Abstract
Injection drug use (IDU) is one of the most significant risk factors for viral hepatitis (B and C) and human immunodeficiency virus (HIV) infections. This study assessed seroprevalence rates of hepatitis B virus (HBV), hepatitis C virus (HCV) and human immunodeficiency virus (HIV) in people who inject drugs (PWID) in Kuwait. We conducted a cross-sectional study from April to September 2017. A total of 521 consecutive subjects, admitted at Al-Sabah Hospital. The serological and virological markers of HBV, HCV, and HIV were tested using automated platforms. The mean age of the participants was 32.26 yrs, and the sex ratio (Male/Female) was 15.28. The prevalence rates of HBsAg, anti-HCV, and anti-HIV antibodies were 0.38% (95% CI: 0.07–1.53%), 12.28% (95% CI: 9.65–15.48), and 0.77% (95% CI: 0.25–2.23%), respectively. HCV-RNA was evident in 51.72% (95% CI: 38.34–64.87%) among anti-HCV positive participants. Multivariate analysis showed that the high prevalence of HCV infection amongst PWID is associated with age. Whereas, multivariate analysis revealed no significant associations with age and gender regarding HIV and HBV infections. The results suggest that high rates of HBV, HCV, and HIV infections among injecting drug users than the general population. These findings emphasize the importance of introducing interventions and harm reduction initiatives that have a high impact on reducing needle sharing.
Similar content being viewed by others
Introduction
The hepatitis B virus (HBV), hepatitis C virus (HCV) and the human immunodeficiency virus (HIV) are a major global public health issue. Recent reports from the World Health Organization (WHO) have reported that 257 million persons live with chronic HBV infection, 71 million have chronic HCV infection and approximately 36.7 million people living with HIV1. People who inject drugs (PWID) are more prone to acquiert blood-borne viruses including HCV, HBV, and HIV through unsafe behaviors such as sharing needles, other injection equipments and drug use paraphernalia2. Worldwide, in 2015 an estimated 15.6 million people (10.2–23.7 million) injected drugs of those aged 15–64 years. Among them, 3.2 million (1.6–5.1 million) are women and 12.5 million men (7.5–18.4 million)2. Globally, 2.8 million (1.5–4.5 million) PWID are living with HIV, 8.2 million (4.7–12. 4 million) people might be anti-HCV positive and 1.4 million (0.7–2.4 million) people have chronic HBV infection (HBsAg positive)2. Furthermore, a recent mathematical modelling analysis shows that several Middle East and North Africa (MENA) countries, PWID contribute dominantly to HIV incidence3. To date, no study has reported population-based estimates of HBV, HCV and HIV among PWID in Kuwait. This epidemiological information will add value in creating a national policy for government and policy-makers to allocate appropriate resources and strategically plan for HBV, HCV and HIV prevention interventions. The aim of this study was to determine the prevalence of HBV, HCV and HIV as well as co- and triple-infection rates among PWID in Kuwait.
Materiel and Methods
Study population
Between April and September 2017, a cross-sectional study was carried out among injection drug users (IDUs). Inclusion criterias are age more than 17 years, had a history of injecting drug use, be able to provide informed consent and to provide a blood samples for serological and molecular testing. Eligible participants were interviewed confidentially and data were collected. The study protocol was approved by the permanent Committee for Coordination of Medical and Health Research, Ministry of health, Kuwait and the study was conducted in accordance with the ethical guidelines of the 1975 Declaration of Helsinki as reflected in a priori approval by the institution’s human research committee. Written informed consent was obtained from all participants in this study. All methods were performed in accordance with the relevant guidelines and regulations mentioned above.
Serologic assays
Sera were used for testing HBV, HCV and HIV status. Serum specimens were assessed for antibodies to HIV types 1 and 2 (anti-HIV1/2), HBV surface antigen (HBsAg) and antibody to HCV (anti-HCV) by electrochemical luminescence ECL technology (Cobas 6000 analyser, Roche Diagnostics). Samples showing reactive results were tested using chemiluminescence microparticle immunoassays on the ARCHITECT i2000SR (Abbott Diagnostics, Wiesbaden, Germany).
