Abstract
Benefit of physical activity in prevention of aneurysmal subarachnoid hemorrhage (SAH) is unclear. We aimed to clarify this by studying how different types of physical activity associate with SAH risk. By following 65 521 population-based FINRISK participants prospectively from medical and autopsy registries since 1972 until 2014, we detected 543 incident SAHs. At baseline, we measured leisure-time physical activity (LTPA), occupational physical activity (OPA), and commuting physical activity (CPA) levels. The Cox model adjusted for all well-known SAH risk factors and for socioeconomic status, provided hazard ratios (HRs) for physical activity variables. Every 30-minute increase in weekly LTPA decreased SAH risk linearly in men and women HR = 0.95 (95% CI = 0.90–1.00). CPA reduced SAH risk as well, but the association diminished as participants retired. In contrast, individuals with moderate (1.41, 1.04–1.92) and high OPA (1.34, 0.99–1.81) had elevated SAH risk. Protective association of LTPA persisted in all age and hypertension groups, and was even greater in current smokers 0.88 (0.81–0.96) than non-smokers (p = 0.04 for difference). Commuting and leisure time physical activity seem to reduce SAH risk in men and women and is most beneficial for smokers. Future intervention studies should investigate whether physical activity can reduce the rupture risk of intracranial aneurysms.
Similar content being viewed by others
Introduction
Aneurysmal subarachnoid hemorrhage (SAH) has remained a serious disease with a case fatality of approximately 40%1,2. In development of SAH, lifestyle risk factors play the main role3. The risk factors include smoking, hypertension, increasing age, and possibly adverse lipid profile and female sex4,5,6,7,8,9.
Leisure-time physical activity (LTPA) protects against ischemic stroke10,11 but only few studies exist on SAH and physical activity12,13,14,15,16,17,18,19 with inconsistent results. Recently, a study19 combining all physical activity types suggested that high overall physical activity elevates SAH risk. However, the study did not differentiate associations by physical activity type, even though these types may associate differently with cardiovascular diseases10. Our aim was to study whether LTPA, commuting physical activity (CPA), and occupational physical activity (OPA) decrease SAH risk and whether the effects distinct by different types of physical activities.
Results
Between 1972 and 2014 the follow-up included 65 521 participants and provided 1.52 million person-years. There were 543 incident SAHs (215 men), of which 98 were fatal outside the hospital or in emergency rooms. Of the whole cohort, 24.3% used motorized transport for commuting and 18.5% had low occupational activity (Table 1). These proportions did not differ significantly by sex.
LTPA
Every 30-minute increase in leisure-time exercise (leading to mild sweating and mild breathlessness) per week associated linearly with a decreased SAH risk of 5% in both men and women (Table 2). This association remained the same even 40 years after answering the questionnaire. This association also persisted in all SBP and age groups, and remained the same after OPA and CPA were included in the model. The protective association of every 30-minute increase in weekly exercise was stronger among smokers (0.88, 0.81–0.96) and ex-smokers (0.93, 0.86–1.03) in comparison with never-smokers (p = 0.04) (Fig. 1). In line with this, smokers with less than 90 minutes of LTPA per week had a relative excess risk (RERI) of 2.03 (0.10–3.96).
Every 30-minute increase in weekly leisure time physical activity reducing subarachnoid hemorrhage risk more in smokers and ex-smokers than in never-smokers (p = 0.04), with no difference between hypertensives and normotensives (p = 0.29). Y-axis describes HR and the points describe point estimates, the whiskers describe 95% CIs.
According to the PAF analysis, a weekly rise in LTPA from 0 to >90 minutes among inactive smokers associated with, 11% decrease in smokers’ SAH events. In comparison, the same analysis showed that diminishing hypertension in smokers associated with similar magnitude of 9% decrease in SAHs.
