Abstract
This study examined the ability of the extreme lateral interbody fusion (XLIF) procedure to restore coronal and sagittal alignments for patients with adult spinal deformity (ASD) using computed tomography multiplanar reconstruction (CT-MPR). Thirty-eight patients with ASD undergoing correction and fixation with XLIF at 114 levels were studied. The coronal segmental Cobb angle, coronal regional Cobb angle (L1-5), sagittal segmental Cobb angle, sagittal regional Cobb angle (L1-5), intervertebral disc height and, vertebral body rotation (VBR) were measured before and after of XLIF surgery using CT-MPR. The mean sagittal segmental Cobb angle, the coronal segmental Cobb angle and VBR were corrected from 5.0° to 9.0°, from 6.3° to 4.3° and from 12.2° to 10.8°, respectively. The mean of the intervertebral disc heights increased significantly from 6.0 mm to 10.4 mm postoperatively. Although increases in coronal segmental Cobb, sagittal segmental Cobb, and intervertebral disc height at each level were significant, there were no significant differences in each parameter acquired by spine levels. The results also showed that it was difficult for L4/5 level to obtain the most postoperative coronal Cobb, sagittal Cobb and intervertebral disc height. This study evaluated the alignment improvement effect of stand-alone XLIF in ASD patients using CT-MPR. For the lower lumbar spine, it is difficult to obtain a lordosis more than 10 degrees with stand-alone XLIF for correcting ASD. Therefore, it is thought that correction such as osteotomy or compression technique to the posterior fusion may be necessary during the 2nd stage surgery.
Similar content being viewed by others
Introduction
Adult spinal deformity (ASD) is defined as an angular deformity presenting after skeletal maturity, and can be divided into coronal (scoliosis), sagittal (kyphosis), and mixed1. With the sagittal alignment of the spine being one of the most important factors influencing disorders of lower back, the surgical correction of ASD is meant to restore spinal balance and improve the patient’s health-related quality of life (QOL)2,3,4. Traditional approaches to surgically correct ASD include anterior-posterior approaches or, more commonly, posterior-only approaches5. Moreover, more rigid and severe deformities may need a combined anterior-posterior approach or a posterior three-column reconstruction technique to obtain correction of spinal alignment. However, ASD surgery remains extremely challenging, and these procedures are invasive for the elderly6.
The extreme lateral interbody fusion (XLIF) technique, which uses a transpsoas retroperitoneal approach, has been popularized as a minimally invasive alternative surgical option for anterior column reconstruction and arthrodesis7. One of the advantages of XLIF is that a wide interbody cage can be inserted into the narrowed intervertebral disc space, correcting the scoliosis and achieving stable fixation. Therefore, XLIF has been reported to achieve good clinical and radiological results when employed in the correction surgery for ASD patients8,9. XLIF has also been shown to be safe in elderly patients and is said to be superior to open transforaminal lumbar interbody fusion (TLIF) historical controls in terms of complication rate, blood loss, and transfusion rate10. To date, numerous radiographic studies have shown significant improvement in foraminal height, intervertebral disc height, and the cross-sectional area with indirect decompression through the XLIF procedure. Multicenter studies on the XLIF procedure have reported improvements in coronal segmental angles, segmental lordosis, and disc height11,12,13. According to formulas for ASD correction, the restoration of lumbar lordosis is considered to be most important. Since L4- S1 lordosis accounts for 2/3 of the total lumbar lordosis, emphasis is placed on restoring lordosis in the lower lumbar levels14.
Recently, it has been reported that significant improvements in sagittal and coronal alignments can be obtained in correction surgery for ASD patients by using a two-stage combined approach15. However, most past studies have evaluated alignment after posterior spinal surgery and not lateral lumbar interbody fusion (LLIF) alone16,17. Therefore, an analysis on the corrective capability of LLIF alone is important to consider if osteotomies are necessary in addition to segmental LLIF fixation to achieve the planned correction. The purpose of this study is to evaluate the spinal alignments of ASD patients treated by XLIF alone using computed tomography multiplanar reconstruction (CT-MPR) immediately after the first stage of surgery.
Results
The spine-pelvic parameters in the preoperative standing full-length x-rays of the 38 patients are shown in Table 1.
