that is, prior to acquisition of movement abnormalities. Our RNAseq data analysis revealed a predominant genetic signature of innate immune response genes immediately following PQ exposure. Interestingly, we found that PQ treatment leads to Relish and JNK activation, and that targeted knockdown of relish in DA neurons confers increased resistance to PQ. Moreover, PQ-induced toxicity involves components of the IMD signaling pathway. Strikingly, activated Relish fails to induce AMP expression leading to increased sensitivity to Gram-negative bacterial infection following PQ treatment. Together, our findings indicate a novel link between environmental-toxin induced PD pathogenesis and innate immune gene regulation, leading to increased susceptibility to bacterial infection.
Results
Transcriptomic profiling reveals early gene expression responses to transient paraquat exposure
In order to capture the early, pre-degenerative phase of differential gene responses to PQ exposure, we performed RNAseq profiling of heads from the wild type (WT) Drosophila strain, Canton S, that were fed either 2.5% sucrose (control) or 5 mM PQ in 2.5% sucrose for 12 h (Fig. S1). We used male flies in our study because we previously determined that they are more sensitive to PQ toxicity during the first week of adult life and display parkinsonian symptoms earlier than female flies3. Gene ontology (GO) enrichment analysis was performed on the list of differentially expressed mRNAs using Database for Annotation, Visualization and Integrated Discovery (DAVID) v6.8 (Fig. 1a). We identified 124 genes that were differentially regulated in response to PQ (Supplemental Table 1); these were enriched for biological processes (BP) that include innate immunity, oxidation-reduction, glutathione metabolic process, response to heat and response to nicotine based on Gene ontology (GO) analysis (Fig. 1a and Table 1). The GO terms represented in Table 1 were filtered with cut-off thresholds that only show BP categories with 3 or more associated genes. Using quantitative real time polymerase chain reaction (qRT-PCR), we subsequently validated PQ-responding candidate genes that were either upregulated or downregulated for the following enriched BP categories, including, the innate immune response and antibacterial defense response (nimrodB5, sr-CI, totA, hemolectin, diptericinB, attacin-B, attacin-C, drosomycin, defensin), oxidation-reduction (Gadd45) and response to heat (Hsp68) (Fig. 1b,c).
Paraquat exposure regulates innate immune genes in a Drosophila model of PD. (a) Gene ontology (GO) biological processes enrichment analysis of PQ exposure in adult male Drosophila heads show extensive regulation of innate immune response genes. RNAseq analysis were performed using four independent biological samples from both sucrose (control) and PQ-fed flies. GO analysis was performed on the differentially expressed mRNAs between sucrose (control) and PQ-fed samples using DAVID v6.8 functional annotation analysis. Bars represent the percentage of associated genes for enriched biological processes in the PQ-fed group. (b,c) Validation of selected RNAseq-based candidate gene expression that are either upregulated or downregulated by qRT-PCR. RNA was isolated from heads of Canton S WT male flies following treatment with 2.5% sucrose (control) or 5 mM PQ in 2.5% sucrose for 12 h. Selected differentially expressed transcripts identified through RNAseq profiling were verified using qRT-PCR and plotted after normalization with rp49 levels as the internal control. Each data point represents the mean +/− SEM. Values represent the fold-change of indicated genes after PQ exposure compared with sucrose-fed controls. mRNA fold changes are normalized to the sucrose-fed flies (assigned a value of 1). Statistical significance between the sucrose and PQ-fed groups were calculated using the Mann-Whitney U test. *P < 0.05; **P < 0.01. The results are the averages of at least three independent biological replicates.
Among the induced expression of genes appearing in the GO terms of enriched BP of oxidation-reduction was the Growth Arrest and DNA-Damage-inducible protein 45 (Gadd45) transcript (Table 1 and Fig. 1b). Mammalian GADD45 functions in DNA-repair in response to environmental and physiological stress, and overexpression of its Drosophila ortholog in the nervous system has been shown to extend life span, delay age dependent neurodegeneration and increase resistance to oxidative stress40,41,42. A targeted knockdown of Gadd45 in dopaminergic neurons revealed decreased survival following PQ exposure (Fig. S2), suggesting a neuroprotective effect of GADD45 against PQ induced neurotoxicity, in parallel with prior reports of Drosophila GADD45 function in the nervous system. Consistent with our findings, GADD45α has also been shown to protect M17 human dopamine neuroblastoma cells against 1-methyl-4-phenylpyridinium (MPP+) toxicity43.
We also identified genes encoding three heat shock proteins (Hsp 22, 26, and 68), and three other turandot genes (totA, tot M, and totX) in the response to heat category, that were induced in response to PQ exposure (Table 1). There is increasing evidence that Hsps play protective roles in response to neurodegenerative diseases including PD, due to their function as molecular chaperones regulating proper protein folding44. Neuron-specific expression of Hsp26 increases life span and enhances stress resistance in Drosophila45. In addition, Hsp70 has been shown to be protective against both PQ-induced neurodegeneration in Drosophila and α-synuclein-induced cellular toxicity in mammalian models46,47.
Another subset of five genes (glutathione-S-transferase D2, D5, E4, E7 and E8) that were differentially regulated by PQ belong to the BP category of glutathione metabolic process. Glutathione S-Transferases (GSTs) are a family of enzymes that catalyze the addition of glutathione to a variety of cellular substrates, including products of reactive oxygen species48. Consistent with our findings, increase in GST activities have been reported in the brains of human PD patients, possibly due to increased levels of oxidative stress in PD pathogenesis48,49.
Another candidate gene, metallothionein D (mtnD), that was not included in Table 1 since it was eliminated due to the three or more associated gene number cut-off threshold for BP categories, is worth noting, as it was strongly induced (9.5 fold compared to sucrose-fed) by PQ ingestion. Metallothioneins are metal-binding proteins shown to be associated with neuroprotection and increased expression of mtnD has been reported in the substantia nigra and cortex of sporadic PD cases50,51. These findings underscore the similarities between the mammalian and the Drosophila disease model due to highly conserved gene responses linked to PD pathogenesis.
The GO term enrichment analysis of biological processes also revealed extensive regulation of innate immune response genes following PQ feeding, with a total of 17 genes that were either induced or repressed (Fig. 1a and Table 1). Many of these genes are expressed predominantly in Drosophila hemocytes, which are functional analogs of mammalian macrophages that show inflammatory responses to PQ exposure. Induced genes expressed in the hemocytes included hemese (he), hemolectin (hml), two nimrod genes, (nimB5 and nimC4), three turandot genes (totA, tot M, and totX), and scavenger receptor C1 (sr-C1), suggesting involvement of the Drosophila immune response in PQ-induced neurodegeneration (Fig. 1b and Table 1). The Nimrod and Turandot gene families are important components of the Drosophila innate immune response that participate in defense mechanisms through phagocytosis of either bacteria, apoptotic cells or both52,53,54,55.
Surprisingly, the RNAseq screen also revealed PQ-mediated down-regulation of multiple anti-microbial peptide (AMPs) genes downstream of both the IMD and the Toll innate immune pathways (Supplemental Table 1 and Fig. 1c). As part of innate immunity, AMPs are defensive peptides that are induced during inflammation or microbial challenge24,56. This result suggests that PQ-mediated suppression of AMPs might impair the host defense system as part of the aberrant neuroinflammatory responses. Taken together, these findings parallel growing evidence of the contribution of the dysregulated immune system in PD pathogenesis.
Paraquat exposure results in activation of Relish/NF-kB and basket/JNK
Based on the transcriptomic analysis data that revealed differential regulation of immune system genes, with an upregulation of cellular immunity genes but a suppression of AMPs, we next explored the effect of PQ exposure on the levels of the transcription factor Relish, the fly ortholog of mammalian NF-κB. In Drosophila, Relish/NF-κB is mainly activated via the IMD pathway, where it plays a key role in regulating the humoral immune response through the production of AMPs, such as attacin, diptericin, and cecropin. Relish regulates the inducible immune response in Drosophila through signal-dependent translocation into the nucleus; its functions are highly conserved with mammalian NF-κB56. The full-length, 110 kD Relish protein is activated in the cytoplasm by proteolytic cleavage resulting in a 68 kD protein, which is subsequently translocated to the nucleus leading to the regulation of gene expression, and a 49 kDa protein, which remains cytosolic. To determine whether Relish is activated and translocated to the nucleus upon PQ treatment, we performed cellular fractionation using heads of Canton S WT male flies treated with either sucrose (control) or PQ (5 and 10 mM). Nuclear fractionation and immunoblotting data revealed that PQ exposure resulted in a dose-dependent induction of the active nuclear form, p68, while the cytoplasmic full-length p110 form decreased, confirming that PQ elicits activation and nuclear translocation of Relish (Fig. 2a). In addition, relish transcript levels were also induced (around 3-fold) in response to 5 mM PQ treatment in two different wild type strains, Canton S and y w1118 as shown in Fig. 2b. Since 5 mM PQ was capable of inducing Relish activation, we used this concentration for subsequent experiments to better distinguish early responses to PQ exposure. Although relish transcripts were not differentially regulated in our single time point RNAseq screen after normalization, we did observe a marginal induction in the raw RPKM values in the PQ-fed group compared to the sucrose-fed flies.
