Abstract
Engineering chlorophyll (Chl) pigments that are bound to photosynthetic light-harvesting proteins is one promising strategy to regulate spectral coverage for photon capture and to improve the photosynthetic efficiency of these proteins. Conversion from the bacteriochlorophyll (BChl) skeleton (7,8,17,18-tetrahydroporphyrin) to the Chl skeleton (17,18-dihydroporphyrin) produces the most drastic change of the spectral range of absorption by light-harvesting proteins. We demonstrated in situ selective oxidation of B800 BChl a in light-harvesting protein LH2 from a purple bacterium Rhodoblastus acidophilus by 2,3-dichloro-5,6-dicyano-1,4-benzoquinone. The newly formed pigment, 3-acetyl Chl a, interacted with the LH2 polypeptides in the same manner as native B800. B850 BChl a was not oxidized in this reaction. CD spectroscopy indicated that the B850 orientation and the content of the α-helices were unchanged by the B800 oxidation. The nonameric circular arrangement of the oxidized LH2 protein was visualized by AFM; its diameter was almost the same as that of native LH2. The in situ oxidation of B800 BChl a in LH2 protein with no structural change will be useful not only for manipulation of the photofunctional properties of photosynthetic pigment-protein complexes but also for understanding the substitution of BChl to Chl pigments in the evolution from bacterial to oxygenic photosynthesis.
Similar content being viewed by others
Introduction
Light-harvesting antenna proteins play crucial roles in the primary process in photosynthesis. In these proteins, cyclic tetrapyrrole pigments such as chlorophyll (Chl) and bacteriochlorophyll (BChl) are embedded in the protein matrix and engaged in the efficient capture of photons and the transfer of excitation energy to the reaction center complexes. The spectral coverage of pigments in light-harvesting proteins is the basis for efficient photosynthetic ability in photosynthetic organisms. Generally, oxygenic photosynthetic organisms utilize Chl pigments (17,18-dihydroporphyrin), whereas purple photosynthetic bacteria and heliobacteria exploit BChls a, b, and g (7,8,17,18-tetrahydroporphyrin)1,2,3. The skeletons of (B)Chl pigments are primarily responsible for their spectral properties. The lowest energy absorption bands (Qy bands) of the 17,18-dihydroporphyrin pigments are positioned at approximately 100-nm shorter wavelength regions than those of the 7,8,17,18-tetrahydroporphyrin pigments.
Light-harvesting complex 2 (LH2), which is a peripheral antenna protein in purple photosynthetic bacteria, has BChl a and carotenoids as light-harvesting pigments. LH2 proteins have symmetric circular arrangements of pigment-polypeptide subunits4,5,6. BChl a pigments in LH2 proteins are classified into two types termed as B800 and B850 due to their Qy bands around 800 and 850 nm. B800 BChl a is present as the monomeric form in LH2 proteins. In contrast, B850 BChl a pigments form a dimer in one subunit composed of transmembranous α- and β-polypeptides and excitonically interact with each other in the circular arrangement. The excitation energy captured by B800 BChl a is transferred to B850 BChl a in LH2 proteins7,8,9.
Changing BChl a to Chl-type pigments in bacterial photosynthetic proteins is one excellent strategy to control light-harvesting and energy transfer abilities. This is helpful not only for the elucidation of the mechanisms of energy transfer and photoprotection in the target photosynthetic proteins but also for the development of novel photofunctional proteins. In this regard, the exchange of B800 BChl a with Chl pigments in LH2 proteins by B800-removal and the subsequent insertion of exogenous Chls have been studied10,11,12,13,14,15,16,17.
In contrast to the exchange of B800 BChl a, there is little information on in situ conversion of BChl a into Chl-type pigments in LH2 proteins. Especially, no information is available about the isolation and the detailed characterization of LH2 proteins whose B800 BChl a is selectively converted to Chl pigments by in situ oxidation, although BChl oxidation has been reported in studies on photoprotection in light-harvesting proteins in purple bacteria18,19,20,21,22,23,24 and the functional roles of BChl pigments in proteins in green sulfur bacteria25 and heliobacteria26. Generally bacteriochlorin pigments (7,8,17,18-tetrahydroporphyrins) are chemically oxidized by 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (DDQ) to give corresponding chlorins (17,18-dihydroporphyrins)27. We apply this reaction to in situ conversion of B800 BChl a in LH2 as a novel methodology to regulate the photofunctional abilities.