Virological testing
Samples showing positive from serology assays results were investigated by molecular assay. Serum HIV-1 and HCV RNA were confirmed using Cobas® TaqMan® 6800 (Roche Diagnostics Deutschland GmbH, Mannheim, Germany) with detection limits of 20 copies/ ml (HIV-1) and 15 IU/ml (HCV).
Statistical analysis
Qualitative variables were described using percentages and their 95% confidence interval (CI). For categorical variables, the Fisher’s exact test was used. Comparisons of continuous variables between groups were conducted using Student’s t-test. Multivariate logistic regression analyses was used to identify independent risk factors for infections and their respective 95% confidence interval. Statistical procedures were performed with R software. All p-values were two-sided and p < 0.05 was considered statistically significant.
Results
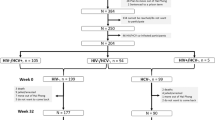
A total of 521 PWID were screened for HBV, HCV and HIV and during the study period. Of them, 489 (93.9%) were male, and 32 (6.1%) were female. The mean age of the participants in the study was 32.26 years (median 30 years, range 17–82 years) with a significant gender difference (females: mean 28.78 years, range 19–44 years; males: mean 32.48 years, range 17–82 years, p = 0.034) (Table 1).
The HIV prevalence in the cohort was 0.77% (95% CI: 0.25–2.23%) (Table 1). The median HIV-RNA viral load was 49015 copies/mL. The differences between males and females were not statistically significant (p = 1.000) (Table 1).
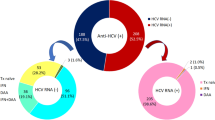
Anti-HCV prevalence was 12.28% (95% CI: 9.65–15.48). The prevalence rates of anti-HCV were 12.88% for males and 3.12% for females (p = 0.159). Of the 64 subjects who were reactive for anti-HCV antibodies, 30 (51.72%, 95% CI: 38.34–64.87%) were HCV-RNA positive while 48.28% had spontaneous HCV-clearance (anti-HCV positive and HCV-RNA negative) (Table 2). In addition, HCV-RNA prevalence correlated with age (Fig. 1).
The prevalence of HBV (HBsAg positive) was 0.38% (95% CI: 0.07–1.53%) with a non-significant difference between males and females (p = 1.000) (Table 3).
Only one of the 521 participants (0.19%; 95% CI: 0.01–1.24%) was found to have an HBsAg-positive, anti-HCV positive and HCV RNA negative. HIV/HBV, HIV/HCV co-infection or HIV/HBV/HCV trible infections were not found among IDUs.
Next, we performed multivariate analyses between positive HBV, HCV and HIV status and socio-demographic characteristics (gender and age). As a result, only age showed a significant association with HCV infection; HCV-positive IDUs were significantly older [mean = 42.58 years; standard deviation (SD) = 9.1] than HCV-negative IDUs (mean = 30.81 years; SD = 8.7) (P < 0.0001). In contrast, HCV-positive and HCV-negative IDUs were comparable in terms of gender. Moreover, no demographic factors were associated with HIV or HBV infection (Table 4).
Discussion
Drug use remains a major in both developed and developing countries, and is a pivotal factor for a plethora of other social problems4. Drug using populations continue to demonstrate a significantly higher prevalence of blood-borne virus infections compared to the general population5,6,7. It is well known that the development of an effective strategy for the control of HBV, HCV, and HIV infections among IDUs is a serious issue for government health authorities. However, there are no data on the rate of blood-borne diseases among PWID in Kuwait. In this cohort, the mean age of the participants (32.26 years) is similar to to previous studies in PWID8,9,10,11,12. In addition, most PWID are males (93.9%) and this data corroborate with previous reports highlighting that the prevalence of IDU among men was far higher than in women2,12,13,14.