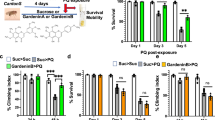
Paraquat activates immune signaling pathways. (a) Relish activation and nuclear translocation in response PQ treatment. Nuclear and cytoplasmic fractionation of heads from Canton S WT flies after sucrose or PQ (5 mM and 10 mM) ingestion for 12 h followed by immunoblot analysis using anti-Relish antibodies. The same blots were probed with anti-Lamin and anti-Tubulin as the nuclear and cytoplasmic markers, respectively for the fractionation protocol. The full-length Relish p110 and p68 are indicated by arrows and ns refers to non-specific band. Band intensities of nuclear Relish p68 were quantitated using the LI-COR C-DiGit Blot scanner software and plotted after normalization with corresponding Lamin band intensities. *p < 0.05 based on Student’s t test. Data are representative of three independent experiments. (b) PQ induces the expression of relish transcript. RNA was isolated from the heads of Canton S and y w1118 wild type flies following 2.5% sucrose or 5 mM PQ treatment for 12 h. The transcript levels of relish were analyzed using qRT-PCR and plotted after normalization with rp49 levels as the internal control. Each data point represents the mean +/− SEM. mRNA fold changes are normalized to the sucrose-fed flies (assigned a value of 1) for each of the wild type strains. *P < 0.05 between the sucrose and PQ-fed group based on Mann-Whitney U test. (c) Paraquat induces JNK activation. Whole cell lysates were prepared using the heads of Canton S flies after 2.5% sucrose or 5 mM PQ ingestion for 12 h followed by immunoblot analysis using phospho-JNK antibodies. The same blots were probed with total JNK and Tubulin antibodies as the loading control. Band intensities were quantitated using the LI-COR C-DiGit Blot scanner software and plotted after normalization with total JNK band intensities. *P < 0.05 based on Student’s t test. Data are representative of three independent experiments. (d) Inhibition of JNK confers PQ resistance. Canton S flies were fed either 2.5% sucrose, 10 mM PQ, 10 μM JNK inhibitor (SP600125) or cofed PQ with the JNK inhibitor diluted in 2.5% sucrose, and survival was scored at the specified time points (hours). Log-rank test was used for survival analysis and statistically significant differences (P < 0.001) were observed between the PQ and PQ + JNKin group. Data are representative of at least five independent biological replicates.
Activation of the Drosophila IMD pathway also triggers an inflammatory stress response via Jun N-terminal kinase (JNK), encoded by the gene basket, as the signaling pathway bifurcates from the IMD signaling cascade at the level of transforming growth factor β-activating kinase 1 (Tak1)57. We therefore investigated whether JNK signaling is affected since PQ exposure is known to induce oxidative stress. Our immunoblotting results show that JNK is activated through phosphorylation in response to PQ feeding as compared to sucrose-fed control flies (Fig. 2c). The same blots were probed with antibodies against total JNK and Tubulin as the loading control. In mammalian PD models, JNK signaling pathway has been shown to play an important role in regulating the cellular processes like oxidative stress and apoptosis14. However, JNK has been shown to be either protective or detrimental in diverse PD models, probably due to differences in cell type specificity, type of stimulus, duration of treatments and genetic backgrounds13,14,47,58. In order to decipher the role of JNK in PQ-mediated toxicity, Canton S WT flies were fed the JNK inhibitor (SP600125) alone or together with PQ and the survival was monitored. Consistent with earlier studies59, flies gained resistance to PQ in the presence of the JNK inhibitor, SP600125 (Fig. 2d). These data indicate that PQ exposure leads to activation of Relish and the stress response JNK pathway and that pharmacological inhibition of JNK enhances survival against PQ toxicity.
Knockdown of relish in dopaminergic neurons confers paraquat resistance
Drosophila innate immune signaling pathways are well-characterized and are highly conserved with vertebrate innate immune pathways22,24. Our studies show that NF-κB transcription factor, Relish is activated in response to PQ feeding without up-regulating the expression of AMPs downstream of the IMD pathway. In fact, the expression of the downstream AMPs was suppressed after PQ exposure. In order to determine the exact role of Relish in PQ-induced PD pathogenesis, we used UAS-RNAi transgenes under the control of tyrosine hydroxylase (TH)-Gal4 to knockdown relish specifically in the DA neurons. As controls, we used both TH-Gal4>+ and UAS-relish-RNAi in the presence or absence of PQ in 2.5% sucrose. We observed that relish knockdown specifically in the DA neurons (TH-Gal4 > UAS-relish-RNAi) leads to significantly increased survival (4 days in average) following PQ exposure compared to sucrose-fed control flies (p < 0.001) (Fig. 3a). To rule out the possibility that the knockdown of relish in the DA neurons could regulate food intake, we measured feeding rates using the blue food dye (1% FD&C Blue#1) mixed with either sucrose or PQ. These treatments did not lead to significant differences in the food intake abilities of the relish knockdown flies as compared to the corresponding controls (Fig. S4).
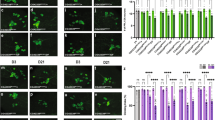
Targeted relish knockdown is protective against paraquat-mediated neurotoxicity. (a) Knockdown of relish in the DA neurons increases survival following PQ exposure. Survival assays were set up using male flies with relish knockdown in DA neurons (TH-Gal4 > UAS-relish-RNAi) and the corresponding controls (TH-Gal4>+ and UAS-relish-RNAi) following sucrose or 5 mM PQ treatment. Log-rank test was used for survival analysis and statistically significant differences (P < 0.001) were observed between the relish knockdown (TH-Gal4 > UAS-relish-RNAi) and the corresponding control groups (TH-Gal4>+ and UAS-relish-RNAi) in response to PQ exposure. Data are representative of at least five independent experiments with 10 flies per group. (b) Relish is involved in PQ-induced mobility defects. The effect of PQ on the mobility of controls (TH-Gal4/+ and UAS-relish-RNAi) and relish knockdown (TH-Gal4 > UAS-relish-RNAi) flies was determined by negative geotaxis assays. The number of flies able to climb 5 cm within 20 s was recorded using 10 flies per group. Data are representative of at least five independent experiments with 10 male flies per group. **P < 0.01; ***P < 0.001 based on one-way ANOVA between indicated genotypes in response to PQ. (c) Relish is associated with PQ-induced loss of DA neurons. The effect of PQ on DA neuron clusters in controls (TH-Gal4/+) and relish knockdown (TH-Gal4 > UAS-relish-RNAi) flies was determined by confocal imaging. GFP-positive green signal depicts DA neuron clusters of the Drosophila brain. White boxes indicate DA neuron PPM3 cluster. Scale bars 100 μm. Enlarged representative views of the posterior DA clusters, protocerebral posterior medial (PPM3) and PPM2 are shown in the side panel. (d) Average number of dopaminergic neurons per cluster in controls (TH-Gal4/+) and relish knockdown (TH-Gal4 > UAS-relish-RNAi) flies. DA neuron clusters: PPM2, PPM3 and protocerebral posterolateral (PPL1). Data are representative of at least three independent experiments. **P < 0.01; ***P < 0.001 based on two-way ANOVA with Tukey’s post-hoc test for multiple comparisons.
PQ exposure has been previously linked to altered movement phenotypes in Drosophila PD models3,13. To evaluate whether reduction of Relish levels in DA neurons could rescue the mobility defects induced by PQ, we employed a negative geotaxis assay, which is an indicator of the onset of PD pathogenesis-linked movement dysfunction. After ingestion of 5 mM PQ for 24 h, both the control groups (TH-Gal4>+ and UAS-relish-RNAi) exhibited resting tremors and bradykinesia, which are characteristic clinical symptoms associated with PD in human patients (Fig. 3b). We observed that relish knockdown in the DA neurons (TH-Gal4 > UAS-relish-RNAi) significantly rescued flies against PQ-induced mobility defects as shown in Fig. 3b, consistent with improved survival during PQ exposure.
We have previously shown that PQ feeding causes the loss of specific clusters of DA neurons3. Since relish knockdown in DA neurons confers PQ resistance in terms of survival and mobility defects (Fig. 3a,b), we determined the relative contribution of Relish on PQ-induced loss of DA neurons in the Drosophila brain. The effect of 5 mM PQ exposure on DA neurons was detected after 48 h using GFP expression driven by TH promoter in TH-Gal4 > UAS-GFP transgenic lines. The total number of DA neurons was significantly reduced in response to PQ ingestion in the WT flies (Fig. 3c). Consistent with earlier findings3, we also noticed that certain DA neuron clusters (protocerebral posterior medial: PPM2, PPM3, protocerebral posterolateral: PPL1) were more sensitive to PQ treatment (Fig. 3d). Targeted reduction of Relish protects against loss of DA neurons in response to PQ (Fig. 3c). Together, these data suggest that Relish activation is detrimental to survival following PQ exposure in PD pathogenesis.