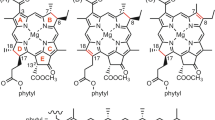
In this study, we performed selective conversion of B800 BChl a to a corresponding chlorin pigment called 3-acetyl Chl a (Fig. 1) in LH2 protein isolated from a purple photosynthetic bacterium Rhodoblastus (Rbl.) acidophilus by oxidation with DDQ. In contrast to the complete oxidation of B800 BChl a, no oxidation of B850 BChl a occurred. The newly formed pigment, 3-acetyl Chl a, was accommodated in the B800 site in essentially the same manner as native BChl a and transferred the excitation energy to B850 BChl a. Circular dichroism (CD) spectroscopy and atomic force microscopy (AFM) indicated that the protein structure was preserved even if B800 BChl a is converted to 3-acetyl Chl a.
Results
Spectral properties
Incubation of native LH2 with DDQ followed by purification with anion exchange chromatography gave LH2 protein whose B800 BChl a was oxidized (denoted as oxidized LH2). We compared the electronic absorption spectrum of this protein with that of the native LH2 (Fig. 2). The native LH2 protein showed two intense Qy absorption bands of B800 and B850 BChl a at 802 and 859 nm. In contrast, the oxidized LH2 protein had no Qy band of B800 BChl a but exhibited a new absorption band at 694 nm. This peak position was the same as that of 3-acetyl Chl a, which was inserted into the B800 site in B800-depleted LH216. The peak position and the bandwidth of the Qy band of B850 BChl a in the oxidized LH2 was identical to those in the native LH2 (Fig. 2, insert). The Soret band of 3-acetyl Chl a in the oxidized LH2 overlapped with the absorption bands of a carotenoid (rhodopin glucoside) at 453 nm.
Electronic absorption spectra of native LH2 (black) and oxidized LH2 (red) in 20 mM Tris-HCl buffer containing 0.1% n-dodecyl-β-D-maltoside (pH = 8.0). Spectra were normalized at Qy peaks of B850 BChl a. Fluorescence emission spectrum of oxidized LH2 by excitation at 680 nm (blue) is shown with its absorption spectrum. Insert shows overlapped absorption spectra of native LH2 and oxidized LH2 in Qy region.
The Qx band of BChl a in the oxidized LH2 protein was detected at 593 nm, which was slightly red-shifted from the Qx band in the native LH2 at 591 nm. This shift is ascribable to the disappearance of the Qx band of B800 BChl a by the conversion of this pigment to 3-acetyl Chl a, since the Qx band of BChl a is derived from both B800 and B850 BChl a, whose Qx positions slightly differed, in native LH216.
The absorption bands of rhodopin glucoside in the native LH2 were positioned at 524, 489, and 461 nm. The positions of the two former bands in the oxidized LH2 were the same as those in the native LH2. Note that the band of rhodopin glucoside at 461 nm overlapped with the Soret band of 3-acetyl Chl a in the spectrum of the oxidized LH2. The coincidence of the peak positions of rhodopin glucoside between the oxidized LH2 and the native LH2 indicates that 3-acetyl Chl a is properly accommodated in the B800 site, since its peak positions are sensitive to the presence of the chlorophyllous pigment in the B800 site in LH210,11.
A CD spectrum of the oxidized LH2 is compared with that of the native LH2 (Fig. 3). The negative signal of B800 BChl a in the native LH2 around 800 nm disappeared accompanying a new negative signal around 695 nm in the CD spectrum of the oxidized LH2 (Fig. 3, right). The position of this new CD signal corresponds to the Qy absorption band of 3-acetyl Chl a. The reversed S-shaped CD signal of B850 BChl a in the CD spectrum of the oxidized LH2 between 820 and 900 nm closely resembled that of the native LH2, indicating that the treatment of the native LH2 with DDQ barely disturbed the orientation and electronic structures of B850 BChl a. The shape and intensity of the negative CD signal in the oxidized LH2 in the UV region is also quite analogous to that of the native LH2, indicating that the content of the α-helices in the oxidized LH2 was identical to that in the native LH2. Therefore, the local structure around B850 BChl a and the secondary structure in LH2 were preserved even if B800 BChl a was oxidized in situ.
