Abstract
Typically, multisensory illusion paradigms emphasise the importance of synchronous visuotactile integration to induce subjective embodiment towards another body. However, the extent to which embodiment is due to the ‘visual capture’ of congruent visuoproprioceptive information alone remains unclear. Thus, across two experiments (total N = 80), we investigated how mere visual observation of a mannequin body, viewed from a first-person perspective, influenced subjective embodiment independently from concomitant visuotactile integration. Moreover, we investigated whether slow, affective touch on participants’ own, unseen body (without concomitant touch on the seen mannequin) disrupted visual capture effects to a greater degree than fast, non-affective touch. In total, 40% of participants experienced subjective embodiment towards the mannequin body following mere visual observation, and this effect was significantly higher than conditions which included touch to participants own, unseen body. The velocity of the touch that participants received (affective/non-affective) did not differ in modulating visual capture effects. Furthermore, the effects of visual capture and perceived pleasantness of touch was not modulated by subthreshold eating disorder psychopathology. Overall, this study suggests that congruent visuoproprioceptive cues can be sufficient to induce subjective embodiment of a whole body, in the absence of visuotactile integration and beyond mere confabulatory responses.
Similar content being viewed by others
Introduction
Body ownership, the feeling that our body belongs to us and is distinct from other people’s bodies, is a fundamental component of our sense of self1,2. Intuitively, this feeling appears stable and durable amongst humans, yet scientific studies have demonstrated that the sense of body ownership is a fragile outcome of integrating multiple sensory signals. Such signals originate via exteroceptive modalities (i.e. outside the body) such as vision and touch3,4, specifically within the boundaries of peripersonal space surrounding the body5,6. Additionally, incoming signals emerge via interoceptive modalities (i.e. within the body) such as heart rate, and via proprioceptive pathways7,8,9. Together, exteroceptive and interoceptive sensory signals are integrated to create a coherent sense of body ownership through which we interact with our environment2.
Experimental paradigms have been successfully used to investigate how body ownership is shaped by the integration of incoming multisensory information. For example, in the Rubber Hand Illusion (RHI)10, individuals experience ownership over a fake (rubber) hand when placed in a congruent anatomical position and stroked in temporal synchrony with their own hand, which is hidden from view. This has been recently extended to ownership over an entire body (Full Body Illusion), of which different variations exist. Participants can perceive a change in self-location which induces an illusory experience of being in a position outside of their physical body11, or an illusory ownership towards another’s body from a third-person perspective12 or first-person perspective13,14. In such illusions, the source of tactile stimulation on one’s own, unseen body (part) is attributed to the location of the visually perceived fake body (part) when the two are stroked synchronously, which is argued to give rise to subjective self-reports of illusory body ownership and a mislocation in one’s own sense of body position (i.e. proprioceptive drift)4. Importantly, such effects typically occur within the constraints of top-down contextual factors, including the orientation3,15, visual perspective16,17,18, and appearance13,19,20 of the embodied body (part). Indeed, research has shown that the strength of the illusion is modulated by the distance between the real and fake body (part), with greater spatial discrepancies decreasing the likelihood of integration between visuoproprioceptive signals21,22,23.
Importantly, it has long been argued that the synchrony of the perceived touch with vision is a necessary condition for illusory ownership to occur, rather than asynchrony which is typically used as a control condition within multisensory illusion paradigms15. However, the role of synchronous visuotactile integration as a necessary component to trigger illusory embodiment remains debated24,25. Research has shown that illusory embodiment could still be induced based purely on visual information of a fake body (part) in the absence of visuotactile stimulation25,26,27, or based on merely expected but not experienced synchronous tactile stimulation28, and even following asynchronous visuo-tactile stimulation, provided that spatial congruence is adhered to between the real and fake body (part)29 (see24 for review). Such evidence highlights that synchronous visuotactile input can strengthen illusory embodiment, by contributing to the downregulation in the weighting of proprioceptive signals regarding one’s own limb position in relation to vision30. However, from a computational perspective, congruent visuoproprioceptive cues may be sufficient to induce such embodiment, suggesting that subsequent visuotactile input may not be a necessary component to trigger this process25,26,31 (see32 for review).
Studies which have investigated illusory body ownership in the absence of tactile stimulation have predominantly investigated this effect during the RHI (e.g.25,33,34), with little research conducted towards a whole body16. Among the latter, some have argued that synchronous visuotactile integration is a necessary condition to elicit illusory ownership in the full body illusion13, while studies using virtual reality have found evidence to the contrary, following illusory ownership towards a virtual body in the absence of visuotactile integration14,16. Therefore, in the present study we wished to investigate whether subjective visual capture of embodiment could occur towards a real mannequin body with a static field of view, from a first-person visual perspective in the ‘physical world’. In this context, ‘visual capture’ is defined as the degree of embodiment due solely to passive, visual perception of the fake body (part) viewed from a first-person perspective, independently from tactile stimulation (hereafter referred to as ‘visual capture of embodiment’)35,36.