CPA
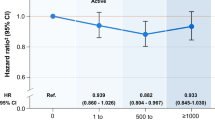
Active commuter (≥30 minutes per day) had reduced SAH risk when compared to those who used motorized transport. This reduced risk persisted until participants reached retirement age (Fig. 2). The SAH risk associated with smoking decreased in active commuters when compared to the low-CPA group (Fig. 3). Similarly, an increase in SBP (standard deviation increase of 21.4 mmHg) did not associate with SAH risk in active commuters (Fig. 4).
OPA
In contrast to LTPA and CPA, an adjusted model including both sexes showed that moderate OPA elevated SAH risk and high OPA may elevate SAH risk (Table 2). In subgroup analysis, moderate OPA was associated with elevated SAH risk only among female smokers. In this group of women, an additive interaction emerged between female heavy smokers (over 10 cigarettes per day) and moderate OPA with RERI of 2.92 (0.30–5.53). Moreover, every SD increase (21.4 mmHg) in SBP was associated with elevated SAH risk in individuals doing physical work (high- and moderate-OPA groups) but not in the low-OPA group.
Competing risks analysis
When weekly LTPA in minutes was divided into quartiles, SAH risk decreased with increasing physical activity also in presence of competing risk of death (Supplementary Fig. 1). No differences in HRs emerged between sudden-death SAH and hospitalized SAH patients in any physical activity category.
Discussion
Every 30-minute increase in weekly LTPA, such as brisk walking, swimming, cycling, or jogging, associated with 5% reduced SAH risk in both men and women. In theory, by increasing LTPA by up to 5 hours a week, one could reduce the risk of SAH by 40%. The protective association of LTPA remained the same not only in all age groups but also in all SBP groups, suggesting that presence of SAH risk factors does not cancel out the protective association of LTPA. Interestingly, the protective association of LTPA was even stronger among ex-smokers and smokers. In fact, smokers with less than 90 minutes of LTPA per week had a relative excess SAH risk of 203%. Thus, motivating smokers to be physically more active, not only quitting smoking, may be both a new and a promising approach in preventing SAH.
Similar to LTPA, high commuting activity, often by foot or by bicycle, associated with reduced SAH risk in men and women and this association persisted until retirement age. Interestingly, smoking and hypertension, the most important SAH risk factors5,6, increased the risk of SAH less in individuals with moderate or high CPA than in individuals with low CPA. In other words, similar to LTPA, CPA may counteract the hazardous effects of smoking and hypertension. The reasons behind the potential protective effect of LTPA and CPA remain speculative but aerobic physical activity reduces systemic inflammation20 and can also reduce inflammation in intracranial vessel walls21. Our finding on LTPA and CPA are further supported by studies on myocardial infarction and physical activity that have shown a decreased risk in high LTPA10,11 and CPA11 groups.
Moderate OPA (work including much walking but not lifting heavy objects) and high OPA (work that includes much walking and frequent lifting of heavy objects or climbing stairs) associated with elevated SAH risk when compared to office work (low OPA). These findings are also consistent with studies on myocardial infarct and stroke10,22,23. The reasons behind elevated SAH risk in moderate and high OPA groups may relate to different health behavior regarding hypertension and smoking in different OPA groups as our results to some extent suggest. Other explanations include residual confounding from inaccurate SES-variable, or an unknown work-related external risk factor. In terms of the high-OPA, the elevated risk may also stem in part from differences in health effects between high OPA and high LTPA23. One possible explanation for harmful effects of OPA in SAH risk may be the different nature of physical activities. LTPA and CPA are relatively short term aerobic cardiovascular exercises, whereas OPA may be more long term and consists more often of lifting of weights, thus resembling anaerobic weight training. The prolonged physical stress in high OPA group may thus become harmful if the workload surpasses the limits of individual’s cardiorespiratory system and may even act as trigger of cardiovascular diseases22,24. Therefore, also cardiovascular effects between LTPA, CPA and OPA may differ. However, in our observational study we can only speculate the possible causal mechanisms between OPA and SAH and further intervention studies on this topic is needed.