As seen in Table 2, the mean sagittal segmental Cobb angle measured 5.0° preoperatively, and 9.0° postoperatively (p < 0.001). The mean preoperative regional lumbar lordosis angle increased from 27.0° (range, kyphosis 7.1° to lordosis 78.1°) to 36.0° (range, lordosis 9.9° to 78.5°). The mean coronal segmental Cobb angle was 6.3° preoperatively and 4.3° postoperatively (p < 0.01). The mean pre- and postoperative regional lumbar coronal Cobb angles were 16.0° and 12.6°, respectively (p = 0.05). The mean disc height was 6.0 mm preoperatively, and 10.4 mm postoperatively (p < 0.001). The ⊿ sagittal segmental Cobb angle, ⊿ coronal segmental Cobb angle, and ⊿ disc height were 4.0 ± 5.2°, 2.0 ± 4.1°, 4.4 ± 2.5 mm, respectively.
XLIF was performed for a total of 114 levels in the 38 patients as follows: 1-level procedure (n = 1), 2-level (n = 10), 3-level (n = 15), and 4-level (n = 12). The position of the XLIF cage was 40.6 ± 14.0%, and from the front of less than 60% to the center were 105 levels (92.1%). Regarding the position of the XLIF cages, there was a tendency for the cages to be placed more posteriorly as the levels descended through L1/2 to L4/5, but there was no statistically significant difference between spinal levels (data not shown).
We compared the results between the 10 degrees cages (10°, n = 88 levels) and 15 degrees cages (15°, n = 26 levels), but cage obliquity was not significantly associated with scoliosis, lordosis, or disc height change (Table 3).
The segmental alignment after the XLIF surgery showed that it was most smallest for the L4/5 level to gain postoperative coronal Cobb, sagittal Cobb, and intervertebral disc height. Although increases in coronal segmental Cobb, sagittal segmental Cobb, and intervertebral disc height at each level were significant (Table 4), ⊿ sagittal segmental Cobb angle, ⊿ coronal segmental Cobb angle, and ⊿ disc height were not statistically significant by spinal levels (Fig. 1).
In addition, the mean vertebral body rotation (VBR) was 12.2 ± 9.4° preoperatively and 10.8° ± 8.9° postoperatively (p < 0.001). When compared to the spine levels, we found that there was no statistically significant difference between spine levels (Table 5).
Analysis of the correlation between ⊿ sagittal segmental Cobb and cage position of the 114 levels showed a negative weak correlation (r = −0.269, P < 0.001). The correlation between ⊿ sagittal segmental Cobb and pre-sagittal segmental Cobb (r = −0.486, P < 0.001) and pre-intervertebral disc height (r = −0.251, P < 0.01) were found (Table 6). That is, the smaller the sagittal segmental Cobb and intervertebral disc height were before XLIF surgery, the greater the increase in sagittal segmental Cobb obtained through surgery.
Discussion
Various formulas have been reported to plan the corrective surgery for ASD. The most respected formula is that proposed by Schwab, but his formula was based on analysis of cases 60 years old, and the applicability of his formula to Japanese patients is often questioned due to the differences in body type between Westerners and Japanese18,19,20,21.
The first report of the corrective properties that XLIF has on alignment was reported by Pimenta, who described the insertion of the cages into the relatively rigid vertebral lateral rim after anterior disc dissection22. Especially in cases of lumbar degenerative scoliosis accompanied by osteophytes, the narrowed disc space can be accessed after resection of the osteophytes with dedicated bone chisels. Insertion of the large XLIF cage into the intervertebral disc naturally corrects the segmental coronal alignment. Restoration of lumbar lordosis is also important in surgeries to correct ASD, and LLIF can correct sagittal deformities with larger lateral interbody cages. Regarding the installation position of cages, Kepler and colleagues reported that segmental lordosis was obtained by putting the cage in the anterior position. They reported significant lordosis correction with a mean segmental increase of 3.7° at levels in which XLIF cages were inserted, which is similar to the correction angle found in the current study23. Several studies have reported that the more the cage is positioned anteriorly, the greater the lordosis obtained23,24. From the correlation analysis, our current study also suggests that the more anterior the cage position, the greater the lordosis that is obtained. On the other hand, there is also study that reports cage position is not a factor7, and the effect of cage position is still under discussion. According to the systematic review by Phan et al. published in 201525, only three studies examined changes in coronal segmental Cobb angles. They reported that the average coronal segmental Cobb angles changed from 3.6° preoperatively to 1.1 ° after surgery. The average preoperative and postoperative coronal regional Cobb angles were 19.1° and 10.0°, respectively. Sagittal segmental Cobb and regional lumbar lordosis also significantly improved after the XLIF surgery. Furthermore, previous reports demonstrated that although XLIF significantly improves lumbar scoliosis and segmental lordosis, there was no effect on global lordosis. These results were different from our study. We think that global lordosis is influenced by the number of levels at which XLIF was performed and by the angle of preoperative global lordosis14,17. Unlike previous reports, many patients in our series had 3-level fusions carried out and had relatively small preoperative lumbar lordosis. However, these reports did not evaluate spinal alignment immediately after XLIF surgery.