It was conspicuous that HIV prevalence among IDUs was remarkably high than national infectious level2. In this study, we found that the HIV prevalence is 0.77%, which is relative low compared to the prevalence observed among IDUs in other parts of the world. HIV prevalence among PWID varied substantially across geographical regions, from 1.1% in Australasia, 3.6% in the MENA, and 4.5% in western Europe, to 24.7% in eastern Europe and 35.7% in Latin America2. In contrast, earlier study and according to the World Bank collection of development indicators, compiled from officially recognized sources, the prevalence of HIV in general population Kuwait was reported at 0–0.1%15,16.
The prevalence of HCV among PWID studied reached 12.28%, which was low compared to the rate found among IDUs in other geographical regions. For example, in sub-Saharan Africa, 21.8% of PWID had anti-HCV, 38.6% in the South Asia, 48.1% in the MENA region2 and 34.5% in Iran17. The lower prevalence relative to other countries is probalbly linked the low levels of sharing of needles, syringes and other drug injection equipment. Moreover, previous studies have reported that the prevalence rate of HCV is up to 10 fold higher than that of HIV18,19,20. In the same line, previous studies carried out in the general popualion showed a prevalence ranging from 0 to 0.8%21,22. In contrast, high prevalence was observed among hemodialysis patients (27 to 71%)23, thalassemic patients (33%)24 and 3% in Kuwaiti individuals with type 2 diabetes21. Viremic HCV infections in Kuwaiti PWID were found to 51.72%. This rate is consistent with HCV viremia prevalence among IDUs reported in other countries8,25. We found that the factor significantly associated with HCV was older age. This result seems to be in line with pervious study8. For HBV and HIV infections, there were no significant associatons with age and gender.
In this study, we found a low HBV prevalence among IDUs (0.38%). In a recent report the highest estimated HBsAg prevalence among PWID was in Thailand (30.5%), China (23.4%) and Myanmar (17.1%). Although countries with the highest prevalence also included the Czech Republic (15.1%), Egypt (13.5%), Belarus (11.2%), Lithuania (10.5%), the Côte d’Ivoire (10.5%) and Azerbaijan (10.4%). Furthermore, the lowest HBsAg prevalence among PWID was in Ireland (0%), Montenegro (0%), Seychelles (0.3%), Bosnia and Herzegovina (0.5%), Syria (0.5%), Maldives (0.5%), Germany (0.7%) and France (0.8%)2. Intersetingly, the lower prevalence of HBsAg among PWID in Kuwait could be explained in part by HBV immunity prior injecting drugs. HBV vaccine had been introduced in the national immunization program from 1990 in Kuwait. Interstingly, 37% (195/521) of PWID in our sample were born since 1990 are likely to have been immunized and thus protected against HBV infection. The prevalence rate of HBV carriers is estimated less than 1%, in the general Kuwait population22,26.
In conclusion, our results showed that there is a high prevalence of HCV infection, but a low prevalence of HIV, HBV and co-infections among PWID in Kuwait. Moreover, the rate of HBV, HCV, and HIV observed among IDUs in Kuwait is very low compared to other parts of the world. However, these rates are higher than observed among the general poupultion thus underscoring the critical need to improve harm-reduction measures. In addition, effective prevention, public education and comprehensive screening programs with a specific focus on high-risk population should be considered as the top priorities of any health policy decision to reduce blood-borne infections. Finally, we are confident that our work significant impact on future efforts on the HBV, HCV, and HIV transmission among IDUs in Kuwait. Whereas, we are fully aware that the limitation of the current study is the lack of some epidemiological data. Other surveys warrant the enrolment of a larger cohort in the future including other risk behaviors related to unsafe sex, frequency of injecting drug use, needles/syringes sharing, duration of injecting drug use and the number of sexual partner.
References
WHO. In WHO guidelines on hepatitis B and C testing. Geneva: World Health Organization; 2017. Licence: CC BY-NC-SA 3.0 IGO WHO Guidelines Approved by the Guidelines Review Committee (2017).