Interestingly, a tendency to weight visual information over other somatosensory signals has been recently observed in neuropsychological, right hemisphere patients with body representation deficits (e.g.36,37,38,39). Moreover, alterations in the weighting and integration of sensory information has been implicated within neurodevelopmental disorders such as autism40,41, and psychiatric disorders such as schizophrenia42,43,44, and eating disorders45,46. Importantly, such alterations are argued to reflect an instability in the bodily self within these populations47,48. However, whilst ‘pure’ visual capture conditions have been tested in right hemisphere patients, evidence for heightened visual dominance within eating disorder patients derives from multisensory illusion studies finding that both synchronous and asynchronous visuotactile stimulation led to alterations in an individual’s body representation45,46,49,50. Thus, direct investigation of visual capture of embodiment from congruent visuoproprioceptive cues alone (i.e. in the absence of tactile stimulation) has been less studied with regard to eating disorder psychopathology.
Importantly, greater illusory embodiment in acute eating disorder patients has been shown to persist to some degree amongst recovered patients, suggesting that such heightened sensitivity to visual information pertaining to the body may be a trait phenomenon49. Therefore, such visual dominance over other sensory information may be independent from a status of malnutrition, and may occur prior to illness onset which could influence an individual’s body perception and body satisfaction51,52,53. Thus, it may be that healthy individuals who display an increased visual capture of embodiment towards a fake body (part) show an increased visual dominance over other sensory information, which may link with a greater risk of developing distortions in one’s body image. Consequently, the present study aimed to investigate whether subthreshold eating disorder psychopathology and body concerns may modulate the subjective embodiment shown towards a fake body as a result of mere visual capture.
In addition to research investigating visuoproprioceptive integration, the importance of interoception in multisensory integration and body ownership has only recently been investigated9,54,55. Interoception refers to information about the internal states of the body, processing sensations from within the body (e.g. hunger, thirst, heart rate), but also outside the body (e.g. itch, pain, pleasure from touch), which is conveyed by a particular afferent pathway8. Affective touch - i.e. slow, caress-like touch – is associated with increased pleasantness and has been found to activate specific C-Tactile (CT) afferents found only in the hairy skin, responding maximally to stroking velocities between 1 and 10 cm/sec.56. Importantly, affective tactile stimulation appears to be dissociable from exteroceptive, discriminatory stimulation such as non-affective touch57. Such CT afferents are hypothesised to take a distinct pathway to the posterior insular cortex58,59, an area associated with the early convergence of interoceptive information with exteroceptive bodily signals8,60,61.
Increasing evidence has shown that the velocity of perceived touch during visuotactile integration plays an influential role within the sense of body ownership. Specifically, touch delivered at CT-optimal velocities has been shown to increase embodiment during the RHI paradigm compared with fast, non-affective touch35,62,63,64, however, evidence of this effect in the full body illusion remains equivocal65. Moreover, recent research has shown that individuals with anorexia nervosa (AN) display a reduced subjective pleasantness to touch, relative to healthy controls60; however, it is yet to be investigated how eating disorder psychopathology may modulate the extent to which individuals show alterations in their experience of touch, or vice versa. Therefore, within our second experiment, individual differences in the perception of touch will be investigated in relation to subthreshold eating disorder psychopathology.
In addition to enhancement of embodiment via interoceptive signals, evidence from patient populations with chronic pain has shown how feelings of body ownership can be disturbed66,67 (but see68 for review). Changes in interoceptive information (e.g. increased limb temperature) has been shown to disrupt the feelings of embodiment by decreasing the strength of the effect within multisensory illusions69. Therefore, in addition to mere visual capture towards subjective embodiment (visual capture condition), the present study aimed to investigate the effects of tactile stimulation administered to participants’ own, unseen arm during visual observation of the mannequin body, as a control condition designed to ‘disrupt’ visual capture by introducing sensory information that is incongruent with participants’ visual input (tactile disruption condition). Furthermore, we aimed to investigate whether CT-optimal, affective touch (i.e. touch administered in CT-optimal velocities) would provide additional interoceptive information on one’s own body which would be expected to disrupt visual capture of embodiment to a greater extent compared with discriminatory, non-affective touch. Previous research has suggested that the perception of interoceptive signals depends on an individual’s ability to regulate the balance between interoceptive and exteroceptive information in ambiguous contexts9,35,70. Thus, differences in an individual’s sensitivity and balance between these two streams of information may determine the degree of embodiment change shown during tactile disruption conditions.
In brief, we investigated whether mere visual observation of a mannequin body would lead to subjective embodiment when visuoproprioceptive cues are congruent with one’s own body. Based on previous research16,25, we predicted that a compatible first-person perspective of a mannequin body would be sufficient to elicit subjective embodiment amongst participants, independent of concomitant tactile stimulation. In addition, we investigated the extent to which subjective embodiment towards the mannequin body was reduced when visual capture of proprioceptive cues are disrupted by tactile stimulation to participant’s own, unseen arm. We manipulated the velocity of tactile stimulation that participants received, to investigate whether slow, affective touch had a differential effect on the disruption of embodiment compared with fast, non-affective touch. Specifically, we predicted that the increased interoceptive information associated with affective touch would disrupt the downregulation of proprioceptive signals by visual capture to a greater extent compared to non-affective touch. Finally, we investigated whether subthreshold eating disorder psychopathology modulated any individual differences in subjective embodiment from visual capture. We hypothesised that higher eating disorder vulnerability would be associated with an increased weighting of visual information, and thus increased visual capture of embodiment. The above measures were replicated across two experiments, with the addition of a separate touch task in Experiment 2, designed to investigate the role of subjective pleasantness of touch in relation to subthreshold eating disorder psychopathology. Extending upon findings from clinical populations60, we expected to observe a negative relationship between the above two measures, such that individuals with higher eating disorder psychopathology were hypothesised to display a reduced pleasantness to both affective touch and non-affective touch.