When the alignment after XLIF surgery was analyzed by CT-MPR, XLIF surgery improved segmental coronal, sagittal alignment, and VBR with an average correction angle of 2.0°, 4.0°, 1.4° respectively, at the instrumented segments where XLIF cages were inserted. There was no statistical difference in the sagittal segmental Cobb angle obtained for each XLIF level. Despite the increase in intervertebral disc height obtained by XLIF surgery, possible reasons for the low restoration of lordosis at the L4/5 level include arthropathic changes of the posterior components of L4/5 (the ankylosis of the facet joint), selection of the cage size, and position of the cage. Each of these points need to be studied in the future to address this issue.
For kyphotic cases, it is difficult to obtain the proposed 10° lordosis in the lower lumbar spine with XLIF alone. We propose that this is an important finding because XLIF alone cannot greatly change the lordosis at the L4/5 level. Therefore, additional correction procedures including osteotomies and compression techniques from the posterior are believed to be necessary.
There are several limitations in this study. First, this was a retrospective study, so a future prospective study with a more detailed evaluation should be considered to determine the best strategy for correction of spinal alignment. Second, there is a wide spectrum of ASD cases that may be difficult to analyze as one group. Mild ASD cases have deformities mainly limited to the lateral side of the intervertebral disc whereas cases with severe kyphoscoliosis not only have deformity in the posterior components but may also have contraction of the anterior longitudinal ligaments as well as osteophytes and ossifications of the intervertebral discs. Many of our patients were kyphotic with a mean SVA of 181 mm in which sagittal alignment is markedly shifted anteriorly, and the possibility that this affected our results cannot be denied. Third, CT can only be obtained in the supine position and cannot evaluate standing alignment. Low-dose EOS system® (EOS Imaging, Paris, France) is considered an innovative slot-scanning radiograph system that provides valuable data for spinal surgery26. It can obtain radiographic images even when the patient is standing, sitting or squatting. We think that it is useful for evaluation in standing position if we use this. Furthermore, to properly evaluate the multiple factors that affect alignment, future studies will require more cases so that the relationship between cage position and the various Cobb angles as well as intervertebral disc heights can be examined for each level. Finally, most cages used in this study were 10° cages, but there were some 15° cages and the cage heights were also different. The selection of the cage angle as well as height may also be affecting the results.
Conclusion
The XLIF technique does improve scoliosis and segmental kyphosis in patients with ASD. However, there is a limit to the sagittal and coronal correction that can be gained with XLIF alone. To further evaluate the effect of XLIF on sagittal balance, long-term follow-up with a larger cohort will be required.
Patients and Methods
Ethics
The study protocol was reviewed and approved by The Committee on Ethics and the Institutional Review Board of Tokai University School of Medicine (18R-253), the House Clinical Study Committee, and Profit reciprocity Committee. With this study being retrospective, the requirement for informed consent was not deemed necessary.
Included patients
Patients were eligible for study enrollment if they were over 50 years old and were diagnosed with ASD that was unresponsive to conservative treatment of at least 6 months. Before surgery, all patients were radiologically evaluated with full length postero-anterior and lateral standing x-rays and CT-MPR. Generally, surgical treatment of thoracolumbar kyphoscoliosis is selected according to whether the deformity is mobile or fixed. Thus, we performed the supine bending such as lateral flexion, extension, and traction radiographs to examine the flexibility of the spinal deformity. CT can also be used to evaluate the status of the facet joints. Facet joint degeneration on the approach side was evaluated using the CT grading system27. In this series, patients with severe fixed anterior column that required three-column osteotomy were excluded.
