Abstract
Sequences and structural attributes of mitochondrial genomes have played a critical role in the clarification of relationships among Cnidaria, a key phylum of early-diverging animals. Among the major lineages of Cnidaria, Ceriantharia (“tube anemones”) remains one of the most enigmatic in terms of its phylogenetic position. We sequenced the mitochondrial genomes of two ceriantharians to see whether the complete organellar genome would provide more support for the phylogenetic placement of Ceriantharia. For both Isarachnanthus nocturnus and Pachycerianthus magnus, the mitochondrial gene sequences could not be assembled into a single circular genome. Instead, our analyses suggest that both species have mitochondrial genomes consisting of multiple linear fragments. Linear mitogenomes are characteristic of members of Medusozoa, one of the major lineages of Cnidaria, but are unreported for Anthozoa, which includes the Ceriantharia. The inferred number of fragments and variation in gene order between species is much greater within Ceriantharia than among the lineages of Medusozoa. We identify origins of replication for each of the five putative chromosomes of the Isarachnanthus nocturnus mitogenome and for each of the eight putative chromosomes of the Pachycerianthus magnus mitogenome. At 80,923 bp, I. nocturnus now holds the record for the largest animal mitochondrial genome reported to date. The novelty of the mitogenomic structure in Ceriantharia highlights the distinctiveness of this lineage but, because it appears to be both unique to and diverse within Ceriantharia, it is uninformative about the phylogenetic position of Ceriantharia relative to other Anthozoa. The presence of tRNAMet and tRNATrp in both ceriantharian mitogenomes supports a closer relationship between Ceriantharia and Hexacorallia than between Ceriantharia and any other cnidarian lineage, but phylogenetic analysis of the genes contained in the mitogenomes suggests that Ceriantharia is sister to a clade containing Octocorallia + Hexacorallia indicating a possible suppression of tRNATrp in Octocorallia.
Similar content being viewed by others
Introduction
Analyses of the mitochondrial genome have played a pivotal role in understanding relationships among Cnidaria. Foundational studies1,2 pointed to a clear division between Anthozoa and Medusozoa, with medusozoans sharing the derived feature of linear mitogenomes. Subsequent studies have confirmed a circular mitochondrial genome in diverse octocorals (reviewed in3,4,5,6 and hexacorals (reviewed by3,7,8,9,10,11,12,13,14 and a linear mitogenome in additional diverse medusozoans (reviewed by3,15,16,17 but see)18,19. Some studies have identified other characters that support a fundamental split within Cnidaria between Medusozoa and Anthozoa (e.g.20,21,22,23).
Comparative analyses of anthozoan mitogenomes have revealed structural genomic features like introns, transpositions, gene losses, homing endonucleases, and gene order rearrangements (e.g.7,10,12,14,24,25,26,27,12). This structural diversity is unexpected because anthozoan mitogenomes have some of the lowest reported rates of sequence evolution among animals (e.g.28,29,30,31). Within Anthozoa, the sequences and structures of the mitogenome have been used to tease apart relationships that had been controversial, such as those between scleractinians and corallimorphs7,32, those among Actiniaria10, and the relationship of zoantharians and antipatharians to other hexacorallians8,9.
Although mitogenomes have been more thoroughly studied in hexacorallians than in any other group of non-bilaterian metazoans16,33, the taxonomic sampling is highly skewed towards Actiniaria (14 of 89 mitogenomes) and Scleractinia (59 of 89)3 and no complete mitogenomes have been reported for any members of order/subclass Ceriantharia. Regions of the mitogenome of ceriantharians appear to evolve under different models than those of other Anthozoa3,21,34, suggesting that there are important differences between the mitochondrial genome of ceriantharians and those of other anthozoans.
Ceriantharia, tube anemones (Fig. 1A,B), has been an especially challenging lineage to resolve in the broader cnidarian phylogeny. Historically, they were considered sibling to the Antipatharia and grouped with them as subclass Ceriantipatharia based on similarities in the larval stage35. This relationship was contested based on anatomical features36 and later based on DNA sequence data37. At present, the most commonly cited relationship for Ceriantharia based on DNA sequences is as the sister to all other hexacorallians (e.g.21,37,38,39,40,41,42,43). However, Ceriantharia has also been reconstructed as the sister to Octocorallia21 and as the sister to all other Anthozoa34.
The phylogenetic position of the Ceriantharia has been difficult to test because there is little sequence data published for this group, which has the fewest sequences in GenBank of any hexacorallian order (411 sequences in nr database, 06/2018). In the phylogenomic analyses of21, Ceriantharia had the lowest percent recovery of genes of any anthozoan and was equally well supported in two phylogenetic positions (sister to all other Hexacorallia or sister to Octocorallia). Some of these difficulties may stem from significant differences in evolutionary rate between Ceriantharia and other Anthozoa34. Taxon sampling of Ceriantharia was low in the analyses of22 and43 and the group is generally represented by one or two exemplars in higher-level phylogenies (e.g.22,23,38,41,42,43). This low representation is especially significant and problematic if it is the sister lineage of a much larger group, as implied by most interpretations of its phylogeny.
The Partial Mitogenome of Ceriantheopsis americana
In 2004, Sanger sequencing of the Ceriantheopsis americana mitogenome44 yielded results that suggested the presence of several linear molecules, a highly speculative and controversial conclusion at the time, and one that would remain anecdotal and unresolved until now. Using primer walking and Sanger sequencing, Brugler completely sequenced four genes (nad1, cox1, cox2, and rns) and partially sequenced two genes (nad6 and rnl) and inferred their arrangement as (1) nad1-cox1-cox2, (2) rns-rnl, and (3) nad6-rnl. Unusual features noted at the time that led to the hypothesis that the cerianthid mitogenome was made up of several linear molecules included (1) PCRs that routinely yielded 3+ bands of different size per lane when viewed on an agarose gel (including the traditional LCO1490-HCO2198 primers used to amplify the cox1 ‘Barcode of Life’), (2) the 3′-end of rnl could not be extended regardless of the cerianthid-specific primer combinations employed, (3) depending on the forward primers employed (i.e., rnl vs. cox2), two different NCRs were obtained upstream of nad1 that both converge on the same nad1 sequence, and (4) sequence downstream of rns and nad6 converged on the same rnl sequence (coming from nad6, convergence occurred 178 bp into rnl; coming from rns, convergence occurred 124 bp into rnl). Two additional unique features of the C. americana mitogenome were that (1) cox1 and cox2 were fused into a single gene (2,355 bp in length) and thus continuously encoded, and (2) there was an unusually long intergenic region (1,117 bp) separating nad6 and rnl. Our new data allow us to interpret and contextualize these early observations.
