Abstract
Marijuana smoke contains cannabinoids, immunosuppressants, and a mixture of potentially-mutagenic chemicals. In addition to systemic disease, it is thought to contribute to oral disease, such as tooth loss, tissue changes in the gums and throat, and possibly oral pharyngeal cancer. We used a cross-sectional study of 20 marijuana users and 19 control non-users, to determine if chronic inhalation-based exposure to marijuana was associated with a distinct oral microbiota at the two most common sites of head and neck squamous cell carcinoma (HNSCC), the lateral border of the tongue and the oral pharynx. At the tongue site, genera earlier shown to be enriched on HNSCC mucosa, Capnocytophaga, Fusobacterium, and Porphyromonas, were at low levels in marijuana users, while Rothia, which is found at depressed levels on HNSCC mucosa, was high. At the oral pharynx site, differences in bacteria were distinct, with higher levels of Selenomonas and lower levels of Streptococcus which is what is seen in HNSCC. No evidence was seen for a contribution of marijuana product contaminating bacteria to these differences. This study revealed differences in the surface oral mucosal microbiota with frequent smoking of marijuana.
Similar content being viewed by others
Introduction
Marijuana is the most commonly used recreational drug in the United States. In 2015, it was reported that 22.2 million U.S. individuals ≥12 years old had used marijuana in the past month. With a shift toward legalization of the drug, questions arise regarding the health implications of marijuana utilization1. Besides the well-known cognitive effects of marijuana usage, it is suspected to have a role in a number of other types of conditions based on its component properties. For example, marijuana smoke may have carcinogenic capabilities as it contains potentially carcinogenic aromatic hydrocarbons2,3. In addition, marijuana contains cannabinoids which can bind both CB1 and CB2 cannabinoid receptors on immune cells and have been shown to have strong effects on immune cell function and can alter inflammation4,5,6,7.
The term “cannabis stomatitis” has been used to describe the oral epithelial changes that occur with inhalation and chewing of cannabis8. These changes include leukoedema of the oral mucosa, and hyperkeratosis and leukoplakia at various oral sites. In addition, gingival inflammation can occur with chronic use8,9,10. Intraorally, frequent recreational cannabis use, including marijuana and hashish, was associated with higher risk of severe periodontitis, including deeper probing depths and more clinical attachment loss. Even after excluding former and current tobacco users, poor periodontal status was twice as likely in frequent cannabis users versus non-users11. Chronic usage is associated with reduced airway function, increased airway infection, and impairment of macrophage activity, though the evidence for the latter effects is limited7,12,13. Marijuana usage has been linked to increased incidence of precancerous mucosal histology in both the head and neck and bronchi in some studies10. Some epidemiological studies suggest increased risk of head and neck and lung cancer with marijuana usage, however the risk is lower than that with usage of tobacco and the increased risk is currently debatable12,14,15,16. At this point, the evidence for effects of marijuana on systemic disease is poor in part due to limited data on usage as a result of its illegal nature. With legalization, this will change and further research can come about.
Studies regarding gut microbiome, and more recently the oral cavity, have shown linkages between differences in microbiome at these sites and incidence of a number of diseases including cancer, diabetes, and autoimmune disorders. The state of the oral microbiome can cause dental caries and play a role in periodontal disease initiation and progression. Evidence is beginning to accumulate that changes in oral bacteria can be associated with head and neck cancer and other distal cancers17. Researchers have noted taxonomic differences in samples collected from lesions in patients with oral squamous cell carcinoma (OSCC) versus those from normal mucosa18,19,20,21. When compared to healthy controls, OSCC lesions showed increased bacterial diversity and increased relative abundances of certain taxa at the phylum (Spirochaetes, Fusobacteria, and Bacteroidetes) and genus (Fusobacterium, Treponema, Dialister, Catonella, Filifactor, Peptococcus, Parvimonas, Peptostreptococcus, Campylobacter, and Pseudomonas) levels18,21,22. It is apparent that the oral mucosal microbiome is different in the presence of disease, such as OSCC, and even in precancerous lesions versus healthy tissue. However, a causative role for bacteria in OSCC incidence or progression is speculative19. The upper and lower digestive tract microbiome which can be altered due to diet, air quality, or lifestyle23,24,25 may be an important link between environment and disease.