Methods
Experiment 1
Participants
Forty-one healthy female participants (mean age = 20.10, SD ± 2.48, range = 18–31) were recruited via the University of York research participation scheme and received course credit for a single 60-minute testing session. Participants had a mean BMI of 21.48 (SD ± 2.40, range = 18.30–28.60), no current or previous neurological or psychological disorders (self-report), and normal or corrected-to-normal vision. Exclusion criteria included any specific skin conditions (e.g. eczema, psoriasis) or any scarring or tattoos on the left arm. All participants gave informed consent to take part in the study. The study received ethical approval from the University of York Departmental Ethics Committee and was conducted in accordance with the Declaration of Helsinki. One participant was later excluded following a self-reported previous psychological condition, therefore, the final sample consisted of forty participants (mean age = 20.15, SD ± 2.49, range = 18–31). Post-hoc power analyses using G*Power 3.1.9.271 indicated that this number of participants resulted in 99% and 97% power to obtain significant effects following visual capture and tactile disruption conditions, respectively.
Design
The experiment employed a within-subjects design to investigate the effects of visual and tactile signals towards the subjective embodiment of a mannequin body. First, during visual capture trials participants visually observed the mannequin body for 30 seconds, from a first-person perspective, independent of any tactile stimulation. Second, participants also undertook trials identical to the visual capture condition, but with the addition of tactile stimulation applied (only) to participant’s own, unseen arm, designed to disrupt such visual capture (tactile disruption condition) for 60 seconds. In the context of the RHI, previous research has shown that 15 seconds is sufficient to elicit visual capture in some participants, as a two-way sensory integration between vision and proprioception34,36. Furthermore, 60 seconds has been shown to be sufficient to induce changes in measures of body ownership in classic RHI and full body illusions involving synchronous touch – i.e. a three-way sensory integration between vision, proprioception, and touch51,62. Therefore, owing to the additional use of head-mounted displays in the present study, we allowed participants 30 seconds in the two-way sensory integration ‘visual capture’ conditions and 60 seconds in the three-way ‘tactile disruption’ conditions.
Stimulation was administered at two different velocities to give rise to affective (3 cm/s) and non-affective (18 cm/s) tactile disruption. The dependent variable was the subjective embodiment experienced by participants, rated after each trial via an embodiment questionnaire (see Measures section and Table 1 for details). The same embodiment questionnaire was completed for both visual capture and tactile disruption conditions. Participants completed two visual capture trials, each followed by an affective or non-affective tactile disruption trial in counterbalanced order between participants, resulting in a total of 4 trials per participant (see Figure 1).
Timeline of experimental procedure. Participants completed two visual capture (30 secs) conditions and two tactile disruption (60 secs) conditions (1x affective touch; 1x non-affective touch). Tactile disruption order was counterbalanced across participants. Participants removed the HMDs following each trial and completed the Embodiment Questionnaire on a separate computer.
Measures
Embodiment Questionnaire: Following each trial, participants rated their subjective embodiment via an embodiment questionnaire (see Table 1) along a 7-point Likert scale (−3 strongly disagree to +3 strongly agree). This questionnaire (adapted from Longo et al., 200872) was composed of two subcomponents: ownership (i.e. the feeling that the mannequin body belongs to them) and location (i.e. the feeling that the mannequin body was in the position of their own body). An overall embodiment score was calculated by averaging the above two subcomponent scores. The final two statements were control statements, in which an overall control score was similarly calculated by averaging across the two control items. These scores served to control for task compliance, suggestibility, and confabulation within the visual capture condition to compare with embodiment scores. Such control statements are similar, body-related items to those of the embodiment statements, but importantly do not capture the phenomenological experience of embodiment. Consequently, comparisons between embodiment and control scores acted to indicate whether a significant sense of embodiment occurred following the visual capture condition.
Eating Disorder Examination Questionnaire (EDE-Q): The EDE-Q is a 28-item questionnaire used as a self-report measure of eating disorder psychopathology73 amongst community and clinical populations. The questionnaire assesses disordered eating behaviours and attitudes (22 items) within the past 28 days, along four subscales: Restraint, Eating Concern, Weight Concern and Shape Concern, which are also averaged for a Global EDE-Q Score. Items are rated along a 7-point (0–6) Likert scale, with higher scores signifying greater eating disorder psychopathology. This measure has good internal consistency, with Cronbach’s alpha ranging from 0.78 to 0.93 in a non-clinical sample74. The overall global EDE-Q measure in the present study had a Cronbach’s alpha of 0.95 in both Experiment 1 and Experiment 2.