A recent study19 suggested that when OPA, CPA, and LTPA were combined into a continuous variable of MET hours per day, high physical activity (>33 MET hours) elevated SAH risk. The 33 MET hours correspond to ~4.5 hours of jogging (LTPA) per day, whereas people in moderate and high OPA groups reach 33 MET hours during a normal working day. Therefore, the elevated SAH risk observed in the Japanese study19 may reflect elevated SAH risk in the moderate and high OPA groups – not harmful effects of physical activity per se.
To our knowledge, only two studies on physical activity and SAH exist that include sudden deaths from SAH and control properly for confounding factors16,17. The current study is an update of a study by Hu et al.17 published in 2005 and describes similar results. Our updated data set includes a longer follow-up, more participants, more physical activity variables, and twice as many first-ever SAH cases, thus providing more detailed, reliable, and novel results. A Norwegian study16 found no association between physical activity and SAH in an analysis including 108 SAHs, although their HR trends for physical activity were similar to ours.
Because association of physical activity with SAH seems less strong when compared to classical SAH risk factors, namely smoking and hypertension, the number of SAHs needs to be relatively high when studying physical activity.
The study may have a few strengths. We analyzed physical activity in three different types of activity categories, enabling us to perform the most detailed analysis of physical activity and SAH to date. The follow-up of up to 40 years is one of the longest among cardiovascular risk factor studies25. The relatively large number (543) of first-ever SAH cases enabled reliable subgroup analyses. Our prospective set-up reduced the risk of informational bias and reverse causality. Our analysis included SES, the known risk factors of SAH, and outside-hospital deaths from SAH with high diagnostic accuracy26. The majority of physical activity studies12,13,14,15,18 have not included the substantial fraction of SAH patients dying suddenly outside hospitals. Though sudden-death SAH individuals have worse risk-factor profiles than do hospitalized SAH patients27, we found no differential effect of any physical activity categories on the two types of outcome. As the Finnish incidence of SAH does not differ from the incidence found in other countries reporting reliable (autopsy reports included) incidence rates28, our results may be generalizable at least to other Caucasian populations.
The study has also limitations. Even though our positive predictive value for aneurysmal SAHs was high, our dataset is likely to include small proportion of non-aneurysmal SAHs (perimesencephalic bleed, arteriovenous malformation, fistula, tumor, or trauma related SAHs). However, given the high positive predictive value of aneurysmal SAH diagnoses, and lack of robust association between physical activity and these non-aneurysmal SAHs, they are unlikely to have major contribution to our results. Our study is observational and strong inference about causality cannot be made. Participants’ risk factor profiles were collected at enrolment, and therefore changes in risk factors during the follow-up have evidently taken place. However, we were able to take into account the changing effects of CPA during follow-up; other variables did not show strong evidence of changes. We measured physical activity by questionnaire, and responses are prone to recall and reporting bias. However, our questionnaire has been validated against accelerometer29 and mortality and morbidity data11. Our questionnaire-measured physical activity correlates well with mortality and morbidity whereas the correlation with accelerometer-measured physical activity revealed a moderate correlation. In accelerometer study, physical activity was consistently over-reported in the questionnaires29, suggesting that our results likely underestimate the potential benefits of LTPA and CPA.
In conclusion, increases in leisure-time and commuting physical activity are likely to reduce SAH incidence in all age- and SBP groups and especially among smokers. Increasing commuting and leisure time physical activity could be a novel non-invasive preventive method for reducing the rupture risk of intracranial aneurysms. However, intervention studies are needed to confirm this. The reasons and mechanisms for elevated SAH risk in those doing strenuous physical work remain to be studied.