The aim of this study was to evaluate the hypothesis that marijuana use via inhalation is associated with differences in the oral mucosal microbiome compared to nonusers. In this study two major sites of head and neck cancer, the lateral tongue and oral pharyngeal mucosa, were examined in marijuana users and nonusers in order to determine if marijuana-specific differences occur in the makeup of these biofilms. It is well established that the oral biofilm on nonpathological mucosa is at least in part dependent on the nature of the mucosal cell character and the associated extracellular matrix23,26,27. This would suggest that monitoring of microbiota on oral surfaces provides a window into histology and biochemistry of the mucosa at that site, perhaps even before obvious histological changes occur17. Differences in bacteria at these cancer-prone sites may serve as a marker of marijuana usage associated change in oral mucosa that might precede cancer. The analysis provides insight on how daily or almost daily usage of combusted marijuana may be pathological at these sites.
Results
Selected subjects
There were twenty marijuana-user subjects, comprised of 17 males and 3 females. The control group, non-marijuana-users, consisted of 16 males and 3 females. The marijuana group subjects ranged from 18 to 49 years of age with mean age 25.7 and median age 24. That of the control group ranged from 18 to 58, with the mean age 27.3, and median age 25.5. None of the subjects had active caries or visible signs of more than mild gingival inflammation, nor did they have mucosal lesions at sample collection sites.
Bacteria communities at oropharynx and lateral border of the tongue in marijuana users
Profiles of bacteria communities from two cancer prone sites, the mucosal surface of the lateral border of the tongue and the oral pharynx, collected with a cotton swab were generated using 16s rDNA sequencing. This consisted of 1,950,345 raw reads for tongue and 2,053,618 raw reads from the oral pharynx. Bray-Curtis dissimilarity (non-phylogenetic) metric was used to perform analysis of similarity (ANOSIM), which indicated minimal overall differences between the two populations at both lateral border of the tongue (R = 0.033, p = 0.159) and the oral pharynx (R = 0.056, p = 0.075). Similarly, alpha diversity analysis revealed no differences in distributions in marijuana users and controls at either mucosal site, indicating that if there were differences in the populations they were limited to a minority of taxa (Fig. 1). The barplot in Fig. 2 reveals the 20 most common genera in marijuana users and nonusers at both sites.
Boxplots of Shannon Diversity Index, Margalef Richness, and Pielou’s Evenness compare marijuana nonusers and marijuana (MJ) users for taxa identified at the two mucosal sites. The Student’s t test compared the significance of taxa differences for users and nonusers using each test. (A) Shannon, tongue t < 0.831 and oral pharynx t < 0.249. (B) Margalef, tongue t < 0.717 and oral pharynx t < 0.204. (C) Evenness, tongue t < 0.819 and oral pharynx, t < 0.507.
Examination of Individual taxa in the marijuana users and controls
16s rDNA based HOMD database taxonomic assignments revealed 67 genera detected at lateral border of the tongue and 65 at the oral pharynx. DESeq2 of these genera revealed potential differences at both sites between controls and marijuana users (Table 1), though with this test only those for tongue were significant after correction for multiple testing.
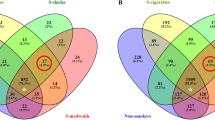
Alternatively, to identify taxa that distinguish marijuana users from nonusers at both sites, the linear discriminant analysis (LDA) effect size (LEfSe) method was used to determine statistically differentially represented taxa while also taking account of biological consistency and performing effect size estimations (Fig. 3). Use of this approach revealed a number of species and genera that were potential markers of marijuana exposure. At the lateral border of the tongue, several species, Rothia mucilaginosa, Delftia acidovorans, Veillonella atypica and Bosea vestrisii, were higher in marijuana users, while three genera, Fusoabcteria, Porphymonas, and Capnocytophaga, were lower (Fig. 3C). A cladogram (Fig. 3B) was used to represent the predominant bacteria and the taxonomic relationship of the microbiota in both groups at the site. At the oral pharynx on the genus level, the abundance of Veillonella (p = 0.0001) was higher in marijuana users along with Mogobacterium and Selenomonas, while Streptococcus was lower (Fig. 3C,D).
Taxa identified as distinct between marijuana, M, and control, C, groups at the two sites using LEfSe analysis. (A) LDA scores show significant differences in bacteria at the lateral border of the tongue in the marijuana and control nonusers. (B) Cladogram constructed shows the phylogenetic distribution of the differentially abundant taxa. (C) LDA scores show differences at the oral pharynx in the marijuana group and the controls. (D) Cladogram reveals phylogenetic distribution of differentially abundant taxa at the oral pharynx site in the marijuana and control groups.