Recognizing the power of mitochondrial genomes to illuminate anthozoan relationships, we sequenced and characterized the mitogenome of the ceriantharians Isarachnanthus nocturnus and Pachycerianthus magnus. With the exception of preliminary sequence data from the mitogenome of Ceriantheopsis americana, these are the first reports of putatively complete mitochondrial genomes for members of this lineage. The genomes of I. nocturnus and P. magnus are unlike those of all other Anthozoa in being linear, but differ from one another in the arrangement of the genes and the inferred number of linear chromosomes. This surprising finding reinforces the uniqueness of Ceriantharia and underscores the difficulty in interpreting its relationship to other major groups within Cnidaria. Phylogenetic analysis based only on coding regions of these mitogenomes supports interpreting Ceriantharia as the sister to Octocorallia and Hexacorallia and thus as a third major lineage within Anthozoa. The presence of tRNA Methionine and Tryptophan in both mitogenomes of Ceriantharia recalls the situation in Hexacorallia and contrasts to the situation in Octocorallia (without tRNATRP) and Medusozoa (which lack these tRNAs) and aligns with the interpretation of relationship based on genomic datasets (see more in23).
Material and Methods
Specimen sampling
The two focal species (Fig. 1) are representatives of the two orders of Ceriantharia. Isarachnanthus nocturnus (Hartog, 1977), order Penicillaria, was collected in São Sebastião Channel (23°49.596′S 45°25.292′W), São Paulo, Brazil (MZUSP 1478) (Ministério do Meio Ambiente - SISBIO 55566-1). Pachycerianthus magnus, order Spirularia, was collected from Chinwan Inner Bay (23°31.896′N 119°33.460′E), Penghu, Taiwan (MZUSP 1951) by Drs. C. Allen Chen and Hernyi Justin Hsieh. Specimens were preserved directly in 92% ethanol. Pieces of marginal tentacles were used for DNA extraction.
Methods for obtaining and assembling mitogenomes
Libraries were prepared using an Illumina TruSeq PCR-free protocol and sequenced on an Illumina HiSeq 2500 platform yielding 250 bp paired-end reads, with an average insert size of 350 bp for I. nocturnus and 550 bp for P. magnus. The sequencing runs produced 14.2 million (m) mate-pairs for I. nocturnus and 15.3 m mate-pairs for P. magnus. The reads were evaluated for quality and adapter-contamination using FastQC45 and cleaned using Trimmomatic46 to remove adapters and low-quality regions. 12.7 m pairs were retained for I. nocturnus (86.1%) and 14.9 m pairs were retained for P. magnus (97.7%). De novo assembly was performed using DISCOVAR de novo v. 5248847 which is optimized for this type of Illumina data. The resulting assembly was converted to a BLAST database, and mitochondrial contigs identified by querying with a set of known cnidarian mitochondrial coding sequences. Trimmed reads were mapped back to the identified mitochondrial contigs using the Geneious 7.1 read mapper48 using High Sensitivity (Medium) default settings, and the mapped reads were reassembled de novo in Geneious to validate assembly and evaluate evenness of coverage and read-agreement. We concatenated species-specific mitochondrial contigs into a single “pseudo contig” and mapped raw reads to determine if paired end reads would map to different mitochondrial chromosomes. Regions of sequence similarity across chromosomes were identified using LASTZ v.1.02.0049 and GC content calculated for each chromosome50. The data are available via GenBank BioSample as SAMN10291198 (Isarachnanthus nocturnus) and SAMN10291199 (Pachycerianthus magnus).
Our assembly pipeline was validated by the current authors through application to other anthozoan mitochondrial genomes using identical methods of data generation. In those cases, mitochondrial genomes assembled into a single DISCOVAR contig10. However, in our study of these two ceriantharians, the assembly for both samples unexpectedly yielded numerous linear chromosomes. Since none of the contigs circularized and no paired-end reads bridged contigs, we attempted to extend contigs using both IMAGE51 with various kmer settings and the Geneious 7.148 iterative read mapper using various parameter settings that balanced extensibility and accuracy. In no case did the contigs for I. nocturnus or P. magnus significantly extend: reads either falsely assembled into highly discordant, non-homologous low-complexity regions or abruptly terminated. We also independently assembled the data using NOVOPlasty v 2.5.952, which is explicitly designed to assemble circular, organellar genomes. In one case, this assembler extended a single contig and circularized it; however, mapping reads back to this contig revealed that an incorporated direct repeat occurs immediately after a 3,000 bp region of minimal mapping quality, casting doubt on this assembly. We used the Phobos tandem repeat search tool53 but found no definitive evidence of telomeric repeats at the ends of any linear fragment. In order to check for the presence of inverted terminal repeats (ITRs) of the type found in the linear mitochondrial genomes of Medusozoans and Cubozoans we aligned each fragment to itself using LASTZ. This method successfully identifies the previously annotated ITRs in Medusozoans and Cubozoans15. Again, there was no evidence of telomeric structure at the ends of any assembled fragment.
Contigs were annotated using DOGMA54 and MITOS55, carefully examining MITOS scores across loci to rule out false positives, and determining ORF boundaries by transferring homologous gene annotations in Geneious from a representative selection of anthozoan and medusozoan sequences from GenBank.