Materials
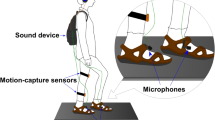
A life-size female mannequin was used within the experimental set-up. The mannequin was dressed in a white t-shirt, blue jeans, and black socks, with the head removed at the neckline to enable correct positioning of the video cameras. The body had a waist circumference of 62 cm and was in a standing position with arms placed by their side (see Figure 2). During all trials, participants wore a set of head-mounted displays (HMDs) (Oculus Rift DK2, Oculus VR, Irvine, CA, USA), with a resolution of 1200 × 1080 pixels per eye, a refresh rate of 75 Hz, and a corresponding nominal visual field of 100°. The HMDs were connected to a stereoscopic camera (Ovrvision Pro USB 3.0 VR stereo camera, Japan), presenting a real-time, video image to participants. The cameras were mounted and positioned downwards, at the eyeline of the mannequin, capturing a first-person perspective of the body, compatible with looking down towards one’s own body. During tactile disruption trials, tactile stimulation was applied using a cosmetic make-up brush (Natural hair Blush Brush, N◦7, The Boots Company). All experimental trials and responses were completed using PsychoPy 275 on an Apple iMac desktop computer (1.6 GHz dual-core Intel Core i5 processor).
Experimental Procedure
Prior to the experiment, two adjacent 9 cm × 4 cm stroking areas were marked on the hairy skin of each participants’ left forearm, using a washable marker pen62,76. This provided a specific area for which to administer tactile stimulation for participants. Stimulation alternated between these two stroking areas within each tactile disruption trial, to minimise habituation, and provide the experimenter with an assigned area to control the pressure of each stroke. For all experimental trials, participants stood to the right of the mannequin body, separated by an office screen divider (see Figure 2a), whilst wearing the HMDs. Participants were instructed to remain still, place their arms by their side, and look down as though towards their own body. A live video image (delay ~60 ms) of the mannequin body, viewed from a first-person perspective, appeared in place of their own body through the HMDs (see Figure 2b).
For visual capture trials, participants visually observed the mannequin body for a 30-second period, without any tactile stimulation. Immediately after the trial, participants removed the HMDs and rated their subjective embodiment towards the mannequin via the embodiment questionnaire (see Table 1) on a separate computer. Removing the HMDs following each trial also served as a rest period for participants to move freely and dissociate their subjective experience between trials. For tactile disruption trials, participants identically visually observed the mannequin body, with the experimenter stroking participants’ own, unseen arm for a 60-second period. Stroking velocity was manipulated by administering slow, affective touch (3 cm/s), and fast, non-affective touch (18 cm/s). The experimenter was trained to administer each stroke at the precise speed within the assigned stroking area (9 cm × 4 cm), by counting the number of strokes within a window of 3 seconds per individual stimulation (i.e. one 3s-long stroke for 3 cm/s velocity, and six 0.5s-long strokes for 18 cm/s velocity). Identically, immediately after tactile disruption trials, participants removed the HMDs and rated their subjective embodiment towards the mannequin via the embodiment questionnaire. Individual questionnaire items were presented in a randomised order across all trials.
Experiment 2
Participants
Forty-three healthy female participants (mean age = 18.98, SD ± 0.74, range = 18–20) were recruited via the University of York research participation scheme and received course credit for a single 60-minute testing session. Participants had a mean BMI of 21.89 (SD ± 2.67, range = 16.66–28.32), no current or previous neurological or psychological disorders (self-report), and normal or corrected-to-normal vision. Exclusion criteria included any specific skin conditions (e.g. eczema, psoriasis) or any scarring or tattoos on the left arm. All participants gave informed consent to take part in the study. The study received ethical approval from the University of York Departmental Ethics Committee and was conducted in accordance with the Declaration of Helsinki. Three participants were later excluded; one following a self-reported previous psychological condition; one excluded with scarring on their arms, and one excluded following poor comprehension with the experimental procedure. Therefore, the final sample consisted of forty participants (mean age = 18.98, SD ± 0.77, range = 18–20). Post-hoc power analyses using G*Power 3.1.9.271 indicated that this number of participants resulted in 99% power to obtain significant effects following both visual capture and tactile disruption conditions (see Section 2.3).
Design, Materials, Measures, Procedure
Design, Materials, Measures and Procedures were identical to Experiment 1, with the addition of a separate Touch Task completed prior to the Full Body Illusion, which explored subjective pleasantness ratings of affective vs. non-affective touch based solely on tactile input. This measure was also investigated in relation to subthreshold eating disorder psychopathology amongst healthy females.
Touch Task
Participants were asked to place their left arm on the table with their palm facing down, and wore a blindfold over their eyes to prevent any visual feedback to tactile stimulation. Tactile stimulation was administered using an identical cosmetic make-up brush (see Materials above) for 3 seconds per trial, at the same velocities as those in the tactile disruption conditions (affective touch − 3 cm/sec and non-affective touch − 18 cm/sec). There was a total of six trials per velocity condition, for a total of 18 trials, with all trials presented in a randomised order for each participant. Following each trial, participants verbally reported the pleasant of the touch, using the pleasantness rating VAS scale, anchored from 0 (Not at all pleasant) to 100 (Extremely pleasant)60. An average score across the six trials was calculated to obtain a single score, per participant, for each of the two tactile conditions.