Fluorescence spectroscopy demonstrates the intracomplex energy transfer from the oxidized pigments in the B800 sites to B850 BChl a. The predominant excitation of 3-acetyl Chl a in the oxidized LH2 produced a strong emission from B850 BChl a at 868 nm with little emission from 3-acetyl Chl a around 700 nm (Fig. 2, blue curve). The apparent fluorescence quantum yields of B850 BChl a by the excitation of 3-acetyl Chl a in the oxidized LH2 was 8.7 ± 0.6% (the average and standard deviation of five samples). This value is consistent with that of native LH2 by the excitation of B800 BChl a (9.1%)17. These results indicate that 3-acetyl Chl a in the B800 sites can efficiently transfer the excitation energy to B850 BChl a in the oxidized LH2 like in the native LH2.
Pigment analysis
Chlorophyllous pigments in the oxidized LH2 protein were analyzed by reverse-phase HPLC. In the chromatogram of the pigments in the oxidized LH2, which were detected at 770 nm, a major fraction was eluted at 11.3 min (Fig. 4B). The retention time of this fraction was identical to that of BChl a in the native LH2 (Fig. 4A). Electrospray ionization mass spectrometry (ESI-MS) also confirmed that this pigment was BChl a; the observed ion peaks (m/z = 911.6 and 933.6) were identical to the calculated values of BChl a for [MH+] and [M + Na+] (911.55 and 933.54). A new fraction was observed at 12.2 min in the 677-nm detected chromatogram of the chlorophyllous pigments in the oxidized LH2 (Fig. 4C). This fraction exhibited Soret and Qy bands at 441 and 685 nm, respectively, in its on-line absorption spectrum. The ion peaks of this fraction by ESI-MS were detected at m/z = 909.6 and 931.6, which correspond to the calculated values of 3-acetyl Chl a for [MH+] and [M + Na+] (909.54 and 931.52). Few fractions, which are ascribable to the by-products via oxidation, were observed in Fig. 4B,C. Note that further oxidized porphyrin-type pigment of BChl a, namely 3-acetyl protochlorophyll a, is absent in the current HPLC analysis. These results indicate that 3-acetyl Chl a is predominantly formed by the in situ oxidation of the native LH2 protein.
HPLC chromatograms of chlorophyllous pigments in native LH2 (A) and oxidized LH2 (B,C). Chromatograms (A–C) were recorded at 770, 770, and 677 nm, respectively. LH2 proteins were directly injected and eluted on a reverse-phase column 5C18-AR-II (6 mm i.d. × 250 mm) with a guard column 5C18-AR-II (4.6 mm i.d. × 10 mm) with methanol at the flow rate of 1.0 mL min–1. Signals denoted by × were due to direct injection of LH2 solutions.
Resonance Raman spectra
The interactions of 3-acetyl Chl a with the polypeptides in the oxidized LH2 protein were examined by resonance Raman spectroscopy. The native LH2 showed the 3-C=O stretching vibrational bands of BChl a at 1623 cm−1 (Fig. 5A). The 3-C=O band of B800 BChl a overlapped with that of B850 BChl a; the difference Raman spectrum between the native LH2 and the B800-depleted LH2 revealed the 3-C=O band of B800 BChl a at 1621 cm−1 (Fig. 5D). This position indicates that the 3-acetyl group in B800 BChl a is hydrogen-bonded with the β-Arg20 in LH212. The oxidized LH2 protein exhibited the 3-C=O stretching vibrational band of 3-acetyl Chl a at 1620 cm−1 in the difference Raman spectrum between the oxidized LH2 and the B800-depleted LH2 (Fig. 5E), indicating that the 3-acetyl group in the oxidized pigment is also hydrogen-bonded with the LH2 polypeptide.