Phylogenetic mitogenome and distance analysis
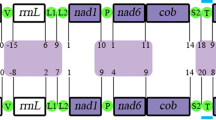
Mitochondrial genomes representing all accepted cnidarian clades – except Myxozoa – were obtained from GenBank (Table 1). Sampling aimed to increase the representation of taxonomic diversity across groups where possible. Represented taxa with complete mitogenomes from each major group were chosen to maintain equal sampling across lineages where possible. Mitochondrial genomes were partitioned by genes for the subsequent assembly of the matrix with the equivalent order of the genes. Differences in the organization of the gene sequence (Fig. 2) were not considered in the present analysis.
The combined dataset was created for all genes from the mitochondrial genomes after alignment of individual genes with MAFFT (parameter: FFT-NS-2)56; this dataset was analyzed under Maximum Likelihood criteria (PhyML 3.0 and RAxML v8)57,58. To evaluate nodal support and to detect if support values were biased, two parametric (aLRT and aBAYES) and non-parametric (Bootstrap) bootstrap values were computed in RAxML (500 pseudoreplicates, same parameters as the original phylogenetic analysis) and additional statistical tests were performed using PhyML with SMS to infer tree model59. The duplicated genes (characterized by sequence similarity) were individualized and aligned in MUSCLE60 and compared by p-distance model in order to calculate their respective genetic distances.
Identification of transfer RNAs
We utilized ARWEN v1.261 and tRNAscan-SE v2.062 to search each individual linear chromosome of I. nocturnus and P. magnus for putative tRNAs. Search parameters for ARWEN were as follows: metazoan mitochondrial tRNA genes, assuming circular topology, search wraps around ends, search both strands, use composite metazoan mitochondrial genetic code. Search parameters for tRNAscan-SE were as follows: sequence source = other mitochondrial, search mode = default (organellar), genetic code = mold/protozoan, cutoff score = 15. Only tRNAs that were located by both programs are reported.
Identification of the origin of replication
We utilized the ‘DNA Walker’ graphing option within GraphDNA63 available online via the Viral Bioinformatics Resource Center [virology.uvic.ca]) to search each mitogenomic chromosome for abrupt changes in base composition bias that are characteristic of both the heavy (OriH) and light (OriL) strand origins of replication (see64 for usage of the DNA Walk within bamboo corals and black corals). We also searched each mitogenomic chromosome for repeats using the Tandem Repeats Finder online server (v 4.0965) and E-QuickTandem (v 6.5.066). After locating putative origins of replication, we used the mfold web server67) to locate stable stem-loop configurations containing characteristic T-rich loops (see64 for an in-depth analysis of features typically associated origins of replication).
Results
The linear and fragmented mitochondrial genomes of Ceriantharia
The mitochondrial genomes we present of the ceriantharians Pachycerianthus magnus and Isarachnanthus nocturnus are the first linear and fragmented mitochondrial genomes described in Anthozoa. The obtained genomes (Fig. 2) are comprised of 77,828 bp and 80,923 bp, respectively, and are organized into eight (P. magnus) and five (I. nocturnus) contigs that likely represent chromosomes. Because we did not detect a telomere sector or something similar at the end of each set of genes, we consider these putative chromosomes, rather than definitive chromosomes.
A small percentage of the paired-end (PE) reads mapped across the possible chromosomes: 1,238 PE reads (1%) for P. magnus and 1,341 PE reads (0.5%) for I. nocturnus. These mismatches resulted in paired ends being mapped consistently to distinct positions within the chromosomes for each species (Supplemental Fig. 1). Due to the position of these mismatches, high sequence similarity across AT-rich chromosomes, and potentially duplicated chromosomal regions and associated genes, this is likely an artifact of the mapping due to their relatively low occurrence.
The ceriantharian mitochondrial genomes we have sequenced are, on average, three to four times longer than those of other cnidarians, with I. nocturnus having the largest animal mitochondrial genome reported to date. The size (but not content) of the mitogenome of these ceriantharians is very similar to those reported for Choanoflagellata68 and exceed the 21,500 bp that Brugler44 estimated for the mitogenome of C. americana. The length and organization help explain several failed attempts of Long-Range PCRs with diverse primers to obtain long mitochondrial sectors with previous mtDNA isolation (Abcam kit (AB65321); SNS & M. Maronna, pers. comm.). Previous attempts by SNS (with M. Maronna) to sequence the mitogenome based on the isolation of mitochondria, DNA extraction and subsequent sequencing on a Roche 454 GS Jr resulted in similar data for Isarachnanthus nocturnus, but the absence or low number of reads in some sectors meant that not all chromosome sequences could be reconstructed without breaks.
We interpret the size of ceriantharian mitogenomes as generally due to an increase in non-coding regions. Despite differences in mitogenome organization and size, the genes in the mitogenomes of I. nocturnus and P. magnus are similar in size to their homologues in other cnidarians, with the exception of ND4L (which is as much as twice the length of that in other Anthozoa) and ND6 (approximately three times the length compared to other Anthozoa). The percent of each of the ceriantharian mitogenomes that encodes proteins and RNAs was low: 19.6% in I. nocturnus and 20.6% in P. magnus. These estimates are much lower than the values obtained by Brugler44 for the partial mitogenome of C. americana, for which 6,894 bp of a total 11,225 bp (~60%) were interpreted to be coding, but the primer walking methods used in that study may have under-captured the non-coding regions and over-estimated the coding regions.
Perhaps surprising given their considerable length, we found some genes that are common in cnidarian mitogenomes are absent in these ceriantharians. In both I. nocturnus and P. magnus, we did not find the open reading frames (ORFs) polB and ORF314 (which have been reported in other anthozoans). In P. magnus, ATP6, CYTb, and ND1 are duplicated, with the copies differing at 34% (ATP6), 28% (CYTb), and 19% (ND1).
Thus, although the mitogenomes of I. nocturnus and P. magnus share some characteristics (linear organization and larger size) when compared to other cnidarian lineages, the organization of the genes was quite different between the two species and it was difficult to identify any conserved gene blocks between them (Fig. 3).