Paraquat-induced toxicity involves the IMD signaling pathway
Our data indicate that Relish, which is the critical transcription factor downstream of the IMD signaling pathway, mediates PQ-induced PD pathogenesis in Drosophila. To determine whether additional components of the IMD pathway play a role in PQ-induced toxicity, we monitored the survival of Imd and Dredd mutant flies upon PQ ingestion. IMD is a death domain protein that mediates defense against Gram-negative bacterial infection through Relish-dependent induction of downstream AMPs and Dredd encodes a caspase downstream of IMD that mediates immune responses to Gram-negative bacteria and apoptotic pathways in Drosophila60,61. We observed significantly improved median survival of Imd1 (Imd-null) mutants in comparison to wild type Canton S controls following PQ treatment (Fig. 4a). To characterize Dredd’s role in mediating PQ-induced toxicity, we used two available mutant alleles: the hypomorphic DreddEP1412 mutation and a Dredd-null (DreddB118) mutant line that harbors a stop codon in the DREDD pro-domain61. Both mutants showed significantly improved median survival to PQ in comparison to their respective wild type controls, with slightly higher protection observed for DreddB118 mutants (Fig. 4b,c). These data suggest that both IMD and the downstream caspase, DREDD are involved in mediating survival upon PQ exposure.
Paraquat toxicity is mediated through components of the IMD pathway. (a) Loss of imd improves survival upon PQ exposure. Survival assays were set up using Imd-null male flies along with the corresponding wild-type (Canton S) control following sucrose or 5 mM PQ treatment. Median survival (days) of flies are indicated. *P < 0.05 based on Mann-Whitney U test. Data are average of at least three independent experiments with 10–15 flies per group. (b) Improved survival in Dredd mutants in response PQ ingestion. Survival assays were set up using Dredd mutant (DreddEP1412 and DreddB118) male flies along with the corresponding wild-type controls following sucrose or 5 mM PQ treatment. Median survival (days) of flies are indicated. *P < 0.05 based on Mann-Whitney U test. Data are average of at least three independent experiments with 10–15 flies per group. (c) Effect of PQ exposure on AMP expression in Imd and Dredd mutant flies. RNA was isolated from the heads of wild type and mutant flies as indicated following 5 mM PQ or sucrose treatment for 12 h. Relative transcript levels of diptericinB and attacinC were analyzed using qRT-PCR and plotted after normalization with rp49 levels as the internal control. Each data point represents the mean +/− SEM. mRNA fold changes are normalized to the sucrose-fed flies (assigned a value of 1). **P < 0.01 based on Mann Whitney’s U-test.
Because PQ treatment leads to suppression of AMP-coding genes downstream of the IMD pathway, we next evaluated the role of IMD and DREDD in PQ-mediated suppression of AMPs. Both the Imd-null and Dredd-null mutant flies along with their corresponding wild type flies were exposed to either sucrose alone or 5 mM PQ for 12 h, prior to RNA extraction from the heads followed by qRT-PCR using AMP-specific primers. As shown in Fig. 4d, the expression of both diptericinB and attacinC transcripts were suppressed in both the WT controls and Imd-null mutant flies in response to PQ, indicating that the upstream adaptor protein, IMD, is not required for PQ-mediated regulation of the AMPs. Surprisingly, these AMP genes were not suppressed in the DreddB118 mutant flies in response to PQ, suggesting the involvement of the downstream caspase, DREDD in regulating PQ-mediated AMP expression.
Paraquat-induced AMP regulation is mediated through Pirk
We have shown that PQ-mediated Relish activation does not lead to AMP induction downstream of the classical IMD pathway, as is typically the case following pathogen exposure23. Instead, we observed that AMP expression was suppressed more than 2-fold upon PQ feeding (Fig. 1c). Negative regulation plays an important role to ensure proper immune response against pathogens while preventing hyperactivation of the immune response that results in tissue damage and chronic inflammation28. We found that PQ exposure to 5 mM PQ for 12 h results in a 2.5-fold induction of pirk, which encodes a negative regulator of the IMD pathway that is itself regulated by Relish (Fig. 5a). Earlier studies have shown that Pirk overexpression suppresses the IMD pathway response following infection with Gram-negative bacteria62. To evaluate the role of Pirk in PQ-mediated suppression of AMPs, pirk-null mutant flies were exposed to 5 mM PQ for 12 h. As shown in Fig. 5b, diptericinB expression was reduced in the WT controls but not in pirk-null mutant flies in response to PQ. Our data indicate that pirk is required for suppression of AMP (diptericinB) expression in response to PQ treatment. To determine the effect of Pirk on PQ-mediated toxicity, we monitored the survival of pirk-null mutant flies upon PQ treatment. As shown in Fig. 5c, the survival rates of the control sucrose-fed WT and pirk-null mutant flies did not differ. However, pirk-null mutant flies had significantly (p < 0.01) lower survival rates compared with the WT control in response to PQ exposure, thereby indicating that loss of pirk increases PQ susceptibility. Since Pirk is a known negative regulator of the IMD pathway, we looked at the effect of Pirk mutation on relish expression upon PQ treatment. As shown in Fig. 5d, loss of pirk resulted in hyper-induction (more than 8-fold) of relish transcripts in response to PQ. Interestingly, relish-null mutant flies were not resistant to PQ toxicity in survival assays (Fig. S3). The higher sensitivity of relish-null mutant flies to PQ is likely due to the absence of functional relish during development which would affect multiple downstream genes affecting developmentally important cellular processes. These data suggest a novel role for Pirk with relevance to PQ-induced toxicity in Drosophila PD model.
Pirk is required for paraquat-mediated AMP suppression. (a) PQ induces the expression of the negative regulator pirk. RNA was isolated from the heads of Canton S wild type flies following 5 mM PQ or sucrose treatment for 12 h. Relative transcript levels of pirk were analyzed using qRT-PCR and plotted after normalization with rp49 levels as the internal control. Each data point represents the mean +/− SEM. mRNA fold changes are normalized to the sucrose-fed flies (assigned a value of 1). *P < 0.05 based on Mann Whitney U-test. (b) PQ-mediated AMP suppression requires Pirk. RNA was isolated from the heads of wild type y w1118 and pirk-null mutant flies following 5 mM PQ or sucrose treatment for 12 h. Relative transcript levels of diptB were analyzed using qRT-PCR and plotted after normalization with rp49 levels as the internal control. Each data point represents the mean +/− SEM. mRNA fold changes are normalized to the sucrose-fed flies (assigned a value of 1) for each of the wild type and mutant strains. **P < 0.01 based on Mann-Whitney U-test. The results are representative of at least three independent experiments. (c) Loss of pirk increases PQ toxicity. Survival assays were set up using pirk-null male flies along with the corresponding wild-type (y w1118) control following sucrose or 5 mM PQ treatment. Log-rank test was used for survival analysis and statistically significant differences (P < 0.01) were observed between the pirk-null and the corresponding control group in response to PQ exposure. Data are average of at least five independent experiments with 10 flies per group. (d) Loss of pirk causes hyper-induction of relish transcript in response to PQ exposure. RNA was isolated from the heads of pirk-null mutant flies following 5 mM PQ or sucrose treatment for 12 h. Relative transcript levels of relish were analyzed using qRT-PCR and plotted after normalization with rp49 levels as the internal control. Each data point represents the mean +/− SEM. mRNA fold changes are normalized to the sucrose-fed flies (assigned a value of 1). **P < 0.01 based on Mann Whitney U-test.
Paraquat exposure confers increased sensitivity to bacterial infection
Drosophila mounts an robust immune response to combat and destroy bacterial infection by inducing AMP expression22,25. The failure to induce AMPs to efficiently clear bacterial infections results in higher mortality rates25. Based on our data showing PQ-mediated suppression of AMPs, we hypothesized that PQ exposure would increase susceptibility to Gram-negative bacterial infection. To test this hypothesis, we poked into the thorax of Canton S WT flies with a concentrated culture of the Gram-negative bacteria Escherichia coli (E. coli) to activate the innate immune response and then monitored flies’ survival. The Canton S WT flies were fed either sucrose or 5 mM PQ containing sucrose for 12 h prior to E. coli infection and the number of flies was scored daily for survival. In addition, sterile injections performed on sucrose and PQ-fed flies show that inflammation resulting from the needle injury alone is not sufficient to kill the flies. The results shown in Fig. 6a demonstrate that PQ pre-treatment leads to a significantly increased sensitivity to E. coli infection as compared to the sucrose-fed flies. Since inducible AMPs are important for the immune defense in Drosophila, we next investigated whether the increased susceptibility could be due to the reduced induction of AMPs following infection in PQ treated flies. Strikingly, PQ pre-treatment abrogated AMP induction (diptericinB and attacinC) in a dose-dependent manner following E. coli infection as compared to the sucrose-fed control flies (Fig. 6b). The data suggest that PQ-mediated suppression of AMPs leads to a compromised immune response against bacterial infection. The higher mortality in the PQ-fed flies is likely due to their inability to clear the infection effectively. Since Relish is required for AMP expression downstream of the Imd pathway, we sought to detect the effect of PQ exposure on the relish transcript levels post-infection. In addition, we examined the levels of pirk in the sucrose and PQ fed flies post-infection as Pirk is a known downstream target of Relish in the IMD pathway. As depicted in Fig. 6c, the transcript levels of relish and pirk were induced in response to E. coli infection in both the sucrose and PQ-fed flies. However, relish induction does not translate to the activation of AMP expression downstream of the IMD pathway. We further explored the localization of the active Relish p68 in the sucrose and PQ-fed flies in response to E. coli infection. As shown in Fig. 6d, the active nuclear Relish p68 is induced in the sucrose-fed flies in response to E. coli infection (compare sucrose-fed sterile vs E. coli). PQ treatment alone causes nuclear translocation of the active Relish p68 and a gradual decrease in the full-length cytoplasmic Relish p110 form (Fig. 6d). However, upon PQ pre-treatment, the nuclear Relish p68 fails to initiate AMP gene transcription following E. coli infection. Taken together, the data suggest that PQ exposure increases susceptibility to Gram-negative bacterial infection due to the inability of active Relish to induce AMP expression downstream of the IMD pathway, thereby leading to a compromised immune response and more rapid death of infected flies.