Degenhardt, L. et al. Global prevalence of injecting drug use and sociodemographic characteristics and prevalence of HIV, HBV, and HCV in people who inject drugs: a multistage systematic review. Lancet Glob Health 5, e1192–e1207, https://doi.org/10.1016/S2214-109X(17)30375-3 (2017).
Mumtaz, G. R., Awad, S. F., Feizzadeh, A., Weiss, H. A. & Abu-Raddad, L. J. HIV incidence among people who inject drugs in the Middle East and North Africa: mathematical modelling analysis. J Int AIDS Soc 21, e25102, https://doi.org/10.1002/jia2.25102 (2018).
Edwards, G., Arif, A. & Hadgson, R. Nomenclature and classification of drug- and alcohol-related problems: a WHO Memorandum. Bull World Health Organ 59, 225–242 (1981).
Abdala, N. et al. Sexually transmitted infections, sexual risk behaviors and the risk of heterosexual spread of HIV among and beyond IDUs in St. Petersburg, Russia. Eur Addict Res 14, 19–25, https://doi.org/10.1159/000110407 (2008).
Kamarulzaman, A. et al. Prevention of transmission of HIV, hepatitis B virus, hepatitis C virus, and tuberculosis in prisoners. Lancet 388, 1115–1126, https://doi.org/10.1016/S0140-6736(16)30769-3 (2016).
Christensen, P. B., Krarup, H. B., Niesters, H. G., Norder, H. & Georgsen, J. Prevalence and incidence of bloodborne viral infections among Danish prisoners. Eur J Epidemiol 16, 1043–1049 (2000).
Kinkel, H. T. et al. Prevalence of HIV, Hepatitis B and C Infections and an Assessment of HCV-Genotypes and Two IL28B SNPs among People Who Inject Drugs in Three Regions of Nepal. PLoS One 10, e0134455, https://doi.org/10.1371/journal.pone.0134455 (2015).
Ramezani, A. et al. HCV, HBV, and HIV seroprevalence, coinfections, and related behaviors among male injection drug users in Arak, Iran. AIDS Care 26, 1122–1126, https://doi.org/10.1080/09540121.2014.882485 (2014).
Zhou, Y. H. et al. Comparison of HIV-, HBV-, HCV- and co-infection prevalence between Chinese and Burmese intravenous drug users of the China-Myanmar border region. PLoS One 6, e16349, https://doi.org/10.1371/journal.pone.0016349 (2011).
Weissenbacher, M. et al. High seroprevalence of bloodborne viruses among street-recruited injection drug users from Buenos Aires, Argentina. Clin Infect Dis 37(Suppl 5), S348–352, https://doi.org/10.1086/377560 (2003).
Wu, J. et al. Infection status and risk factors of HIV, HBV, HCV, and syphilis among drug users in Guangdong, China–a cross-sectional study. BMC Public Health 10, 657, https://doi.org/10.1186/1471-2458-10-657 (2010).
Wenz, B. et al. High variability of HIV and HCV seroprevalence and risk behaviours among people who inject drugs: results from a cross-sectional study using respondent-driven sampling in eight German cities (2011–14). BMC Public Health 16, 927, https://doi.org/10.1186/s12889-016-3545-4 (2016).
Spada, E. et al. Incidence and Risk Factors for Hepatitis C Virus Infection among Illicit Drug Users in Italy. J Urban Health 95, 99–110, https://doi.org/10.1007/s11524-017-0207-5 (2018).
Al-Owaish, R. A., Anwar, S., Sharma, P. & Shah, S. F. HIV/AIDS prevalence among male patients in Kuwait. Saudi Med J 21, 852–859 (2000).
Factbook, C. W. HIV/AIDS - ADULT PREVALENCE RATE (2018).