Methods
Previous studies describe the research protocol in detail1,5,25. The National FINRISK Surveys, conducted every five years since 1972, collected data on independent, population-based, random samples of adults from various geographical areas of Finland. The participation rate in FINRISK surveys in the 70’s and 80’s was >90% and 80% and has remained over 60% in all surveys25. The questionnaire measured physical activity in three main domains of daily activity: CPA, OPA, and LTPA. The analysis of CPA and OPA included only those who were employed. Supplemental Methods describes the data collection, physical activity, and other variables in detail. The local ethics committees at the University of Helsinki and Helsinki University Hospital gave approval for each FINRISK survey in accordance with legislation pertinent to the time of the survey. The World Medical Association’s Declaration of Helsinki on ethical principles for medical research also guided the studies. Each participant provided oral informed consent between 1972 and 1997, whereas from 2002 onwards, participants also gave a written informed consent25.
SAH identification and definition
Follow-up started at enrolment and ended at first-ever SAH, death, or on December 31, 2014, whichever came first. The follow-up was complete for deaths and hospitalization when the subject was resident in Finland; those moving abroad were censored at the time of emigration1. The nationwide Hospital Discharge Register and Causes of Death Register identified nonfatal and fatal SAHs with high accuracy30. Sudden deaths from SAH occurred away from hospitals, in an ambulance, or emergency room and were confirmed in forensic autopsy, medical autopsy, or with clinical or radiological examinations (spinal tap and/or computer tomography), or both. Externally validated positive predictive value of overall aneurysmal SAH diagnoses was 87%, whereas it was 97% for sudden-death aneurysmal SAHs30. The strengthening the Reporting of Observational studies in Epidemiology (STROBE) statement31 guided the reporting.
Statistical analyses
Scatter plots and Spearman’s correlation coefficients studied correlations between physical activity variables measured by the questionnaire. The Cox proportional hazard model calculated hazard ratios (HRs) between physical activity and SAH while Schoenfeld residuals and log-log plots examined proportionality assumptions. Due to long follow-up, we also calculated a competing risks model32. Based on the SAH literature4,5,6,7, the final adjusted model included all known SAH risk factors, (age, sex, SBP, smoking) and one to three physical activity variables. To control the cohort effect and potential confounding, we also included BMI, cholesterol, study year, and study area into the final model. Preliminary models also included SES and alcohol consumption. The likelihood-ratio test examined multiplicative interactions, and cubic splines examined linearity. Additive interactions33 were modelled as relative excess risk (RERI). To avoid over-estimation of population-attributable fraction (PAF), these were calculated by the average attributable fraction method, which restricts overall PAFs to 100%34. Analysis plan in the Supplementary File describes the predefined analysis protocol. All statistical analyses used Stata Corp version 14.2 (Stata Corp, College Station, TX).
References
Korja, M. et al. Cause-specific mortality of 1-year survivors of subarachnoid hemorrhage. Neurology 80, 481–486, https://doi.org/10.1212/WNL.0b013e31827f0fb5 (2013).
Stegmayr, B., Eriksson, M. & Asplund, K. Declining mortality from subarachnoid hemorrhage: changes in incidence and case fatality from 1985 through 2000. Stroke 35, 2059–2063, https://doi.org/10.1161/01.STR.0000138451.07853.b6 (2004).
Korja, M. et al. Genetic epidemiology of spontaneous subarachnoid hemorrhage: Nordic Twin Study. Stroke 41, 2458–2462, https://doi.org/10.1161/STROKEAHA.110.586420 (2010).
Lindbohm, J. V., Kaprio, J., Jousilahti, P., Salomaa, V. & Korja, M. Sex, Smoking, and Risk for Subarachnoid Hemorrhage. Stroke 47, 1975–1981, https://doi.org/10.1161/STROKEAHA.116.012957 (2016).
Korja, M. et al. Risk factors and their combined effects on the incidence rate of subarachnoid hemorrhage–a population-based cohort study. PLoS One 8, e73760, https://doi.org/10.1371/journal.pone.0073760 (2013).