The position of the Raman signal of the 13-C=O stretching vibrational band of 3-acetyl Chl a in the oxidized LH2 at 1698 cm−1 was analogous to that of B800 BChl a in the native LH2 (Fig. 5A,B), indicating that the 13-keto group in 3-acetyl Chl a is free from hydrogen-bonding with polypeptides like native BChl a in the B800 site12. Therefore, 3-acety Chl a is accommodated in the B800 site in essentially the same manner as native BChl a even if B800 BChl a was oxidized.
The signal in the Raman spectrum of the oxidized LH2 protein at 1667 cm−1 is assigned to the 13-C=O stretching vibrational band of B850 BChl a (Fig. 5B). This position is almost the same as those in the native LH2 and the B800-depleted LH2 (Fig. 5A,C). The signals at 1602 cm−1 are assigned to the CC stretching modes of the methine bridges of BChl a and 3-acetyl Chl a.
AFM observations
The overall structure of the oxidized LH2 protein was clearly visualized with a home-built frequency modulation AFM (FM-AFM) in a buffer solution. The ring structure, which is comprised of nine subunits, in the oxidized LH2 was quite analogous to that of the native LH2 (Fig. 6, left and center), indicating that no deformation of the LH2 ring structure was induced by the oxidation of B800 BChl a in the protein. The averaged top-to-top distances of the LH2 ring in both the native and oxidized LH2 were estimated to be 5.2 ± 0.3 and 5.1 ± 0.2 nm (the averages and standard deviations of ten samples) by the height-profiles obtained from the AFM images (Fig. 6, right). Therefore, the ring diameter in the LH2 protein was unchanged by the in situ conversion of BChl a to 3-acetyl Chl a in the B800 sites.
Discussion
In this study, we first demonstrated the selective conversion of B800 BChl a to 3-acetyl Chl a in LH2 protein by in situ oxidation with DDQ and characterized the oxidized protein by spectroscopic and AFM measurements. B800 BChl a was completely changed to 3-acetyl Chl a without oxidation of B850 BChl a. 3-acetyl Chl a was accommodated in the B800 site in essentially the same manner as B800 BChl a. The secondary structure and the nonameric circular arrangement of the LH2 protein remained via this oxidation.
Although chemical, photochemical, and electrochemical oxidation of isolated light-harvesting proteins and chromatophores in some purple photosynthetic bacteria has been reported18,19,20,21,22,23,24, a mixture of oxidized products was formed from BChl a in the light-harvesting proteins and site-specific oxidation of BChl a in LH2 was scarcely observed. These results are in sharp contrast to our results, which clearly indicate that only B800 BChl a was exclusively converted to 3-acetyl Chl a. The differences are mainly ascribable to the oxidants used for the chemical oxidation of LH2 proteins; ferricyanide was commonly used in previous studies18,21,22,23,25. Ferricyanide produces a cation radical of BChl a, which is subsequently converted to oxidized products, since ferricyanide acts as a one-electron oxidant. Such reactions via the cation radical reduce both the selectivity of the oxidized products and the B800/B850 oxidation inside LH2. On the contrary, DDQ used in the current study can withdraw two hydrogen atoms from the bacteriochlorin and chlorin macrocycles27,28,29. This feature is appropriate for the product selectivity by oxidation of BChl a in LH2 protein. In addition, DDQ does not react with B850 BChl a embedded in the protein matrix and only oxidizes B800 BChl a that is exposed on the outside of the protein scaffold, producing the site-selectivity for oxidation of B800 BChl a in LH2. Preferential photooxidation of B850 BChl a in LH2 from a purple sulfur bacterium Ectothiorhodospira haloalkaliphila was reported very recently24. This B850 photooxidation induced the blue-shift of the Qy band of B850 BChl a, and the blue-shift was reproduced by simulations of electronic absorption spectra of successive numbers of oxidized pigments in the B850 sites in LH2. In contrast, the current chemical oxidation produces no shift of the B850 Qy band (Fig. 2), supporting no oxidation of B850 BChl a in LH2.
