Abstract
More than 400 syndromes associated with hearing loss and other symptoms have been described, corresponding to 30% of cases of hereditary hearing loss. In this study we aimed to clarify the mutation spectrum of syndromic hearing loss patients in Japan by using next-generation sequencing analysis with a multiple syndromic targeted resequencing panel (36 target genes). We analyzed single nucleotide variants, small insertions, deletions and copy number variations in the target genes. We enrolled 140 patients with any of 14 syndromes (BOR syndrome, Waardenburg syndrome, osteogenesis imperfecta, spondyloepiphyseal dysplasia congenita, Stickler syndrome, CHARGE syndrome, Jervell and Lange-Nielsen syndrome, Pendred syndrome, Klippel-Feil syndrome, Alport syndrome, Norrie disease, Treacher-Collins syndrome, Perrault syndrome and auditory neuropathy with optic atrophy) and identified the causative variants in 56% of the patients. This analysis could identify the causative variants in syndromic hearing loss patients in a short time with a high diagnostic rate. In addition, it was useful for the analysis of the cases who only partially fulfilled the diagnostic criteria.
Similar content being viewed by others
Introduction
Congenital hearing loss is one of the most common sensory disorders, affecting one out of 500–1000 newborns. Over half of the cases of congenital or early onset sensorineural hearing loss are estimated to be caused by genetic factors1, with 30% of these hereditary hearing loss patients affected by various syndromes. More than 400 syndromes associated with hearing loss and other symptoms have been described2.
The most commonly observed syndromes in clinical settings include Pendred syndrome, BOR syndrome, Waardenburg syndrome, osteogenesis imperfecta, Stickler syndrome, spondyloepiphyseal dysplasia congenita, CHARGE syndrome, Klippel-Feil syndrome, Alport syndrome, Treacher-Collins syndrome, Jervell Lange-Nielsen syndrome, Perrault syndrome, Norrie disease, and auditory neuropathy with optic atrophy. The clinical characteristics and responsible genes for these 14 syndromes are summarized in Table 1.
Branchio-Oto-Renal (BOR) syndrome (OMIM#113650 and #610896) or Branchio-Oto (BO) syndrome (OMIM#602588 and 608389) is characterized by the association of the branchial arch, external ear anomalies, hearing impairment and renal anomalies. BO/BOR syndrome is observed in one out of 40,000 children, and in 2% of profoundly deaf children3,4.
Waardenburg syndrome (WS1 OMIM#193500, WS2 OMIM#193510, #608890 and #611584, WS3 OMIM#148820, WS4 OMIM#277580, #613265 and #613266) is characterized by varying degrees of hearing impairment and pigmentation disturbances in the hair, skin and eyes5,6. WS is classified into four types based on clinical findings. The frequency of WS is 1/20,000–40,000 newborns5,6,7,8,9. Osteogenesis imperfecta type 1 (OMIM#166200) is an autosomal dominant inheritance disorder characterized by fractures with minimal or no trauma, blue sclera, hearing loss and otosclerosis10.
Spondyloepiphyseal dysplasia congenita (OMIM#183900) is an autosomal dominantly inherited chondrodysplasia characterized by a disproportionately short stature (short trunk), abnormal epiphyses and flattened vertebral bodies11.
Stickler syndrome (OMIM#108300, #604841, #614134 and #614284) is an inherited connective tissue disorder associated with myopia, retinal detachment, cleft palate, midfacial hypoplasia, arthritis and hearing impairment12,13,14,15,16,17. Alport syndrome (OMIM#301050, #203780 and #104200) is a progressive disease associated with glomerulonephritis, sensorineural hearing loss, and ocular complications caused by abnormalities in type IV collagen18,19,20.
CHARGE syndrome (OMIM#214800) is an autosomal dominant disorder characterized by congenital multiple anomalies (coloboma, heart defect, choanal atresia, retarded growth and development, genital hypoplasia and ear anomalies/deafness)21,22,23,24.
Jervell and Lange-Nielsen syndrome (OMIM#220400 and #612347) is a rare autosomal recessive cardio-auditory disorder characterized by congenital profound bilateral sensorineural hearing loss and a long QT interval with arrhythmia (torsade de pointes)25,26
Pendred syndrome (OMIM#274600) is an autosomal recessive disorder characterized by congenital hearing loss, goiter, and enlarged vestibular aqueduct27.
In this study, we conducted a comprehensive analysis of 140 Japanese syndromic hearing loss patients to obtain the mutation spectrums and clinical features by using next-generation sequencing (NGS) analysis with a multiple syndromic targeted resequencing panel.
Results
As shown in Table 2, we performed NGS analysis of 36 previously reported genes associated with syndromic hearing loss for 140 probands and identified the causative gene variants in 79 probands (56%). The diagnostic rate by syndrome was 32% (19/59) for BOR syndrome, 78% (18/23) for Waardenburg syndrome, 60% (3/5) for osteogenesis imperfecta, 100% (3/3) for Stickler syndrome, and 89% (32/36) for Pendred syndrome. On the other hand, we could not detect any causative gene variants for Klippel-Feil syndrome, Alport syndrome or Norrie disease cases.
