Abstract
Considerable taxonomic diversity has been recognised among early Miocene catarrhines (apes, Old World monkeys, and their extinct relatives). However, locomotor diversity within this group has eluded characterization, bolstering a narrative that nearly all early catarrhines shared a primitive locomotor repertoire resembling that of the well-described arboreal quadruped Ekembo heseloni. Here we describe and analyse seven catarrhine capitates from the Tinderet Miocene sequence of Kenya, dated to ~20 Ma. 3D morphometrics derived from these specimens and a sample of extant and fossil capitates are subjected to a series of multivariate comparisons, with results suggesting a variety of locomotor repertoires were present in this early Miocene setting. One of the fossil specimens is uniquely derived among early and middle Miocene capitates, representing the earliest known instance of great ape-like wrist morphology and supporting the presence of a behaviourally advanced ape at Songhor. We suggest Rangwapithecus as this catarrhine’s identity, and posit expression of derived, ape-like features as a criterion for distinguishing this taxon from Proconsul africanus. We also introduce a procedure for quantitative estimation of locomotor diversity and find the Tinderet sample to equal or exceed large extant catarrhine groups in this metric, demonstrating greater functional diversity among early catarrhines than previously recognised.
Similar content being viewed by others
Introduction
While catarrhines (the clade including Old World monkeys and apes) of the early Miocene (ca. 23-16 Ma) are thought to have been taxonomically diverse, the range of locomotor diversity in this group has been more difficult to characterise due to a relative lack of fossil evidence. In accord with early and influential narratives positing primate locomotor evolution to have occurred in a series of distinct stages1,2, most early Miocene catarrhines are thought to have practiced a similar and somewhat limited set of positional behaviours3. This inference, derived largely from functional analysis of the extensive postcranial hypodigm of Ekembo heseloni4,5,6,7, is consistent with above-branch palmigrade quadrupedalism with occasional bouts of slow climbing or careful clambering8,9,10,11,12,13,14,15,16,17, and perhaps some leaping18,19. This behavioural repertoire has essentially become the null hypothesis in evaluating early Miocene catarrhine positional behaviour, with most postcrania thought to be insufficiently distinct from E. heseloni to indicate discernable behavioural divergence3,20,21,22. Derived positional behaviours (i.e., orthogrady rather than pronogrady, suspension rather than above-branch positional behaviours, versatile and/or acrobatic climbing involving highly abducted limbs rather than largely parasagittal limb movements, and substantial terrestriality) are not generally considered to have begun emerging until the middle Miocene (ca. 16-12 Ma), as represented by the identification of incipient terrestriality in Equatorius africanus23,24,25 and enhanced climbing ability in Nacholapithecus kerioi26,27, with more substantial locomotor adaptation and diversification occurring only later during the late-middle Miocene of Eurasia28,29.
Efforts to evaluate this narrative are hindered by a lack of fossil evidence, as early Miocene catarrhine postcrania tend to be rare, fragmentary, and unassociated30,31,32,33. Despite these limitations, several studies have argued for the presence of derived positional behaviours in some early Miocene taxa. For example, the stem catarrhines Dendropithecus macinnesi from Rusinga and Simiolus enjiessi from Kalodirr are thought to have some adaptations for climbing20,34 or suspension4,10,11,35,36,37. Among putative apes, Morotopithecus bishopi demonstrates adaptation for orthograde climbing38,39,40,41, and Turkanapithecus kalakolensis may also have had enhanced climbing abilities11. At Songhor, a mid-sized proximal femur has also been interpreted to evince adaptation for climbing and suspension21,30, two medial cuneiforms seem to resemble extant apes more than E. heseloni42, and a sample of hallucal metatarsals displays morphological diversity potentially related to function32. Nevertheless, evidence of derived behaviours is still often deemphasised in reviews of early Miocene catarrhine evolution21,22, and there has been no attempt to quantify locomotor diversity among early Miocene catarrhines.
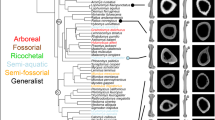
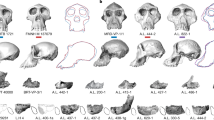
Seven catarrhine capitates (Fig. 1) were recovered between 1931 and 1996 from Songhor, Chamtwara, and Mteitei Valley, penecontemporaneous sites within the Tinderet Miocene sequence of Western Kenya (Fig. S1, Table S1b) dated to around 20 Ma43,44. This sample provides a rare opportunity to evaluate locomotor diversity among the catarrhines of a geographically and temporally constrained period predating the Eurasian hominoid radiation45. While several of these specimens are discussed in the work of previous researchers28,30, none had been formally described. We compare the Tinderet capitates to a broad sample of extant anthropoids as well as Ekembo heseloni (Table S1) using morphometrics derived from 3D models (Table 1, Fig. S4; see also SI and ref. 46). Functional morphology is assessed with positional classifiers built using linear discriminant function analysis (DFA) and glmnet, an elastic net-regularised multinomial logistic regression machine learning algorithm47, and by quantitative estimation of locomotor proportions as well as qualitative comparisons. We attempt to constrain the species identity of each fossil specimen by considering these results in concert with body mass estimates, taxonomic classifiers and hierarchical clustering. Finally, we use an approach derived from two-block partial least squares analysis (PLS)48 to create, for the first time, a quantitative estimate of locomotor diversity in a fossil catarrhine sample relative to that of extant catarrhine groups, and discuss implications for our understanding of catarrhine evolution.
Results and Discussion
Results of this study sort the seven Tinderet capitates into four groups: small- and medium-sized arboreal quadrupeds, a small suspensory form, and a derived, medium-sized morph with great ape affinities. When combined with the large quadruped Proconsul major, also present in this setting but not represented in the sample, at least five niches are currently distinguishable from postcrania, close in number to the 6 genera (8 species) recognised dentally from these sites (Table 2).