The current study will shed new light on the substitution from BChl (7,8,17,18-tetrahydroporphyrin) to Chl pigments (17,18-dihydroporphyrin) in the evolution of photosynthetic organisms. Phylogenic analyses of the genes of the enzymes for (B)Chl synthesis suggest that ancient photosynthetic organisms might possess BChl pigments, although the first appearance of chlorophyllous pigment (Chl or BChl) in the evolutional history has not been accurately unraveled30,31,32,33,34,35,36. Oxygenic conditions might be one type of evolutionary pressure that caused the shift from BChl to Chl pigments by changing from anoxygenic to oxygenic organisms. Indeed, our study demonstrates that an oxidant can change BChl a bound to LH2 protein to a corresponding Chl pigment and the formed Chl pigment can engage in light-harvesting. These results are in line with a working hypothesis called the acquisition and discrimination model for pigment-binding proteins proposed by Mimoro and Tanaka37, in which the incorporation of a new pigment into pre-existing proteins leads to the acquisition of new genes for the biosynthesis of the new pigment and the subsequent evolution of a protein moiety. In the case of evolution from BChl a to Chl a, the loss of the genes of the enzymes for the reduction of the chlorin macrocycle and the conversion of the 3-substituent from the vinyl to acetyl groups might be promoted by the oxidation of BChl a that is bound to ancestor photosynthetic proteins.
In conclusion, B800 BChl a can be selectively converted to a corresponding chlorin-type pigment, 3-acetyl Chl a, in LH2 protein from Rbl. acidophilus without perturbation to the protein structure by oxidation with DDQ. This conversion allows the LH2 protein to collect red-light, which is not absorbed by native LH2 even though the photon number in the red region is maximum in all the wavelength region of the photons that reached the surface on the earth. The in situ oxidation of BChl a in LH2 protein will be useful for engineering photofunctions in natural light-harvesting proteins and for understanding the alteration from BChl pigments in anoxygenic photosynthetic bacteria to Chl pigments in oxygenic organisms in the evolution of photosynthesis.
Materials and Methods
Materials
Native LH2 protein was isolated from the cultured cells of a purple photosynthetic bacterium Rbl. acidophilus DSM145 according to previous reports38,39. B800-depleted LH2 protein was prepared as reported elsewhere39. DDQ and acetone were purchased from Wako Pure Chemical Industries, Ltd. (Osaka, Japan) and used without further purification. DDM was purchased from Dojindo Laboratories, Co. (Kumamoto, Japan).
Preparation of oxidized LH2
A solution of native LH2 in 20 mM Tris buffer containing 0.1% DDM (pH 8.0) was adequately desalted and mixed with 1/10 volume of an acetone solution of DDQ. The relative molar ratio of DDQ to native LH2 was 2000 in the mixed solution. The solution was incubated at 35 °C in the dark. After disappearance of the B800 Qy band using electronic absorption spectral analysis, DDQ was roughly removed by ultrafiltration using Amicon centricon concentrators (30 kDa cutoff, Merk Millipore Ltd., Cork, Ireland). Oxidized LH2 was purified by anion exchange column chromatography using Whatman DE52 resin (GE Healthcare, Little Chalfont, U.K.), followed by ultrafiltration using Amicon centricon concentrators (30 kDa cutoff, Merk Millipore Ltd., Cork, Ireland).
Spectroscopic measurements
Electronic absorption and CD spectra of LH2 proteins were measured with a Shimadzu UV-2450 spectrophotometer (Shimadzu, Kyoto, Japan) and a JASCO J-820 spectropolarimeter (JASCO, Tokyo, Japan). Fluorescence emission spectra were measured with a Hitachi F-7100 spectrophotometer (Hitachi, Tokyo, Japan). The apparent fluorescence quantum yields of B850 BChl a were measured by a Hamamatsu C9920-03G fluorescence measurement system (Hamamatsu Photonics, Shizuoka, Japan) from emission between 840 and 950 nm.
HPLC analysis of chlorophyllous pigments
LH2 proteins in 20 mM Tris buffer containing 0.1% DDM (pH = 8.0) were concentrated by ultrafiltration using Amicon centricon concentrators (30 kDa cutoff, Merk Millipore Ltd., Cork, Ireland), followed by the injection of the concentrated LH2 solution into a HPLC system comprised of a Shimadzu LC-20AT pump, a SPD-M20A photodiode array detector, and a Shimadzu CBM-20A system controller (Shimadzu, Kyoto, Japan). ESI-MS analysis was performed with a Shimadzu LCMS-2020 system (Shimadzu, Kyoto, Japan).