Measurement of bacteria found in commercially prepared marijuana at oral sites
16s rRNA gene sequenced datasets of operational taxonomic units (OTUs) from the tongue and oral pharynx sites were subjected to taxa assignment using the SILVA 132 comprehensive library of taxonomically known 16s rRNA genes28. The Silva 132 library includes 16s genes not included in the HOMD database of taxa that are very rarely found in saliva or other oral samples. Supplemental Figure S1 reveals of the 6 bacteria species shown earlier to be found reproducibly in commercially prepared marijuana, Acinetobacter baumannii, Escherichia coli, Psuedomonas aeruginosa, Ralstonia pickettii, Salmonella enterica, and Stenotrophomonas maltophilia, several were found at low levels at the lateral tongue and oral pharyngeal sites29. Stenotrophomonas maltophilia was discernible on the species level by 16s rDNA sequencing but found only at the tongue site in one marijuana subject and one nonuser. More subjects were positive based on 16s rDNA sequence for the genus Stenotrophomonas at this site but no difference in levels in marijuana versus negative control group was seen. Ralstonia and Psuedomonas were only differentiable at the genus level and based on 16s analysis, few samples had detectable levels, and again there was no difference whether the subject used marijuana or not. A species of Pseudomonas, unidentified marine bacterioplankton, was found in both marijuana and control subjects at both sites, lateral tongue and oral pharyngeal, but at similar low levels. Two additional bacterial species found in marijuana, Acinetobacter bumanni and Escherichi Coli, were not found in any subjects at the tested sites based on this analysis.
Discussion
The results of this study suggest that daily or almost daily inhalation of marijuana in the past month correlates with differentially abundant taxa of the oral microbiome in samples taken from the lateral border of the tongue and from the oral pharynx. Both sites are distinct in regard to the marijuana-associated microbiome. Given that they are cancer-prone sites, one question arises: does marijuana use correlate with differences in the biofilm known to correlate with cancer? Several studies have catalogued, to varying degrees, biofilm differences in malignant HNSCC versus normal head and neck mucosa18,20,21,30,31. These tumor-associated taxa may be indicative of pathology, such as inflammation18, and not just malignancy when compared to healthy tissue. Several genera have been shown to be at higher levels with head and neck squamous cell carcinoma (HNSCC) and oral squamous cell carcinoma (OSCC), including Fusobacterium, Capnocytophaga, Alloprevotella, Treponema, Campylobacter, Selenomonas with other groups at lower levels: Streptococcus, Veillonella, Lautropia, Actinomyces, and Rothia. At the lateral tongue site, none of these differences were replicated in the marijuana users18,21. In fact, Rothia and Lautropia were higher, while Capnocytophaga, Fusobacteria and Porphyromonas were lower, with the latter thought to be mechanistically linked to OSCC32. In contrast, at the oral pharyngeal site, differences in marijuana users versus controls were more consistent with the cancer state. There were lower levels of Streptococcus and higher levels of Selenomonas, though Veillonella was higher21,30,33. It is quite possible that with marijuana use, we see these changes in bacteria levels that correspond with subtle changes in the mucosa that occur as normal tissue progresses toward pathology and ultimately SCC, but do not have a causative role in this process. Further research needs to be done to determine if marijuana shows these correlations in a larger population, whether marijuana is the causative factor for those changes in taxa, and the relationship of those bacteria with oral mucosal disease.
In conclusion, the lateral tongue site showed microbial changes with marijuana usage but these were inconsistent with cancer. Results with the oral pharynx were mixed, but overall more consistent with the malignant state. Interestingly, there has been research that found a 2.6 times more likely association of primary squamous cell carcinoma of the head and neck in marijuana users, once adjusting for cigarette smoking, alcohol use, and other risk factors34. However, this finding has not been consistent amongst all studies15,16,35 and a link between marijuana usage and OSCC is not well supported. There is more support for the observation that marijuana usage is associated epidemiologically with HPV-positive oral pharyngeal SCC14,36. This was corroborated in a second population37. In contrast, a negative correlation between marijuana usage and tongue SCC was seen. It is not clear if marijuana usage is associated with HPV infection38,39. The strong association of marijuana usage and oral pharynx SCC risk is consistent with what was observed in the marijuana users in regard to bacterial taxa at the oral pharynx site in this study.