Group 1: Small arboreal quadrupeds
KNM-MV 4 is the smallest, and in some ways most primitive, specimen of the sample. It has qualitative resemblance to some platyrrhine specimens, including a wide and medially-tilted palmar portion of the Mc4 facet and a mediolaterally expanded and relatively planar Mc3 facet (see SI for detailed morphological descriptions), and is grouped among them by the taxonomic DFA (Fig. S8). The positional classifiers identify it as a palmigrade quadruped (Fig. 2, Tables 3 and S7), a finding corroborated by its estimated locomotor proportions (Tables 3 and S12). Among species recognised dentally at Mteitei Valley, its size comports with L. evansi and K. songhorensis. As these species are unknown postcranially, a more definitive attribution is not currently possible. Of species known from other Tinderet sites, this specimen is also compatible in size with L. legetet and, to a lesser extent, M. clarki and D. macinnesi, although it lacks derived morphology linking it with the latter species.
Discriminant scores based on nine shape variables best distinguishing extant positional classes. Points are coloured according to a priori class and shaped according to predicted class: circles, suspensory; triangles, knuckle-walking; squares, digitigrade; diamonds, palmigrade. Grey lines represent decision boundaries. See Tables S7 and S8 for model details and accuracy metrics.
Group 2: Small suspensors
KNM-SO 1000 and KNM-SO 1001 are estimated to have been substantially reliant on below-branch behaviours, with both grouped among extant suspensors by the positional classifiers (Fig. 2, Table S7). Suspensory estimates for KNM-SO 1000 approach those observed in Ateles (Tables 3 and S11). Estimates of arboreal proportions (QuadA and SuspA) align KNM-SO 1001 with Pongo, but total proportions (Quad and Susp) differ only slightly from those of E. heseloni (Table S12). Both specimens are relatively narrow, with low hamate facet concavity and small, radially-oriented centrale facets, features associated with suspension49. KNM-SO 1000 has additional, qualitative features linking it with suspensory taxa. Its relatively small and discontinuous Mc2 articulation may indicate hypertrophy of the lateral carpometacarpal ligament, hypothesised to aid suspension50, although in the current sample this trait was variably present in each of the anthropoid subfamilies, with no apparent functional correspondence (Table S6a). Qualitative observations have been used to argue that the size of the canal transmitting this ligament is the more diagnostic feature51, a conclusion not examined here. KNM-SO 1000’s hamate facet is also discontinuous (Fig. S6), a trait typical only in brachiators of the extant sample (Ateles and the hylobatids) and otherwise found only in a minority of Pongo and Nasalis (Table S6b). This feature may relate to hypertrophy of the capitohamate interosseous ligament, potentially stabilizing this joint against sudden load transmission gradients experienced during brachiation or other acrobatic arborealism.
KNM-SO 1000 and KNM-SO 1001 are only slightly larger than KNM-MV 4, and therefore potentially compatible with the same set of taxa, although they further exceed the estimated size range of M. clarki. The suspensory features of these specimens are consistent with interpretations of D. macinnesi4,10,11,35,36,37, but confident attribution to this taxon is precluded by the potential for similar adaptations in the yet-unknown postcrania of the other small-bodied catarrhines at Songhor. Furthermore, although a strong suspensory signal is identified in both KNM-SO 1000 and KNM-SO 1001, the discussed dissimilarities (see also Figs 3 and S7) raise the possibility that these specimens are not conspecific, which would suggest derived arborealism in one of the other small-bodied Songhor taxa. These specimens are therefore only tentatively referred to cf. D. macinnesi.
Group 3: Medium arboreal quadrupeds
KNM-CA 409, KNM-SO 31245, and KNM-SO 31246 are all reconstructed as mid-sized palmigrade quadrupeds resembling E. heseloni. The latter two specimens show particular affinity with E. heseloni in functional models (Figs 2 and 3, Table 3). KNM-CA 409 demonstrates less quantitative similarity with the Rusinga specimens (Figs 3b and S7) and its estimated locomotor proportions differ somewhat from those predicted for other medium-bodied quadrupeds, while its body mass estimate is lower than other group 3 specimens. However, it has the greatest qualitative resemblance to E. heseloni of the fossil sample, including the presence of the dorsodistal lip formerly exclusive to this taxon (Fig. S5), and it falls within the lower limit of the size range estimated for the medium-bodied taxa (Table 2).
The prevalence of R. gordoni relative to P. africanus at Songhor has been used to justify assigning mid-sized postcrania from this site to the former taxon30,32,42,52, but R. gordoni dental remains are only about twice as prevalent53, rendering this criterion uncompelling in the allocation of individual specimens. Given the discussed affinities of these specimens with E. heseloni and the close phylogenetic relationship of this taxon with P. africanus21, with which it was until recently thought to be congeneric54, we provisionally attribute the group 3 specimens to P. africanus.
Group 4: Medium derived morph
KNM-SO 1002 is only slightly larger than the group 3 specimens, but its morphology is distinct. While in some ways, particularly its articulations with the Mc2 and Mc3, it remains similar to those of most other early Miocene specimens including E. heseloni (see SI), it pairs these ancestral traits with derived ones, reinforcing the piecemeal nature of catarrhine functional evolution29,55,56. Its large, globular, and laterally-expanded head, highly-waisted neck, medially and laterally expanded body, moderately-expressed dorsal ridge, and concave hamate facet contribute to a profile in dorsal or palmar view that is uniquely great ape-like among early or middle Miocene capitates (Figs 1 and S6), an especially surprising finding given the early date. These observations are born out in classification and hierarchical models, which group this specimen with African apes (Figs 2, 3, S7 and S8; Table S7). It is estimated to have the highest overall reliance on quadrupedalism of the fossil sample, matching the estimated (but not observed) African ape values (Tables S11 and S12). Its suspensory estimates are lower than the other fossils, with proportions corresponding to the baseline values assigned to extant non-suspensors.