Resonance Raman spectroscopy
An aliquot of a LH2 solution was deposited on a stainless plate and dried with nitrogen. Excitation beam (355 nm) from a Nd:YAG laser was focused onto the sample film through ×40 objective lens. The laser intensity at the sample surface was adjusted to ∼0.4 mW and backscattering from the sample was collected at 25 °C. Each spectrum was accumulated for 30 s at a single spot to alleviate sample degradation. To improve the S/N ratio, 25–80 spectra at different points were averaged.
AFM measurements
A 100 μL of a solubilized LH2 solution in 20 mM Tris buffer containing 0.1% DDM and 150 mM NaCl (pH 8.0) was deposited to a cleaved mica substrate with a diameter of 12 mm (SPI Supplies, West Chester, PA, USA), followed by standing for 30 min at room temperature in the dark. Then the sample was rigorously rinsed with 20 mM Tris buffer containing 150 mM NaCl (pH 8.0) and observed in 20 mM Tris buffer containing 150 mM NaCl (pH 8.0) by a home-built FM-AFM40,41,42 with a silicone cantilever (PPP-NCHAuD, Nanoworld, Headquarters, Switzerland), which had a nominal spring constant of 42 N/m. The typical resonance frequency and the Q factor in an aqueous buffer solution were 150 kHz and 7. A commercially available AFM controller (ARC2, Asylum Research, Santa Barbara, CA, USA) was used to control the FM-AFM system.
Data Availability
Data generated or analyzed during the current study are included in this published article and are available from the corresponding author on reasonable request.
References
Croce, R. & van Amerongen, H. Natural strategies for photosynthetic light harvesting. Nature Chem. Biol. 10, 492–501 (2014).
Hashimoto, H. et al. Natural and artificial light-harvesting systems utilizing the functions of carotenoids. J. Photochem. Photobiol. C: Photochem. Rev. 25, 46–70 (2015).
Saer, R. G. & Blankenship, R. E. Light-harvesting in phototrophic bacteria: structure and function. Biochem. J. 474, 2107–2131 (2017).
McDermott, G. et al. Crystal structure of an integral membrane light-harvesting complex from photosynthetic bacteria. Nature 374, 517–521 (1995).
Papiz, M. Z. et al. The structure and thermal motion of the B800–B850 LH2 complex from Rps. acidophila at 2.0 Å resolution and 100 K: new structural features and functionally relevant motions. J. Mol. Biol. 326, 1523–1538 (2003).
Koepke, J. et al. The crystal structure of the light-harvesting complex II (B800–850) from Rhodospirillum molischianum. Structure 4, 581–597 (1996).
Sundström, V., Pullerits, T. & van Grondelle, R. Photosynthetic light-harvesting: reconciling dynamics and structure of purple bacterial LH2 reveals function of photosynthetic unit. J. Phys. Chem. B 103, 2327–2346 (1999).
Cogdell, R. J., Gall, A. & Köhler, J. The architecture and function of the light-harvesting apparatus of purple bacteria: from single molecules to in vivo membranes. Q. Rev. Biophys. 39, 227–324 (2006).
Scholes, G. D., Fleming, G. R., Olaya-Castro, A. & van Grondelle, R. Lessons from nature about solar light harvesting. Nature Chem. 3, 763–774 (2011).
Bandilla, M. et al. Reconstitution of the B800 bacteriochlorophylls in the peripheral light harvesting complex B800–850 of Rhodobacter sphaeroides 2.4.1 with BChl a and modified (bacterio-)chlorophylls. Biochim. Biophys. Acta 1364, 390–402 (1998).
Fraser, N. J. et al. Selective release, removal, and reconstitution of bacteriochlorophyll a molecules into the B800 sites of LH2 complexes from Rhodopseudomonas acidophila 10050. Biochemistry 38, 9684–9692 (1999).