Paraquat exposure leads to increased susceptibility to Gram-negative bacterial infection. (a) Canton S wild type male flies were fed either 2.5% sucrose or 5 mM PQ for 12 h prior to E. coli infection. The pre-treated flies were then injected either with sterile needle or E. coli and survival was scored once every 24 h. Log-rank test was used for survival analysis and statistically significant differences (P < 0.001) were observed between the PQ-pretreated group in response to sterile or E. coli infection. Data are representative of at least three independent experiments with 10 flies per group. (b) PQ exposure suppresses AMP expression in response to Gram-negative bacterial infection. Canton S wild type male flies were fed either sucrose or 5 or 10 mM PQ for 12 h prior E. coli injection. Pre-treated flies were then injected either with sterile needle or E. coli followed by RNA extraction 6 h post-infection. Relative transcript levels of diptB and attacin-C were analyzed using qRT-PCR and plotted after normalization with rp49 levels as the internal control. Each data point represents the mean +/− SEM. mRNA fold changes are normalized to the corresponding sterile-injected flies. *P < 0.05; **P < 0.01 based on Mann Whitney U-test. (c) The effect of PQ on pirk and relish transcripts in response to bacterial infection. Canton S wild type male flies were fed either sucrose or 5 mM PQ for 12 h prior to E. coli infection. The pre-treated flies were then either not infected, injected with sterile needle or E. coli followed by RNA extraction 6 h post-infection. Relative transcript levels of relish and pirk were analyzed using qRT-PCR and plotted after normalization with rp49 levels as the internal control. Each data point represents the mean +/− SEM. mRNA fold changes are normalized to the sucrose-fed uninfected flies (assigned a value of 1). **P < 0.01 based on Mann Whitney U-test. (d) The effect of PQ on Relish localization in response to E. coli infection. Canton S wild type male flies were fed either sucrose or 5 mM PQ for 12 h prior to E. coli infection. The pre-treated flies were then either injected with sterile needle or infected with E. coli followed by nuclear fractionation and immunoblotting using the specified antibodies. The full-length Relish p110 and p68 are indicated by arrows. The same blots were probed with anti-Lamin and anti-Tubulin as the nuclear and cytoplasmic markers for the fractionation protocol. The band intensities were quantitated using the LI-COR C-DiGit Blot scanner software and plotted after normalization with Lamin and Tubulin. *P < 0.05 based on Student’s t test.
Discussion
Neuroinflammation is associated with several neurodegenerative diseases, including AD, PD, and multiple sclerosis (MS), that contribute to neuronal dysfunction and death5. Populations of activated microglia have been observed in areas of neuronal damage and degeneration in post-mortem brains of patients suffering from PD and AD, and subsequent research has shown that inflammatory responses are functionally linked to disease progression6,7,8. The involvement of dysregulated inflammation impedes deployment of therapeutic intervention capable of slowing or blocking disease progression. A serious confounding factor is that most neurodegenerative diseases are not caused by highly penetrant, monogenic variants, but rather, appear to be rooted in combinations of genetic background and exposure to generally ill-defined environmental factors. As the vast majority of PD cases are sporadic, the underlying mechanisms of disease progression remain poorly understood.
In this study, we employed transcriptomic profiling in response to transient PQ exposure in Drosophila to identify pre-symptomatic gene signatures involved in the progression of PD, prior to the manifestation of movement dysfunction. The inflammatory responses to neurodegeneration in the adult Drosophila brain mirror quite strongly the responses to neurodegeneration in mammalian models and in human disease63. Our RNAseq data demonstrate that PQ exposure triggers transcriptional changes in genes involved in innate immune response pathways in Drosophila. We show that PQ treatment activates genes associated with Drosophila hemocytes, which are macrophage-like cells involved in phagocytosis and inflammation. This finding is in agreement with our previous data showing neuroinflammatory responses through the induction of NOS in response to PQ treatment33. Moreover, involvement of hemocytes has also been implicated in the progression of AD in the fly model64. Interestingly, we observed that other innate immune genes, particularly those encoding AMPs, downstream of both the Toll and IMD pathways were suppressed in response to PQ exposure. Contrary to our findings, earlier reports suggest a link between increased expression of AMPs and neurodegeneration27,29,30. One possible explanation for this difference could be that we have employed a pre-symptomatic disease onset model, as opposed to other reported models that mainly focus on degeneration stage processes. A pre-symptomatic strategy can both optimize target manipulation by genetic or drug intervention and highlight key biomarkers with potential for early diagnosis and treatment. Our results indicate that PQ-mediated suppression of AMP expression might impair the host innate immune system leading to decreased ability to fight bacterial infection. In this study, we focused on AMPs downstream of the IMD pathway due to the involvement of Relish in PQ-mediated neurodegeneration of DA neurons. Further dissection of PQ-mediated regulation of the Toll pathway may aid our understanding of the mechanistic link between aberrant innate immune response and PD pathogenesis.
In addition to genes involved in the innate immune response, the transcriptomic analysis identified genes, whose mammalian orthologs are also implicated in PD pathogenesis. For example, we observed increased expression of Gadd45 transcript in fly heads following PQ exposure, while DA neurons-specific knockdown of this gene using RNAi led to increased PQ sensitivity, suggesting a neuroprotective effect of GADD45 against PQ-induced neurotoxicity in Drosophila. In alignment with our findings, GADD45α has been shown to be protective against MPP+ toxicity in human dopamine neuroblastoma cells42. Furthermore, our results revealed induction of chaperone responses through the upregulation of Hsps (Hsp22, Hsp26, Hsp68) upon PQ exposure. Hsps have been shown to play neuroprotective roles in both mammalian and Drosophila models of neurodegenerative disease, including PD44. Interestingly, earlier studies show neuron-specific expression of Hsp26 and Hsp70 improves life span and enhances stress resistance in PQ and α-synuclein-induced cellular toxicity in both Drosophila and mammalian models46,47. Noticeably, we also identified genes involved in the glutathione metabolic responses (GstD2, GstD5, GstE7, GstE8) as PQ-responding genes. The neuroprotective role of GSTs has been established in several models of PD, including Drosophila where overexpression of GST has been shown to ameliorate neurodegeneration48,49. These findings justify the use of our toxin-induced neurodegeneration model system in Drosophila to successfully identify potential therapeutic targets in PD. However, further studies are warranted with different PQ doses and duration to build a temporal landscape of pre-symptomatic gene expression signatures to discover novel molecular targets relevant to prediction of future disease and potential therapeutic targets.
Our data indicate that the NF-κB transcription factor Relish is required for PQ-induced neurodegeneration and suggest a role in environmental toxin-induced PD pathogenesis. Relish regulates the inducible innate immune response in Drosophila through signal-dependent translocation into the nucleus resulting in increased expression of AMPs downstream of the IMD pathway24,25. Activation of Relish in glial cells has been shown to be responsible for neuron loss in a model of ataxia–telangiectasia, while neuron-specific overexpression of Relish-dependent AMP genes is sufficient to cause neurodegeneration29,30,31,65. Here, we demonstrate that PQ treatment mediates nuclear translocation of active Relish. In addition, we show that targeted knockdown of relish specifically in the dopaminergic neurons significantly improves survival, climbing performance and rescues DA neuron loss following PQ exposure. Our results suggest that activation of Relish contributes to environmental toxin-induced neurodegeneration. Consistent with our findings, targeting NF-κB activation has been shown to be an effective therapy against neurodegeneration in murine PD models66. It would be interesting to investigate the role of Relish in different cell types including glial cells and hemocytes in response to PQ-induced neurodegeneration.
