Abstract
We report to evaluate if disorganization of the retinal inner layers (DRIL) obtained by swept-source optical coherence tomography (SS-OCT) predicts the postoperative best-corrected visual acuity (BCVA) to treat proliferative diabetic retinopathy (PDR). Twenty-one eyes of 21 patients who underwent vitrectomy for PDR were studied retrospectively. BCVA and SS-OCT images were obtained until 6 months postoperatively. The associations between BCVA and SS-OCT parameters measured in a 1-mm central foveal area were evaluated. The DRIL length, external limiting membrane disruption, and ellipsoid zone (EZ) disruption 1 month postoperatively were associated positively with the postoperative logarithm of the minimum angle of resolution (logMAR) BCVA at 1, 3, and 6 months (1 month, p = 0.009, p = 0.013, p = 0.001; 3 months, p = 0.03, p = 0.021, p = 0.002; and 6 months, p = 0.021, p = 0.013, and p = 0.005, respectively). The eyes with a 500-µm or longer DRIL 1 month postoperatively (19%, 4/21 eyes) had significantly worse VA at 1, 3, and 6 months postoperatively (p = 0.007, p = 0.008, and p = 0.020, respectively). Multilinear regression analysis of all visits until 6 months postoperatively showed that the DRIL was correlated more significantly (p = 0.0004) with logMAR BCVA than the disrupted EZ length. The DRIL in the early postoperative period may predict the visual outcomes after treating PDR.
Similar content being viewed by others

Introduction
Diabetic retinopathy (DR) is the leading cause of blindness among working-age patients, and the prevalence of diabetes mellitus is predicted to increase globally1,2. Proliferative diabetic retinopathy (PDR) is the advanced stage of DR characterized by preretinal neovascularization3, which causes vitreous hemorrhages and traction retinal detachments. Those complications are possible indications for pars plana vitrectomy (PPV), but the visual prognosis in some cases is poor. Numerous studies have reported the visual outcome after diabetic vitrectomy; however, the factors predictive of the visual outcomes remain to be elucidated4. The identification of reliable biomarkers that predict the visual acuity (VA) after diabetic vitrectomy is important for the care of patients with PDR.
Sun and associates first characterized the disorganization of the retinal inner layers (DRIL) as the inability to distinguish any of the boundaries of the ganglion cell layer-inner plexiform layer (GCL-IPL) complex, inner nuclear layer (INL), and outer plexiform layer (OPL) in the horizontal B-scan of optical coherence tomography (OCT) images5,6. Regarding diabetic macular edema (DME), those investigators reported that the DRIL was associated with worse VA and that the changes in the DRIL predicted the subsequent changes in VA. The purpose of the current study was to evaluate the correlation between the DRIL and postoperative VA in eyes with PDR that required vitrectomy. Because one of the most frequent complications of PDR is vitreous hemorrhage, which prevents retinal evaluation, we used swept-source OCT (SS-OCT) to analyze patients during the first month postoperatively. SS-OCT uses a longer wavelength (1,050 nm) than SD-OCT and is expected to penetrate through opacities or into the deeper tissues and obtain sharp images in the early postoperative period.
Results
Twenty-one eyes of 21 patients (14 men, 7 women; mean age, 62.4 ± 13.0 years; range 41–82 years) who underwent PPV with cataract surgery and a SS-OCT examination were included in this study. All eyes were diagnosed with PDR without any other retinal diseases preoperatively. Table 1 shows the preoperative characteristics of the patients and OCT parameters 1 month postoperatively. The surgical indications included vitreous hemorrhage (12 patients, 57%), traction retinal detachment (5 patients, 24%), and epiretinal membrane (4 patients, 19%). One month postoperatively, seven eyes (33%) had cystoid macular edema (CME) and none had subretinal fluid (SRF). Twenty-one (100%) eyes had hyperreflective foci (HRF); one (5%) eye had HRF only in the inner retina and 20 eyes (95%) had HRF in the inner and outer retina. The mean central subfield thickness (CST) was 289 ± 111 µm (range, 100–633 µm). The mean size of the external limiting membrane (ELM) disruption was 109 ± 187 µm (range, 0–551 µm), and the mean size of the ellipsoid zone (EZ) disruption was 434 ± 379 µm (range, 0–1,000 µm). The total area of CME was 0.11 ± 0.36 mm2 (range, 0–1.67 mm2). Four (19%) eyes were with a mean DRIL length of 500 µm or longer. The mean DRIL length of all B-scans was 350 ± 234 µm (range, 0–1,000 µm). The percentage of the scans with DRIL out of five scans in each eye was 30 ± 32% (range, 0–100%).