While the locomotor behaviour of this specimen was therefore likely distinct from the others of the fossil sample, characterization of these differences is difficult. A waisted neck and dorsal ridge have been interpreted to contribute to a functional midcarpal complex reflecting limited extension and enhanced transmission of loads generated during knuckle-walking57,58,59, but this interpretation has been challenged on the basis of the relative rarity of these and other reputed knuckle-walking traits in Gorilla, their inconsistent presence in Pan, and their variable presence in non-knuckle-walking taxa60,61,62,63,64. The condition of KNM-SO 1002, also present to a lesser extent in KNM-SO 31246, in which the distal extent of the centrale facet is positioned palmarly to a laterally-projecting portion of the body’s dorsal margin (Fig. S7), would intuitively reflect enhanced midcarpal stability at full extension during knuckle-walking. However, this condition appears in sampled Pongo specimens roughly as often as in Gorilla. In most Pan specimens, but only occasionally in Gorilla, an elaborated condition is found in which the entire dorsum of the body projects laterally, reorienting a sizable portion of the scaphoid facet palmarly and creating a sharply-angled margin that is often confluent with a raised dorsal ridge separating the head and body (Fig. S7, inset). The greater prevalence of this morphology in Pan may be explained, as other reputed knuckle-walking traits have been, as reflecting a greater degree of wrist extension during stance phase relative to Gorilla63,65,66,67. However, sampled hylobatids frequently possess morphology matching the most pronounced African ape examples of this trait (Fig. S7h). Therefore, in addition to its plausible association with enhanced stability at limited midcarpal extension during knuckle-walking, this morphology likely has additional utility, perhaps reflecting a need to prevent over-pronation of the midcarpal joint (or over-supination of the forearm below a fixed grasp) during vertical climbing or suspension. While the lower incidence of this morphology in Gorilla could reflect their lesser reliance on these behaviours in adulthood, it would not account for similar reliance on these behaviours in early subadults of the two genera68,69, nor the presumably increasing utility of this morphology with body size63.
Whether this morphology is related to multiple positional behaviours, facilitates some unrecognised kinematic affinity among hominoids, or arose from nonadaptive factors, these findings support the view that classical knuckle-walking traits are not exclusively associated with knuckle-walking, and highlight the continuing difficulty of inferring this behaviour from wrist morphology60,61,62,63,64. Deciphering the positional repertoire of KNM-SO 1002 will therefore require a detailed understanding of differential carpal biomechanics among extant apes. While the presence of a knuckle-walking ape in this early setting would evince the homoplastic evolvability of the behaviour70, a hypothesis that KNM-SO 1002 practiced knuckle-walking is not favoured here. This specimen nevertheless adds to a growing body of evidence supporting the presence of a derived ape at Songhor. Although it falls within the estimated size range of P. africanus, its derived morphology strongly suggests it is taxonomically distinct from the group 3 specimens. It is provisionally attributed to R. gordoni.
Early Miocene locomotor diversity and implications for hominoid evolution
The Tinderet sample occupies a greater proportion of the full-sample PLS shape-space (Fig. 3a) than do the cercopithecoids (6.41 vs. 5.58%; Table S13c), with the great ape value being even higher (8.93%). Because the shape-space structure (discussed in SI) is dominated by the extreme values of the hylobatids, the analysis was repeated with them excluded. In the resulting shape-space (Fig. 3b), the locomotor diversity estimate of the Tinderet capitates (17.18%) exceeds that of the great apes (12.96%) and cercopithecoids (15.43%), despite the extant groups being represented by a larger number of data points. While interpretation is complicated by the likely inclusion of both hominoids and stem catarrhines in the Tinderet sample, as well as the comparison of fossil individuals to extant taxon centroids, it suggests that the catarrhine clade had already undergone substantial functional diversification by 20 million years ago, resulting in the use of a broader range of positional repertoires than previously thought to characterise the clade’s early Miocene representatives.
Particularly intriguing among the range of forms documented in this study is that represented by KNM-SO 1002. Its morphology lends further support to the presence of a mid-sized catarrhine at Songhor with a behavioural repertoire more similar in some ways to that of extant great apes than to E. heseloni. The identity of this taxon cannot be determined with current evidence, but R. gordoni is currently the best candidate. Derived features have previously been identified among the nyanzapithecines11,71, of which R. gordoni is perhaps a basal member72,73,74, and this clade may be more closely related to crown hominoids than to Proconsul or Ekembo74,75,76, although there is not consensus on this point22. Implicit in the provisional allocation of KNM-SO 1002 to R. gordoni is the suggestion that the presence of derived, great ape-like features may be a useful criterion by which to distinguish the postcrania of R. gordoni and P. africanus. This hypothesis predicts that medium-sized postcranial specimens with similarly derived morphology should occur at Songhor and Kapurtay, but not at Koru, Chamtwara or Legetet, from which Rangwapithecus dental specimens have not been recovered.
Whether ape-like traits preserved at Songhor (and Moroto39) offer a glimpse of the ancestral crown hominoid morphotype or only of early examples of the evolutionary experimentation characterizing later hominoid evolution is difficult to address with current evidence. Homology cannot be ruled out, as although the mosaicism of later hominoid evolution has changed the calculus regarding the parsimony of extant ape homology3,45,77,78,79, the hierarchical nature of homoplasy supports the parsimony of some degree of homology in cases of derived morphology shared among extant ape lineages80. Furthermore, the time period represented at these Tinderet localities may coincide with the cladistic event separating hylobatids from great apes (mean: 20.19 Ma, median: 19.43 Ma81). The less derived anatomy of later African catarrhines decreases the likelihood of this explanation, however.