Phylogenomic analysis
The best tree from our maximum likelihood analysis (PHYML - model GTR, Gamma distribution parameter: 1.138, AIC = 333907.267, Log-likelihood: −291618.12823, Unconstrained likelihood: −218549.49064) of the sequences in the mitochondrial genomes of Cnidaria (Fig. 4) is similar in topology to those recovered previously (e.g.37,69,34,23). It includes monophyletic Medusozoa and Anthozoa, with Ceriantharia as the sister group of Hexacorallia + Octocorallia. In this tree, the monophyly of Anthozoa and of the three subgroups within it (Hexacorallia, Octocorallia, and Ceriantharia) are well supported. At the same time, the medusozoan groups have high support values despite only a small number of mitogenomes available from this group (especially in the Cubozoa and Staurozoa). The structure of the tree for Medusozoa had relatively short internal branches and relatively long terminal branches (Fig. 4).
Transfer RNAs
Within the five linear chromosomes making up the I. nocturnus mitogenome, we recovered three tRNAs (Fig. 5), all of which were located in non-coding regions. Transfer RNA Methionine (Met) was located on two different chromosomes (Isa2 and Isa4), while tRNA Tryptophan (Trp) was located on chromosome Isa3. Within the eight linear chromosomes making up the P. magnus mitogenome, we also recovered three tRNAs. Two different copies of tRNAMet were located on the same chromosome (Pac2) while tRNATrp was located on chromosome Pac4.
Origins of Replication
Using a combination of DNA Walker and mfold, we successfully located the putative origin of replication in all five chromosomes of the Isarachnanthus nocturnus mitogenome and in all eight chromosomes of the Pachycerianthus magnus mitogenome. This combination of analyses searched for abrupt changes in base composition bias, stable stem-loop configurations containing T-rich loops, and repeats. While the origin of replication is often associated with inverted repeats or significant tandem repeats, in only two instances was a predicted origin of replication found associated with significant repeats (surrounding the origin of replication on chromosomes Pac5 and Pac7). Interestingly, the DNA Walks were significantly more complex than those produced to date for actiniarians, antipatharians and scleractinians (MRB personal observation). Finding a unique Ori on each individual chromosome provides definitive support that the mitogenomes of Isarachnanthus nocturnus and Pachycerianthus magnus are indeed multipartite. See Table 2 for the location of each predicted origin of replication and Fig. 6 for graphical output from DNA Walker and mfold that typically define the origin of replication.
The origin of replication (Ori) was successfully located in all chromosomes. To demonstrate how we visually located the Ori, we selected the DNA Walk (A,B) and mfold (C,D) analysis of chromosome Pac8 (A,C) and Pac4 (B,D). The DNA Walk shows abrupt changes in base composition bias that are characteristic of Oris, and the mfold analysis shows stem-loop configurations containing T-rich loops that are also characteristics of Oris. For Pac8, the Ori was located at position 4,950, while for Pac4, the Ori was located at position 7,850 (both Oris are denoted by a black box). DNA Walker window size: 50.
Our analyses do not differentiate between the origin of replication on the heavy (OriH) vs. light (OriL) strand; however, based on previous analyses in closely related groups (see63), we hypothesize that the Oris reported here are indicative of OriH.
Discussion
Mitochondrial genome architecture seems to be more variable among members of early-diverging clades of Metazoa than among members of the Bilateria70,71,33. Among non-bilaterian phyla, only Cnidaria includes major lineages for which the mitochondrial genome is not circular1,3,33. In Cnidaria, linear mitochondrial genomes are compelling as a synapomorphy for Medusozoa because this feature is both fairly unusual and highly consistent (reviewed by16). The linear mitogenomes of major medusozoan lineages are largely conserved in terms of gene order and can be related through a relatively straightforward transformation series16 involving fragmentation and gene re-arrangement through recombination.
A striking exception to the pattern in Cubozoa, Hydrozoa, Scyphozoa, and Staurozoa is the Myxozoa. Myxozoans are intracellular parasites with complex life cycles. Although their phylogenetic position has been difficult to assess (reviewed by72), they are inferred to be either highly modified medusozoans73,74,75 or the sister group (Endocnidozoa) to Medusozoa23. The linear mitochondrial genome characteristic among members of Medusozoa does not apply to Myxozoa, since all the myxozoan species studied to date have circular mitochondrial genomes. The mitochondrion of species in the myxozoan genus Kudoa are small, and the genes in them are organized into a single circular genome that is evolving more quickly than those in other Medusozoa18. The order of genes reported for the mitochondrial genome of Kudoa does not correspond to those published for other Medusozoans (cf.16,18). In contrast, in the myxozoan Enteromyxum leei, the mitogenome is organized into eight circular chromosomes69. This high within-lineage variation in genome architecture mirrors what we have discovered here in the Ceriantharia.
The deviation in mitogenome structure in Myxozoa does not refute the value of linear mitochondrial genomes as a synapomorphy for the Medusozoa23, but it does underscore that variation in mitochondrial genome structure is characteristic of Cnidaria. Likewise, the linear mitogenome of Ceriantharia we describe here should not be interpreted as proof for a particularly close relationship between Ceriantharia and Medusozoa; it is merely more evidence of plasticity in mitogenome architecture in the Cnidaria.
The present study presents evidence of the isolation of Ceriantharia in relation to Hexacorallia and Octocorallia but does not support a close relationship between Ceriantharia and Medusozoa. The gene order in the mitogenomes of Medusozoa is largely conserved among medusozoans3,16 and wholly different in Ceriantharia. Mitogenome architecture in anthozoan lineages is generally more variable that that of Medusozoa in structure and gene order, (see, e.g.7,8,9,10,11,12,13,14,19,24,26,27) other than the common occurrence of the mismatch repair gene MutS in the mitogenome of Octocorallia76,77 the nature and evolutionary importance of this variation remains unclear. The variety reported to date suggests that there may be much more diversity than has been appreciated, and a better account of this diversity may improve inference about the evolutionary transformations of the mitogenome. With respect to the phylogenetic evidence in the sequences of genes within the mitogenome, our phylogenetic reconstruction supports Ceriantharia as an isolated branch within Anthozoa, rather than a close ally of the Medusozoa (Fig. 4). This pattern of sequence affinity despite structural difference was also seen for the myxozoans Kudoa and Enteromyxum18,78 phylogenetic analyses of sequences place these species within or sister to the Medusozoa although the structure of their genomes is unlike those of other medusozoans.