Oxidative stress plays a major role in the loss of dopaminergic neurons due to excessive production of ROS67. Noticeably, the RNAseq screen revealed upregulation of genes involved in redox reactions upon PQ exposure (Table 1). In mammalian PD models, JNK signaling pathway plays an important role in regulating the cellular processes such as oxidative stress and apoptosis14. Our data show that JNK is activated by phosphorylation following PQ treatment, while pharmacological inhibition of JNK increases survival in WT flies. In accordance with our data, knockdown of the basket gene in the dopaminergic neurons has been shown to improve survival and climbing ability in Drosophila68. Prolonged JNK activation has been implicated in exacerbating disease phenotypes in both AD and PD models69,70. In contrast, JNK has also been shown to be protective in diverse PD models because JNK can be both pro- and anti-apoptotic depending on cell type, nature and duration of the activation stimulus, interaction with other signaling pathways and genetic backgrounds13,14,58,71. Consistent with our observations, another group demonstrated that inhibition of JNK activity using SP600125 confers protection against PQ toxicity59. Furthermore, increased JNK signaling has been implicated in PQ-induced neurodegeneration and active JNK has been reported in Drosophila parkin mutants14,72.
We demonstrate that PQ exposure induces nuclear translocation of Relish p68 (Fig. 2a). Interestingly, activated Relish fails to induce the expression of downstream target AMPs genes upon PQ treatment, thus compromising survival following infection with Gram-negative bacteria. Our data suggest that PQ-induced toxicity is mediated by components of the IMD pathway, including the adaptor protein, IMD, and the downstream caspase, DREDD23. Interestingly, both the Imd and Dredd mutant flies show increased resistance to PQ toxicity in survival assays. DREDD is required for the activation and nuclear translocation of active Relish in response to bacterial infection. In addition to its role in antibacterial response, the caspase DREDD is involved in regulating apoptotic pathways in Drosophila60. Interestingly, caspase-8 activation has been reported in both PD patients as well as environmental-toxin induced PD model in mice73. In a fly model of retinal degeneration, DREDD but not IMD plays a role in photoreceptor degeneration74. Our work shows that both of these factors mediate survival to PQ-induced toxicity and future studies will determine their involvement in PQ-mediated DA neuronal degeneration.
NF-kB signaling is tightly regulated by negative regulators to avoid prolonged immune responses that are detrimental to the host28. Our results show that PQ induces the expression of pirk, a gene encoding a negative regulator of the IMD pathway. In addition, we show that Pirk is required for PQ-mediated suppression of AMPs suggesting a novel function of Pirk in environmental toxin-induced PD pathogenesis. Elevated levels of the negative regulator Pirk might activate or interact with another unknown factor to prevent nuclear active Relish p68 from inducing downstream AMP targets. In the context of bacterial infection, pirk induction depends on Relish and Pirk regulates IMD signaling at the level of the adaptor protein IMD, which is downstream of the receptor PGRP-LC25,62. Our data suggest that DREDD but not the upstream adaptor protein, IMD is required to regulate AMP expression in response to PQ. Therefore, it is possible that PQ-mediated activation of Relish and AMP expression might be regulated through an alternative noncanonical pathway as observed in ATM mutants30. A recent study demonstrated that Relish becomes activated in response to infection with the invertebrate iridescent virus 6 (IIV6) and that its translocation to the nucleus results in suppression of AMPs gene expression75. The failure of active Relish to induce downstream AMP gene expression upon PQ exposure suggests that transcriptional inhibition could occur at the promoter-binding site. Our results show that loss of pirk results in increased PQ sensitivity leading to higher mortality due to induction of relish levels. We speculate that PQ exposure might directly activate DREDD or additional factors responsible for nuclear translocation of Relish resulting in pirk induction and subsequent inhibition of AMPs as depicted in Fig. 7. Therefore, future studies will focus on elucidating the mechanisms regulating Relish activation upon PQ exposure. In addition, further studies are required to understand the precise role of Pirk in the regulation of innate immune responses following PQ treatment. Since our data show that PQ downregulates AMPs and that loss of relish in the DA neurons improves the PQ-induced parkinsonian phenotypes, further studies are warranted to dissect the exact role of AMPs in PQ-mediated toxicity.
Schematic representation of paraquat-mediated innate immune responses in a Drosophila PD model. Short-term PQ exposure regulates genes involved in the Drosophila innate immunity. PQ activates full-length Relish p110 by proteolytic cleavage and induces nuclear translocation of Relish p68. In the dopaminergic neurons, PQ-induced Relish activation leads to poor survival, mobility defects and loss of specific dopaminergic neuron clusters. PQ-mediated toxicity involves components of the IMD pathway, the adapter protein, IMD and the downstream caspase, DREDD. PQ inhibits Relish-dependent induction of AMPs through activation of pirk, a negative regulator of the IMD pathway. PQ-mediated suppression of AMPs disrupts the innate immune response thereby increasing susceptibility to Gram-negative bacterial infections. On the other hand, PQ might also directly activate other unknown factors which might interact with either Relish, DREDD and/or Pirk and lead to suppression of AMPs. Dotted lines represent hypothetical pathways regulating immune responses downstream of PQ.
There is a growing body of evidence suggesting higher susceptibility to infection in PD patients and in PD disease models, although the precise mechanism remains to be elucidated. Higher infection burden has been associated with PD pathogenesis due to increased levels of serum inflammatory cytokines and α-synuclein76,77. Additionally, high prevalence of small intestinal bacterial overgrowth and higher incidence of respiratory tract and urinary tract infections has been reported in PD patients78,79. Moreover, PD patients exhibit higher susceptibility to infections affecting the central nervous system and sepsis80. Interestingly, Parkin, an E3 ubiquitin ligase associated with early onset PD also modulates innate immune responses; loss of parkin leads to a defective immune response against bacterial infection in flies and murine models81. Genome-wide association studies link leucine rich repeat kinase 2 (LRRK2) variants, related to one of the leading heritable forms of PD, to higher susceptibility to the autoimmune Crohn’s disease and Mycobacterium leprae infection82,83, reinforcing a strong link between elevated risk of infection and PD. Consistent with these findings, we demonstrate that in Drosophila, PQ treatment results in an increased susceptibility to Gram-negative bacterial infection due to suppressed AMP expression. Healthy flies normally survive infection with a non-pathogenic strain of E. coli as observed in the sucrose-fed group84, whereas flies exposed to the environmental toxin PQ exhibit compromised immune response and higher mortality. Moreover, increased bacterial susceptibility has also been reported in another genetic PD model using parkin deficient flies81. Therefore, our observation of suppressed AMP expression as a response to PQ exposure suggests a possible mechanism for the increased risk of infections in PD patients.
Our study provides strong evidence for the involvement of the innate immune response in environmental toxin-induced PD pathogenesis. We also show that PQ-mediated dysregulation of the innate immune response is associated with increased susceptibility to E.coli infection. Further studies of pre-symptomatic immune systems are warranted for new therapies aimed at modulating the immune system’s response during PD, thereby leading to improved clinical outcomes for disease.
Materials and Methods
Drosophila strains and culture maintenance
Stocks were raised and maintained at 25 °C on standard medium (Bloomington Stock Center Recipe) containing cornmeal, corn syrup, yeast, and agar. The control genotype for all experiments was Canton S or the appropriate heterozygous control, and flies used for all adult assays were three to five-day old males unless otherwise noted. The following stocks were purchased from the Bloomington Drosophila Stock Center at Indiana University: Canton S, y w1118, w1118, UAS-2XEGFP, pirkEY00723, w1118; RelE20, UAS-GFP, UAS-RelRNAi#33661, and UAS-RelRNAi#28943, Imd1, DreddB118, DreddEP1412. TH-GAL4 was a gift from J. Hirsh (University of Virginia, Charlottesville, VA).
RNAseq analysis
Transcriptomic profiling was performed using four independent biological replicates per feeding. Age-matched adult Canton S male flies were fed either 2.5% sucrose (control) or 5 mM PQ in 2.5% sucrose for 12 h and the heads were collected for RNA extraction. Total RNA was extracted using TRIzol Reagent (Thermo Fisher Scientific) from the fly heads following sucrose or PQ ingestion (thirty males/group). The concentration and integrity of the extracted total RNA were estimated using the Qubit® 2.0 Fluorometer (Invitrogen) and Agilent 2100 Bioanalyzer (Applied Biosystems, Carlsbad, CA, USA), respectively. For paired-end library preparation, NEBNext mRNA Library Prep Reagent Set for Illumina was used and the library concentration was analyzed by utilizing a DNA 1000 Chip on an Agilent 2100 Bioanalyzer. Paired-end sequencing (25 million, 50-bp, paired-end reads) was performed using a 200 Cycle TruSeq SBS HS v4 Kit on an Illumina HiSeq2500 sequencer (Illumina, Inc., San Diego, CA, USA) at the HudsonAlpha Institute for Biotechnology, Huntsville, AL. TopHat v2.0 were used to map raw reads to the reference Drosophila melanogaster genome dm3 (Fig. S1). The transcript abundance was quantified for the final read list using Trimmed Means of M-values as the normalization method. The p-value of the differentially expressed gene list were estimated by z-score calculations using Benjamini Hochberg corrections of 0.05 for false-discovery rate. Differential gene expression was calculated on the basis of fold changes between the control sucrose and PQ-fed groups. A gene ontology (GO) analysis for enriched biological process was performed on the list of differentially expressed mRNAs between samples using DAVID v6.8 and the GO data represented in Table 1 were filtered with thresholds that only show enriched biological processes categories with 3 or more associated genes.