Mutation spectrum and clinical features of BOR syndrome patients
We conducted genetic analysis of 59 probands with clinical findings of BO/BOR syndrome (16 typical cases, 43 atypical cases, Supplementary Table S1), and identified causative heterozygous variants in 19 probands (diagnostic rate 32%). We identified the causative variants in 12/16 typical cases, but the causative variants were identified in only 7/43 atypical cases. Table 3 summarizes the identified variants and clinical features of the probands and all affected family members for 18 families with EYA1 mutations, and one family with a SIX1 mutation. There were no BO/BOR cases caused by the SIX5 gene variant. Among the 12 EYA1 variants, 8 were truncating variants (five were nonsense, one was frameshift, two were splice site), three were missense variants and one was a copy number variation (one copy number loss). Four of them were novel variants and 8 of them were previously reported. JHLB4043 had one copy loss detected using NGS read depth data, which seemed to be deleted in all of the EYA1 gene, confirmed by array Comparative Genomic Hybridization (aCGH). The mutations identified in this study were located in exon 6 to exon 13, and frequently observed in exon 8 and exon 12. Two or more cases carried the same variants (p.R264X, p.R275X, c.867 + 5 G > A, p.R328X and p.R407Q). EYA1 variants were mainly identified from autosomal dominant families (10/18 cases); however, we also identified variants from 7 sporadic cases. Among them we confirmed de novo mutations in four families (Supplementary Fig. S1). The case with a SIX1 mutation was also caused by de novo mutation (Supplementary Fig. S1). In terms of the clinical features of all BO/BOR-affected patients with EYA1 and SIX1 gene variants (19 probands and their family members who carried the same variants; 34 patients in total), the most frequent symptom was hearing loss (31/ 32, 97%). Unilateral hearing loss was observed in 2 cases. The most frequent type of hearing loss was moderate mixed hearing loss. Middle and/or inner ear anomalies were observed in 22 of 23 cases who underwent CT imaging (96%). Twenty-seven of 31 cases had preauricular pits (87%), and 14 of 25 cases for whom information was available had branchial anomalies (56%). Renal anomalies, on the other hand, were revealed in only one of 7 cases for whom kidney abnormalities were examined (14%). It is noteworthy that there were only a limited number of cases (7/34) with renal ultrasonographic information available in our cohort, thus the frequencies of renal anomalies may be underestimated. The presence of branchial or renal anomalies was not correlated with the severity of hearing loss. Furthermore, no relationship was found between genotype and clinical findings. As rare symptoms, one patient had hemifacial palsy, and 3 cases had eye symptoms.
Mutation spectrum and clinical features of Waardenburg syndrome patients
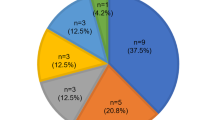
We conducted genetic analysis of 23 probands with hearing loss and one or more clinical findings typical of Waardenburg syndrome, and identified the causative heterozygous variants in 18 probands (diagnostic rate 78%). Table 4 and Supplementary Fig. S2 provides a summary of the identified variants and clinical features of probands and all family members (four families with PAX3 mutations, five families with MITF mutations, 8 families with SOX10 mutations, and one family with a EDNRB mutation). No pathogenic variants were found in SNAI2 or EDN3. Most of the identified variants were truncating variants (four were nonsense, 7 were frameshift, one was splice site) and only three cases had missense variants, one each in PAX3, MITF and SOX10. In addition, we also identified three cases with one copy number loss of the SOX10 gene identified from NGS read depth data and confirmed by aCGH. Thirteen variants were novel and five variants (three PAX3 mutations, and one each with MITF and SOX10 mutations) were previously reported. Computer prediction scores, allele frequency information and the pathogenicity classification for novel variants are listed in Supplementary Table S2. Autosomal dominant inherited cases were 2/4 in PAX3 cases, 4/5 in MITF cases, 1/8 in SOX10 cases, and 1/1 in EDNRB cases. The other 10 cases were sporadic cases, with confirmed de novo mutations in SOX10 in five cases (Table 4). In terms of the clinical features of the probands and all family members harboring the same causative gene variants (29 patients from 18 families in total), the most frequent symptom was hearing loss (27/29, 93%), followed by heterochromia iridis (23/28, 82%). The severity of hearing loss for each gene is shown in Fig. 1, with the frequency of the profound hearing loss higher in cases with MITF and SOX10 mutations. Two cases with PAX3 mutations had bilateral normal hearing and three cases with MITF mutations had unilateral hearing loss. Only a limited number of patients showed discoloration of the hair and skin: hair discoloration was seen in two cases (with SOX10 and MITF mutations), leukoderma in one case with a SOX10 mutation, and excessive freckles in three cases with MITF mutations. No abnormal musculoskeletal findings were observed in any case. Dystopia canthorum was seen in two cases with PAX3 mutations, and one each with MITF and SOX10 mutations. The other associated symptoms observed in SOX10 cases were ptosis (JHLB4270, JHLB4310), developmental delay (JHLB4310) and Asperger syndrome (JHLB3480). In addition, inner ear anomalies, including hypoplasia of the semicircular canal, cochlea, cochlear nerve, and saccular vestibule, were observed. It is suggested that there is no obvious correlation between the type of mutation and its location and the severity of the symptoms. Most of the clinical findings for cases associated with each gene were in agreement with previous reports; however, we identified phenotype-genotype disagreement in two Waardenburg syndrome 1 (WS1) cases (JHLB2469 with a MITF mutation and JHLB5132 with a SOX10 mutation).