In contrast to the conservation of gene order generally characterizing medusozoan mitochondrial genomes, we did not identify any conserved gene blocks among ceriantharians and other anthozoans. We found no consistency between our two ceriantharian species and any other published gene order from a cnidarian mitochondrial genome. The absence of any relation to the patterns observed in Octocorallia or Hexacorallia may be an indication of the phylogenetic isolation of Ceriantharia from these two groups. These differences in gene order underscore the differences in rate of gene evolution between ceriantharians and other anthozoans34.
The nucleotide composition of protein genes in each of the ceriantharian mitogenomes did not deviate from the general pattern seen in other Cnidaria (Table 3). In some cases (e.g. ATP6, NAD4), the nucleotide composition appears to be at an intermediate stage between Medusozoa and Anthozoa. The nucleotide composition is distinct in each of these groups and the values we found for Ceriantharia lies between them. Nevertheless, conclusive interpretations will require a greater number of species of Ceriantharia and greater sampling of Medusozoa (e.g., Cubozoa, Staurozoa).
The non-coding regions in Ceriantharia are very long and account for almost 80% of the mitochondrial genome. It is in these regions that the differences between Ceriantharia and other Cnidaria are most notable. In general, non-coding regions tend to be larger in Anthozoa than in Medusozoa and represent as much as 10% of the mitochondrial genome (Octocorallia79). The marked increase in non-coding DNA in the mitochondrial genome of Ceriantharia is noteworthy even though the increases seem not to have been the result of a single event in the two ceriantharians studied here, because the recovered patterns are quite distinct and may indicate different events. The higher rate of mitochondrial gene evolution in Ceriantharia compared to other Anthozoa34 may help to explain the generation and accumulation of the non-coding regions in Ceriantharia.
Foox and colleagues10 posit that mitochondrial genomes may be under selective pressure to place rns and rnl in close proximity to one another. Montoya et al.80 noted that animals with adjacent rRNA genes use a “transcriptional attenuator” system to produce a nearly 50-fold excess of ribosomal RNA compared with other genes. This accelerated rate of transcription resulting from gene rearrangement may increase the productivity of a mitochondrion and, therefore, be selectively advantageous. In the I. nocturnus mitogenome, rns and rnl are adjacent to one another on chromosome Isa2. Additionally, rnl is found on three separate chromosomes (Isa1, Isa2 and Isa3), which can be explained with a similar rationale (i.e., more copies = more transcripts). In the P. magnus mitogenome, rns and rnl are adjacent to one another on chromosome Pac5. Evidence for increased transcription of rns and rnl is seen in the increased read coverage for those sections of the chromosomes that contain adjacent ribosomal genes (Suppl Fig. 1). These findings meet the expectations of the hypothesis that there is selective advantage to the association of rns and rnl¸ but in the absence of an ancestral gene order for the ceriantharian mitogenome or a well-resolved phylogeny for Anthozoa, we cannot determine whether these are ancestral similarities or evidence of convergent re-arrangements or speculate on the sequence of rearrangements that generated the observed gene orders.
Even though the I. nocturnus mitogenome is made up of five chromosomes and the P. magnus mitogenome consists of eight chromosomes, both mitogenomes have two tRNAMet and a single tRNATrp. Unlike the Octocorallia, which only have tRNAMet, to date, all hexacorallians have tRNAMet and tRNATrp. Thus, finding both tRNAMet and tRNATrp in both species analyzed herein suggests that the loss of tRNATrp in octocorals is a derived condition. However, interpretation of evolutionary gain or loss of tRNAs requires a well-supported phylogeny, which remains elusive for Anthozoa. Quattrini and collegues43 recently developed probes to target Ultra Conserved Elements (UCEs) across a broad range of anthozoans, including the cerianthids Cerianthus membranaceus and Pachycerianthus sp. Quattrini et al. (in prep) have now sequenced 100 s of UCE across the anthozoan Tree of Life and produced the first phylogenomic tree for this group. These results, along with ongoing whole mitochondrial genome, nuclear genome and transcriptome sequencing efforts, will hopefully provide resolution as to the placement of cerianthids.
Data Availability
The data that support the findings of this study are available from GenBank as SAMN10291198 (Isarachnanthus nocturnus) and SAMN10291199 (Pachycerianthus magnus). These will be released upon acceptance of the manuscript. Access for editors and reviewers prior to publication can be obtained through https://ftp-trace.ncbi.nlm.nih.gov/sra/review/SRP166847_20190125_103955_37d5c0b6b354bc3c790d2696b42756c9.
References
Bridge, D., Cunningham, C. W., Schierwater, B., DeSalle, R. & Buss, L. W. Class-level relationships in the phylum Cnidaria: evidence from mitochondrial genome structure. Proc. Nat. Acad. Sci. 89, 8750–8753 (1992).
Bridge, D., Cunningham, C. W., DeSalle, R. & Buss, L. W. Class-level relationships in the phylum Cnidaria: molecular and morphological evidence. Mol. Biol. Evol. 12, 679–689 (1995).
Kayal, E., Roure, B., Philippe, H., Collins, A. G. & Lavrov, D. V. Cnidarian phylogenetic relationships as revealed by mitogenomics. BMC Evol. Biol. 13, 5 (2013).
Figueroa, D. F. & Baco, A. R. Octocoral mitochondrial genomes provide insights into the phylogenetic history of gene order rearrangements, order reversals, and cnidarian phylogenetics. Genome Biol. Evol. 7, 391–409 (2014).