The cross-sectional nature of this study makes it possible that any component of marijuana lifestyle may be responsible for the differences in oral pharynx and lateral border of the tongue biofilms. While we excluded tobacco users and controlled for age and gender, there are other potential confounders that may correlate with marijuana usage. However, if marijuana itself is directly responsible it is reasonable to assume reactive oxidative chemicals2,3, some produced in the burning process, can damage cellular and bacterial macromolecules and play a role in changing mucosa or bacteria so to alter biofilm microbes. The immunomodulatory activity of marijuana may also play a role in the makeup of the biofilm. Marijuana cannabinoids can alter apoptotic rates, cell proliferation and/or chemotaxis in T and B lymphocytes, and macrophage and dendritic cells4,5,6,7. Finally, marijuana, depending on the method of preparation, can contain a range of potentially pathogenic bacteria as live contaminants29,40. While we saw no evidence for elevated levels of marijuana product-associated bacteria contaminants at the two mucosal sites tested (Supplemental Fig. S1), oxidative and immunomodulatory effects of marijuana were not examined. Limitations of the study are the slight differences in of races and ethnicities in the two groups, with 8 Asians, 5 Caucasians, 4 Hispanics and 1 African American and three unknowns in the control group and 5 Asians, 9 Caucasions and 5 Hispanics and 1 African American in the marijuana group. (Supplemental Table 1). Differences in subgingival and salivary microbiota between Caucasian Americans and African Americans, Chinese Americans and Latin Americans have been noted41 while others have suggested the environment plays a larger role than genetics in oral microbiota42,43 It is also possible oral hygiene level may differ between the marijuana and control groups which can contribute to differences in mucosal microbiota. Finally, undetected periodontal disease, with minimal inflammation due to marijuana usage4,5, may be present in some patients and could contribute to taxa found at distal sites such as tongue and oral pharynx in those patients.
Both sites tested, oral pharynx and lateral border of the tongue, showed microbial differences with marijuana usage. This finding is consistent with the speculative model that mucosal biofilm constituents may provide a window to the state of mucosal health given the known effects of long-term marijuana usage on oral mucosa histology in some users. At the lateral border of the tongue the differences may not be carcinogenic, while for oral pharynx they may indeed be. Given the association of marijuana usage with HPV-induced SCC, and possibly HPV infections themselves, one model is that marijuana-related biofilm organism changes are manifestations of the same phenomenon – the immune state. Future experiments will test the effect of marijuana induction to a naive host and the effect on immune state and oxidative changes at these same sites. It will also be important to confirm the taxa differences seen at both mucosal sites in additional marijuana users and controls, and examine other variables so to better establish the relationship between marijuana usage and the taxa differences.
Methods
Study subjects
This pilot study consisted of 20 frequent marijuana users with 17 of these daily or almost daily users of marijuana in the past month (defined as using marijuana on 20 or more days in the past month)44. Exclusion criteria were systemic disease, overt oral disease such as active caries or moderate periodontitis, concurrent tobacco usage, usage of antibiotics in the last month45, usage of bactericidal mouthwash in the last 24 hours, and age less than 18 years. Controls who had never used marijuana nor tobacco were matched based on gender and age. Subjects who had clinically visible lesions at sites of sample collection were excluded. Participants were recruited from the dental clinics of the University of Illinois at Chicago College of Dentistry. All subjects provided written informed consent to participate in accordance with guidelines of the Office for the Protection of Research Subjects of the University of Illinois at Chicago, with formal approval of the study protocol by the Institutional Review Board 1 of the University of Illinois at Chicago.
Sampling procedure
A cotton swab Fisherbrand 23-400-114 was used to collect samples from two intraoral sites (lateral border of the tongue and oral pharynx). Samples were placed into (TE) buffer solution (10 mmol Tris-HCl and 1 mmol EDTA) with a pH of 8.0 and frozen until DNA extraction could be completed.