While the depositional and diagenetic environments of Miocene Tinderet sediments seem to have been unconducive to the preservation of associated crania and postcrania, continued work at these and other early Miocene sites leading to additional sets of overlapping postcranial elements like that presented here will be important in further characterizing functional diversity among early catarrhines. The morphological diversity of this sample offers a snapshot of differing lifestyles among a community of penecontemporaneous catarrhines and demonstrates a degree of functional diversity beyond what is generally accepted to have been present in early Miocene catarrhines. Although confident conclusions cannot be made based on a single skeletal element, the morphological diversity among these capitates raises the possibility of early hominoids and their contemporaries having diversified to fill multiple ecological niches early in their evolutionary career, well before the previously-inferred locomotor diversification of the later Miocene of Eurasia.
Materials and Methods
Shape data and body mass estimation
15 metrics characterizing articular surface area ratios, angles between regions of interest, and other aspects of shape (Table 1) were extracted from 3D models derived from μCT or laser scans of the Tinderet sample, four E. heseloni specimens, and a sample of 343 capitates from 28 extant taxa (detailed in SI; see ref.46 for further details and additional discussion. See refs82,83,84 for use of similar metrics). Body mass was estimated based on capitate volume, whose suitability for this purpose was evaluated via log-log ordinary least squares (OLS) regression against sex-specific body mass data gathered from museum records of sampled specimens when available, supplemented by published data85,86. Homogeneity of regression slopes between sexes and superfamilies was confirmed via analysis of covariance. Because sex-specific means were used to predict individual body masses, confidence intervals are not meaningful87. Prediction error was therefore characterised with residual standard error (0.147), percent mean prediction error (12.36%), and percent standard error of the estimate (%SEE; 16.15%) after 100 repetitions of 10-fold cross validation (CV).
Functional and systematic affinities
Extant taxa were assigned to one of four broad classes characterizing the dominant positional behaviour in each taxon’s repertoire: KW = knuckle-walking; S = orthograde climbing, clambering, suspension and/or brachiation; PG = arboreal palmigrade quadrupedalism; DG = terrestrial digitigrade quadrupedalism (Table S1; see also SI text). Two positional classifiers were built from the shape variables found to covary with extant positional classes using phylogenetic generalised linear mixed modelling88,89,90,91 (detailed in SI). Linear DFA was chosen for its interpretability and ease of visualization, while glmnet was chosen for being less prone to overfitting and bias due to collinearity, less stringent in its assumptions regarding heteroscedasticity, and its ability to detect non-linear relationships92,93. The accuracy of each classifier was calculated after 100 repetitions of 10-fold CV with random, non-stratified sampling, chosen for its favorable combination of variance reduction and low bias relative to other CV techniques94. Taxonomic classifiers were also built following the same methods (Fig. S8). Fossil systematic affinities were further explored via BIONJ95, a neighbour-joining hierarchical clustering algorithm (Fig. S9).
Locomotor behaviour in the extant sample was also characterised by continuous variables based on published observations available for 22 of 28 sampled taxa (Table S1a). These metrics represent the proportion of locomotor time spent in four different modes: Quad = quadrupedalism/tripedalism; Susp = orthograde suspension, including brachiation, forelimb swinging, orthograde clambering/transferring, and inverted walking/running (after ref.96); Climb = vertical climbing, quadrupedal climbing and scrambling, vertical descent, bridging, sliding, and swaying (after ref.97); Leap = leaping and dropping (see ref.98 for definitions and discussion of behavioural terms). Each mode is represented by an additional variable (e.g., SuspA) representing its use as a proportion of the taxon’s arboreal locomotion. A final locomotor variable, Arb, represents the proportion of locomotion occurring on arboreal rather than terrestrial substrates. Bipedal proportions were also compiled but are not included here due to an expected lack of morphological association. The total and arboreal proportions of some taxa therefore do not sum to 1.
Models to estimate locomotor proportions were built in a multi-step process. After eliminating shape variables found not to covary with the locomotor variable under consideration using PGLS with size as a covariate, subsets of the remaining shape variables were ranked by ascending second-order Akaike Information Criterion (AICc), a metric useful in balancing the opposing concerns of model accuracy and reduced generalisability due to overfitting99. The best three or four variable subsets, depending on similarity of delta-AICc values, were used to predict that locomotor proportion in the extant sample using quasibinomial logistic regression. The most accurate of these models, as judged by %SEE after 100 repetitions of 10-fold CV, was chosen for use in estimating that locomotor proportion in the fossil specimens. Climbing, leaping, and arboreality were found to have insufficient covariance with capitate morphology to produce accurate predictive models, so only those for the quadrupedal and suspensory proportions are reported.
Assessment of locomotor diversity
Two-block PLS analysis was used to create a two-dimensional shape space in which covariance with locomotor behaviour is maximised along each axis. The locomotor block consists of the four arboreal proportions along with the proportion of arboreality, a combination selected as having the strongest relationship with carpal morphology (detailed in ref.46). The fossil specimens and centroids of extant taxa for which locomotor proportions are unavailable were projected into PLS shape-space by multiplying their scaled shape variable matrix by the singular vectors of the PLS shape block. The functional diversity of the Tinderet sample was then estimated relative to that of extant groups by calculating the Euclidean area of the convex hull enveloping the constituent data points of each group as a proportion of PLS shape-space. See SI for additional analysis, results, and discussion.