Table 2 shows the associations between the OCT parameters 1 month postoperatively and the postoperative logarithm of the minimal angle of resolution (logMAR) best corrected visual acuity (BCVA) at 1, 3, and 6 months postoperatively. No significant correlations was seen between the CST at 1 month and the postoperative logMAR BCVA at each visit (1 month, p = 0.39; 3 months, p = 0.16; and 6 months, p = 0.40) or between the total area of the CME at 1 month and the postoperative logMAR BCVA at each visit (1 month, p = 0.40; 3 months, p = 0.41; and 6 months, p = 0.49). The mean DRIL length at 1 month was correlated positively with the postoperative logMAR BCVA at each visit (1 month, p = 0.009; 3 months, p = 0.030; and 6 months, p = 0.021). The percentages of the five scans with DRIL in each eye at 1 month were correlated positively with the postoperative logMAR BCVA at each visit (1 month, p = 0.002; 3 months, p = 0.009; and 6 months, p = 0.008). The mean length of the ELM disruption at 1 month was correlated positively with the postoperative logMAR BCVA at each visit (1 month, p = 0.013; 3 months, p = 0.021; and 6 months, p = 0.013). The mean length of the EZ disruption at 1 month was correlated positively with the postoperative logMAR BCVA at each visit (1 month, p = 0.001; 3 months, p = 0.002; and 6 months p = 0.005).
We divided the patients into two groups: those with a mean DRIL length less than 500 µm 1 month postoperatively (DRIL−) and those with a mean DRIL length of 500 µm or longer 1 month postoperatively (DRIL+). The postoperative BCVA was significantly better in the DRIL− group than in the DRIL+ group at each visit (1 month, p = 0.007; 3 months p = 0.008; and 6 months p = 0.020) (Table 3).
Multilinear regression was performed to predict the VA based on the length of the DRIL length and EZ disruption across all visits up to 6 months postoperatively (Table 4). The mean length of the DRIL was a significant (p = 0.0004) predictor of the BCVA, whereas the mean length of the EZ disruption was not associated significantly (p = 0.45) with the BCVA.
Discussion
The current study evaluated the association between SS-OCT parameters and the postoperative BCVA in PDR. The horizontal and vertical extents of the DRIL, ELM disruption, and EZ disruption 1 month postoperatively were correlated negatively with the logMAR BCVA at 1, 3, and 6 months postoperatively. The eyes with a DRIL 500 µm or longer 1 month postoperatively were associated significantly with worse BCVA at 1, 3, and 6 months postoperatively compared to eyes with a DRIL less than 500 µm long. Multilinear regression analysis showed that the length of the DRIL was correlated significantly with the BCVA at all visits until 6 months postoperatively. These results suggested that the DRIL 1 month postoperatively may predict the visual outcomes of surgeries performed to treat PDR and that the DRIL may reflect the VA at the same time. To the best of our knowledge, this is the first study to investigate the association between the DRIL and VA after surgery to treat PDR using SS-OCT.
Both the reports on PDR after surgery and numerous studies of various macular diseases have reported that disruption of the outer retina is a significant parameter to predict the visual prognosis7,8,9,10,11. In the current study, simple linear regression analysis, the presence or length of the DRIL, and the integrity of the ELM and EZ were considered as the factors that might predict the visual outcomes; however, multilinear regression analysis showed that the DRIL, but not the EZ, was associated with the visual outcomes. Radwan et al. reported previously that the length of the DRIL was associated significantly with the VA after resolution of center-involved DME, whereas the disrupted lengths of the ELM and EZ were not associated significantly12. The reason that the inner retina was more impaired compared to the outer retina is unclear. Several studies have suggested that the inner retinal status was more important for assessing the visual prognosis in eyes with an ERM13,14,15. Govetto et al. reported that the presence of ectopic inner foveal layers in ERMs is a newly identified factor associated with significant visual loss14. Cho et al. reported that the inner retinal irregularity index was the factor that was most significantly correlated with visual outcomes before and after ERM surgery13.