Mutation spectrum and clinical features of other syndromic hearing loss patients
We also conducted genetic analysis of other syndromic hearing loss patients (five osteogenesis imperfecta cases, one spondyloepiphyseal dysplasia congenita case, three Stickler syndrome cases, three CHARGE syndrome cases, one Jervell and Lange-Nielsen syndrome case, one auditory neuropathy with optic atrophy case, and 36 Pendred syndrome cases). The diagnostic rate for each syndrome was 60% for osteogenesis imperfecta with COL1A1 variants (3/5), 100% for spondyloepiphyseal dysplasia congenita with a COL2A1 variant (1/1), 100% for Stickler syndrome with COL11A1, COL11A2 variants (2/3, 1/3), 33% for CHARGE syndrome with a CHD7 variant (1/3), 100% for Jervell and Lange-Nielsen syndrome with a KCNQ1 variant (1/1), 100% for auditory neuropathy with a OPA1 mutation (1/1), and 89% for Pendred syndrome with SLC26A4 variants (32/36). Tables 5, 6 provide summaries of the identified variants and clinical features of the probands and all family members harboring the same variants (the pedigrees and audiograms of these cases are shown in Supplementary Figs S3, S4). The identified variants in all three probands with osteogenesis imperfecta were previously reported truncating variants. All four affected cases had easily fractured bones, blue sclera and hearing loss. The severity of hearing loss varied from mild to severe with air-bone gap. All three probands were from autosomal dominant families.
The proband with spondyloepiphyseal dysplasia congenita had a novel truncating variant in COL2A1. She and her father, who harbored the same variant, had characteristic clinical features (cleft palate, short stature and short extremities). Their hearing level was severe to profound sensorineural hearing loss.
With regard to Stickler syndrome, we identified pathogenic variants in the COL11A1 (two cases) and COL11A2 (one case) genes. All identified variants were truncating (two were splice site, one was frameshift), with the two variants in COL11A1 being novel. One COL11A1 and one COL11A2 variant were identified from an autosomal dominant family, and one COL11A1 variant was identified from a sporadic case (de novo). As to the clinical features of the probands and all family members harboring the same causative gene variants (8 patients in total), hearing loss was observed in 75% of cases (3/3 with COL11A1 variants, 3/5 with COL11A2 variants), with the severity of hearing loss being mild to moderate. Two children of the proband with a COL11A2 variant (JHLB4181) carried the same variant but had normal hearing. Seventy-five percent of cases (6/8) had a cleft palate or uvula bifida (3/3 with COL11A1 variants, 3/5 with COL11A2 variants), and all three cases with COL11A1 variants had congenital myopia. One case harboring a COL11A2 variant, who was the son of the proband, had no symptoms.
A novel OPA1 variant was identified in one case who suffered auditory neuropathy with optic atrophy. Two other pathogenic amino acid substitutions have been previously identified in the same position. The proband had amblyopia since infancy, and bilateral moderate sensorineural hearing loss. OAE (Otoacoustic emission) presented a normal response, the ABR (Auditory Brainstem Response) threshold was out of scale, and MRI (magnetic resonance imaging) showed bilateral cochlear nerve hypoplasia. The proband’s mother had similar symptoms (no DNA sample was available).
With regard to Pendred syndrome, we identified SLC26A4 variants in 32 probands with autosomal recessive inheritance or sporadic cases. No variants in KCNJ10 and FOXI1 were identified in cases with heterozygous SLC26A4 variants.
Discussion
In this study, we conducted a comprehensive analysis of Japanese syndromic hearing loss patients to clarify mutation spectrums and clinical features by using NGS analysis with a multiple syndromic targeted resequencing panel. This analysis had a high diagnostic rate (56%) and was suitable for comprehensive analysis. Further, it allowed us to clarify the types and frequency of causative genes in Japanese syndromic hearing loss patients. In addition, it was particularly useful in cases that only partially fulfilled the respective diagnostic criteria. To the best of our knowledge, this is the first study using targeted resequencing panel analysis for multiple syndromic hearing loss patients.