References
Napier, J. R. Evolutionary aspects of primate locomotion. Am. J. Phys. Anthropol. 27, 333–342 (1967).
Fleagle, J. G. & Lieberman, D. E. Major transformations in the evolution of primate locomotion. In Great Transformations in Vertebrate Evolution (eds K. P. Dial, N. Shubin, & E. L. Brainerd) 257–278 (University of Chicago Press, 2015).
Ward, C. V. Postcranial and locomotor adaptations of hominoids. In Handbook of Paleoanthropology (eds W. Henke & I. Tattersall) 1363–1386 (Springer-Verlag, 2015).
Napier, J. R. & Davis, P. R. The forelimb skeleton and associated remains of Proconsul africanus. Fossil Mammals Afr. 16, 1–69 (1959).
Walker, A. C. & Pickford, M. New postcranial fossils of Proconsul africanus and Proconsul nyanzae. In New Interpretations of Ape and Human Ancestry (eds Russell L. Ciochon & Robert S. Corruccini) 325–351 (Plenum Press, 1983).
Walker, A., Teaford, M. F., Martin, L. & Andrews, P. A new species of Proconsul from the early Miocene of Rusinga/Mfangano Islands, Kenya. J. Hum. Evol. 25, 43–56 (1993).
Walker, A. Proconsul function and phylogeny. In Function, Phylogeny, and Fossils: Miocene Hominoid Evolution and Adaptations (eds David R. Begun, Carol V. Ward, & Michael D. Rose) 209–224 (Plenum Press, 1997).
McHenry, H. M. & Corruccini, R. S. The wrist of Proconsul africanus and the origins of hominoid postcranial adaptations. In New Interpretation of Ape and Human Ancestry (eds R. L. Ciochon & R. S. Corruccini) 353–367 (Plenum Press, 1983).
Cartmill, M. & Milton, K. The lorisiform wrist joint and the evolution of “brachiating” adaptations in the Hominoidea. Am. J. Phys. Anthropol. 47, 249–272 (1977).
Rose, M. D. Miocene hominoid postcranial morphology: monkey-like, ape-like, neither, or both? In New Interpretations of Ape and Human Ancestry (eds R. L. Ciochon & R. S. Corruccini) 405–417 (Plenum Press, 1983).
Rose, M. D. Locomotor anatomy of Miocene hominoids. In Postcranial Adaptation in Nonhuman Primates (ed. D. L. Gebo) 252–272 (Northern Illinois University Press, 1993).
Rose, M. D. Functional morphological similarities in the locomotor skeleton of Miocene catarrhines and platyrrhine monkeys. Folia Primatol. 66, 7–14 (1996).
Beard, K. C., Teaford, M. F. & Walker, A. New wrist bones of Proconsul africanus and P. nyanzae from Rusinga Island, Kenya. Folia Primatol. 47, 97–118 (1986).
Beard, K. C., Teaford, M. F. & Walker, A. New hand bones of the early Miocene hominoid Proconsul and their implications for the evolution of the hominoid wrist. In Hands of Primates (eds Preusehoft Holger & David J. Chivers) 387–403 (Springer, 1993).
Kelley, J. Paleobiological and phylogenetic significance of life history in Miocene hominoids. In Function, Phylogeny, and Fossils (eds David R. Begun, Carol V. Ward, & Michael D. Rose) 173–208 (Plenum Press, 1997).
Ward, C. V. Afropithecus, Proconsul, and the primitive hominoid skeleton. In Primate Locomotion: Recent Advances (eds E. Strasser, J. Fleagle, A. Rosenberger, & H. McHenry) 337–352 (Springer, 1998).
Daver, G. & Nakatsukasa, M. Proconsul heseloni distal radial and ulnar epiphyses from the Kaswanga Primate Site, Rusinga Island, Kenya. J. Hum. Evol. 80, 17–33 (2015).
Ruff, C. B. Long bone articular and diaphyseal structure in old world monkeys and apes. I: locomotor effects. Am. J. Phys. Anthropol. 119, 305–342 (2002).
Ryan, T. M., Silcox, M. T. & Walker, A. Evolution of locomotion in Anthropoidea: the semicircular canal evidence. Proc. R. Soc. Lond. B. 279, 3467–3475 (2012).
Rose, M. D. Quadrupedalism in some Miocene catarrhines. J. Hum. Evol. 26, 387–411 (1994).
Harrison, T. Dendropithecoidea, Proconsuloidea, and Hominoidea. In Cenozoic Mammals of Africa (eds L. Werdelin & W. J. Sanders) 429–469 (University of California Press, 2010).
Begun, D. R. Fossil record of Miocene hominoids. In Handbook of Paleoanthropology (eds W. Henke & I. Tattersall) 1261–1332 (Springer-Verlag, 2015).
McCrossin, M. L., Benefit, B. R., Gitau, S. N., Palmer, A. K. & Blue, K. T. Fossil evidence for the origins of terrestriality among Old World higher primates. In Primate Locomotion: Recent Advances (eds E. Strasser, J. Fleagle, A. Rosenberger, & H. McHenry) 353–396 (Plenum Press, 1998).
Ward, S., Brown, B., Hill, A., Kelley, J. & Downs, W. Equatorius: a new hominoid genus from the Middle Miocene of Kenya. Science 285, 1382–1386 (1999).
Sherwood, R. J. et al. Preliminary description of the Equatorius africanus partial skeleton (KNM-TH 28860) from Kipsaramon, Tugen Hills, Baringo District, Kenya. J. Hum. Evol. 42, 63–73 (2002).
Rose, M. D., Nakano, Y. & Ishida, H. Kenyapithecus postcranial specimens from Nachola, Kenya. African Study Monographs Supplement 24, 3–56 (1996).