Patient characteristics and operative details are given in Table 7. We conducted our retrospective review using a retrospective cohort. The study included ASD patients who underwent LLIF procedure using XLIF PEEK cages (NuVasive Inc., San Diego, CA, USA) at a single institute from January 2016 to December 2018. In total, 38 patients (aged 70.7 ± 7.8 years; 3 males and 35 females) who underwent XLIF with a transpsoas approach in a minimally invasive fashion, at one or more intervertebral levels, were enrolled in this study. Five of the 38 patients had degenerative kyphosis (DK), while the remaining 33 patients had degenerative kyphoscoliosis (DKS). The patients diagnosed with ASD were classified according to the SRS Schwab classification system28. In terms of the type of curve, 24 cases (63.2%) had a Lumbar curve (Type L), 1 case (2.6%) had a Double curve (Type D), and 13 cases (34.2%) did not have a curve in the coronal plane (Type N), but only a deformity in the sagittal plane. The average grade of facet joint degeneration on the approach side was 1.4; 17 (14.9%) levels were not affected by facet joint degeneration (grade 0), 55 (48.2%) levels presented with grade 1, 24 (21.1%) levels with grade 2, and 18 (15.8%) levels with grade 3. XLIF was performed across an average of 3.0 levels per patient on intervertebral levels L1–2 to L4–5.
Operative data
The operations were performed by four spine surgeons. A two-stage surgery was adopted: the first stage was the minimally invasive multilevel XLIFs via transpsoas approach, using the 10 or 15-degree lordotic XLIF cages. The second stage was the posterior corrective fusion with TLIF performed at L5-S1 and/or L4-L5 and posterior instrumentation using pedicle screws.
XLIF was performed through a right-sided approach in 13 patients and through a left-sided approach in 25 patients. Basically, in cases without significant coronal deformity, a left-sided approach was performed. In many cases, we performed the XLIFs on the concave side of the scoliosis with the hinge in the radiolucent table flexed opposite the curve to help reduce the scoliosis. As a result, 19 (76%) out of 25 patients except type N group have approached from the concave side.
Interbody fusion was completed using the XLIF technique as described by Ozgur et al.7. Briefly, the patient is positioned in the lateral decubitus position with the concave side of the lumbar coronal curve facing up. This facilitates access to the largest number of disc spaces with a relatively small incision. Blunt dissection is then used to access the disc spaces under fluoroscopic guidance. After removal of the disc material with a rongeur, a Cobb elevator was advanced gently under fluoroscopy guidance along the endplates to release the contralateral annulus. Cage size trials were followed by additional disc curettage and rasping of the endplates. All cages were inserted using two containment sliders to protect the endplates and to keep graft material inside the cage. For all patients, side-to side cage size was decided by width of the endplates at that level based on intraoperative fluoroscopic guidance, and titanium cages of a standard 18-mm width were used. The maximum distraction achieved during discectomy using the trial inserts provided guidance as to the height of the cage. The choice of these XLIF cages (CoRoent XL; NuVasive Inc. San Diego, CA, USA) was decided by the surgeon. Cage lengths ranged from 40 to 55 mm and heights from 8 to 12 mm.
For graft material, artificial bone material comprising hydroxyapatite and collagen (Refit; HOYA Techno-surgical, Tokyo, Japan) soaked in autologous bone marrow aspirate.
Radiological parameters
Radiographic assessment was performed preoperatively and postoperatively (within 7 days) using CT-MPR in contiguous 1-mm slices. CT-MPR was performed with the patients in spine position.
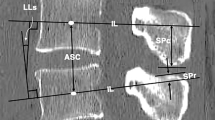
The radiographic parameter studied were coronal segmental Cobb angle, coronal regional Cobb angle (L1-L5), coronal disc height, sagittal segmental Cobb angle, and regional lumbar lordosis (L1-L5) (Fig. 2A). We also examined the pre and postoperative values for VBR (Fig. 2B). The rotation for each curve was measured from the pre- and postoperative axial CT-MPR using the methods described by Aaro and Dahlborn29.
(A) Diagram showing the measurement of regional lumbar lordosis (L1-L5) (ab), sagittal segmental Cobb angle (cd), coronal regional Cobb angle (L1-L5) (ef), coronal segmental Cobb angle (gh), and disc height (arrow; i or j). (B) The rotation for each curve was measured from the pre- and postoperative axial CT. The measured vertebral body rotation (VBR) was expressed relative to a vertical line which corresponded to VBR of 0°. (C) A midsagittal CT scan showing an XLIF cage implanted into the disc space. The cage center is located by the midpoint between the anterior and posterior radiomarkers of the cage. The arrow (a) indicates the distance between the anterior vertebral border (AVB) of the inferior endplate and the center of the cage. The arrow (b) indicates the anteroposterior width of the inferior end plate. Cage position = 100 × a/b (%).