Knekt, P. et al. Risk factors for subarachnoid hemorrhage in a longitudinal population study. J Clin Epidemiol 44, 933–939 (1991).
Sandvei, M. S. et al. Risk factors for aneurysmal subarachnoid hemorrhage - BMI and serum lipids: 11-year follow-up of the HUNT and the Tromso Study in Norway. Acta Neurol Scand 125, 382–388, https://doi.org/10.1111/j.1600-0404.2011.01578.x (2012).
Lindbohm, J. V., Kaprio, J. & Korja, M. Cholesterol as a Risk Factor for Subarachnoid Hemorrhage: A Systematic Review. PLoS One 11, e0152568, https://doi.org/10.1371/journal.pone.0152568 (2016).
Lindbohm, J., Korja, M., Jousilahti, P., Salomaa, V. & Kaprio, J. Adverse lipid profile elevates risk for subarachnoid hemorrhage: A prospective population-based cohort study. Atherosclerosis 274, 112–119, https://doi.org/10.1016/j.atherosclerosis.2018.05.011 (2018).
Li, J., Loerbroks, A. & Angerer, P. Physical activity and risk of cardiovascular disease: what does the new epidemiological evidence show? Curr Opin Cardiol 28, 575–583, https://doi.org/10.1097/HCO.0b013e328364289c (2013).
Hu, G. et al. Occupational, commuting and leisure-time physical activity in relation to coronary heart disease among middle-aged Finnish men and women. Atherosclerosis 194, 490–497, https://doi.org/10.1016/j.atherosclerosis.2006.08.051 (2007).
Armstrong, M. E. G., Green, J., Reeves, G. K., Beral, V. & Cairns, B. J. Frequent physical activity may not reduce vascular disease risk as much as moderate activity: Large prospective study of women in the United Kingdom. Circulation 131, 721–729, https://doi.org/10.1161/CIRCULATIONAHA.114.010296 (2015).
Abbott, R. D., Rodriguez, B. L., Burchfiel, C. M. & Curb, J. D. Physical activity in older middle-aged men and reduced risk of stroke: the Honolulu Heart Program. Am J Epidemiol 139, 881–893 (1994).
Fann, J. R., Kukull, W. A., Katon, W. J. & Longstreth, W. T. Jr. Physical activity and subarachnoid haemorrhage: a population based case-control study. J Neurol Neurosurg Psychiatry 69, 768–772 (2000).
Hu, F. B. et al. Physical activity and risk of stroke in women. JAMA 283, 2961–2967 (2000).
Sandvei, M. S., Romundstad, P. R., Muller, T. B., Vatten, L. & Vik, A. Risk factors for aneurysmal subarachnoid hemorrhage in a prospective population study: the HUNT study in Norway. Stroke 40, 1958–1962, https://doi.org/10.1161/STROKEAHA.108.539544 (2009).
Hu, G. et al. Leisure time, occupational, and commuting physical activity and the risk of stroke. Stroke 36, 1994–1999, https://doi.org/10.1161/01.STR.0000177868.89946.0c (2005).
Aberg, N. D. et al. Influence of Cardiovascular Fitness and Muscle Strength in Early Adulthood on Long-Term Risk of Stroke in Swedish Men. Stroke 46, 1769–1776, https://doi.org/10.1161/STROKEAHA.115.009008 (2015).
Kubota, Y. et al. Daily Total Physical Activity and Incident Stroke: The Japan Public Health Center-Based Prospective Study. Stroke 48, 1730–1736, https://doi.org/10.1161/STROKEAHA.117.017560 (2017).
Borodulin, K., Laatikainen, T., Salomaa, V. & Jousilahti, P. Associations of leisure time physical activity, self-rated physical fitness, and estimated aerobic fitness with serum C-reactive protein among 3,803 adults. Atherosclerosis 185, 381–387, https://doi.org/10.1016/j.atherosclerosis.2005.06.015 (2006).