Characterization of microbial community structure
Genomic DNA was extracted from saliva samples using ZR Fungal/Bacterial DNA MiniPrep D6005 (Zymo Research Corp, Irvine, CA, USA) as recommended. Amplification reactions were performed targeting the V1-V3 variable regions of bacterial 16S ribosomal RNA (rRNA) genes using the primer sets 27F/534R, followed by a second amplification with barcoding and Illumina MiSeq sequencer as described earlier46,47. Library preparation and sequencing were performed at the University of Illinois at Chicago Sequencing Core48.
Bioinformatics analysis
For taxa assignment and measurement, reverse sequences from the FASTQ files were analyzed using the software package QIIME249,50,51. Sequences were trimmed if the average quality was lower than 20. As a result, the forward read sequences were truncated at 250 nt. and the reverse at 225 nt. Dada2-plugin in QIIME2 was used to sequence denoise and generate feature data and feature tables for the dataset49,52. It has earlier been shown that sequencing of V3 of 16S rDNA can be used to differentiate oral taxa when aligned to the HOMD annotated sequences53. Taxonomy assignment was done by classify-consensus-blast function with 98% match identity to the Human Oral Microbiome Database54. Of the 81 samples there were on average 32,746 reverse sequence reads generated per sample, ranging from 9884 to 46,125 reads. Samples with sequence reads below 9000 were eliminated prior to this analysis. For the alignment to the SILVA database of 16s rDNA sequences28 QIIME1.8 was used with merged paired reads of the V1-V3 region as described in the supplement and taxonomic assignment using the SILVA 132 database as described in the supplement49,55,56.
Alpha and beta diversity analysis were performed using Primer757. This analysis included calculation of alpha diversity, Shannon’s diversity index of both species number and their distribution, Margalef’s of numbers, and Pielou’s of evenness of distribution58,59,60 and the significance of the differences between marijuana users and nonusers was derived using an unpaired Student’s t test. Beta diversity analysis was done using Bray Curtis dissimilarity (non-phylogentic) metric. ANOSIM) tests were performed to determine if microbial communities were significantly different between groups47,57. Differences in specific microbiota taxonomic abundance between the groups were tested using DESeq2 based on the negative binomial distribution61. Both this and the LEfSe analysis were performed after eliminating taxa that appeared in 15% or fewer subjects at >3 reads62.
Data Availability
The sequencing data from this study is deposited in the Sequence Read Archive at NCBI.
Change history
10 February 2022
A Correction to this paper has been published: https://doi.org/10.1038/s41598-022-06597-7
References
Behavioral Health Trends in the United States: Results from the 2014 National Survey on Drug use and Health Vol. SMA 15-4927, NSDH Series H-50 (ed Center for Behavioral Health Statistics and Quality) (Human Health Services, 2015).
Hoffmann, D., Brunnemann, K. D., Gori, G. B. & E. L., W. In Recent Advances in Phytochemsitry 63–81 (Srpinger, US, 1975).
Novotny, M., Merli, F., Wiesler, D., Fenci, M. & Saeed, T. Fractionation and capillary gas chromatographic - mass spectrometric characterization of the neutral compents of mairijuana and tobacco smoke condensates. J Chromatogr. 238, 141–150 (1982).
Cabral, G. A., Rogers, T. J. & Lichtman, A. H. Turning Over a New Leaf: Cannabinoid and Endocannabinoid Modulation of Immune Function. Journal of neuroimmune pharmacology: the official journal of the Society on NeuroImmune Pharmacology 10, 193–203, https://doi.org/10.1007/s11481-015-9615-z (2015).
Karsak, M. et al. Attenuation of allergic contact dermatitis through the endocannabinoid system. Science 316, 1494–1497, https://doi.org/10.1126/science.1142265 (2007).
Katchan, V., David, P. & Shoenfeld, Y. Cannabinoids and autoimmune diseases: A systematic review. Autoimmun Rev 15, 513–528, https://doi.org/10.1016/j.autrev.2016.02.008 (2016).
Volkow, N. D., Compton, W. M. & Weiss, S. R. Adverse health effects of marijuana use. N Engl J Med 371, 879, https://doi.org/10.1056/NEJMc1407928 (2014).
Rawal, S. Y., Tatakis, D. N. & Tipton, D. A. Periodontal and oral manifestations of marijuana use. J Tenn Dent Assoc 92, 26–31; quiz 31–22 (2012).
Boyce, S. H. & Quigley, M. A. Uvulitis and partial upper airway obstruction following cannabis inhalation. Emerg Med (Fremantle) 14, 106–108 (2002).