Data Analysis
All statistical analyses were conducted using SPSS version 23.0 (IBM, Chicago, IL, USA). Data from the embodiment questionnaire were ordinal and found to be non-normal via a Shapiro-Wilk test (p < 0.05), thus, appropriate non-parametric tests were used for analysis. Data for pleasantness ratings in the Touch Task were normally distributed (p > 0.05), therefore parametric tests were used to analyse this data. Effect sizes for parametric tests are indicated by Cohen’s d, and non-parametric Wilcoxon signed-rank tests are indicated by r values (r) which are equivalent to Cohen’s d77.
First, to indicate whether a significant sense of subjective embodiment occurred following mere visual observation of a mannequin body (i.e. visual capture effect), we used a Wilcoxon signed-rank test to compare embodiment scores with control scores within the embodiment questionnaire (see Table 1 for embodiment questionnaire items). Such comparisons were made to ensure that positive subjective embodiment was specific to the visual capture effect and not due to task compliance or suggestibility, with control items not expected to score highly, irrespective of illusory experience. Second, to investigate whether subjective embodiment was significantly reduced when visual capture was disrupted by tactile stimulation to participant’s own, unseen arm (tactile disruption), a further Wilcoxon signed-rank test was conducted to compare subjective embodiment scores between visual capture and tactile disruption conditions. Moreover, we assessed whether slow, affective touch on participants own arm led to greater disruption in subjective embodiment within participants compared with fast, non-affective touch, using a Wilcoxon signed-rank test to compare embodiment scores between the two stroking velocities (affective vs. non-affective tactile disruption). The above analyses were also conducted for individual Ownership and Location subcomponents within the embodiment questionnaire (see Supplementary Materials, Sections 1 and 2). In addition, in Experiment 2 we examined the effect of stroking velocity on pleasantness ratings using a paired-samples t-test, to first establish whether slow, affective touch was indeed perceived as significantly more pleasant that fast, non-affective touch (manipulation check). The perception of touch was then investigated in relation to subthreshold eating disorder psychopathology (as measured by the EDE-Q), using a non-parametric Spearman’s correlation.
To establish individual differences in reported visual capture of embodiment, we calculated percentage frequencies across the combined samples of Experiment 1 and 2, of those who reported visual capture of embodiment (average scores of ≥+1 in response to the embodiment questionnaire3,78), those who neither affirmed or denied embodiment (average scores of <+1 and >−1 in response to the embodiment questionnaire) and those who denied visual capture (average scores of <−1 in the embodiment questionnaire). Finally, we wished to explore whether such individual differences in subjective embodiment from visual capture related to subthreshold eating disorder psychopathology (EDE-Q). Therefore, we conducted a non-parametric Spearman’s correlational analysis between the psychometric EDE-Q measure and subjective embodiment scores from visual capture.
Results
Experiment 1
Embodiment Questionnaire
Preliminary analysis showed that there was no effect of trial order across visual capture trials, with a Wilcoxon signed-rank test revealing no significant difference in embodiment scores between visual capture trial 1 vs. trial 2 (Z = −0.084, p = 0.933). Therefore, embodiment questionnaire scores were collapsed across the two visual capture trials to provide an overall visual capture embodiment score, per participant.
Main effect: Visual Capture: To examine the effects of mere visual capture towards subjective embodiment of the mannequin body, we compared embodiment scores with control scores in the embodiment questionnaire. A Wilcoxon signed-rank test revealed a main effect of visual capture, with significantly higher embodiment scores compared with control scores (Z = −4.04, p < 0.001, r = 0.64) (see Figure 3).
Main effect: Tactile Disruption: In order to determine whether tactile disruption to participants’ own unseen arm would disrupt subjective embodiment, we compared embodiment scores between tactile disruption and visual capture conditions. A Wilcoxon signed-rank test revealed a main effect of condition, in which participants showed significantly lower subjective embodiment following tactile disruption trials (median = −0.38) compared with visual capture trials (median = 0.82) (Z = −3.74, p < 0.001, r = 0.59).
Main effect: Tactile Velocity: Next, we examined whether tactile velocity had an effect in disrupting the subjective embodiment towards the mannequin body within tactile disruption trials. A Wilcoxon signed-rank test revealed that there was no significant difference in embodiment scores between affective and non-affective tactile disruption trials (Z =−0.104, p = 0.918, r = 0.02), which suggests that interoceptive affective touch did not disrupt visual capture of embodiment to a greater degree than exteroceptive, non-affective touch.