Frosen, J. et al. Saccular intracranial aneurysm: pathology and mechanisms. Acta Neuropathol 123, 773–786, https://doi.org/10.1007/s00401-011-0939-3 (2012).
Krause, N., Brand, R. J., Arah, O. A. & Kauhanen, J. Occupational physical activity and 20-year incidence of acute myocardial infarction: results from the Kuopio Ischemic Heart Disease Risk Factor Study. Scand J Work Environ Health 41, 124–139, https://doi.org/10.5271/sjweh.3476 (2015).
Gram, B. et al. Occupational and leisure-time physical activity and workload among construction workers - a randomized control study. Int J Occup Environ Health 22, 36–44, https://doi.org/10.1080/10773525.2016.1142724 (2016).
Kivimaki, M. & Steptoe, A. Effects of stress on the development and progression of cardiovascular disease. Nat Rev Cardiol 15, 215–229, https://doi.org/10.1038/nrcardio.2017.189 (2018).
Borodulin, K. et al. Forty-year trends in cardiovascular risk factors in Finland. Eur J Public Health 25, 539–546, https://doi.org/10.1093/eurpub/cku174 (2015).
Leppala, J. M., Virtamo, J. & Heinonen, O. P. Validation of stroke diagnosis in the National Hospital Discharge Register and the Register of Causes of Death in Finland. Eur J Epidemiol 15, 155–160 (1999).
Lindbohm, J. V., Kaprio, J., Jousilahti, P., Salomaa, V. & Korja, M. Risk Factors of Sudden Death From Subarachnoid Hemorrhage. Stroke 48, 2399–2404, https://doi.org/10.1161/STROKEAHA.117.018118 (2017).
Korja, M., Lehto, H., Juvela, S. & Kaprio, J. Incidence of subarachnoid hemorrhage is decreasing together with decreasing smoking rates. Neurology 87, 1118–1123, https://doi.org/10.1212/WNL.0000000000003091[doi] (2016).
Fagt, S. et al. Nordic Monitoring of diet, physical activity and overweight. Validation of indicators. Nordic Council of Ministers, 48–53 (2011).
Tolonen, H. et al. The validation of the Finnish Hospital Discharge Register and Causes of Death Register data on stroke diagnoses. Eur J Cardiovasc Prev Rehabil 14, 380–385, https://doi.org/10.1097/01.hjr.0000239466.26132.f2 (2007).
Vandenbroucke, J. P. et al. Strengthening the Reporting of Observational Studies in Epidemiology (STROBE): explanation and elaboration. Int J Surg 12, 1500–1524, https://doi.org/10.1016/j.ijsu.2014.07.014 (2014).
Fine, J. P. & Gray, R. J. A Proportional Hazards Model for the Subdistribution of a Competing Risk. Journal of the American Statistical Association 94, 496–509, https://doi.org/10.1080/01621459.1999.10474144 (1999).
Andersson, T., Alfredsson, L., Kallberg, H., Zdravkovic, S. & Ahlbom, A. Calculating measures of biological interaction. Eur J Epidemiol 20, 575–579 (2005).
Greenland, S. & Drescher, K. Maximum likelihood estimation of the attributable fraction from logistic models. Biometrics 49, 865–872 (1993).
Author information
Authors and Affiliations
Contributions
J.V.L., I.R., J.K. and M.K. designed the study. J.V.L. and I.R. performed the statistical analyses. All authors participated in interpreting of the results, in writing of the manuscript and in critical revisions of the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lindbohm, J.V., Rautalin, I., Jousilahti, P. et al. Physical activity associates with subarachnoid hemorrhage risk– a population-based long-term cohort study. Sci Rep 9, 9219 (2019). https://doi.org/10.1038/s41598-019-45614-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-45614-0
This article is cited by
-
A two-sample Mendelian randomization analysis of modifiable risk factors and intracranial aneurysms
Scientific Reports (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.