Ishida, H., Kunimatsu, Y., Takano, T., Nakano, Y. & Nakatsukasa, M. Nacholapithecus skeleton from the Middle Miocene of Kenya. J. Hum. Evol. 46, 69–103 (2004).
Rose, M. D. Hominoid postcranial specimens from the middle Miocene Chinji formation, Pakistan. J. Hum. Evol. 13, 503–516 (1984).
Moya-Sola, S., Kohler, M., Alba, D. M., Casanovas-Vilar, I. & Galindo, J. Pierolapithecus catalaunicus, a new Middle Miocene great ape from Spain. Science 306, 1339–1344 (2004).
Harrison, T. Small-bodied Apes from the Miocene of East Africa Ph.D. Dissertation, University College London (1982).
Deane, A. S. & Begun, D. R. Broken fingers: retesting locomotor hypotheses for fossil hominoids using fragmentary proximal phalanges and high-resolution polynomial curve fitting (HR-PCF). J. Hum. Evol. 55, 691–701 (2008).
Patel, B. A., Yapuncich, G. S., Tran, C. & Nengo, I. O. Catarrhine hallucal metatarsals from the early Miocene site of Songhor, Kenya. J. Hum. Evol. 108, 176–198 (2017).
Langdon, J. H. A Comparative Functional Study of the Miocene Hominoid Foot Remains Ph.D. Dissertation, Yale University (1984).
Rose, M. D., Leakey, M. G., Leakey, R. E. F. & Walker, A. C. Postcranial specimens of Simiolus enjiessi and other primitive catarrhines from the early Miocene of Lake Turkana, Kenya. J. Hum. Evol. 22, 171–237 (1992).
Preuschoft, H. Body posture and locomotion in some East African Miocene Dryopithecinae. In Human Evolution (ed. M. H. Day) 13–46 (Taylor & Francis Ltd, 1973).
Simons, E. L. & Fleagle, J. G. The history of extinct gibbon-like primates. In Gibbon and Siamang, Vol. 2: Anatomy, Dentition, Taxonomy, Molecular Evolution and Behavior (ed. D. M. Rumbaugh) 121–148 (Karger, 1973).
Fleagle, J. G. Locomotor adaptations of Oligocene and Miocene hominoids and their phyletic implications. In New Interpretations of Ape and Human Ancestry (eds R. L. Ciochon & R. S. Corruccini) 301–324 (Plenum Press, 1983).
Sanders, W. J. & Bodenbender, B. E. Morphometric analysis of lumbar vertebra UMP 67-28: implications for spinal function and phylogeny of the Miocene Moroto hominoid. J. Hum. Evol. 26, 203–237 (1994).
Gebo, D. L. et al. A hominoid genus from the early Miocene of Uganda. Science 276, 401–404 (1997).
MacLatchy, L., Gebo, D., Kityo, R. & Pilbeam, D. Postcranial functional morphology of Morotopithecus bishopi, with implications for the evolution of modern ape locomotion. J. Hum. Evol. 39, 159–183 (2000).
Maclatchy, L. The oldest ape. Evol. Anthropol. 13, 90–103 (2004).
Nengo, I. O. & Rae, T. C. New hominoid fossils from the early Miocene site of Songhor, Kenya. J. Hum. Evol. 23, 423–429 (1992).
Bishop, W. W., Miller, J. A. & Fitch, F. J. New potassium-argon age determinations relevant to the Miocene fossil mammal sequence in East Africa. Am. J. Sci. 267, 669–699 (1969).
Cote, S., McNulty, K. P., Stevens, N. J. & Nengo, I. O. A detailed assessment of the maxillary morphology of Limnopithecus evansi with implications for the taxonomy of the genus. J. Hum. Evol. 94, 83–91 (2016).
Casanovas-Vilar, I., Alba, D. M., Garces, M., Robles, J. M. & Moya-Sola, S. Updated chronology for the Miocene hominoid radiation in Western Eurasia. Proc. Natl. Acad. Sci. 108, 5554–5559 (2011).
Wuthrich, C. Computational Relationships among Form, Function, and Phylogeny in the Catarrhine Ulnar Carpus, and the Evolutionary History of Ape and Human Locomotion Ph.D. Dissertation, University of Michigan (2017).
Friedman, J., Hastie, T. & Tibshirani, R. Regularization paths for generalized linear models via coordinate descent. J. Stat. Softw. 33, 1–22 (2010).
Rohlf, F. J. & Corti, M. Use of two-block partial least-squares to study covariation in shape. Syst. Biol. 49, 740–753 (2000).
Kivell, T. L. The primate wrist. In The Evolution of the Primate Hand (eds T. L. Kivell, P. Lemelin, B. G. Richmond, & D. Schmitt) 17–54 (Springer, 2016).
Lovejoy, C. O., Simpson, S. W., White, T. D., Asfaw, B. & Suwa, G. Careful climbing in the Miocene: The forelimbs of Ardipithecus ramidus and humans are primitive. Science 326(70), e71–78 (2009).
Selby, M. S., Simpson, S. W. & Lovejoy, C. O. The functional anatomy of the carpometacarpal complex in anthropoids and its implications for the evolution of the hominoid hand. Anat. Rec. 299, 583–600 (2016).
Gebo, D. L., Malit, N. R. & Nengo, I. O. New proconsuloid postcranials from the early Miocene of Kenya. Primates 50, 311–319 (2009).
Cote, S. Sampling and Ecology in Three Early Miocene Catarrhine Assemblages from East Africa Ph.D. Dissertation, Harvard (2008).
McNulty, K. P., Begun, D. R., Kelley, J., Manthi, F. K. & Mbua, E. N. A systematic revision of Proconsul with the description of a new genus of early Miocene hominoid. J. Hum. Evol. 84, 42–61 (2015).