With regard to BO/BOR syndrome, the causative variants were identified in 32% (19/59) of probands (16 typical, 43 atypical). The diagnostic rate was increased to 75% when we restricted the analysis to typical BO/BOR cases (12/16). Krug et al. reported the results of genetic analysis for a large number of BO/BOR patients and identified the causative variants in 36% of cases. Similar to this study, the diagnostic rate was increased to 76% when they restricted subjects to typical BO/BOR cases28. Unzaki et al. analyzed 36 Japanese families with clinically diagnosed BO/BOR syndrome and identified causative genes in 72% of them29. Thus, the diagnostic rate in this study was similar to the rates in these previous reports. EYA1 variants account for 95% of the causative gene variants identified in this study. Similarly, EYA1 was commonly identified in BO/BOR cases in previous studies; 85% in Japanese patients29 and 93% in French patients28. SIX1 variants were identified in 5% (1/19) of the genetically diagnosed cases in this study. This percentage was similar to the results of previous reports28,29. No causative gene variants were identified in 25% of the typical BO/BOR syndrome cases in this study. There is a possibility that variants in other genes (such as SALL1) or genomic rearrangement (inversion or translocation in chromosome 8) may contribute to these cases. In this study, we also identified one copy number loss with a 2.8 Mb deletion of 8q13.2-q13.3 including the EYA1 gene in one familial case. The frequency of one copy number loss of the EYA1 gene was 6% (1/18) in this study. In other reports, copy number loss of the EYA1 gene was also involved in BO/BOR syndrome, with 7% to 10% or more of cases caused by EYA1 copy number loss28,29. The most frequent clinical feature was hearing loss, which was observed in 97% of cases (31/32), followed by preauricular pits in 88% (29/33). In other reports, the most frequent clinical feature was also hearing loss; however, the frequencies of other symptoms varied, with the frequency of renal symptoms higher in some reports28,29,30. Chen et al. reported renal anomalies in 67% of affected individuals31, with about 6% of them progressing to renal failure32. Some of them were asymptomatic in the first decade but required dialysis or renal transplantation in adulthood33,34. In this study, only one case showed congenital renal anomalies. One plausible reason for this lower rate of renal anomalies was that we enrolled BO/BOR candidate patients, and information regarding renal anormalies was available for only a limited number of patients (renal ultrasonographic information was available for only 7/34 cases). Therefore, more cases may have had renal symptoms. In cases in which BO/BOR syndrome is suspected clinically or genetically, even in the absence of renal dysfunction in early childhood, renal examination may be important.
It is noteworthy that three cases from two unrelated families with EYA1 variants presented visual symptoms (progressive disturbance of vision, amblyopia and hypermetropia), but visual symptoms are not typically associated with BOR syndrome. EYA1 is needed for the formation of the anterior portion of the eye35. Azuma et al. reported one case who presented with congenital cataracts with a BOR phenotype (cervical fistula, unilateral multicystic kidney and conductive hearing loss due to ossicular malformations), and others have also reported cases with visual symptoms (dysopia, cataract, micrognathia, and iris coloboma)28,29,36,37,38. The frequency of amblyopia is reported to be 3.0% to 3.2% in the general population39,40, but the frequency of visual symptoms in the EYA1-related BO/BOR patients in this study was a little higher (9%). There is a possibility that visual symptoms actually represent a rare clinical feature of BO/BOR syndrome.
Waardenburg syndrome was subdivided into four types based on the clinical findings, and each causative gene was identified. We successfully identified the genetic causes in 80% of WS1 probands (4/5), 71% of WS2 probands (10/14), and 100% of WS4 probands (2/2). Hoth et al. reported that point mutations in PAX3 have been identified in more than 90% of affected individuals with WS1 or WS341,42. In this study, we identified one case each with MITF and SOX10 variants from WS1. MITF and SOX10 variants were generally identified from WS2 or WS4 patients. Similarly, MITF, EDNRB, and SOX10 variants were identified from WS1 patients in previous reports42,43,44. The cause of this inconsistency between phenotype and genotype may be 1) a new genotype-phenotype correlation or 2) the wider distance between the inner canthus in the Japanese population. In the Japanese literature, Motomura reported the inter-inner canthal, inter-outer canthal and inter-pupillary distance for each age group among Japanese (published in Japanese)45. It appears that the W-index calculated from these data may exceed 1.95 in many age groups (Supplementary Fig. S5). In future, it may be necessary to consider ethnic differences when evaluating the W-index. Among the WS2 cases, we identified the causative variants in 29% of cases with MITF, in 36% with SOX10, and in 7% with EDNRB. Pingault et al. reported that MITF mutations were involved in about 15% of cases, 15% with SOX10, and EDNRB and SNAI2 are a small percentage among WS2 patients46. Bocángel et al. reported that MITF variants and SOX10 variants were observed in 12% and 20% of South-eastern Brazilian WS2 cases, respectively47. Sun et al. also reported that the rates of causative genes observed in Chinese WS2 cases were 34% for MITF and 45% for SOX10, respectively48. Taken together, these results indicate that SOX10 variants may be more frequently identified in East Asian WS2 cases.
It is worth noting that we also identified one copy number loss of the SOX10 gene using NGS read depth data and confirmed by aCGH in three cases. Two probands had a large deletion within the chromosome 22q13.1, a proband had the whole SOX10 gene deletion, and the other proband in a familial case had a partial deletion of SOX10. To date, more than 20 cases caused by copy number variation in PAX3 or SOX10 have been reported42,47,49,50,51,52,53,54. We identified one SOX10-assiociated WS case with developmental delay and one with Asperger syndrome. Both of these cases carried truncation variants; however, no cases were observed with developmental delay among the SOX10 CNV cases. Thus, the association between genotype and developmental delay phenotype remains unclear.
In addition, we also identified one familial WS case with variations in phenotype among family members. In the MITF family (JHLB1623), the father had only unilateral hearing loss and excessive freckles, and her younger brother had only bilateral severe sensorineural hearing loss, but both had the same variant. It is usually difficult to suspect WS from clinical findings and family history; therefore, the comprehensive syndromic hearing loss panel was useful in such cases who only partially fulfilled the diagnostic criteria.
In conclusion, this analysis using NGS with a multiple syndromic targeted resequencing panel was useful for identifying the causative genes in multiple syndromic hearing loss patients in a short time and with a high diagnostic rate.