Although the mechanism of development of the DRIL is unknown, previous studies have reported that the DRIL was identified as a potential biomarker for macular edema caused by DR, retinal vein occlusion, and uveitis6,16,17,18. The diseases that involve retinal vascular dysfunction can disrupt the blood retinal barrier, which is followed subsequently by accumulation of extracellular fluid in the intra- or subretinal space. The DRIL obviously is a result of destruction of the neural structure due to compromised retinal microcirculation; however, PDR also includes a fibrovascular membrane that creates tractional stress on the macula. Zur et al. reported recently that the DRIL also can be a reliable biomarker for eyes that underwent surgery for ERM15. Thus, the combined mechanisms that include vascular and mechanical stress-related factors might have exaggerated formation of DRIL in eyes with PDR.
The inability to visualize the boundaries of the inner retinal layers may indicate anatomic collapse in the visual transmission pathway. Because the inner retinal layers include the bipolar, amacrine, or horizontal cells, which correspond to the second neuron in the visual system, this may indicate that the DRIL disrupts pathways that transmit visual information from the photoreceptors to the ganglion cells. These anatomic alterations also might be responsible for less robust associations observed with other outer retinal parameters observed by OCT5.
The current study had several limitations. This study was retrospective and the sample size was relatively small. As mentioned previously, the reason that the DRIL and not the EZ was identified as a crucial factor for visual outcomes is unknown; however, the current sample size might have affected this. On the other hand, the sample size were still appropriate because of following reason. First, because the current study has a pilot nature, we were not able to hypothesize a difference to be detected in the analysis. Second, for example, outcomes comparing visual acuity between DRIL− and DRIL+ which are shown in Table 3 demonstrated median logMAR approximately 0.3 and 1.0, respectively. Those values still seem to be appropriate and the differeces were reasonable when we see PDR patients who received surgery. That strengthens the fact that as long as we performed appropriate statistical analysis, the data we detected in the current study were significant. Another limitation of the current study is that there is controversy regarding the inter-observer difference in DRIL evaluation19. To evaluate DRIL objectively, an automated detection system of DRIL should be developed in future20,21. Nonetheless, the horizontal and vertical extents of the DRIL 1 month postoperatively were correlated negatively with the logMAR BCVA postoperatively. Moreover, the length of the DRIL was correlated significantly with the BCVA at all visits until 6 months postoperatively. These results demonstrated that the DRIL evaluated 1 month postoperatively may be a prognostic factor that present the visual outcomes after PDR surgeries. The detailed definition of DRIL and the technology to detect DRIL automatically is required for future application in the clinic where the visual prognosis can be predicted during the early postoperative stage in eyes with PDR.
Methods
This single-site, retrospective study adhered to the tenets of the Declaration of Helsinki; the institutional review board of Osaka University Hospital approved the study. Informed consent was obtained from all individual participants. We enrolled 21 eyes of 21 consecutive patients who had undergone both 25-gauge PPV for PDR from May 2015 to March 2018 and an SS-OCT examination at the Department of Ophthalmology of Osaka University Hospital. The inclusion criteria were eyes with PDR and an SS-OCT examination 1 month postoperatively. Eyes were excluded that had any other retinal diseases, a history of other intraocular surgeries, postoperative poor-quality OCT images, or an additional surgery within 6 months after the initial surgery.
PPV was performed using a 25-gauge vitrectomy system (Constellation Vision System; Alcon Laboratories, Inc., Fort Worth, Texas, USA) and a wide-angle viewing system (Resight, Carl Zeiss Meditec, Oberkochen, Germany). Phacoemulsification and intraocular lens implantation were performed simultaneously in all phakic eyes. A core vitrectomy was performed with intravitreal injection of triamcinolone acetonide to visualize the vitreous gel. A peripheral vitrectomy was performed under scleral indentation. Fibrovascular membranes were removed with a vitreous cutter, vitreoretinal forceps, and scissors. Panretinal photocoagulation was applied up to the vitreous base under scleral indentation in all eyes. Internal limiting membrane peeling was performed at the surgeons’ discretion. Fluid-gas exchange was performed in eyes with retinal breaks or a rhegmatogenous retinal detachment. Air or 20% sulfur hexafluoride was used in these cases.