The obtained sagittal segmental Cobb angle, coronal segmental Cobb angle, and disc height were expressed as ⊿ sagittal segmental Cobb angle (post-pre), ⊿ coronal segmental Cobb angle (post-pre), and ⊿ Intervertebral disc height (post-pre), respectively. Disc height was measured as an average of left and right disc heights.
We also used standard measurements reported elsewhere30 to assess coronal Cobb angle, sagittal vertical axis (SVA), lumbar lordosis (LL; T12–S1), thoracic kyphosis (TK; T5–12), pelvic incidence (PI), pelvic tilt (PT), and sacral slope (SS) by x-ray of the whole spine in standing position. The PI was measured as the angle between a line drawn perpendicular to the sacral end plate at its midpoint and a line drawn from the midpoint of the sacral end plate to the midpoint of the femoral head axis. The LL is the sagittal cobb angle measured between the superior end plate of T12 and the superior end plate of S131.
For CT evaluation, cases in which angle measurements were not possible due to a high degree of kyphosis were excluded from this study.
We also examined whether the position of the cage correlates with each evaluation angle. Cage positioning was evaluated using pre and postoperative CT scans in the midsagittal plane. The distance between the anterior vertebral border (AVB) of the inferior end plate and the center of the cage was measured and normalised to the AP width of the inferior end plate. The center of the cage was defined as the midpoint between the anterior and posterior radio-markers of the cage (Fig. 2C).
Statistical analysis
Statistical analyses were performed using IBM SPSS Statistics version 20.0 (IBM Corp., Armonk, NY). All values are expressed as mean ± standard deviation. An analysis of variance with a posthoc test (Mann–Whitney U test) was used for comparisons. The correlations between cage position and radiological parameters were analyzed using Spearman’s product-moment correlation coefficient.
To identify the minimum number of participants required for adequate statistical power, we used the G-Power Analysis software program (G Power 3.1.9, University of Düsseldorf, Germany, http://www.gpower.hhu.de/)32.
A power analysis performed to calculate the minimum sample size necessary to detect a difference between two dependent groups (calculated with Cohen’s d = 0.5, alpha = 0.05, power = 0.8) indicated a required sample size of 34 participants. A power analysis performed to calculate the minimum sample size necessary to detect a correlation (calculated with Correlation ρ H1 = 0.3, alpha = 0.05, power = 0.8) indicated a required total sample size of 84 participants. For all statistical analyses, the type 1 error was set at 5% and p < 0.05 was considered significant.
References
Aebi, M. The adult scoliosis. European spine journal: official publication of the European Spine Society, the European Spinal Deformity Society, and the European Section of the Cervical Spine Research Society 14, 925–948, https://doi.org/10.1007/s00586-005-1053-9 (2005).
Glassman, S. D. et al. The impact of positive sagittal balance in adult spinal deformity. Spine 30, 2024–2029 (2005).
Glassman, S. D., Berven, S., Bridwell, K., Horton, W. & Dimar, J. R. Correlation of radiographic parameters and clinical symptoms in adult scoliosis. Spine 30, 682–688 (2005).
Schwab, F. J. et al. Adult scoliosis: a quantitative radiographic and clinical analysis. Spine 27, 387–392 (2002).
Crandall, D. G. et al. Transforaminal lumbar interbody fusion with rhBMP-2 in spinal deformity, spondylolisthesis, and degenerative disease–part 1: Large series diagnosis related outcomes and complications with 2- to 9-year follow-up. Spine 38, 1128–1136, https://doi.org/10.1097/BRS.0b013e31828864e6 (2013).
Good, C. R., Auerbach, J. D., O’Leary, P. T. & Schuler, T. C. Adult spine deformity. Current reviews in musculoskeletal medicine 4, 159–167, https://doi.org/10.1007/s12178-011-9101-z (2011).
Ozgur, B. M., Aryan, H. E., Pimenta, L. & Taylor, W. R. Extreme Lateral Interbody Fusion (XLIF): a novel surgical technique for anterior lumbar interbody fusion. The spine journal: official journal of the North American Spine Society 6, 435–443, https://doi.org/10.1016/j.spinee.2005.08.012 (2006).