Paraquat treatment, JNK inhibitor and survival assay
Ten males of specified genotype, aged 3–5 days post-eclosion, were fed daily on filter paper saturated with specified concentrations of paraquat (Methyl viologen dichloride hydrate, Sigma-Aldrich) in 2.5% sucrose (Sigma-Aldrich) or with 2.5% sucrose only. Mortality was monitored daily for ten days. PQ treatment was performed with at least five independent biological replicates of 10 males each for each genotype tested. The JNK inhibitor, SP600125 was purchased from Sigma-Aldrich.
Climbing assay
The mobility of adult male flies from each treatment group was recorded using a negative geotaxis climbing assay. Ten flies per treatment group were placed in an empty plastic vial and gently tapped to the bottom. The percentage of flies that crossed a line 5 cm from the bottom of the vial in 20 sec were calculated. Each biological replicate was assayed three times at 1 min intervals and the percentages averaged. Statistical significance between different genotypes was calculated using One-way analysis of variance (ANOVA) for P < 0.05.
Confocal microscopy
Brains from adult flies were processed for confocal imaging as described earlier3. Briefly, brains were dissected in 1x phosphate buffered saline with Tween20 (PBST), fixed in 4% paraformaldehyde for 20 min in a 24 well plate, and washed three times using 1x PBST. GFP-positive dopaminergic neurons were detected using a chicken polyclonal anti-GFP antibody (Abcam) and Alexa fluor® 488 goat anti-chicken secondary antibody (Thermo Fisher Scientific). Brains were mounted on slides using ProLong Gold Antifade Mountant with DAPI (Thermo Fisher Scientific). Whole mounts of dissected brains were imaged using a Nikon C2 DUVb confocal laser scanning microscope. The GFP-positive cells of specific DA clusters were counted based on confocal Z-stacks of whole mount brains.
Western blot analysis
25–30 adult fly heads were homogenized in RIPA buffer (Amresco) supplemented with 2 mM DTT and 1x protease inhibitor cocktail (Amresco). The homogenates were centrifuged at 12,000 × g for 5 min at 4 °C. Equal amounts of samples were mixed with 4X laemmli sample buffer (LDS) and heated at 70 °C for 10 min, briefly centrifuged, and subjected to SDS-PAGE using 4–12% NuPage Bis-Tris minigel and MOPS SDS Running buffer. Proteins were transferred to nitrocellulose or PVDF membranes (Bio-Rad) using iBlot Transfer Stacks, blocked in 2.5% dry nonfat milk and immunoblotted using one of the following antibodies: anti-Relish (RayBiotech Inc) (1:1000), anti-Tubulin (Sigma-Aldrich) (1:2500), anti-Lamin (Development Studies Hybridoma Bank) (1:2000), anti-JNK (Cell Signaling) (1:1000), anti-phosphoJNK (Cell Signaling) (1:1000). The appropriate anti-mouse or anti-rabbit horseradish peroxidase-conjugated secondary antibody was used, and signal was detected using Supersignal West Pico Chemiluminescent Substrate (Thermo Fisher Scientific) and LI-COR C-DiGit Blot scanner.
Cell fractionation
Approximately 50 adult fly heads were collected per experimental group and nuclear fractionation was performed using NE-PER Nuclear and Cytoplasmic Extraction Kit (Thermo Fisher Scientific) according to the manufacturer’s protocol. Briefly, the heads were homogenized in 100 μL Cytoplasmic Extraction Reagent I, containing protease inhibitors, and samples were vortexed and allowed to incubate on ice for 10 min, followed by the addition of 5.5 μL of Cytoplasmic Extraction Reagent II and centrifugation at 10,000 rpm for 5 min at 4 °C. The supernatant was saved at −80 °C as the cytoplasmic fraction. The pellet was resuspended in 50 μL of Nuclear Extraction Reagent I (1X Protease Inhibitor, Thermo Scientific, IL USA). The samples were incubated on ice for 40 mins followed by centrifugation at 10,000 rpm for 10 mins at 4 °C. The resulting supernatant (nuclear fraction) was collected and stored at −80 °C for Western blot analysis.
Quantitative Real time RT-PCR
Total RNA was extracted from 30 adult fly heads using TRIzol Reagent (Thermo Fisher Scientific), and cDNA was prepared from 0.5–1 μg total RNA using the High Capacity cDNA Reverse Transcription kit (Applied Biosystems). Triplicate cDNA samples were amplified using the iQ SYBR Green Supermix (Biorad) in a StepOnePlus Real time PCR System according to the manufacturer’s protocols. The relative levels of specified transcripts were calculated using the ΔΔCt method, and the results were normalized based on the expression of ribosomal protein L32 (RpL32/RP49) as the endogenous control within the same experimental setting.
Bacterial infection
Canton S adult flies were infected by poking the thorax with a fine needle dipped into a concentrated overnight grown culture of Escherichia coli (ATCC#11775 strain). For gene expression analysis, triplicates of male flies were snap frozen in liquid nitrogen 6 h post-infection. Frozen heads were homogenized in TRIzol reagent (Invitrogen Life Technologies), and the total RNAs extracted according to the manufacturer’s instructions. For survival assays, 30 flies were infected per experimental group as described above and their survival was scored daily at 25 °C.
Statistical analysis
Data were analyzed using GraphPad Prism 8 software (GraphPad Software, Inc., La Jolla, CA). Statistical significances of gene expression were determined using a non-parametric Mann Whitney U-test. Statistically significant differences between survival curves were determined by log-rank test. Student’s t test was used as a parametric test for comparing differences between two groups. Results are expressed as mean ± SEM. p < 0.05 was considered statistically significant. Details of analysis are specified in the figure legends.
Data Availability
All the data generated or analyzed during this study are included in this published article or available from the corresponding author upon request. RNAseq expression data are available in the Gene Expression Omnibus (GEO) repository through Accession number: GSE135919.
References
Poewe, W. et al. Parkinson disease. Nat Rev Dis Primers 3, 17013, https://doi.org/10.1038/nrdp.2017.13 (2017).
Barnum, C. J. & Tansey, M. G. Neuroinflammation and non-motor symptoms: the dark passenger of Parkinson’s disease? Curr Neurol Neurosci Rep 12, 350–358, https://doi.org/10.1007/s11910-012-0283-6 (2012).
Chaudhuri, A. et al. Interaction of genetic and environmental factors in a Drosophila parkinsonism model. J Neurosci 27, 2457–2467, https://doi.org/10.1523/jneurosci.4239-06.2007 (2007).
Warner, T. T. & Schapira, A. H. Genetic and environmental factors in the cause of Parkinson’s disease. Ann Neurol 53(Suppl 3), S16–23; discussion S23–15, https://doi.org/10.1002/ana.10487 (2003).
Chen, W. W., Zhang, X. & Huang, W. J. Role of neuroinflammation in neurodegenerative diseases (Review). Mol Med Rep 13, 3391–3396, https://doi.org/10.3892/mmr.2016.4948 (2016).
McGeer, P. L., Itagaki, S., Boyes, B. E. & McGeer, E. G. Reactive microglia are positive for HLA-DR in the substantia nigra of Parkinson’s and Alzheimer’s disease brains. Neurology 38, 1285–1291 (1988).
Tansey, M. G., McCoy, M. K. & Frank-Cannon, T. C. Neuroinflammatory mechanisms in Parkinson’s disease: potential environmental triggers, pathways, and targets for early therapeutic intervention. Exp Neurol 208, 1–25, https://doi.org/10.1016/j.expneurol.2007.07.004 (2007).
Kim, Y. S. & Joh, T. H. Microglia, major player in the brain inflammation: their roles in the pathogenesis of Parkinson’s disease. Exp Mol Med 38, 333–347, https://doi.org/10.1038/emm.2006.40 (2006).
Drouin-Ouellet, J. et al. Toll-like receptor expression in the blood and brain of patients and a mouse model of Parkinson’s disease. Int J Neuropsychopharmacol 18, https://doi.org/10.1093/ijnp/pyu103 (2014).
Fellner, L. et al. Toll-like receptor 4 is required for alpha-synuclein dependent activation of microglia and astroglia. Glia 61, 349–360, https://doi.org/10.1002/glia.22437 (2013).
Kurkowska-Jastrzebska, I. et al. Dexamethasone protects against dopaminergic neurons damage in a mouse model of Parkinson’s disease. Int Immunopharmacol 4, 1307–1318, https://doi.org/10.1016/j.intimp.2004.05.006 (2004).
Wu, D. C. et al. Blockade of microglial activation is neuroprotective in the 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine mouse model of Parkinson disease. J Neurosci 22, 1763–1771 (2002).
Inamdar, A. A., Chaudhuri, A. & O’Donnell, J. The Protective Effect of Minocycline in a Paraquat-Induced Parkinson’s Disease Model in Drosophila is Modified in Altered Genetic Backgrounds. Parkinsons Dis 2012, 938528, https://doi.org/10.1155/2012/938528 (2012).
Peng, J., Mao, X. O., Stevenson, F. F., Hsu, M. & Andersen, J. K. The herbicide paraquat induces dopaminergic nigral apoptosis through sustained activation of the JNK pathway. J Biol Chem 279, 32626–32632, https://doi.org/10.1074/jbc.M404596200 (2004).