Gall, A. et al. Bacteriochlorin-protein interactions in native B800-B850, B800 deficient and B800-Bchla P-reconstituted complexes from Rhodopseudomonas acidophila, strain 10050. FEBS Lett. 449, 269–272 (1999).
Herek, J. L. et al. B800→B850 energy transfer mechanism in bacterial LH2 complexes investigated by B800 pigment exchange. Biophys. J. 78, 2590–2596 (2000).
Gall, A. et al. Probing the binding sites of exchanged chlorophyll a in LH2 by Raman and site-selection fluorescence spectroscopies. FEBS Lett. 491, 143–147 (2001).
Saga, Y., Amari, K. & Miyagi, K. Insertion of chlorophyll a derivatives into the binding sites of B800 bacteriochlorophyll a in light-harvesting complex 2 from the purple photosynthetic bacterium Rhodoblastus acidophilus. J. Photochem. Photobiol. A: Chem. 353, 591–596 (2018).
Saga, Y. & Miyagi, K. Characterization of 3-acetyl chlorophyll a and 3-acetyl protochlorophyll a accommodated in the B800 binding sites in photosynthetic light-harvesting complex 2 in the purple photosynthetic bacterium Rhodoblastus acidophilus. Photochem. Photobiol. 94, 698–704 (2018).
Saga, Y., Yamashita, M., Imanishi, M. & Kimura, Y. Reconstitution of chlorophyll d into the bacterial photosynthetic light-harvesting protein LH2. Chem. Lett. 47, 1071–1074 (2018).
Picorel, R., Lefebvre, S. & Gingras, G. Oxido-reduction of B800–B850 and B880 holochromes isolated from three species of photosynthetic bacteria as studied by electron-paramagnetic resonance and optical spectroscopy. Eur. J. Biochem. 142, 305–311 (1984).
Kropacheva, T. N. & Hoff, A. J. Electrochemical oxidation of bacteriochlorophyll a in reaction centers and antenna complexes of photosynthetic bacteria. J. Phys. Chem. B 105, 5536–5545 (2001).
Makhneva, Z. K., Erokhin, Y. E. & Moskalenko, A. A. Carotenoid-photosensitized oxidation of bacteriochlorophyll dimers in light-harvesting complexes B800-B850 in Allochromatium minutissimum cells. Dokl. Biochem. Biophys. 416, 256–259 (2007).
Fiedor, J. et al. Antioxidant effects of carotenoids in a model pigment–protein complex. Acta Biochim. Pol. 59, 61–64 (2012).
Makhneva, Z. K., Ashikhmin, A. A., Bolshakov, M. A. & Moskalenko, A. A. 3-Acetyl-chlorophyll formation in light-harvesting complexes of purple bacteria by chemical oxidation. Biochemistry (Moscow) 81, 176–186 (2016).
Law, C. J. & Cogdell, R. J. The effect of chemical oxidation on the fluorescence of the LH1 (B880) complex from the purple bacterium Rhodobium marinum. FEBS Lett. 432, 27–30 (1998).
Leiger, K. et al. Controlling photosynthetic excitons by selective pigment photooxidation. J. Phys. Chem. B 123, 29–38 (2019).
Bina, D. & Blankenship, R. E. Chemical oxidation of the FMO antenna protein from Chlorobaculum tepidum. Photosynth. Res. 116, 11–19 (2013).
Ferlez, B. et al. The effect of bacteriochlorophyll g oxidation on energy and electron transfer in reaction centers from Heliobacterium modesticaldum. J. Phys. Chem. B 119, 13714–13725 (2015).
Smith, J. S. R. & Calvin, M. Studies on the chemical and photochemical oxidation of bacteriochlorophyll. J. Am. Chem. Soc. 88, 4500–4506 (1966).
Tamiaki, H., Watanabe, T. & Miyatake, T. Facile synthesis of 131-oxo-porphyrins possessing reactive 3-vinyl or 3-formyl group, protochlorophyll-a/d derivatives by 17,18-dehydrogenation of chlorins. J. Porphyrins Phthalocyanines 3, 45–52 (1999).
Saga, Y., Hojo, S. & Hirai, Y. Comparison of demetalation properties between zinc chlorin and zinc porphyrin derivatives: effect of macrocyclic structures. Bioorg. Med. Chem. 18, 5697–5700 (2010).