Almecija, S., Alba, D. M., Moya-Sola, S. & Kohler, M. Orang-like manual adaptations in the fossil hominoid Hispanopithecus laietanus: first steps towards great ape suspensory behaviours. Proc. R. Soc. Lond. B. 274, 2375–2384 (2007).
Kivell, T. L., Kibii, J. M., Churchill, S. E., Schmid, P. & Berger, L. R. Australopithecus sediba hand demonstrates mosaic evolution of locomotor and manipulative abilities. Science 333, 1411–1417 (2011).
Corruccini, R. S. Comparative osteometrics of the hominoid wrist joint, with special reference to knuckle-walking. J. Hum. Evol. 7, 307–321 (1978).
Richmond, B. G., Begun, D. R. & Strait, D. S. Origin of human bipedalism: The knuckle-walking hypothesis revisited. Am. J. Phys. Anthropol. 33(Suppl), 70–105 (2001).
Begun, D. R. Knuckle-walking and the origin of human bipedalism. In From Biped to Strider: The Emergence of Modern Human Walking, Running and Resource Transport (eds D. J. Meldrum & Charles E. Hilton) 9–33 (Kluwer Academic/Plenum Publishers, 2004).
Shea, B. T. & Inouye, S. E. Knuckle-walking ancestors. Science 259, 293–294 (1993).
McCrossin, M. L. & Benefit, B. R. On the relationships and adaptations of Kenyapithecus, a large-bodied hominoid from the Middle Miocene of Eastern Africa. In Function, Phylogeny, and Fossils: Miocene Hominoid Evolution and Adaptations (eds David R. Begun, Carol V. Ward, & Michael D. Rose) 241–267 (Plenum Press, 1997).
Richmond, B. G. Functional morphology of the midcarpal joint in knuckle-walkers and terrestrial quadrupeds. In Human Origins and Environmental Backgrounds (eds H. Ishida et al.)105–122 (Springer, 2006).
Kivell, T. L. & Schmitt, D. Independent evolution of knuckle-walking in African apes shows that humans did not evolve from a knuckle-walking ancestor. Proc. Natl. Acad. Sci. 106, 14241–14246 (2009).
Orr, C. M. Adaptations to Knuckle-walking and Digitigrady: a Three-dimensional Kinematic and Morphometric Analysis of the Anthropoid Wrist Ph.D. Dissertation, Arizona State University (2010).
Inouye, S. E. Ontogeny and allometry of African ape manual rays. J. Hum. Evol. 23, 107–138 (1992).
Inouye, S. E. Ontogeny of knuckle-walking hand postures in African apes. J. Hum. Evol. 26, 459–485 (1994).
Inouye, S. E. & Shea, B. T. The implications of variation in knuckle-walking features for models of African hominoid locomotor evolution. J. Anthropol. Sci. 82, 67–88 (2004).
Doran, D. M. Ontogeny of locomotion in mountain gorillas and chimpanzees. J. Hum. Evol. 32, 323–344 (1997).
Sarringhaus, L. A., MacLatchy, L. M. & Mitani, J. C. Locomotor and postural development of wild chimpanzees. J. Hum. Evol. 66, 29–38 (2014).
Begun, D. R. & Kivell, T. L. Knuckle-walking in Sivapithecus? The combined effects of homology and homoplasy with possible implications for pongine dispersals. J. Hum. Evol. 60, 158–170 (2011).
McCrossin, M. L. An oreopithecid proximal humerus from the middle Miocene of Maboko Island, Kenya. Int. J. Primatol. 13, 659–677 (1992).
Rossie, J. B. & MacLatchy, L. A new pliopithecoid genus from the early Miocene of Uganda. J. Hum. Evol. 50, 568–586 (2006).
Hill, A., Nengo, I. O. & Rossie, J. B. A Rangwapithecus gordoni mandible from the early Miocene site of Songhor, Kenya. J. Hum. Evol. 65, 490–500 (2013).
Nengo, I. et al. New infant cranium from the African Miocene sheds light on ape evolution. Nature 548, 169–174 (2017).
McNulty, K. Geometric Morphometric Analyses of Extant and Fossil Hominoid Craniofacial Morphology Ph.D. Dissertation, City University of New York (2003).
Stevens, N. J. et al. Palaeontological evidence for an Oligocene divergence between Old World monkeys and apes. Nature 497, 611–614 (2013).
Begun, D. R. New catarrhine phalanges from Rudabanya (Northeastern Hungary) and the problem of parallelism and convergence in hominoid postcranial morphology. J. Hum. Evol. 24, 373–402 (1993).
Almecija, S., Alba, D. M. & Moya-Sola, S. Pierolapithecus and the functional morphology of Miocene ape hand phalanges: paleobiological and evolutionary implications. J. Hum. Evol. 57, 284–297 (2009).
Morgan, M. E. et al. A partial hominoid innominate from the Miocene of Pakistan: Description and preliminary analyses. Proc. Natl. Acad. Sci. 112, 82–87 (2015).
Begun, D. R. How to identify (as opposed to define) a homoplasy: examples from fossil and living great apes. J. Hum. Evol. 52, 559–572 (2007).
Hedges, S. B., Marin, J., Suleski, M., Paymer, M. & Kumar, S. Tree of life reveals clock-like speciation and diversification. Mol. Biol. Evol. 32, 835–845 (2015).
Tocheri, M. W. et al. The primitive wrist of Homo floresiensis and its implications for hominin evolution. Science 317, 1743–1745 (2007).
Tocheri, M. W. et al. Functional capabilities of modern and fossil hominid hands: three-dimensional analysis of trapezia. Am. J. Phys. Anthropol. 122, 101–112 (2003).