Subjects and Methods
Subjects
In this study we enrolled total 140 probands with possible syndromic hearing loss who carried hearing loss with one or more associated symptoms typical of each syndrome from our hearing loss cohort of 5,137 patients gathered from 67 cooperative research institutes in Japan as described elsewhere55 (Detailed numbers for each syndrome are listed in Table 2). We also collected data on the hearing level of each proband and their family members. The severity of hearing was classified as mild (20–40 dB), moderate (41–70 dB), severe (71–95 dB), or profound (>95 dB). With regard to BO/BOR syndrome, we enrolled the patients who fulfilled the criteria (typical and atypical) described previously4. Regarding auditory neuropathy, the probands with pathogenic variants in OTOF and DFNB59 were excluded from this study.
Written informed consent was obtained from all patients or their guardians. This study was approved by the Shinshu University Ethical Committee as well as the respective Ethical Committees of the other participating institutions listed below. Akita University Ethical Committee, Iwate Medical University Ethical Committee, Tohoku Rosai Hospital Ethical Committee, Fukushima Medical University Ethical Committee, Yamagata University Ethical Committee, Dokkyo Medical University Ethical Committee, TAKASAKI Ear Nose & Throat Clinic Ethical Committee, Niigata University Ethical Committee, Tokyo Medical University Ethical Committee, Jikei University Ethical Committee, Toranomon Hospital Ethical Committee, Kitasato University Ethical Committee, International University of Health and Welfare Mita Hospital Ethical Committee, National Rehabilitation Center for Persons with Disabilities Ethical Committee, Keio University Ethical Committee, Hamamatsu University Ethical Committee, Shiga University Ethical Committee, Shiga Medical Center for Children Ethical Committee, Osaka University Ethical Committee, Kobe City Medical Center General Hospital Ethical Committee, Hyogo College of Medicine Ethical Committee, Kyoto Prefectural University Ethical Committee, Okayama University Ethical Committee, Yamaguchi University Ethical Committee, Ehime University Ethical Committee, Kyushu University Ethical Committee, Kanda ENT Clinic Ethical Committee, Nagasaki University Ethical Committee, Miyazaki University Ethical Committee, Kagoshima University Ethical Committee, Ryukyus University Ethical Committee, Sapporo Medical University Ethical Committee, Tohoku University Ethical Committee, Jichi Medical University Ethical Committee, Gunma University Ethical Committee, Jyuntendo University Ethical Committee, Yokohama City University Ethical Committee, Mejiro University Ethical Committee, Saitama Medical University Ethical Committee, Abe ENT Clinic Ethical Committee, Tokyo Medical Center Institute of Sensory Organs Ethical Committee, Jichi University Saitama Medical Center Ethical Committee, Aichi Children’s Health Medical Center Ethical Committee, Chubu Rosai Hospital Ethical Committee, Kyoto University Ethical Committee, Mie University Ethical Committee, Kansai Medical University Ethical Committee, Kobe University Ethical Committee, Osaka Medical Center and Research Institute for Maternal and Children Health Ethical Committee, Wakayama Medical University Ethical Committee, Kouchi University Ethical Committee, Hiroshima University Ethical Committee, Hiroshima City Hiroshima Citizen Hospital Ethical Committee, Fukuoka University Ethical Committee, Kurume University Ethical Committee, National Defense Medical College Ethical Committee, Tokai University Ethical Committee, Hokkaido University Ethical Committee, Kanagawa Children’s Medical Center Ethical Committee, Tokyo Medical and Dental University Ethical Committee, Hirosaki University Ethical Committee, Tokyo Metropolitan Children’s Medical Center Ethical Committee, Hakodate Central General Hospital Ethical Committee, Osaka Red Cross Hospital Ethical Committee, Hiroshima Prefectural Hospital Ethical Committee, Nara Medical University Ethical Committee, Tsukuba University Ethical Committee. All methods were performed in accordance with the Guidelines for Genetic Tests and Diagnoses in Medical Practice of the Japanese Association of Medical Sciences and the Declaration of Helsinki as required by Shinshu University.
Methods
Amplicon Library Preparation
An Amplicon library was prepared with an Ion AmpliSeqTM Custom Panel (Applied Biosystems, Life Technologies) for 36 target genes reported to cause syndromic hearing loss. We selected the 36 genes associated with 14 types of syndromic hearing loss commonly observed in practical settings. We also referred to the hereditary hearing loss homepage (https://hereditaryhearingloss.org) to select these genes. The responsible genes for Usher syndrome were not included in our syndromic hearing loss targeting panel as these genes were included in the non-syndromic hearing loss panel reported in a previous paper55. To avoid any overlap between these two panels, we removed the genes associated with Usher syndrome from our panel. The panel contained the following genes: EYA1-SIX1-SIX5 for BOR syndrome; PAX3-MITF-SNAI2-EDNRB-EDN3-SOX10 for Waardenburg syndrome; COL2A1-COL11A1-COL11A2-COL9A1-COL9A2-COL9A3-COL1A1-COL4A3-COL4A4-COL4A5 for connective tissue disorder including osteogenesis imperfecta, spondyloepiphyseal dysplasia congenita, Stickler syndrome, and Alport syndrome; CHD7-SEMA3E for CHRGE syndrome; SLC26A4- FOXI1-KCNJ10 for Pendred syndrome; KCNQ1-KCNE1 for Jervell Lange-Nielsen syndrome; NDP for Norrie disease; TCOF1-POLR1C for Treacher-Collins syndrome, HSD17B4-HARS2-CLPP-LARS2 for Perrault syndrome; OPA1 for auditory neuropathy with optic atrophy and GDF6-MEOX1 for Klippel Feil syndrome.