Experiment 2
Touch Task (Manipulation Check)
A further one participant was later excluded within the Touch Task analysis as an extreme outlier, scoring more than 2 SD below the group mean in pleasantness ratings of affective touch (3 cm/s velocity)34. Therefore, the final sample for this analysis consisted of 39 participants. As expected, a paired samples t-test revealed an effect of stroking velocity within the Touch Task, with significantly higher subjective pleasantness ratings following affective touch (3 cm/s) (mean = 74.27) compared with non-affective touch (18 cm/s) (mean = 52.94) (t (38) = 7.93, p < 0.001, d = 1.27). Moreover, correlational analyses were conducted to investigate the relationship between pleasantness ratings and subthreshold eating disorder psychopathology (measured by the Eating Disorder Examination Questionnaire; EDE-Q). First, a Spearman’s rank correlation revealed an approaching significant correlation between pleasantness ratings (average affective/non-affective touch) and global EDE-Q score (r = −0.316, p = 0.05). Next, difference scores were calculated between affective and non-affective touch pleasantness ratings to determine whether those with higher subthreshold eating disorder psychopathology were less sensitive to differences in the affectivity of touch. However, a Spearman’s rank correlation revealed no significant correlation between touch difference score and global EDE-Q (r = 0.014, p = 0.935). Thus, the results suggest a trend in which those scoring higher in subthreshold eating disorder psychopathology may show a reduced pleasantness to all tactile stimulation, however this may not be further modulated by the affectivity of the touch that they receive.
Embodiment Questionnaire
Preliminary analysis showed that there was no effect of trial order across visual capture trials, with a Wilcoxon signed-rank test revealing no significant difference in embodiment scores between visual capture trial 1 vs. trial 2 (Z = −0.958, p = 0.338). Therefore, embodiment questionnaire scores were collapsed across the two visual capture trials to provide an overall visual capture embodiment score, per participant.
Main effect: Visual Capture: To examine the effects of mere visual capture towards subjective embodiment of the mannequin body, we compared embodiment scores with control scores in the embodiment questionnaire. A Wilcoxon signed-rank test revealed a main effect of visual capture, with significantly higher embodiment scores compared with control scores (Z = −4.30, p < 0.001, r = 0.68) (see Figure 3).
Main effect: Tactile Disruption: In order to determine whether tactile disruption to participants’ own unseen arm would disrupt subjective embodiment, we compared embodiment scores between tactile disruption and visual capture conditions. A Wilcoxon signed-rank test revealed a main effect of condition, in which participants showed significantly lower subjective embodiment following tactile disruption trials (median = −0.23) compared with visual capture trials (median = 0.59) (Z = −4.08, p < 0.001, r = 0.65).
Main effect: Tactile Velocity: Next, we examined whether tactile velocity had an effect in disrupting the subjective embodiment towards the mannequin body within tactile disruption trials. A Wilcoxon signed-rank test revealed that there was no significant difference in embodiment scores between affective and non-affective tactile disruption trials (Z = −0.354, p = 0.723, r = 0.06), which suggests that interoceptive affective touch did not disrupt embodiment to a greater degree than exteroceptive, non-affective touch.
Combined Samples
Visual Capture of Embodiment – Individual Differences
Across the combined, total sample (N = 80), 32 participants (40%) experienced a degree of embodiment over the mannequin from mere visual capture, with average scores of ≥+1 in response to the embodiment questionnaire (hereafter referred to as ‘visual capture’ (VC) group). To confirm this percentage was not a consequence of participant compliance, a Wilcoxon signed rank test was conducted which revealed a significant difference between embodiment and control scores (Z = −4.71, p < 0.001, r = 0.74), with only 4 participants (12.5%) of the VC group scoring ≥+1 in response to control items. 36 participants (45%) seemed to neither affirm or deny embodiment over the mannequin, with average scores of <+1 and >−1 in response to the embodiment questionnaire (hereafter referred to as ‘borderline’ group). 12 participants (15%) of the total sample denied any subjective embodiment from visual capture, with average scores of < −1 in the embodiment questionnaire (hereafter referred to as ‘no visual capture’ (no-VC) group).
Subthreshold Eating Disorder Psychopathology
Finally, correlational analyses were conducted to investigate the relationship between visual capture effects and subthreshold eating disorder psychopathology (measured by the EDE-Q). EDE-Q subscale and global scores across both experiments are presented in Table 2. A Spearman’s rank correlation revealed no significant correlation between visual capture embodiment scores and global EDE-Q scores (r = 0.030, p = 0.79), or any EDE-Q subscale scores (all ps > 0.05). Similarly, no significant correlations were observed when analysing subcomponent (Ownership and Location) scores within the embodiment questionnaire with EDE-Q scores (see Supplementary Materials, Section 3). This suggests that subthreshold attitudes and behaviours regarding to eating and body image did not relate to the degree of subjective embodiment of a mannequin body due to mere visual capture.
Discussion
The present study investigated the extent to which mere visual observation of a mannequin body, viewed from a first-person perspective, influenced subjective embodiment independently from concomitant visuotactile integration. Across two experiments, our results showed that congruent visuoproprioceptive cues between one’s own physical body and a mannequin body was sufficient to induce subjective embodiment in 40% of our total sample. Furthermore, as expected, embodiment was significantly reduced when ‘visual capture’ of embodiment was disrupted by tactile stimulation to participant’s own, unseen arm, confirming that the visual capture effect on embodiment was not due to confabulatory or social desirability responses. Contrary to our secondary hypothesis regarding interoception, this tactile disruption effect was not modulated by stroking velocity, with comparable changes in embodiment following slow, affective (CT-optimal) and fast, non-affective touch. Finally, subthreshold eating disorder psychopathology was not found to modulate the effects of embodiment in visual capture or the subjective pleasantness of perceived touch.