Dakwar, E., Cardona, R. F., Smith, D. A. & Uribe, J. S. Early outcomes and safety of the minimally invasive, lateral retroperitoneal transpsoas approach for adult degenerative scoliosis. Neurosurgical focus 28, E8, https://doi.org/10.3171/2010.1.focus09282 (2010).
Phillips, F. M. et al. Adult degenerative scoliosis treated with XLIF: clinical and radiographical results of a prospective multicenter study with 24-month follow-up. Spine 38, 1853–1861, https://doi.org/10.1097/BRS.0b013e3182a43f0b (2013).
Rodgers, W. B., Gerber, E. J. & Rodgers, J. A. Lumbar fusion in octogenarians: the promise of minimally invasive surgery. Spine 35, S355–360, https://doi.org/10.1097/BRS.0b013e3182023796 (2010).
Elowitz, E. H., Yanni, D. S., Chwajol, M., Starke, R. M. & Perin, N. I. Evaluation of indirect decompression of the lumbar spinal canal following minimally invasive lateral transpsoas interbody fusion: radiographic and outcome analysis. Minimally invasive neurosurgery: MIN 54, 201–206, https://doi.org/10.1055/s-0031-1286334 (2011).
Isaacs, R. E., Sembrano, J. N., Tohmeh, A. G. & Group, S. D. S. Two-Year Comparative Outcomes of MIS Lateral and MIS Transforaminal Interbody Fusion in the Treatment of Degenerative Spondylolisthesis: Part II: Radiographic Findings. Spine 41(Suppl 8), S133–144, https://doi.org/10.1097/brs.0000000000001472 (2016).
Oliveira, L., Marchi, L., Coutinho, E. & Pimenta, L. A radiographic assessment of the ability of the extreme lateral interbody fusion procedure to indirectly decompress the neural elements. Spine 35, S331–337, https://doi.org/10.1097/BRS.0b013e3182022db0 (2010).
Johnson, R. D., Valore, A., Villaminar, A., Comisso, M. & Balsano, M. Pelvic parameters of sagittal balance in extreme lateral interbody fusion for degenerative lumbar disc disease. Journal of clinical neuroscience: official journal of the Neurosurgical Society of Australasia 20, 576–581, https://doi.org/10.1016/j.jocn.2012.05.032 (2013).
Hsieh, M. K. et al. Combined anterior lumbar interbody fusion and instrumented posterolateral fusion for degenerative lumbar scoliosis: indication and surgical outcomes. BMC surgery 15, 26, https://doi.org/10.1186/s12893-015-0006-4 (2015).
Acosta, F. L. et al. Changes in coronal and sagittal plane alignment following minimally invasive direct lateral interbody fusion for the treatment of degenerative lumbar disease in adults: a radiographic study. Journal of neurosurgery. Spine 15, 92–96, https://doi.org/10.3171/2011.3.spine10425 (2011).
Sharma, A. K. et al. Lateral lumbar interbody fusion: clinical and radiographic outcomes at 1 year: a preliminary report. Journal of spinal disorders & techniques 24, 242–250, https://doi.org/10.1097/BSD.0b013e3181ecf995 (2011).
Yamato, Y. et al. Calculation of the Target Lumbar Lordosis Angle for Restoring an Optimal Pelvic Tilt in Elderly Patients With Adult Spinal Deformity. Spine 41, E211–217, https://doi.org/10.1097/brs.0000000000001209 (2016).
Schwab, F. J. et al. Radiographical spinopelvic parameters and disability in the setting of adult spinal deformity: a prospective multicenter analysis. Spine 38, E803–812, https://doi.org/10.1097/BRS.0b013e318292b7b9 (2013).
Rose, P. S. et al. Role of pelvic incidence, thoracic kyphosis, and patient factors on sagittal plane correction following pedicle subtraction osteotomy. Spine 34, 785–791, https://doi.org/10.1097/BRS.0b013e31819d0c86 (2009).
Inami, S. et al. Optimum pelvic incidence minus lumbar lordosis value can be determined by individual pelvic incidence. European spine journal: official publication of the European Spine Society, the European Spinal Deformity Society, and the European Section of the Cervical Spine Research Society 25, 3638–3643, https://doi.org/10.1007/s00586-016-4563-8 (2016).