Emulsion PCR and sequencing
The emulsion PCR and NGS (next-generation sequencing) were performed with an Ion Proton system using the Ion Proton 200 sequencing Kit and an Ion P1 Chip (ThermoFisher Scientific, Waltham, MA, USA) according to the manufacturer’s instructions.
Base call and data analysis
The sequence data were mapped against the human genome sequence (build GRCh37/hg19) with the Torrent Mapping Alignment Program. After sequencing mapping, the DNA variant regions were piled up with Torrent Variant Caller plug-in software. After variant detection, variant effects were analyzed using the ANNOVAR software56,57.
Direct sequencing
After the filtering process, described previously55, we performed confirmation of the identified variant and family segregation analysis by Sanger sequencing.
CNV (Copy Number Variation) analysis
CNV analysis was performed with NGS analysis read depth data according to the method described in a previous report58.
aCGH (Array Comparative Genomic Hybridization)
To confirm the CNVs identified from NGS read depth data, we performed array CGH analysis with the Agilent 8 × 60 K whole genome array (Agilent Technologies, Santa Clara, CA). We used the same DNA samples as for the amplicon re-sequencing, and quality assessment was also carried out. Ten microliters of genomic DNA solution (0.5ug of DNA) were fragmented, labeled with cyanine-3 for reference DNA samples and cyanine-5 for subjects, and then hybridized. Scanning of the array was carried out according to the manufacturer’s recommended protocols. Scanned aCGH data were analyzed using CytoGenomics software version 3.0.6.6 (Agilent Technologies).
References
Morton, C. C. & Nance, W. E. Newborn hearing screening-a silent revolution. N. Engl. J. Med. 354, 2151–2164 (2006).
Alford, R. L. et al. American college of medical genetics and genomics guideline for the clinical evaluation and etiologic diagnosis of hearing loss. Genet. Med. 16, 347–355 (2014).
Fraser, F. C., Sproule, J. R. & Halal, F. Frequency of the branchio-oto-renal (BOR) syndrome in children with profound hearing loss. Am. J. Med. Genet. 7, 341–349 (1980).
Chang, E. H. et al. Branchio-oto-renal syndrome: the mutation spectrum in EYA1 and its phenotypic consequences. Hum. Mutat. 23, 582–589 (2004).
Waardenburg, P. J. A new syndrome combining developmental anomalies of the eyelids, eye brows and nose root with pigmentary defects of the iris and head hair and with congenital deafness. Am. J. Hum. Genet. 3, 195–253 (1951).
Read, A. P. & Newton, V. E. Waardenburg syndrome. J. Med. Genet. 34, 656–665 (1997).
Arias, S. Genetic heterogeneity in the Waardenburg syndrome. Birth Defects Orig. Artic. Ser. 7, 87–101 (1971).
Klein, D. Historical background and evidence for dominant inheritance of the Klein-Waardenburg syndrome (type III). Am. J. Med. Genet. 14, 231–239 (1983).
Fraser, G. R. The causes of profound deafness in childhood. Baltimore: Johns Hopkins university press (1976).
Sillence, D. O., Senn, A. & Danks, D. M. Genetic heterogeneity in osteogenesis imperfecta. J. Med. Genet. 16, 101–116 (1979).
Anderson, I. J., Goldberg, R. B., Marion, R. W., Upholt, W. B. & Tsipouras, P. Spondyloepiphyseal dysplasia congenita: genetic linkage to type II collagen (COL2A1). Am. J. Hum. Genet. 46, 896–901 (1990).
Stickler, G. B. et al. Hereditary progressive arthro-ophthalmopathy. Mayo Clinic Proc. 40, 433–455 (1965).
Ahmad, N. N. et al. Stop codon in the procollagen II gene (COL2A1) in a family with the Stickler syndrome (arthro-ophthalmopathy). Proc. Natl. Acad. Sci. USA 88, 6624–6627 (1991).
Richards, A. J. et al. A family with Stickler type 2 has a mutation in the COL11A1 gene resulting in the substitution of glycine 97 by valine in alpha 1(XI) collagen. Hum. Mol. Genet. 5, 1339–1343 (1996).
Van Camp, G. et al. A new autosomal recessive form of Stickler syndrome is caused by a mutation in the COL9A1 gene. Am. J. Hum. Genet. 79, 449–457 (2006).
Baker, S. et al. A loss of function mutation in the COL9A2 gene causes autosomal recessive Stickler syndrome. Am. J. Med. Genet. A. 155A, 1668–1672 (2011).
Vikkula, M. et al. Autosomal dominant and recessive osteochondrodysplasias associated with the COL11A2 locus. Cell 80, 431–437 (1995).
Pirson, Y. Making the diagnosis of Alport’s syndrome. Kidney Int. 56, 760–775 (1999).