Xiong, J. & Bauer, C. E. Complex evolution of photosynthesis. Annu. Rev. Plant Biol. 53, 503–521 (2002).
Olson, J. M. & Blankenship, R. E. Thinking about the evolution of photosynthesis. Photosynth. Res. 80, 373–386 (2004).
Hohmann-Marriot, M. F. & Blankenship, R. E. Evolution of photosynthesis. Annu. Rev. Plant Biol. 62, 515–548 (2011).
Xiong, J., Inoue, K. & Bauer, C. E. Tracking molecular evolution of photosynthesis by characterization of a major photosynthesis gene cluster from Heliobacillus mobilis. Proc. Natl. Acad. Sci. USA 95, 14851–14856 (1998).
Xiong, J. et al. Molecular evidence for the early evolution of photosynthesis. Science 289, 1724–1730 (2000).
Raymond, J., Siefert, J. L., Staples, C. R. & Blankenship, R. E. The natural history of nitrogen fixation. Mol. Biol. Evol. 21, 541–544 (2003).
Cardona, T. Origin of bacteriochlorophyll a and the early diversification of photosynthesis. Plos One 11, e0151250 (2016).
Mimuro, M. & Tanaka, A. The in vivo and in vitro reconstitution of pigment-protein complexes, and its implication in acquiring a new system. Photosynth. Res. 81, 129–137 (2004).
Saga, Y. & Hirota, K. Determination of the molar extinction coefficients of the B800 and B850 absorption bands in light-harvesting complexes 2 derived from three purple photosynthetic bacteria Rhodoblastus acidophilus, Rhodobacter sphaeroides, and Phaeospirillum molischianum by extraction of bacteriochlorophyll a. Anal. Sci. 32, 801–804 (2016).
Saga, Y. et al. Reversible Changes in the Structural Features of Photosynthetic Light-harvesting Complex 2 by Removal and Reconstitution of B800 Bacteriochlorophyll a Pigments. Biochemistry 56, 3484–3491 (2017).
Fukuma, T. et al. Development of low noise cantilever deflection sensor for multienvironment frequency-modulation atomic force microscopy. Rev. Sci. Instrum. 76, 053704–053711 (2005).
Fukuma, T. Wideband low-noise optical beam deflection sensor with photothermal excitation for liquid-environment atomic force microscopy. Rev. Sci. Instrum. 80, 023707–023714 (2009).
Asakawa, H. et al. Submolecular-scale imaging of alpha-helices and C-terminal domains of tubulins by frequency modulation atomic force microscopy in liquid. Biophys. J. 101, 1270–1276 (2011).
Acknowledgements
We thank Ms. Madoka Yamashita, Kindai University, for her experimental assistance and Prof. Takeshi Fukuma, Kanazawa University, for his kind advice for building of the FM-AFM instrument. This work was partially supported by JST PRESTO (JPMJPR1416 to Y.S. and JPMJPR1411 to H.A.) and JSPS KAKENHI (JP17K05940 and JP18H05182 to Y.S.).
Author information
Authors and Affiliations
Contributions
Y.S. conceived the project. K.K. and Y.O. prepared native and oxidized LH2 proteins. Y.S., K.K. and Y.O. contributed to spectroscopic analysis of LH2 proteins and pigment analysis. M.I. and Y.K. performed RR measurements. S.M. and H.A. performed AFM analysis. Y.S., Y.K. and H.A. analyzed the data, interpreted the results, and wrote the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Saga, Y., Kawano, K., Otsuka, Y. et al. Selective oxidation of B800 bacteriochlorophyll a in photosynthetic light-harvesting protein LH2. Sci Rep 9, 3636 (2019). https://doi.org/10.1038/s41598-019-40082-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-40082-y
This article is cited by
-
Atomic force microscopic analysis of the light-harvesting complex 2 from purple photosynthetic bacterium Thermochromatium tepidum
Photosynthesis Research (2023)
-
In situ formation of photoactive B-ring reduced chlorophyll isomer in photosynthetic protein LH2
Scientific Reports (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.