Tocheri, M. W., Razdan, A., Williams, R. C. & Marzke, M. W. A 3D quantitative comparison of trapezium and trapezoid relative articular and nonarticular surface areas in modern humans and great apes. J. Hum. Evol. 49, 570–586 (2005).
Smith, R. J. & Jungers, W. L. Body mass in comparative primatology. J. Hum. Evol. 32, 523–559 (1997).
Delson, E. et al. Body mass in Cercopithecidae (Primates, Mammalia): Estimation and scaling in extinct and extant taxa. Anthropol. Pap. Am. Mus. Nat. Hist. 83 (2000).
Smith, R. J. The present as a key to the past: body weight of Miocene hominoids as a test of allometric methods for paleontological inference. In Size and Scaling in Primate Biology (ed. W. L. Jungers) 437–448 (Plenum Press, 1985).
Hadfield, J. D. & Nakagawa, S. General quantitative genetic methods for comparative biology: phylogenies, taxonomies and multi-trait models for continuous and categorical characters. J. Evol. Biol. 23, 494–508 (2010).
Hadfield, J. D. MCMC methods for multi-response generalized linear mixed models: The MCMCglmm R package. J. Stat. Softw. 33, 1–22 (2010).
Carter, K. E. & Worthington, S. The evolution of anthropoid molar proportions. BMC Evol. Biol. 16, 110 (2016).
Gelman, A. Prior distributions for variance parameters in hierarchical models (comment on article by Browne and Draper). Bayesian Analysis 1, 515–534 (2006).
Kovarovic, K., Aiello, L. C., Cardini, A. & Lockwood, C. A. Discriminant function analyses in archaeology: Are classification rates too good to be true? J. Archaeol. Sci. 38, 3006–3018 (2011).
Mitteroecker, P. & Bookstein, F. Linear discrimination, ordination, and the visualization of selection gradients in modern morphometrics. Evol. Biol. 38, 100–114 (2011).
Kuhn, M. & Johnson, K. Applied Predictive Modeling. (Springer, 2013).
Gascuel, O. BIONJ: An improved version of the NJ algorithm based on a simple model of sequence data. Mol. Biol. Evol. 14, 685–695 (1997).
Thorpe, S. K. & Crompton, R. H. Orangutan positional behavior and the nature of arboreal locomotion in Hominoidea. Am. J. Phys. Anthropol. 131, 384–401 (2006).
Doran, D. M. Comparative positional behavior of the African apes. in Great Ape Societies (eds W. McGrew, L. Marchant, & T. Nishida) 213–224 (Cambridge University Press, 1996).
Hunt, K. D. et al. Standardized descriptions of primate locomotion and postural modes. Primates 37, 363–387 (1996).
Burnham, K. P. & Anderson, D. R. Model Selection and Inference: A Practical Information-Theoretical Approach, 2nd Ed., (Springer-Verlag, 2002).
Rafferty, K. L., Walker, A., Ruff, C. B., Rose, M. D. & Andrews, P. J. Postcranial estimates of body weight in Proconsul, with a note on a distal tibia of P. major from Napak, Uganda. Am. J. Phys. Anthropol. 97, 391–402 (1995).
Andrews, P. J. A revision of the Miocene Hominoidea of East Africa. Bull. Br. Mus. Nat. Hist. 30, 85–224 (1978).
Harrison, T. Catarrhine origins. In A Companion to Paleoanthropology (ed. David R. Begun) 376–396 (Blackwell Publishing Ltd., 2013).
Bilsborough, A. & Rae, T. C. Hominoid cranial diversity and adaptation. In Handbook of Paleoanthropology (ed. Winfried Henke) 1031–1105 (Springer, 2007).
Fleagle, J. G. & Simons, E. L. Micropithecus clarki, a small ape from the Miocene of Uganda. Am. J. Phys. Anthropol. 49, 427–440 (1978).
Ruff, C. B. Long bone articular and diaphyseal structure in Old World monkeys and apes. II: Estimation of body mass. Am. J. Phys. Anthropol. 120, 16–37 (2003).
Acknowledgements
Thanks to Mark Omura and Judy Chupasko at the Museum of Comparative Zoology at Harvard University, Eileen Westwig at the American Museum of Natural History, Lyman Jelema at the Cleveland Museum of Natural History, Darrin Lunde at the Smithsonian National Museum of Natural History, and Stephen Hinshaw and Cody Thompson at the University of Michigan Museum of Zoology for access to extant specimens in their care. For access to fossil specimens and casts thereof, we thank Emma Mbua, Fredrick Manthi, and Blasto Onyango at the National Museums of Kenya. Thanks to Caley Orr and Matt Tocheri for laser scans of KNM-SO 1002. This manuscript was improved by helpful comments from Susanne Cote, Caley Orr, Biren Patel, Craig Byron, John Kingston, Bill Sanders, and Jacinta Beehner. Support was provided by the Leakey Foundation, the Boise Fund, the Foothill-De Anza Foundation, and the Fulbright Program. This manuscript is publication #9 supporting Research on East African Catarrhine and Hominoid Evolution (REACHE).
Author information
Authors and Affiliations
Contributions
C.W. designed the research and collected and analysed the data. L.M.M. and I.O.N. supervised the study, provided feedback, and shaped the research. All authors contributed to the final manuscript. An earlier iteration of this work was included in the Ph.D. thesis of C.W.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wuthrich, C., MacLatchy, L.M. & Nengo, I.O. Wrist morphology reveals substantial locomotor diversity among early catarrhines: an analysis of capitates from the early Miocene of Tinderet (Kenya). Sci Rep 9, 3728 (2019). https://doi.org/10.1038/s41598-019-39800-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-39800-3
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.