All patients underwent comprehensive ophthalmic examinations at all visits until 6 months postoperatively. The examinations included measurements of BCVA and intraocular pressure, slit-lamp biomicroscopy, fundus photography, and SS-OCT (DRI-OCT, Topcon Medical Systems, Tokyo, Japan). This SS-OCT system operates at a speed of 100,000 A-scans/second using a 1,050-nm wavelength light source and has an 8-µm axial resolution and 20-µm lateral resolution. For each eye, we used the 5 Line Cross mode with a 12-mm scan length and 0.1-mm spacing to acquire five horizontal B-scan images centered on the fovea.
Image analysis
For each study eye, a 1-mm overlay centered on the fovea was placed over each of the five horizontal scans. We investigated the following in this area as previously reported18: the presence of CME, SRF, and HRF; CST (µm); DRIL length (µm); vertical extent of the DRIL (defined as the number of scans out of five with DRIL calculated as a percentage); ELM disruption (µm); EZ disruption (µm); and total area of the CME (mm2). ImageJ software (National Institutes of Health, Bethesda, Maryland, USA) was used to manually measure each OCT parameter. The means of the CST, DRIL, ELM disruption, and EZ disruption were calculated from the five horizontal scans at each visit.
Definition of DRIL
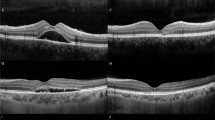
DRIL was defined as the inability to distinguish any of the boundaries of the GCL-IPL complex, INL, and OPL within 1 mm centered on the fovea (Fig. 1). The length of the DRIL was measured in five horizontal B-scans, and the measurements were averaged for each eye at each visit. Two masked investigators (TI, SS) independently evaluated the DRIL. When there was disagreement, the evaluation was discussed to reach a consensus.
Disorganization of retinal inner layers (DRIL) is evaluated within 1 mm centered on the fovea. (A,B) A swept-source optical coherence tomography (SS-OCT) image of a 64-year-old woman whose decimal visual acuity (VA) is 1.0 1 month postoperatively. The boundaries of the inner nuclear layer (INL) are almost clearly identified despite the presence of cystoid macular edema and hyperreflective foci at the outer nuclear layer. (C,D) A SS-OCT image of a 56-year-old man whose decimal VA is 0.06 1 month postoperatively. The boundaries of the INL cannot be identified clearly.
Statistical analysis
The BCVA were converted to the logarithm of the minimal angle of resolution (logMAR) for all analyses. Counting fingers and hand motions VAs were converted as previously reported22. All statistical analyses were conducted using Statcel4 software (OMS, Saitama, Japan). Simple linear regression and multilinear regression analyses were performed to determine the relationship between the BCVA and OCT parameters. The Mann-Whitney U-test was used for group comparisons. P < 0.05 was considered significant.
References
Klein, R., Klein, B. E., Moss, S. E., Davis, M. D. & DeMets, D. L. The Wisconsin epidemiologic study of diabetic retinopathy. III. Prevalence and risk of diabetic retinopathy when age at diagnosis is 30 or more years. Arch Ophthalmol. 102, 527–532 (1984).
Guariguata, L. et al. Global estimates of diabetes prevalence for 2013 and projection for 2035. Diabetes Res Clin Pract. 103, 137–149 (2014).
Stitt, A. W. et al. The progress in understanding and treatment of diabetic retinopathy. Prog Retin Eye Res. 51, 156–186 (2016).
Newman, D. K. Surgical management of the late complication of proliferative diabetic retinopathy. Eye. 24, 441–449 (2010).
Sun, J. K. et al. Disorganization of the retinal inner layers as a predictor of visual acuity in eyes with center-involved diabetic macular edema. JAMA Ophthalmol. 132, 1309–1316 (2014).
Sun, J. K. et al. Neural retinal disorganization as a robust marker of visual acuity in current and resolved diabetic macular edema. Diabetes. 64, 2560–2570 (2015).
Alasil, T. et al. Relationship between optical coherence tomography retinal parameters and visual acuity in diabetic macular edema. Ophthalmology. 117, 2379–2386 (2010).
Ito, S., Miyamoto, N., Ishida, K. & Kurimoto, Y. Association between external limiting membrane status and visual acuity in diabetic macular oedema. Br J Ophthalmol. 97, 228–232 (2013).
Shin, H. J., Lee, S. H., Chung, H. & Kim, H. C. Association between photoreceptor integrity and visual outcome in diabetic macular edema. Graefes Arch Clin Exp Ophthalmol. 250, 61–70 (2012).