Wu, J. S., Ju, Y. M., Hsiao, S. T. & Hsu, C. H. Complete mitochondrial genome of Junceella fragilis (Gorgonacea, Ellisellidae). Mitochondrial DNA 27, 1229–1230 (2016).
Poliseno, A., Altuna, A., Cerrano, C., Wörheide, G. & Vargas, S. Historical biogeography and mitogenomics of two endemic Mediterranean gorgonians (Holaxonia, Plexauridae). Org. Div. Evol. 17, 365–373 (2017).
Medina, M., Collins, A. G., Takaoka, T. L., Kuehl, J. V. & Boore, J. L. Naked corals: skeleton loss in Scleractinia. Proc. Nat. Acad. Sci. 103, 9096–9100 (2006).
Brugler, M. R. & France, S. C. The complete mitochondrial genome of the black coral Chrysopathes formosa (Cnidaria: Anthozoa: Antipatharia) supports classification of antipatharians within the subclass Hexacorallia. Mol. Phylogenet. Evol. 42, 776–788 (2007).
Sinniger, F., Chevaldonné, P. & Pawlowski, J. Mitochondrial genome of Savalia savaglia (Cnidaria, Hexacorallia) and early metazoan phylogeny. J. Mol. Evol. 64, 196–203 (2007).
Foox, J., Brugler, M., Siddall, M. E. & Rodríguez, E. Multiplexed pyrosequencing of nine sea anemone (Cnidaria: Anthozoa: Hexacorallia: Actiniaria) mitochondrial genomes. Mitochondrial DNA Part A. 27, 2826–2832 (2016).
Shi, X. et al. Complete mitochondrial genome of disc coral Turbinaria peltata (Scleractinia, Dendrophylliidae). Mitochondrial. DNA 27, 962–963 (2016).
Chi, S. I. & Johansen, S. D. Zoantharian mitochondrial genomes contain unique complex group I introns and highly conserved intergenic regions. Gene 628, 24–31 (2017).
Zhang, L. & Zhu., Q. Complete mitochondrial genome of the sea anemone, Anthopleura midori (Actiniaria: Actiniidae). Mitochondrial DNA 28, 335–336 (2017).
Zhang, B., Zhang, Y. H., Wang, X., Zhang, H. X. & Lin, Q. The mitochondrial genome of a sea anemone Bolocera sp. exhibits novel genetic structures potentially involved in adaptation to the deep‐sea environment. Ecol. Evol. 7, 4951–4962 (2017).
Smith, D. R. et al. First complete mitochondrial genome sequence from a box jellyfish reveals a highly fragmented linear architecture and insights into telomere evolution. Genome Biol Evol. 4, 52–58 (2012).
Kayal, E. et al. Phylogenetic analysis of higher-level relationships within Hydroidolina (Cnidaria: Hydrozoa) using mitochondrial genome data and insight into their mitochondrial transcription. PeerJ 3, e1403 (2015).
Li, H. H., Sung, P. J. & Ho, H. C. The complete mitochondrial genome of the Antarctic stalked jellyfish, Haliclystus antarcticus Pfeffer, 1889 (Staurozoa: Stauromedusae). Genome. Data 8, 113–114 (2016).
Takeuchi, F. et al. The mitochondrial genomes of a myxozoan genus Kudoa are extremely divergent in Metazoa. PLoS One 10, e0132030 (2015).
Dubin, A., Chi, S. I., Emblem, Å. Moum, T. & Johansen, S. D. Deep-water sea anemone with a two-chromosome mitochondrial genome. Gene 692, 195-200 (2019).
Marques, A. C. & Collins, A. G. Cladistic analysis of Medusozoa and cnidarian evolution. Invert Biol. 123, 23-42 (2004).
Daly., M. et al. The phylum Cnidaria: A review of phylogenetic patterns and diversity 300 years after Linnaeus. Zootaxa 1668, 127–182 (2007).
Zapata, F. et al. Phylogenomic analyses support traditional relationships within Cnidaria. PLoS One 10, e0139068 (2015).
Kayal, E. et al. Phylogenomics provides a robust topology of the major cnidarian lineages and insights on the origins of key organismal traits. BMC Evol Biol. 18, 68 (2018).
Beagley, C. T., Okimoto, R. & Wolstenholme, D. R. The mitochondrial genome of the sea anemone Metridium senile (Cnidaria): introns, a paucity of tRNA genes, and a near-standard genetic code. Genetics 148, 1091–1108 (1998).
Fukami, H., Chen, C. A., Chiou, C. Y. & Knowlton, N. Novel group I introns encoding a putative homing endonuclease in the mitochondrial cox1 gene of scleractinian corals. J. Mol. Evol. 64, 591–600 (2007).
Emblem, Å. et al. Sea anemones possess dynamic mitogenome structures. Mol. Phylogenet. Evol. 75, 184–193 (2014).
Emblem, Å., Karlsen, B. O., Evertsen, J. & Johansen, S. D. Mitogenome rearrangement in the cold-water scleractinian coral Lophelia pertusa (Cnidaria, Anthozoa) involves a long-term evolving group I intron. Mol Phylogenet Evol 61, 495–503 (2011).
Shearer, T. L., Van Oppen, M. J. H., Romano, S. L. & Wörheide, G. Slow mitochondrial DNA sequence evolution in the Anthozoa (Cnidaria). Mol Ecol. 11, 2475–2487 (2002).
Huang, D., Meier, R., Todd, P. A. & Chou, L. M. Slow mitochondrial COI sequence evolution at the base of the metazoan tree and its implications for DNA barcoding. J. Mol. Evol 66, 167–174 (2008).
Chen, I. P. et al. Comparative analyses of coding and noncoding DNA regions indicate that Acropora (Anthozoa: Scleractina) possesses a similar evolutionary tempo of nuclear vs. mitochondrial genomes as in plants. Mar Biotechnol. 11, 141–152 (2009).