Cho, C. M., Hirsch, R. & Johnstone, S. General and oral health implications of cannabis use. Aust Dent J 50, 70–74 (2005).
Shariff, J. A., Ahluwalia, K. P. & Papapanou, P. N. Relationship Between Frequent Recreational Cannabis (Marijuana and Hashish) Use and Periodontitis in Adults in the United States: National Health and Nutrition Examination Survey 2011 to 2012. J Periodontol 88, 273–280, https://doi.org/10.1902/jop.2016.160370 (2017).
Tashkin, D. P. Marijuana and Lung Disease. Chest 154, 653–663, https://doi.org/10.1016/j.chest.2018.05.005 (2018).
Ghasemiesfe, M. et al. Marijuana Use, Respiratory Symptoms, and Pulmonary Function: A Systematic Review and Meta-analysis. Ann Intern Med 169, 106–115, https://doi.org/10.7326/M18-0522 (2018).
Berthiller, J. et al. Marijuana smoking and the risk of head and neck cancer: pooled analysis in the INHANCE consortium. Cancer Epidemiol Biomarkers Prev 18, 1544–1551, https://doi.org/10.1158/1055-9965.EPI-08-0845 (2009).
de Carvalho, M. F. et al. Head and neck cancer among marijuana users: a meta-analysis of matched case-control studies. Arch Oral Biol 60, 1750–1755, https://doi.org/10.1016/j.archoralbio.2015.09.009 (2015).
Osazuwa-Peters, N., Adjei-Boakye, E., Loux, T. M., Varvares, M. A. & Schootman, M. Insufficient Evidence to Support or Refute the Association between Head and Neck Cancer and Marijuana Use. J Evid Based Dent Pract 16, 127–129, https://doi.org/10.1016/j.jebdp.2016.06.004 (2016).
Zarco, M. F., Vess, T. J. & Ginsburg, G. S. The oral microbiome in health and disease and the potential impact on personalized dental medicine. Oral Dis 18, 109–120, https://doi.org/10.1111/j.1601-0825.2011.01851.x (2012).
Al-Hebshi, N. N. et al. Inflammatory bacteriome featuring Fusobacterium nucleatum and Pseudomonas aeruginosa identified in association with oral squamous cell carcinoma. Scientific reports 7, 1834, https://doi.org/10.1038/s41598-017-02079-3 (2017).
Shin, J. M., Kamarajan, P., Fenno, J. C., Rickard, A. H. & Kapila, Y. L. Metabolomics of Head and Neck Cancer: A Mini-Review. Front Physiol 7, 526, https://doi.org/10.3389/fphys.2016.00526 (2016).
Wang, H. et al. Microbiomic differences in tumor and paired-normal tissue in head and neck squamous cell carcinomas. Genome Med 9, 14, https://doi.org/10.1186/s13073-017-0405-5 (2017).
Zhao, H. et al. Variations in oral microbiota associated with oral cancer. Scientific reports 7, 11773, https://doi.org/10.1038/s41598-017-11779-9 (2017).
Shin, J. M. et al. Microbial Communities Associated with Primary and Metastatic Head and Neck Squamous Cell Carcinoma - A High Fusobacterial and Low Streptococcal. Signature. Scientific reports 7, 9934, https://doi.org/10.1038/s41598-017-09786-x (2017).
Lloyd-Price, J., Abu-Ali, G. & Huttenhower, C. The healthy human microbiome. Genome Med 8, 51, https://doi.org/10.1186/s13073-016-0307-y (2016).
Rosenfeld, C. S. Gut Dysbiosis in Animals Due to Environmental Chemical Exposures. Front Cell Infect Microbiol 7, 396, https://doi.org/10.3389/fcimb.2017.00396 (2017).
Singh, R. K. et al. Influence of diet on the gut microbiome and implications for human health. J Transl Med 15, 73, https://doi.org/10.1186/s12967-017-1175-y (2017).
Mark Welch, J. L., Rossetti, B. J., Rieken, C. W., Dewhirst, F. E. & Borisy, G. G. Biogeography of a human oral microbiome at the micron scale. Proc Natl Acad Sci USA 113, E791–800, https://doi.org/10.1073/pnas.1522149113 (2016).
Segata, N. et al. Composition of the adult digestive tract bacterial microbiome based on seven mouth surfaces, tonsils, throat and stool samples. Genome Biol 13, R42, https://doi.org/10.1186/gb-2012-13-6-r42 (2012).