Our findings support previous research which argues that synchronous visuotactile stimulation is not a necessary condition amongst all individuals in triggering subjective embodiment within bodily illusions. Research has shown that visual capture of proprioception can be sufficient to elicit embodiment towards a fake hand25,36 and whole body16 in some individuals. Indeed, whilst Maselli and Slater (2013) have shown this effect using a full body within an immersive, virtual environment, the present study is the first to explore this effect towards a full body in the ‘physical world’. Our results suggest that multisensory illusion paradigms would benefit from a baseline measure based on the mere visual observation of the fake body (part) (i.e. visual capture effect), which is unbiased by concomitant visuotactile stimulation35,62. Indeed, this is in support of research which argues that asynchronous stimulation in multisensory illusion paradigms is not strictly a neutral, control condition within multisensory body illusions29,33, with visuotactile asynchrony instead providing somatosensory conflict29,79.
The present data showed that a substantial percentage of participants displayed a degree of subjective embodiment towards the mannequin body following mere visual observation. Indeed, it was confirmed that such individuals who did display visual capture of embodiment were not simply complying with all items in the embodiment questionnaire, indicated by significantly higher responses in embodiment scores compared with control scores (see Results section). Nevertheless, congruent visuoproprioceptive signals did not induce subjective embodiment amongst all individuals to the same degree. We speculate that such individual differences may be due to a number of processes; for example, some individuals may have weaker proprioceptive signals which would give rise to greater sensory weighting towards the salient visual cues of the mannequin body within the illusion. However, our own hypothesis that individual differences in visual capture may relate to subthreshold eating disorder psychopathology was not confirmed (see below for further discussion). Typically, bodily illusions which involve a three-way interaction between vision, proprioception and synchronous touch are interpreted as the result of visual capture of touch and proprioception due to the synchronicity of the touch2. However, as it is now increasingly recognised that a two-way integration of vision and proprioception can affect ownership in some individuals following mere observation of a realistic body (part)25,34,80, future studies should test whether such two-way effects also affect the perception of synchrony itself in some individuals. Indeed, it may be that previous research which has reported ownership changes even in asynchronous conditions may be explained by the two-way integration of proprioception and vision influencing the perception of asynchrony. Thus, further research is required to establish how individual differences in the weighting of distinct sensory cues contribute to modulating body ownership in mere visual capture conditions, and how increased visual weighting may thus influence the perception of visuotactile synchrony within typical multisensory illusion paradigms.
Furthermore, our results showed that tactile stimulation to participants own, unseen arm significantly disrupted subjective embodiment towards the mannequin body, by delivering somatosensory information that was incongruent with participants’ visuoproprioceptive cues. This result further highlights that embodiment shown from visual capture conditions was not due to participant compliance, as disruption to such visual capture resulted in significantly lower embodiment scores. From a computational approach to multisensory integration25,30,81, such incongruent tactile information is likely to have disrupted the sensory weighting that is occurring between visual and proprioceptive body signals32. Indeed, predictive coding accounts of multisensory illusions argue that illusory embodiment typically occurs by the brain downregulating the precision of conflicting, bottom-up somatosensory signals, which allows top-down predictions to resolve any sensory ambiguity about the body (i.e. the body (part) I see is mine)30. Therefore, in the present study, additional tactile input to participants’ own, unseen arm added further somatosensory information which could not be downregulated or “explained away” by top-down predictions, given its incongruency with the visually perceived mannequin body82, thus leading to reduced subjective embodiment.
Moreover, it was expected that the interoceptive properties associated with slow, affective touch35 would disrupt subjective embodiment to a greater degree than fast, non-affective touch. This is following evidence that affective touch led to enhanced embodiment during RHI paradigms62,63,64, which is argued to be due to the additional interoceptive information conveyed by this CT-optimal touch83. Further, research has shown that manipulation of interoceptive information (e.g. changes in body temperature) can disrupt feelings of body ownership69. However, contrary to our predictions and previous findings, our results showed that the interoceptive, affective tactile stimuli did not appear to disrupt visual capture of embodiment to a greater extent than non-affective tactile stimuli. Such findings may be because the salience of incongruent visuotactile information was sufficient in disrupting embodiment towards the mannequin, with the subtlety of increased interoceptive information from the arm following affective touch providing no additional value to multisensory integration in this context. Furthermore, the previously observed effects of affective touch in enhancing body ownership during the RHI (which involves concomitant felt and seen touch on the rubber hand) may also be explained by the vicarious affectivity of the seen touch in addition to the interoceptive nature of the felt touch (Filippetti et al., submitted). Indeed, CT-optimal velocities have been shown to have distinct vicarious touch effects in behavioural84 and neuroimaging61 studies. However, visual cues of affective touch were not present in the current study, therefore the felt affectivity of the touch within the illusion set-up may have been attenuated by participants receiving only tactile stimulation that was not visually observed.