Rahimi-Movaghar, A., Razaghi, E. M., Sahimi-Izadian, E. & Amin-Esmaeili, M. HIV, hepatitis C virus, and hepatitis B virus co-infections among injecting drug users in Tehran, Iran. Int J Infect Dis 14, e28–33, https://doi.org/10.1016/j.ijid.2009.03.002 (2010).
Vickerman, P., Hickman, M., May, M., Kretzschmar, M. & Wiessing, L. Can hepatitis C virus prevalence be used as a measure of injection-related human immunodeficiency virus risk in populations of injecting drug users? An ecological analysis. Addiction 105, 311–318, https://doi.org/10.1111/j.1360-0443.2009.02759.x (2010).
Kretzschmar, M. et al. Regional differences in HIV prevalence among drug users in China: potential for future spread of HIV? BMC Infect Dis 8, 108, https://doi.org/10.1186/1471-2334-8-108 (2008).
Zhou, Y. H. et al. High prevalence of HIV, HCV, HBV and co-infection and associated risk factors among injecting drug users in Yunnan province, China. PLoS One 7, e42937, https://doi.org/10.1371/journal.pone.0042937 (2012).
Chehadeh, W. et al. Hepatitis C virus infection in a population with high incidence of type 2 diabetes: impact on diabetes complications. J Infect Public Health 4, 200–206, https://doi.org/10.1016/j.jiph.2011.05.004 (2011).
Ameen, R. et al. Prevalence of viral markers among first-time Arab blood donors in Kuwait. Transfusion 45, 1973–1980, https://doi.org/10.1111/j.1537-2995.2005.00635.x (2005).
Mohamoud, Y. A., Riome, S. & Abu-Raddad, L. J. Epidemiology of hepatitis C virus in the Arabian Gulf countries: Systematic review and meta-analysis of prevalence. Int J Infect Dis 46, 116–125, https://doi.org/10.1016/j.ijid.2016.03.012 (2016).
al-Fuzae, L., Aboolbacker, K. C. & al-Saleh, Q. beta-Thalassaemia major in Kuwait. J Trop Pediatr 44, 311–312 (1998).
Fraser, H. et al. Model projections on the impact of HCV treatment in the prevention of HCV transmission among people who inject drugs in Europe. J Hepatol 68, 402–411, https://doi.org/10.1016/j.jhep.2017.10.010 (2018).
Schweitzer, A., Horn, J., Mikolajczyk, R. T., Krause, G. & Ott, J. J. Estimations of worldwide prevalence of chronic hepatitis B virus infection: a systematic review of data published between 1965 and 2013. Lancet 386, 1546–1555, https://doi.org/10.1016/S0140-6736(15)61412-X (2015).
Acknowledgements
The authors would like to acknowledge all patients for their participation in this study.
Author information
Authors and Affiliations
Contributions
H.A. and S.E. conceived, designed the experiments, analysis and interpretation of data. H.A. and S.E. wrote the paper in consultation with W.A. H.A., S.ES. carried out the experiment. All authors discussed the results and contributed to the final manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Altawalah, H., Essa, S., Ezzikouri, S. et al. Hepatitis B virus, hepatitis C virus and human immunodeficiency virus infections among people who inject drugs in Kuwait: A cross-sectional study. Sci Rep 9, 6292 (2019). https://doi.org/10.1038/s41598-019-42810-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-42810-w
This article is cited by
-
Multiplex Detection of RNA Viruses Based on Ligation Reaction and Universal PCR Amplification
Current Microbiology (2024)
-
Prevalence of Hepatitis B Virus infection in the Gulf Cooperation Council: a systematic review and meta-analysis
BMC Infectious Diseases (2022)
-
Harm reduction program and hepatitis C prevalence in people who inject drugs (PWID) in Iran: an updated systematic review and cumulative meta-analysis
Harm Reduction Journal (2021)
-
The Past, Present, and Future of PrEP implementation Among People Who Use Drugs
Current HIV/AIDS Reports (2021)
-
A systematic review and meta-analysis of the prevalence of hepatitis C virus infection in people who inject drugs in Iran
BMC Public Health (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.