Quast, C. et al. The SILVA ribosomal RNA gene database project: improved data processing and web-based tools. Nucleic Acids Res 41, D590–596, https://doi.org/10.1093/nar/gks1219 (2013).
McKernan, K. et al. Metagenomic analysis of medicinal Cannabis samples; pathogenic bacteria, toxigenic fungi, and beneficial microbes grow in culture-based yeast and mold tests. F1000Res 5, 2471, https://doi.org/10.12688/f1000research.9662.1 (2016).
Schmidt, B. L. et al. Changes in abundance of oral microbiota associated with oral cancer. PloS one 9, e98741, https://doi.org/10.1371/journal.pone.0098741 (2014).
Yang, S. F. et al. Compositional and functional variations of oral microbiota associated with the mutational changes in oral cancer. Oral Oncol 77, 1–8, https://doi.org/10.1016/j.oraloncology.2017.12.005 (2018).
Perera, M., Al-Hebshi, N. N., Speicher, D. J., Perera, I. & Johnson, N. W. Emerging role of bacteria in oral carcinogenesis: a review with special reference to perio-pathogenic bacteria. J Oral Microbiol 8, 32762, https://doi.org/10.3402/jom.v8.32762 (2016).
Wolf, A. et al. The salivary microbiome as an indicator of carcinogenesis in patients with oropharyngeal squamous cell carcinoma: A pilot study. Scientific reports 7, 5867, https://doi.org/10.1038/s41598-017-06361-2 (2017).
Zhang, Z. F. et al. Marijuana use and increased risk of squamous cell carcinoma of the head and neck. Cancer Epidemiol Biomarkers Prev 8, 1071–1078 (1999).
Hayes, R. B. et al. Association of Oral Microbiome With Risk for Incident Head and Neck Squamous Cell Cancer. JAMA. Oncol 4, 358–365, https://doi.org/10.1001/jamaoncol.2017.4777 (2018).
Gillison, M. L. et al. Distinct risk factor profiles for human papillomavirus type 16-positive and human papillomavirus type 16-negative head and neck cancers. Journal of the National Cancer Institute 100, 407–420, https://doi.org/10.1093/jnci/djn025 (2008).
Marks, M. A. et al. Association of marijuana smoking with oropharyngeal and oral tongue cancers: pooled analysis from the INHANCE consortium. Cancer Epidemiol Biomarkers Prev 23, 160–171, https://doi.org/10.1158/1055-9965.EPI-13-0181 (2014).
Kahn, J. A. et al. Prevalence and risk factors for oral DNA tumor viruses in HIV-infected youth. Journal of medical virology 88, 1944–1952, https://doi.org/10.1002/jmv.24555 (2016).
Ortiz, A. P. et al. Association of marijuana use with oral HPV infection and periodontitis among Hispanic adults: Implications for oral cancer prevention. J Periodontol 89, 540–548, https://doi.org/10.1002/JPER.17-0372 (2018).
Thompson, G. R. 3rd et al. A microbiome assessment of medical marijuana. Clin Microbiol Infect 23, 269–270, https://doi.org/10.1016/j.cmi.2016.12.001 (2017).
Mason, M. R., Nagaraja, H. N., Camerlengo, T., Joshi, V. & Kumar, P. S. Deep sequencing identifies ethnicity-specific bacterial signatures in the oral microbiome. PloS one 8, e77287, https://doi.org/10.1371/journal.pone.0077287 (2013).
Gupta, V. K., Paul, S. & Dutta, C. Geography, Ethnicity or Subsistence-Specific Variations in Human Microbiome Composition and Diversity. Frontiers in microbiology 8, 1162, https://doi.org/10.3389/fmicb.2017.01162 (2017).
Hoffman, K. L. et al. Oral microbiota reveals signs of acculturation in Mexican American women. PloS one 13, e0194100, https://doi.org/10.1371/journal.pone.0194100 (2018).
Azofeifa, A. et al. National Estimates of Marijuana Use and Related Indicators — National Survey on Drug Use and Health, United States, 2002–2014. MMWR Surveill Summ 2016 65, 1–25, https://doi.org/10.15585/mmwr.ss6511a1 (2016).