Chen, C. Y. et al. (G2019S) LRRK2 activates MKK4-JNK pathway and causes degeneration of SN dopaminergic neurons in a transgenic mouse model of PD. Cell Death Differ 19, 1623–1633, https://doi.org/10.1038/cdd.2012.42 (2012).
Ugur, B., Chen, K. & Bellen, H. J. Drosophila tools and assays for the study of human diseases. Dis Model Mech 9, 235–244, https://doi.org/10.1242/dmm.023762 (2016).
Knight, A. L. et al. The glycolytic enzyme, GPI, is a functionally conserved modifier of dopaminergic neurodegeneration in Parkinson’s models. Cell Metab 20, 145–157, https://doi.org/10.1016/j.cmet.2014.04.017 (2014).
Chege, P. M. & McColl, G. Caenorhabditis elegans: a model to investigate oxidative stress and metal dyshomeostasis in Parkinson’s disease. Front Aging Neurosci 6, 89, https://doi.org/10.3389/fnagi.2014.00089 (2014).
Courtney, E., Kornfeld, S., Janitz, K. & Janitz, M. Transcriptome profiling in neurodegenerative disease. J Neurosci Methods 193, 189–202, https://doi.org/10.1016/j.jneumeth.2010.08.018 (2010).
Miller, J. R. et al. RNA-Seq of Huntington’s disease patient myeloid cells reveals innate transcriptional dysregulation associated with proinflammatory pathway activation. Hum Mol Genet 25, 2893–2904, https://doi.org/10.1093/hmg/ddw142 (2016).
Schlachetzki, J. C. & Winkler, J. The innate immune system in Parkinson’s disease: a novel target promoting endogenous neuroregeneration. Neural Regen Res 10, 704–706, https://doi.org/10.4103/1673-5374.156958 (2015).
De Gregorio, E., Spellman, P. T., Tzou, P., Rubin, G. M. & Lemaitre, B. The Toll and Imd pathways are the major regulators of the immune response in Drosophila. EMBO J 21, 2568–2579, https://doi.org/10.1093/emboj/21.11.2568 (2002).
Lemaitre, B. & Hoffmann, J. The host defense of Drosophila melanogaster. Annu Rev Immunol 25, 697–743, https://doi.org/10.1146/annurev.immunol.25.022106.141615 (2007).
Stoven, S. et al. Caspase-mediated processing of the Drosophila NF-kappaB factor Relish. Proc Natl Acad Sci USA 100, 5991–5996, https://doi.org/10.1073/pnas.1035902100 (2003).
Kleino, A. & Silverman, N. The Drosophila IMD pathway in the activation of the humoral immune response. Dev Comp Immunol 42, 25–35, https://doi.org/10.1016/j.dci.2013.05.014 (2014).
Hunot, S. et al. Nuclear translocation of NF-kappaB is increased in dopaminergic neurons of patients with parkinson disease. Proc Natl Acad Sci USA 94, 7531–7536 (1997).
Kounatidis, I. et al. NF-kappaB Immunity in the Brain Determines Fly Lifespan in Healthy Aging and Age-Related Neurodegeneration. Cell Rep 19, 836–848, https://doi.org/10.1016/j.celrep.2017.04.007 (2017).
Lee, K. Z. & Ferrandon, D. Negative regulation of immune responses on the fly. EMBO J 30, 988–990, https://doi.org/10.1038/emboj.2011.47 (2011).
Cao, Y., Chtarbanova, S., Petersen, A. J. & Ganetzky, B. Dnr1 mutations cause neurodegeneration in Drosophila by activating the innate immune response in the brain. Proc Natl Acad Sci USA 110, E1752–1760, https://doi.org/10.1073/pnas.1306220110 (2013).
Petersen, A. J., Katzenberger, R. J. & Wassarman, D. A. The innate immune response transcription factor relish is necessary for neurodegeneration in a Drosophila model of ataxia-telangiectasia. Genetics 194, 133–142, https://doi.org/10.1534/genetics.113.150854 (2013).
Petersen, A. J., Rimkus, S. A. & Wassarman, D. A. ATM kinase inhibition in glial cells activates the innate immune response and causes neurodegeneration in Drosophila. Proc Natl Acad Sci USA 109, E656–664, https://doi.org/10.1073/pnas.1110470109 (2012).
Sulzer, D. et al. T cells from patients with Parkinson’s disease recognize alpha-synuclein peptides. Nature 546, 656–661, https://doi.org/10.1038/nature22815 (2017).
Ajjuri, R. R. & O’Donnell, J. M. Novel whole-tissue quantitative assay of nitric oxide levels in Drosophila neuroinflammatory response. J Vis Exp, 50892, https://doi.org/10.3791/50892 (2013).
Houser, M. C. & Tansey, M. G. The gut-brain axis: is intestinal inflammation a silent driver of Parkinson’s disease pathogenesis? NPJ Parkinsons Dis 3, 3, https://doi.org/10.1038/s41531-016-0002-0 (2017).
Cantera, R. & Barrio, R. Do the genes of the innate immune response contribute to neuroprotection in Drosophila? J Innate Immun 7, 3–10, https://doi.org/10.1159/000365195 (2015).
Di Monte, D. A., Lavasani, M. & Manning-Bog, A. B. Environmental factors in Parkinson’s disease. Neurotoxicology 23, 487–502 (2002).
Liou, H. H. et al. Environmental risk factors and Parkinson’s disease: a case-control study in Taiwan. Neurology 48, 1583–1588, https://doi.org/10.1212/wnl.48.6.1583 (1997).
McCormack, A. L. et al. Environmental risk factors and Parkinson’s disease: selective degeneration of nigral dopaminergic neurons caused by the herbicide paraquat. Neurobiol Dis 10, 119–127 (2002).
Uversky, V. N. Neurotoxicant-induced animal models of Parkinson’s disease: understanding the role of rotenone, maneb and paraquat in neurodegeneration. Cell Tissue Res 318, 225–241, https://doi.org/10.1007/s00441-004-0937-z (2004).
Moskalev, A. et al. The role of D-GADD45 in oxidative, thermal and genotoxic stress resistance. Cell Cycle 11, 4222–4241, https://doi.org/10.4161/cc.22545 (2012).
Plyusnina, E. N., Shaposhnikov, M. V. & Moskalev, A. A. Increase of Drosophila melanogaster lifespan due to D-GADD45 overexpression in the nervous system. Biogerontology 12, 211–226, https://doi.org/10.1007/s10522-010-9311-6 (2011).
Bgatova, N. et al. Gadd45 expression correlates with age dependent neurodegeneration in Drosophila melanogaster. Biogerontology 16, 53–61, https://doi.org/10.1007/s10522-014-9533-0 (2015).
Wang, X. F. et al. Induction of GADD45alpha protects M17 neuroblastoma cells against MPP*. IUBMB Life 66, 786–792, https://doi.org/10.1002/iub.1327 (2014).
Aridon, P. et al. Protective role of heat shock proteins in Parkinson’s disease. Neurodegener Dis 8, 155–168, https://doi.org/10.1159/000321548 (2011).
Liao, P. C., Lin, H. Y., Yuh, C. H., Yu, L. K. & Wang, H. D. The effect of neuronal expression of heat shock proteins 26 and 27 on lifespan, neurodegeneration, and apoptosis in Drosophila. Biochem Biophys Res Commun 376, 637–641, https://doi.org/10.1016/j.bbrc.2008.08.161 (2008).
Klucken, J., Shin, Y., Masliah, E., Hyman, B. T. & McLean, P. J. Hsp70 Reduces alpha-Synuclein Aggregation and Toxicity. J Biol Chem 279, 25497–25502, https://doi.org/10.1074/jbc.M400255200 (2004).
Shukla, A. K. et al. Heat shock protein-70 (Hsp-70) suppresses paraquat-induced neurodegeneration by inhibiting JNK and caspase-3 activation in Drosophila model of Parkinson’s disease. PLoS One 9, e98886, https://doi.org/10.1371/journal.pone.0098886 (2014).
Smeyne, M. & Smeyne, R. J. Glutathione metabolism and Parkinson’s disease. Free Radic Biol Med 62, 13–25, https://doi.org/10.1016/j.freeradbiomed.2013.05.001 (2013).
Whitworth, A. J. et al. Increased glutathione S-transferase activity rescues dopaminergic neuron loss in a Drosophila model of Parkinson’s disease. Proc Natl Acad Sci USA 102, 8024–8029, https://doi.org/10.1073/pnas.0501078102 (2005).
Michael, G. J. et al. Up-regulation of metallothionein gene expression in parkinsonian astrocytes. Neurogenetics 12, 295–305, https://doi.org/10.1007/s10048-011-0294-5 (2011).
Moran, L. B. et al. Whole genome expression profiling of the medial and lateral substantia nigra in Parkinson’s disease. Neurogenetics 7, 1–11, https://doi.org/10.1007/s10048-005-0020-2 (2006).
Kurucz, E. et al. Nimrod, a putative phagocytosis receptor with EGF repeats in Drosophila plasmatocytes. Curr Biol 17, 649–654, https://doi.org/10.1016/j.cub.2007.02.041 (2007).