Daly, M., Gusmão, L. C., Reft, A. J. & Rodríguez, E. Phylogenetic signal in mitochondrial and nuclear markers in sea anemones (Cnidaria, Actiniaria). Integ. Comp. Biol. 50, 371–388 (2010).
Kitahara, M. V. et al. The “Naked Coral” hypothesis revisited – evidence for and against scleractinian monophyly. PLoS One 9, e94774 (2014).
Lavrov, D. V. & Pett, W. Animal mitochondrial DNA as we do not know it: mt-genome organization and evolution in Nonbilaterian lineages. Genome Biol Evol. 8, 2896–2913 (2016).
Stampar, S. N., Maronna, M. M., Kitahara, M. V., Reimer, J. D. & Morandini, A. C. Fast-evolving mitochondrial DNA in Ceriantharia: A reflection of Hexacorallia paraphyly? PLoS One 9, e86612 (2014).
van Beneden, E. Les Anthozoaires de la “Plankton-Expedition”. Ergebn Plankton-Exp Humbolt-Stiftung. 2, 1–222 (1897).
Schmidt, H. On the evolution in the Anthozoa in Proceedings of the 2nd international symposium on coral reefs (ed. Cameron, A. M et al.) 533–560 (The Great Barrier Reef Committee, 1974).
Chen, C. A. et al. Systematic relationships within the Anthozoa (Cnidaria: Anthozoa) using the 5′-end of the 28S rDNA. Mol. Phylogenet. Evol. 4, 175–183 (1995).
France, S. C., Rosel, P. E., Agenbroad, J. E., Mullineaux, L. S. & Kocher, T. D. DNA sequence variation of mitochondrial large-subunit rRNA provides support for a two-subclass organization of the Anthozoa (Cnidaria). Mol Mar Biol Biotech. 5, 15–28 (1996).
Berntson, E. A., France, S. C. & Mullineaux, L. S. Phylogenetic relationships within the class Anthozoa (phylum Cnidaria) based on nuclear 18S rDNA sequences. Mol. Phylog. Evol. 13, 417–33 (1999).
Won, J., Rho, B. & Song, J. A phylogenetic study of the Anthozoa (phylum Cnidaria) based on morphological and molecular characters. Coral Reefs 20, 39–50 (2001).
Daly, M., Fautin, D. G. & Cappola, V. A. Systematics of the Hexacorallia (Cnidaria: Anthozoa). Zool J Linn Soc. 139, 419–37 (2003).
Rodríguez, E. et al. Hidden among sea anemones: the first comprehensive phylogenetic reconstruction of the order Actiniaria (Cnidaria, Anthozoa, Hexacorallia) reveals a novel group of hexacorals. PLoS One 9, e96998 (2014).
Quattrini, A. M. et al. Universal target‐enrichment baits for anthozoan (Cnidaria) phylogenomics: New approaches to long‐standing problems. Mol. Ecol. Res. 18, 281–95 (2018).
Brugler, M. R. The complete mitochondrial DNA sequence of the black coral Chrysopathes formosa (Antipatharia) and six non-contiguous mitochondrial genes of the tube anemone Ceriantheopsis americanus (Ceriantharia): implications for cnidarian phylogeny. Doctoral dissertation, Graduate School of the College of Charleston. 2004.
Andrews, S. FastQC: a quality control tool for high throughput sequence data. Version 1 [FastQC]. Available from, http://www.bioinformatics.babraham.ac.uk/projects/fastqc. 2018 Jun 18.
Bolger, A. M., Lohse, M. & Usadel, B. Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 30, 2114–20 (2014).
Weisenfeld, N. I. et al. Comprehensive variation discovery in single human genomes. Nature Genetics 46, 1350 (2014).
Kearse, M. et al. Geneious Basic: an integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics 28, 1647–9 (2012).
Harris, R. S. Improved pairwise alignment of genomic DNA. PhD thesis. Penn State University, Computer Science and Engineering, 2007.
Richard, D. GC Content in sliding window - GitHub repository. Available from, https://github.com/DamienFr/GC-content-in-sliding-window- 2018 Jun 18.
Tsai, I. J., Otto, T. D. & Berriman, M. Improving draft assemblies by iterative mapping and assembly of short reads to eliminate gaps. Genome Biol. 11, R41 (2010).
Dierckxsens, N., Mardulyn, P. & Smits, G. NOVOPlasty: de novo assembly of organelle genomes from whole genome data. Nucleic Acids Res. 45, e18 (2016).
Mayer, C. Phobos 3.3.11. Available from, http://www.rub.de/spezzoo/cm/cm_phobos.htm 2018 Jun 18.
Wyman, S. K., Jansen, R. K. & Boore, J. L. Automatic annotation of organellar genomes with DOGMA. Bioinformatics 20, 3252–5 (2004).
Bernt, M. et al. MITOS: improved de novo metazoan mitochondrial genome annotation. Mol. Phylog. Evol. 69, 313–9 (2013).
Katoh, K., Rozewicki, J. & Yamada, K. D. MAFFT online service: multiple sequence alignment, interactive sequence choice and visualization. Brief. Bioinform. 2017 Sep 6.
Guindon, S. et al. New algorithms and methods to estimate maximum-likelihood phylogenies: assessing the performance of PhyML 3.0. Syst Biol. 59, 307–21 (2010).
Stamatakis, A. RAxML version 8: a tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 30, 1312–3 (2014).
Lefort, V., Longueville, J. E. & Gascuel, O. SMS: smart model selection in PhyML. Mol Biol Evol. 34, 2422–2424 (2017).
Edgar, R. C. MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucl. Acids Res. 32, 1792–7 (2004).
Laslett, D. & Canbäck, B. ARWEN: a program to detect tRNA genes in metazoan mitochondrial nucleotide sequences. Bioinformatics 24, 172–5 (2007).
Lowe, T. M. & Chan, P. P. tRNAscan-SE On-line: integrating search and context for analysis of transfer RNA genes. Nucleic Acids Res. 44, W54–7 (2016).