Zaura, E. et al. Same Exposure but Two Radically Different Responses to Antibiotics: Resilience of the Salivary Microbiome versus Long-Term Microbial Shifts in Feces. mBio 6, e01693–01615, https://doi.org/10.1128/mBio.01693-15 (2015).
Adami, G. R. et al. Effects of green tea on miRNA and microbiome of oral epithelium. Scientific reports 8, 5873, https://doi.org/10.1038/s41598-018-22994-3 (2018).
Ionescu, D. et al. In Manual of Environmental Microbiology (ed Marylynn, V. and Nakatsu, Yates, Cindy, H. & Miller, Robert V. and Pillai, Suresh D) (2016).
Naqib, A. et al. Making and Sequencing Heavily Multiplexed, High-Throughput 16S Ribosomal RNA Gene Amplicon Libraries Using a Flexible, Two-Stage PCR Protocol. Methods Mol Biol 1783, 149–169, https://doi.org/10.1007/978-1-4939-7834-2_7 (2018).
Caporaso, J. G. et al. QIIME allows analysis of high-throughput community sequencing data. Nat Methods 7, 335–336, https://doi.org/10.1038/nmeth.f.303 (2010).
Hall, M. & Beiko, R. G. 16S rRNA Gene Analysis with QIIME2. Methods Mol Biol 1849, 113–129, https://doi.org/10.1007/978-1-4939-8728-3_8 (2018).
Navas-Molina, J. A. et al. Advancing our understanding of the human microbiome using QIIME. Methods Enzymol 531, 371–444, https://doi.org/10.1016/B978-0-12-407863-5.00019-8 (2013).
Callahan, B. J. et al. DADA2: High-resolution sample inference from Illumina amplicon data. Nat Methods 13, 581–583, https://doi.org/10.1038/nmeth.3869 (2016).
Lazarevic, V. et al. Analysis of the salivary microbiome using culture-independent techniques. J Clin Bioinforma 2, 4, https://doi.org/10.1186/2043-9113-2-4 (2012).
Dewhirst, F. E. et al. The human oral microbiome. J Bacteriol 192, 5002–5017, https://doi.org/10.1128/JB.00542-10 (2010).
Edgar, R. C. Search and clustering orders of magnitude faster than BLAST. Bioinformatics 26, 2460–2461, https://doi.org/10.1093/bioinformatics/btq461 (2010).
Zhang, J., Kobert, K., Flouri, T. & Stamatakis, A. PEAR: a fast and accurate Illumina Paired-End reAd mergeR. Bioinformatics 30, 614–620, https://doi.org/10.1093/bioinformatics/btt593 (2014).
Clarke, K. R., Gorley, R. N., Somerfield, P. J. & Warwick, R. M. Change in marine communities: an approach to statistical analysis and interpretation. (Primer-E: Plymouth, 2014).
Magurran, A. E. Ecological Diversity and its Measurement. (Princeton University Press, 1988).
Pielou, C. E. Introduction ot Mathematical Ecology. (Wiley-Interscience, 1969).
Shannon, C. E. A Mathematical Theory of Communication. Bell System Technical Journal 27, 379–423 (1948).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol 15, 550, https://doi.org/10.1186/s13059-014-0550-8 (2014).
Segata, N. et al. Metagenomic biomarker discovery and explanation. Genome Biol 12, R60, https://doi.org/10.1186/gb-2011-12-6-r60 (2011).
Acknowledgements
We thank Yalu Zhou for help with DESeq2 analysis.
Author information
Authors and Affiliations
Contributions
G.R.A. and T.N. designed the study, and wrote the manuscript. T.N. screened subjects and collected all samples. L.P.K. performed QIIME2 analysis and along with G.R.A. performed additional data analysis. J.L. processed samples, organized records and performed data analysis. All the authors contributed critical discussion and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this Article was revised: The original version of this Article contained an error in the spelling of the author Taylor Newman which was incorrectly given as Taylor M. Newman.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Newman, T., Krishnan, L.P., Lee, J. et al. Microbiomic differences at cancer-prone oral mucosa sites with marijuana usage. Sci Rep 9, 12697 (2019). https://doi.org/10.1038/s41598-019-48768-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-48768-z
This article is cited by
-
Molecular pathways in the development of HPV-induced oropharyngeal cancer
Cell Communication and Signaling (2023)
-
Cannabis related side effects in otolaryngology: A scoping review
Journal of Otolaryngology - Head & Neck Surgery (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.