Longo, I. et al. COL4A3/COL4A4 mutations: from familial hematuria to autosomal-dominant or recessive Alport syndrome. Kidney Int. 61, 1947–1956 (2002).
Marcocci, E. et al. Autosomal dominant Alport syndrome: molecular analysis of the COL4A4 gene and clinical outcome. Nephrol. Dial. Transplant. 24, 1464–1471 (2009).
Hall, B. D. Choanal atresia and associated multiple anomalies. J. Pediatr. 95, 395–398 (1979).
Hittner, H. M., Hirsch, N. J., Kreh, G. M. & Rudolph, A. J. Colobomatous micriphthalmia, heart disease, hearing loss, and mental retardation–a syndrome. J. Pediatr. Ophthalmol. Strabismus 16, 122–128 (1979).
Pagon, R. A., Graham, J. M. Jr., Zonana, J. & Yong, S. L. Coloboma, congenital heart disease, and choanal atresia with multiple anomalies: CHARGE association. J. Pediatr. 99, 223–227 (1981).
Lalani, S. R. et al. SEMA3E mutation in a patient with CHARGE syndrome. J. Med. Genet. 41, e94 (2004).
Neyroud, N. et al. A novel mutation in the potassium channel gene KVLQT1 causes the Jervell and Lange-Nielsen cardioauditory syndrome. Nat. Genet. 15, 186–189 (1997).
Schulze-Bahr, E. et al. KCNE1 mutations cause Jervell and Lange-Nielsen syndrome. Nat. Genet. 17, 267–268 (1997).
Everett, L. A. et al. Pendred syndrome is caused by mutations in a putative sulphate transporter gene (PDS). Nat. Genet. 17, 411–422 (1997).
Krug, P. et al. Mutation screening of the EYA1, SIX1, and SIX5 genes in a large cohort of patients harboring branchio-oto-renal syndrome calls into question the pathogenic role of SIX5 mutations. Hum. Mutat. 32, 183–190 (2011).
Unzaki, A. et al. Clinically diverse phenotypes and genotypes of patients with branchio-oto-renal syndrome. J. Hum. Genet. 63, 647–656 (2018).
Morisada, N., Nozu, K. & Iijima, K. Branchio-oto-renal syndrome: Comprehensive review based on nationwide surveillance in Japan. Pediatr. Int. 56, 309–314 (2014).
Chen, A. et al. Phenotypic manifestations of branchio-oto-renal syndrome. Am. J. Med. Genet. 58, 365–370 (1995).
Misra, M. & Nolph, K. D. Renal failure and deafness: Branchio-oto-renal syndrome. Am. J. Kidney Dis. 32, 334–337 (1998).
Annear, N. M., Gale, D. P., Loughlin, S., Dorkins, H. R. & Maxwell, P. H. End-stage renal failure associated with congenital deafness. NDT Plus 1, 171–175 (2008).
Gigante, M. et al. Branchio-oto-renal syndrome (BOR) associated with focal glomerulosclerosis in a patient with a novel EYA1 splice site mutation. BMC Nephrol. 14, 60 (2013).
Xu, P. X., Woo, I., Her, H., Beier, D. R. & Maas, R. L. Mouse eya homologues of the Drosophila eyes absent gene require Pax6 for expression in lens and nasal placode. Development 124, 219–231 (1997).
Azuma, N., Hirakiyama, A., Inoue, T., Asaka, A. & Yamada, M. Mutations of a human homologue of the Drosophila eyes absent gene (EYA1) detected in patients with congenital cataracts and ocular anterior segment anomalies. Hum. Mol. Genet. 9, 363–366 (2000).
Rickard, S., Boxer, M., Trompeter, R. & Bitner-Glindzicz, M. Importance of clinical evaluation and molecular testing in the branchio-oto-renal (BOR) syndrome and overlapping phenotypes. J. Med. Genet. 37, 623–627 (2000).
Sanchez-Valle, A. et al. HERV-mediated genomic rearrangement of EYA1 in an individual with branchio-oto-renal syndrome. Am. J. Med. Genet. A 152A, 2854–2860 (2010).
Anderson, R. L. & Baumgartner, S. A. Amblyopia in ptosis. Arch. Ophthalmol. 98, 1068–1069 (1980).
Dray, J. P. & Leibovitch, I. Congenital ptosis and amblyopia: a retrospective study of 130 cases. J. Pediatr. Ophthalmol. Strabismus 39, 222–225 (2002).
Hoth, C. F. et al. Mutations in the paired domain of the human PAX3 gene cause Klein-Waardenburg syndrome (WS-III) as well as Waardenburg syndrome type I (WS-I). Am. J. Hum. Genet. 52, 455–462 (1993).
Milunsky, J. M., Maher, T. A., Ito, M. & Milunsky, A. The value of MLPA in Waardenburg syndrome. Genet. Test. 11, 179–182 (2007).
Morimoto, N. et al. Homozygous EDNRB mutation in a patient with Waardenburg syndrome type1. Auris Nasus Larynx 45, 222–226 (2018).
Suzuki, N. et al. A case report of reversible generalized seizures in a patient with Waardenburg syndrome associated with a novel nonsense mutation in the penultimate exon of SOX10. BMC Pediatr. 18, 171 (2018).