Tortorella, P., D’Ambrosio, E., Iannetti, L., De Marco, F. & La Cava, M. Correlation between visual acuity, inner segment/outer segment junction, and cone outer segment tips line integrity in uveitic macular edema. Biomed Res Int. 2015, 853728 (2015).
Kang, J. W., Lee, H., Chung, H. & Kim, H. C. Correlation between optical coherence tomographic hyperreflective foci and visual outcomes after intravitreal bevacizumab for macular edema in branch retinal vein occlusion. Graefes Arch Clin Exp Ophthalmol. 252, 1413–1421 (2014).
Radwan, S. H. et al. Association of disorganization of retinal inner layers with vision after resolution of center-involved diabetic macular edema. JAMA Ophthalmol. 133, 820–825 (2015).
Cho, K. H., Park, S. J., Cho, J. H., Woo, S. J. & Park, K. H. Inner-retinal irregularity index predicts postoperative visual prognosis in idiopathic epiretinal membrane. Am J Ophthalmol. 168, 139–149 (2016).
Govetto, A., Lalane, R. A., Sarraf, D., Figueroa, M. S. & Hubschman, J. P. Insights into epiretinal membranes: presence of ectopic inner foveal layers and a new optical coherence tomography staging scheme. Am J Ophthalmol. 175, 99–113 (2017).
Zur, D. et al. Disorganization of retinal inner layers as a biomarker for idiopathic epiretinal membrane after macular surgery-the DREAM study. Am J Ophthalmol. 196, 129–135 (2018).
Das, R., Spence, G., Hogg, R. E., Stevenson, M. & Chakravarthy, U. Disorganization of inner retina and outer retinal morphology in diabetic macular edema. JAMA Ophthalmol. 136, 202–208 (2018).
Mimouni, M. et al. Disorganization of the retinal inner layers as a predictor of visual acuity in eyes with macular edema secondary to vein occlusion. Am J Ophthalmol. 182, 160–167 (2017).
Grewal, D. S., O’Sullivan, M. L., Kron, M. & Jaffe, G. J. Association of disorganization of retinal inner layers with visual acuity in eyes with uveitic cystoid macular edema. Am J Ophthalmol. 177, 116–25 (2017).
Schmidt-Erfurth, U. & Michl, M. Disorganization of retinal inner layers and the importance of setting boundaries. JAMA Ophthalmol. 137, 46–47 (2019).
Huazhu, F. et al. Automatic optic disc detection in OCT slices via low-rank reconstruction. IEEE Trans. Biomed. Eng. 62, 1151–1158 (2015).
Huazhu, F. et al. A deep learning system for automated angle-closure detection in anterior segment optical coherence tomography images. Am J Ophthalmol. 203, 37–45 (2019).
Schulze-Bonsel, K., Feltgen, N., Burau, H., Hansen, L. & Bach, M. Visual acuities “hand motion” and “counting fingers” can be quantified with the freiburg visual acuity test. Invest Ophthalmol Vis Sci. 47, 1236–40 (2006).
Author information
Authors and Affiliations
Contributions
Study design, data acquisition, and statistical analysis: T.I. and S.S. interpretation of data: T.I., S.S., N.S., K.e.N., H.S. writing (original draft): T.I. writing (review and editing): S.S., N.S., K.e.N., H.S., K.o.N. final approval of manuscript: all authors; supervision: H.S. and K.o.N.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ishibashi, T., Sakimoto, S., Shiraki, N. et al. Association between disorganization of retinal inner layers and visual acuity after proliferative diabetic retinopathy surgery. Sci Rep 9, 12230 (2019). https://doi.org/10.1038/s41598-019-48679-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-48679-z
This article is cited by
-
Changes in optical coherence tomography biomarkers in eyes with advanced idiopathic epiretinal membrane treated with dexamethasone implantation
BMC Ophthalmology (2024)
-
Assessment of area and structural irregularity of retinal layers in diabetic retinopathy using machine learning and image processing techniques
Scientific Reports (2024)
-
Automated assessment of the smoothness of retinal layers in optical coherence tomography images using a machine learning algorithm
BMC Medical Imaging (2023)
-
Association of disorganization of retinal inner layers with optical coherence tomography angiography features in branch retinal vein occlusion
Graefe's Archive for Clinical and Experimental Ophthalmology (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.