Kurucz, E. et al. Hemese, a hemocyte-specific transmembrane protein, affects the cellular immune response in Drosophila. Proc Natl Acad Sci USA 100, 2622–2627, https://doi.org/10.1073/pnas.0436940100 (2003).
Lesch, C. et al. A role for Hemolectin in coagulation and immunity in Drosophila melanogaster. Dev Comp Immunol 31, 1255–1263, https://doi.org/10.1016/j.dci.2007.03.012 (2007).
Ramet, M. et al. Drosophila scavenger receptor CI is a pattern recognition receptor for bacteria. Immunity 15, 1027–1038 (2001).
Ganesan, S., Aggarwal, K., Paquette, N. & Silverman, N. NF-kappaB/Rel proteins and the humoral immune responses of Drosophila melanogaster. Curr Top Microbiol Immunol 349, 25–60, https://doi.org/10.1007/82_2010_107 (2011).
Silverman, N. et al. Immune activation of NF-kappaB and JNK requires Drosophila TAK1. J Biol Chem 278, 48928–48934, https://doi.org/10.1074/jbc.M304802200 (2003).
Wang, M. C., Bohmann, D. & Jasper, H. JNK signaling confers tolerance to oxidative stress and extends lifespan in Drosophila. Dev Cell 5, 811–816 (2003).
Jimenez-Del-Rio, M., Daza-Restrepo, A. & Velez-Pardo, C. The cannabinoid CP55,940 prolongs survival and improves locomotor activity in Drosophila melanogaster against paraquat: implications in Parkinson’s disease. Neurosci Res 61, 404–411, https://doi.org/10.1016/j.neures.2008.04.011 (2008).
Chen, P., Rodriguez, A., Erskine, R., Thach, T. & Abrams, J. M. Dredd, a novel effector of the apoptosis activators reaper, grim, and hid in Drosophila. Dev Biol 201, 202–216, https://doi.org/10.1006/dbio.1998.9000 (1998).
Leulier, F., Rodriguez, A., Khush, R. S., Abrams, J. M. & Lemaitre, B. The Drosophila caspase Dredd is required to resist gram-negative bacterial infection. EMBO Rep 1, 353–358, https://doi.org/10.1093/embo-reports/kvd073 (2000).
Kleino, A. et al. Pirk is a negative regulator of the Drosophila Imd pathway. J Immunol 180, 5413–5422 (2008).
McGurk, L., Berson, A. & Bonini, N. M. Drosophila as an In Vivo Model for Human Neurodegenerative Disease. Genetics 201, 377–402, https://doi.org/10.1534/genetics.115.179457 (2015).
Wu, S. C., Cao, Z. S., Chang, K. M. & Juang, J. L. Intestinal microbial dysbiosis aggravates the progression of Alzheimer’s disease in Drosophila. Nat Commun 8, 24, https://doi.org/10.1038/s41467-017-00040-6 (2017).
Shukla, A. k., Spurrier, J., Kuzina, I. & Giniger, E. Hyperactive Innate Immunity Causes Degeneration of Dopamine Neurons upon Altering Activity of Cdk5. Cell Rep. 26(1), 131–144.e4, https://doi.org/10.1016/j.celrep.2018.12.025 (2019).
Ghosh, A. et al. Selective inhibition of NF-kappaB activation prevents dopaminergic neuronal loss in a mouse model of Parkinson’s disease. Proc Natl Acad Sci USA 104, 18754–18759, https://doi.org/10.1073/pnas.0704908104 (2007).
Blesa, J., Trigo-Damas, I., Quiroga-Varela, A. & Jackson-Lewis, V. R. Oxidative stress and Parkinson’s disease. Front Neuroanat 9, 91, https://doi.org/10.3389/fnana.2015.00091 (2015).
Ortega-Arellano, H. F., Jimenez-Del-Rio, M. & Velez-Pardo, C. Dmp53, basket and drICE gene knockdown and polyphenol gallic acid increase life span and locomotor activity in a Drosophila Parkinson’s disease model. Genet Mol Biol 36, 608–615, https://doi.org/10.1590/s1415-47572013000400020 (2013).
Wang, W., Ma, C., Mao, Z. & Li, M. JNK inhibition as a potential strategy in treating Parkinson’s disease. Drug News Perspect 17, 646–654 (2004).
Yarza, R., Vela, S., Solas, M. & Ramirez, M. J. c-Jun N-terminal Kinase (JNK) Signaling as a Therapeutic Target for Alzheimer’s Disease. Front Pharmacol 6, 321, https://doi.org/10.3389/fphar.2015.00321 (2015).
Liu, J. & Lin, A. Role of JNK activation in apoptosis: a double-edged sword. Cell Res 15, 36–42, https://doi.org/10.1038/sj.cr.7290262 (2005).
Cha, G. H. et al. Parkin negatively regulates JNK pathway in the dopaminergic neurons of Drosophila. Proc Natl Acad Sci USA 102, 10345–10350, https://doi.org/10.1073/pnas.0500346102 (2005).
Hartmann, A. et al. Caspase-8 is an effector in apoptotic death of dopaminergic neurons in Parkinson’s disease, but pathway inhibition results in neuronal necrosis. J Neurosci 21, 2247–2255 (2001).
Chinchore, Y., Gerber, G. F. & Dolph, P. J. Alternative pathway of cell death in Drosophila mediated by NF-kappaB transcription factor Relish. Proc Natl Acad Sci USA 109, E605–612, https://doi.org/10.1073/pnas.1110666109 (2012).
West, C. et al. IIV-6 Inhibits NF-kappaB Responses in Drosophila. Viruses 11, https://doi.org/10.3390/v11050409 (2019).
Bu, X. L. et al. The association between infectious burden and Parkinson’s disease: A case-control study. Parkinsonism Relat Disord 21, 877–881, https://doi.org/10.1016/j.parkreldis.2015.05.015 (2015).
Bu, X. Parkinson disease: High infection burden in patients with Parkinson disease. Nature Reviews. Neurology 11, 370 (2015).
Tan, A. H. et al. Small intestinal bacterial overgrowth in Parkinson’s disease. Parkinsonism Relat Disord 20, 535–540, https://doi.org/10.1016/j.parkreldis.2014.02.019 (2014).
Su, C. M. et al. Manifestations and Outcomes of Patients with Parkinson’s Disease and Serious Infection in the Emergency Department. Biomed Res Int 2018, 6014896, https://doi.org/10.1155/2018/6014896 (2018).
Fang, F. et al. CNS infections, sepsis and risk of Parkinson’s disease. Int J Epidemiol 41, 1042–1049, https://doi.org/10.1093/ije/dys052 (2012).
Manzanillo, P. S. et al. The ubiquitin ligase parkin mediates resistance to intracellular pathogens. Nature 501, 512–516, https://doi.org/10.1038/nature12566 (2013).
Hartlova, A. et al. LRRK2 is a negative regulator of Mycobacterium tuberculosis phagosome maturation in macrophages. EMBO J 37, https://doi.org/10.15252/embj.201798694 (2018).
Hui, K. Y. et al. Functional variants in the LRRK2 gene confer shared effects on risk for Crohn’s disease and Parkinson’s disease. Sci Transl Med 10, https://doi.org/10.1126/scitranslmed.aai7795 (2018).
Shokal, U. et al. Effects of co-occurring Wolbachia and Spiroplasma endosymbionts on the Drosophila immune response against insect pathogenic and non-pathogenic bacteria. BMC Microbiol 16, 16, https://doi.org/10.1186/s12866-016-0634-6 (2016).
Acknowledgements
We thank Drs. Katrina Ramonell and Laura Reed (University of Alabama) for contributing equipment and Dr. Jay Hirsh (University of Virginia) for providing fly stocks. This work was supported by National Institutes of Health (NIH) Grant R15 NS078728 to J.M.O. and start-up funds from the University of Alabama to S.C.
Author information
Authors and Affiliations
Contributions
U.M. and J.M.O. designed research; U.M., M.N.S. and S.C. performed research; U.M., S.C. and J.M.O. analyzed data; and U.M. wrote the manuscript. All authors reviewed and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Maitra, U., Scaglione, M.N., Chtarbanova, S. et al. Innate immune responses to paraquat exposure in a Drosophila model of Parkinson’s disease. Sci Rep 9, 12714 (2019). https://doi.org/10.1038/s41598-019-48977-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-48977-6
This article is cited by
-
Potential of whey protein as a nutritional intervention in alleviating manganese and paraquat-mediated oxidative stress in Apis mellifera meda
Apidologie (2023)
-
Inflammation and immune dysfunction in Parkinson disease
Nature Reviews Immunology (2022)
-
Suppression of intestinal dysfunction in a Drosophila model of Parkinson’s disease is neuroprotective
Nature Aging (2022)
-
Neuroprotective Effects of Resveratrol in In vivo and In vitro Experimental Models of Parkinson’s Disease: a Systematic Review
Neurotoxicity Research (2022)
-
An apocrine mechanism delivers a fully immunocompetent exocrine secretion
Scientific Reports (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.