Pimenta, L., Oliveira, L., Schaffa, T., Coutinho, E. & Marchi, L. Lumbar total disc replacement from an extreme lateral approach: clinical experience with a minimum of 2 years’ follow-up. Journal of neurosurgery. Spine 14, 38–45, https://doi.org/10.3171/2010.9.spine09865 (2011).
Kepler, C. K. et al. Factors influencing segmental lumbar lordosis after lateral transpsoas interbody fusion. Orthopaedic surgery 4, 71–75, https://doi.org/10.1111/j.1757-7861.2012.00175.x (2012).
Park, S. J. et al. The Ideal Cage Position for Achieving Both Indirect Neural Decompression and Segmental Angle Restoration in Lateral Lumbar Interbody Fusion (LLIF). Clinical spine surgery 30, E784–E790, https://doi.org/10.1097/bsd.0000000000000406 (2017).
Phan, K., Rao, P. J., Scherman, D. B., Dandie, G. & Mobbs, R. J. Lateral lumbar interbody fusion for sagittal balance correction and spinal deformity. Journal of clinical neuroscience: official journal of the Neurosurgical Society of Australasia 22, 1714–1721, https://doi.org/10.1016/j.jocn.2015.03.050 (2015).
McKenna, C. et al. EOS 2D/3D X-ray imaging system: a systematic review and economic evaluation. Health technology assessment (Winchester, England) 16, 1–188, https://doi.org/10.3310/hta16140 (2012).
Weishaupt, D., Zanetti, M., Boos, N. & Hodler, J. MR imaging and CT in osteoarthritis of the lumbar facet joints. Skeletal radiology 28, 215–219 (1999).
Schwab, F. et al. Scoliosis Research Society-Schwab adult spinal deformity classification: a validation study. Spine 37, 1077–1082, https://doi.org/10.1097/BRS.0b013e31823e15e2 (2012).
Aaro, S. & Dahlborn, M. Estimation of vertebral rotation and the spinal and rib cage deformity in scoliosis by computer tomography. Spine 6, 460–467 (1981).
Ames, C. P. et al. Impact of spinopelvic alignment on decision making in deformity surgery in adults: A review. Journal of neurosurgery. Spine 16, 547–564, https://doi.org/10.3171/2012.2.spine11320 (2012).
Schwab, F., Patel, A., Ungar, B., Farcy, J. P. & Lafage, V. Adult spinal deformity-postoperative standing imbalance: how much can you tolerate? An overview of key parameters in assessing alignment and planning corrective surgery. Spine 35, 2224–2231, https://doi.org/10.1097/BRS.0b013e3181ee6bd4 (2010).
Faul, F., Erdfelder, E., Lang, A. G. & Buchner, A. G*Power 3: a flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behavior research methods 39, 175–191 (2007).
Author information
Authors and Affiliations
Contributions
A.H., H.K., D.S., M.T., M.S., T.N. and M.W. conceived the study. A.H. and H.K. wrote the main manuscript text. A.H., D.S., T.N. and M.T. collected data. A.H., H.K. and M.W. analyzed results. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hiyama, A., Katoh, H., Sakai, D. et al. Changes in Spinal Alignment following eXtreme Lateral Interbody Fusion Alone in Patients with Adult Spinal Deformity using Computed Tomography. Sci Rep 9, 12039 (2019). https://doi.org/10.1038/s41598-019-48539-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-48539-w
This article is cited by
-
The analysis of segmental lordosis restored by oblique lumbar interbody fusion and related factors: building up preoperative predicting model
BMC Musculoskeletal Disorders (2024)
-
Effectiveness of a two-stage posterior-anterior–posterior surgery using subcutaneously preserved autologous bone grafts for adult spinal deformity: a retrospective observational study
Journal of Orthopaedic Surgery and Research (2024)
-
Short-term and mid-term evaluation of three types of minimally invasive lumbar fusion surgery for treatment of L4/L5 degenerative spondylolisthesis
Scientific Reports (2024)
-
Early outcomes of oblique lateral interbody fusion with posterior fixation versus posterior interbody fusion with fixation for treating adult degenerative scoliosis
Journal of Orthopaedic Surgery and Research (2023)
-
Effects of the cage height and positioning on clinical and radiographic outcome of lateral lumbar interbody fusion: a retrospective study
BMC Musculoskeletal Disorders (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.