Thomas, J. M., Horspool, D., Brown, G., Tcherepanov, V. & Upton, C. GraphDNA: a Java program for graphical display of DNA composition analyses. Bioinformatics 8, 21 (2007).
Brugler, M. R. & France, S. C. The mitochondrial genome of a deep-sea bamboo coral (Cnidaria, Anthozoa, Octocorallia, Isididae): genome structure and putative origins of replication are not conserved among octocorals. J. Mol. Evol. 67, 125 (2008).
Benson, G. Tandem repeats finder: a program to analyze DNA sequences. Nucleic Acids Res. 27, 573–80 (1999).
Rice, P., Longden, I. & Bleasby, A. EMBOSS: the European molecular biology open software suite. Trends. Genetics 16, 276–7 (2000).
Zuker, M. Mfold web server for nucleic acid folding and hybridization prediction. Nucleic Acids Res. 31, 3406–15 (2003).
Burger, G., Forget, L., Zhu, Y., Gray, M. W. & Lang, B. F. Unique mitochondrial genome architecture in unicellular relatives of animals. Proc. Nat. Acad. Sci 100, 892–7 (2003).
Song, J. I. & Won, J. H. Systematic relationship of the anthozoan orders based on the partial nuclear 18S rDNA sequences. Korean J Biol Sci. 1, 43–52 (1997).
Lavrov, D. V. et al. Mitochondrial DNA of Clathrina clathrus (Calcarea, Calcinea): six linear chromosomes, fragmented rRNAs, tRNA editing, and a novel genetic code. Mol Biol Evol. 30, 865–80 (2012).
Osigus, H. J., Eitel, M., Bernt, M., Donath, A. & Schierwater, B. Mitogenomics at the base of Metazoa. Mol Phylogen Evol. 69, 339–51 (2013).
Foox, J. & Siddall, M. E. The road to Cnidaria: history of phylogeny of the Myxozoa. J Parasitol. 101, 269–274 (2015).
Evans, N. M., Lindner, A., Raikova, E. V., Collins, A. G. & Cartwright, P. Phylogenetic placement of the enigmatic parasite, Polypodium hydriforme, within the phylum Cnidaria. BMC Evol Biol. 8, 139 (2008).
Evans, N. M., Holder, M. T., Barbeitos, M. S., Okamura, B. & Cartwright, P. The phylogenetic position of Myxozoa: exploring conflicting signals in phylogenomic and ribosomal data sets. Mol Biol Evol. 27, 2733–46 (2010).
Chang, E. S. et al. Genomic insights into the evolutionary origin of Myxozoa within Cnidaria. Proc. Nat. Acad. Sci. 112, 14912–7 (2015).
Pont-Kingdon, G. A. et al. A coral mitochondrial mutS gene. Nature 375, 109–111 (1995).
Bilewitch, J. P. & Degnan, S. M. A unique horizontal gene transfer event has provided the octocoral mitochondrial genome with an active mismatch repair gene that has potential for an unusual self-contained function. BMC Evol. Biol. 11, 228 (2011).
Yahalomi, D. et al. The multipartite mitochondrial genome of Enteromyxum leei (Myxozoa): eight fast-evolving megacircles. Mol. Biol. Evol. 34, 1551–6 (2017).
Park, E., Song, J. I. & Won, Y. J. The complete mitochondrial genome of Calicogorgia granulosa (Anthozoa: Octocorallia): potential gene novelty in unidentified ORFs formed by repeat expansion and segmental duplication. Gene 486, 81–7 (2011).
Montoya, J., Gaines, G. L. & Attardi, G. The pattern of transcription of the human mitochondrial rRNA genes reveals two overlapping transcription units. Cell 34, 151–9 (1983).
Kayal, E. et al. Evolution of linear mitochondrial genomes in medusozoan cnidarians. Genome Biol. Evol. 4, 1–2 (2011).
Acknowledgements
This work was partly supported by São Paulo Research Foundation (FAPESP 2015/24408-4, 2017/50028-0) and CNPq 404121/2016-0 to SNS, a SPRINT award from UNC Charlotte to J.M. and A.M.R. and by NSF DEB-1257796 to M.D. We are deeply indebted to Drs M. Maronna and M. Kitahara for comments during the development of the study, to Dr. D. Huchon for comments in review and to Prof. CA Chen, Biodiversity Research Centre, Academia Sinica, Taiwan and Dr. HJ Hsieh, Marine Biological Research Centre, Penghu, Taiwan, for collecting specimens of Pachycerianthus magnus.
Author information
Authors and Affiliations
Contributions
The study was conceived by M.B., M.D. and S.S. Samples were obtained and identified by S.S. and prepared for sequencing by J.M. M.B. and M.R.B. analyzed the data. M.R.B. contributed comparative data from his thesis. M.B., M.R.B., M.D., J.M., A.M.R. and S.S. interpreted the results and helped develop secondary methods to explore them. S.S., M.B. and M.D. generated the first draft of the manuscript, to which M.R.B. added significant text. All authors read, commented on, and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Stampar, S.N., Broe, M.B., Macrander, J. et al. Linear Mitochondrial Genome in Anthozoa (Cnidaria): A Case Study in Ceriantharia. Sci Rep 9, 6094 (2019). https://doi.org/10.1038/s41598-019-42621-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-42621-z
This article is cited by
-
Mito-nuclear discordance within Anthozoa, with notes on unique properties of their mitochondrial genomes
Scientific Reports (2023)
-
Unlocking the jar: revealing gastric content in Ceriantharia (Cnidaria, Anthozoa) through whole-genome shotgun sequencing
Aquatic Ecology (2023)
-
Inheritance through the cytoplasm
Heredity (2022)
-
Application of phylogenomic tools to unravel anthozoan evolution
Coral Reefs (2022)
-
Extensive gene rearrangements in the mitogenomes of congeneric annelid species and insights on the evolutionary history of the genus Ophryotrocha
BMC Genomics (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.