Motomura, Y. Clinical and genetic study of Waardenburg-Klein syndrome in Japanese. Audiology Japan 12, 57–92 (1969) (in Japanese).
Pingault, V. et al. Review and update of mutations causing Waardenburg syndrome. Hum. Mutat. 31, 391–406 (2010).
Bocángel, M. A. P. et al. Waardenburg syndrome: Novel mutations in a large Brazilian sample. Eur. J. Med. Genet. 61, 348–354 (2018).
Sun, L. et al. Molecular etiology and genotype-phenotype correlation of Chinese Han deaf patients with type I and type II Waardenburg syndrome. Sci. Rep. 6, 35498 (2016).
Tassabehji, M. et al. The mutational spectrum in Waardenburg syndrome. Hum. Mol. Genet. 4, 2131–2137 (1995).
Bondurand, N. et al. Deletions at the SOX10 gene locus cause Waardenburg syndrome types 2 and 4. Am. J. Hum. Genet. 81, 1169–1185 (2007).
Siomou, E. et al. A 725 kb deletion at 22q13.1 chromosomal region including SOX10 gene in a boy with a neurologic variant of Waardenburg syndrome type 2. Eur. J. Med. Genet. 55, 641–645 (2012).
Jelena, B., Christina, L., Eric, V. & Fabiola, Q. R. Phenotypic variability in Waardenburg syndrome resulting from a 22q12.3-q13.1 microdeletion involving. SOX10. Am. J. Med. Genet. A 164A, 1512–1519 (2014).
Wenzhi, H. et al. Heterozygous deletion at the SOX10 gene locus in two patients from a Chinese family with Waardenburg syndrome type II. Int. J. Pediatr. Otorhinolaryngol. 79, 1718–1721 (2015).
Li, H. et al. Identification of a novel de novo heterozygous deletion in the SOX10 gene in Waardenburg syndrome type II using next-generation sequencing. Genet. Test. Mol. Biomarkers 21, 681–685 (2017).
Kitano, T. et al. POU4F3 mutation screening in Japanese hearing loss patients: Massively parallel DNA sequencing-based analysis identified novel variants associated with autosomal dominant hearing loss. PLos One 12, e0177636 (2017).
Wang, K., Li, M. & Hakonarson, H. ANNOVAR: functional annotation of genetic variants from high-throughput sequencing data. Nucleic. Acids. Res. 38, e164 (2010).
Chang, X. & Wang, K. wANNOVAR: annotating genetic variants for personal genomes via the web. J. Med. Genet. 49, 433–436 (2012).
Nishio, S. Y., Moteki, H. & Usami, S. I. Simple and efficient germline copy number variant visualization method for the Ion AmpliseqTM custom panel. Mol. Genet. Genomic Med. 6, 678–686 (2018).
Tassabehji, M. et al. Mutations in GDF6 are associated with vertebral segmentation defects in Klippel-Feil syndrome. Hum. Mutat. 29, 1017–1027 (2008).
Huang, T., Santarelli, R. & Starr, A. Mutation of OPA1 gene causes deafness by affecting function of auditory nerve terminals. Brain Res. 1300, 97–104 (2009).
Acknowledgements
This study was supported by a Health and Labour Sciences Research Grant for Research on Rare and Intractable Diseases (H29-Nanchito (Nan)-Ippan-031, H29-Nanchito (Nan)-Ippan-007) from the Ministry of Health, Labour and Welfare of Japan (SU), a grant for Practical Research Project for Rare/Intractable Disease from the Japan Agency for Medical Research and Development (AMED) (SU) (18ek0109363h0001), the Program for an Integrated Database of Clinical and Genomic Data from the Japan Agency for Medical Research and Development (AMED) (SU) (16kk0205010h0001) and by a Grant-in-Aid for Scientific Research (A) (15H02565) from the Ministry of Education, Science and Culture of Japan (SU).
Author information
Authors and Affiliations
Contributions
M.I., S.N. and S.U. designed this study. M.I., S.N. and H.M. performed the experiments and contributed to interpretation of the data. M.I. and N.S. drafted the manuscript. Y.T., M.M., T.S., Y.K., K.O., K.O., T.M., T.I., H.S., K.N., S.I. N.N., M.K., K.K., H.T., Y.K., S.I., S.F., K.I., M.F., H.N., J.N., R.H., Y.O., Y.N., M.K., H.S., Y.K., K.S., N.H., T.N., N.T., Y.K., C.K., T.T., I.M. and A.G. participated in sample and data collection. S.U. supervised this study.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ideura, M., Nishio, Sy., Moteki, H. et al. Comprehensive analysis of syndromic hearing loss patients in Japan. Sci Rep 9, 11976 (2019). https://doi.org/10.1038/s41598-019-47141-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-47141-4
This article is cited by
-
Compound heterozygous variants of the SLC26A4 gene in a Chinese family with enlarged vestibular aqueducts
BMC Medical Genomics (2022)
-
Phenotype–genotype correlation in patients with typical and atypical branchio-oto-renal syndrome
Scientific Reports (2022)
-
Discovery of sensorineural hearing loss and ossicle deformity in a Chinese Li nationality family with spondyloepiphyseal dysplasia congenita caused by p.G504S mutation of COL2A1
BMC Medical Genomics (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.