The present results must be considered in relation to the top-down, cognitive constraints within which illusory ownership is argued to occur. Research has shown that the embodied fake body (part) must be in an anatomically plausible position3,15,22,23, must represent a corporeal object13,19,20, and must be viewed from a first-person visual perspective16,17,18. Indeed, it has been shown that when these constraints are violated, illusory effects diminish or disappear24,85,86, suggesting that the perceived fake body (part) is required to fit with a reference model of the body based on top-down, contextual information20. The above conditions were closely adhered to in the present study, which was particularly salient using the HMDs, allowing a high degree of spatial overlap by replacing the first-person perspective of one’s own body with the identical perspective of a mannequin body. This provided a greater congruence of visuoproprioceptive cues which cannot be as closely matched within the RHI set-up without the use of computer-generated technology. However, further research should investigate the specific boundaries within which mere visual capture is sufficient in inducing embodiment towards a whole body, in the absence of visuotactile stimulation16,87, by systematically manipulating the above conditions within which the illusion can typically occur. Methodologically, comparison between visual capture (30 secs) and tactile disruption (60 secs) conditions may be potentially limited in its interpretation, given the difference in trial duration between conditions. However, such a design is widely accepted within previous research which has used two-way and three-way sensory integration conditions34,62. Furthermore, the duration between velocities was identical within tactile disruption conditions, which provided direct comparison between affective and non-affective touch.
Finally, following evidence that acute eating disorder patients display a dominance in weighting to visual information related to the body45,46, which is shown to persist after recovery49, we explored whether this trait phenomenon would exist amongst healthy individuals, in relation to subthreshold eating disorder psychopathology. However, no significant correlations were observed between EDE-Q scores and subjective embodiment following visual capture. This finding is in line with previous research in which those higher in subthreshold eating disorder symptoms did not experience a stronger subjective embodiment within the full body illusion53, despite relationships observed between EDE-Q scores and subsequent behavioural measures (e.g. body satisfaction) following the illusion (see also50 for similar effects in AN patients). This suggests that previous findings which highlight differences in subjective embodiment in relation to eating disorder psychopathology may be body-part specific45,46,88. Nevertheless, studying eating disorder characteristics within healthy individuals remains clinically important to identify factors associated with the development of eating disorders without the confounds of physical consequences of the disorder89,90.
Taken together, the present findings are in accordance with previous research which highlights the dynamic mechanisms that lead to illusory body ownership16. First, there exists a two-way interaction between visual information of the fake body (part) and proprioceptive information of one’s own body (part), which is combined to inform an estimate of an individual’s current spatial position. When the fake body (part) is in an anatomically plausible position with one’s own body, sensory information between competing visual and proprioceptive cues is weighted in favour of the salient visual information81,91, which for many is sufficient to induce feelings of embodiment to occur prior to visuotactile integration16,25. Subsequently, the addition of synchronous visuotactile information creates a three-way weighted interaction between vision, touch and proprioception, with the visually perceived touch processed in a common reference frame based on the visuoproprioceptive cues. The subsequent ‘visual capture’ of synchronous visuotactile stimulation acts to further weaken one’s own proprioceptive signals, which can lead to increased illusory embodiment24,86. Thus, future studies which compare the two-way vs. three-way interaction between sensory inputs would be informative in quantifying the additive effect that visuotactile stimulation plays within such paradigms. This could also be used to further investigate individual differences in the susceptibility to integrate visuoproprioceptive information to a greater degree than the additional integration of tactile stimuli during the illusion.
In conclusion, the present study suggests that mere visual observation of a mannequin body, viewed from a first-person perspective, can elicit subjective embodiment amongst individuals. Congruent visuoproprioceptive cues between one’s own physical body (part) and a fake body (part) was shown to be sufficient to induce subjective embodiment in 40% of our total sample in the absence of concomitant visuotactile stimulation, which is typically used to induce illusory embodiment within multisensory illusion paradigms. In addition, tactile stimulation delivered to participants own, unseen arm acted to disrupt reported subjective embodiment, however, this was not influenced to a greater degree by slow, affective touch compared with fast, non-affective touch. This suggests that interoceptive information about one’s body does not have the potency of discriminatory tactile signals, when the integration of vision and proprioception need to be moderated by touch. Future studies should explore this possibility using other interoceptive modalities such as cardiac awareness, and further investigate how the perception and integration of different sensory signals might be implicated within a distorted sense of self amongst clinical eating disorder populations.
Data Availability
The datasets analysed during the current study are available from the corresponding author on reasonable request.
References
Gallagher, S. Philosophical conceptions of the self: Implications for cognitive science. Trends Cogn. Sci. 4, 14–21 (2000).
Tsakiris, M. The multisensory basis of the self: from body to identity to others. Q. J. Exp. Psychol. 0218, 1–28 (2016).
Ehrsson, H. H., Spence, C. & Passingham, R. That’s My Hand! Acriviy in Premotor Cortex Reflects Feelings of Ownership of a Limb. Science (80-.). 305, 875–877 (2004).
Tsakiris, M. My body in the brain: A neurocognitive model of body-ownership. Neuropsychologia 48, 703–712 (2010).
Serino, A. et al. Body part-centered and full body-centered peripersonal space representations. Sci. Rep. 5, 1–14 (2015).
Salomon, R. et al. Unconscious integration of multisensory bodily inputs in the peripersonal space shapes bodily self-consciousness. Cognition 166, 174–183 (2017).
Craig, A. D. How do you feel? Interoception: the sense of the physiological condition of the body. Nat. Rev. Neurosci. 3, 655 (2002).
Craig, A. D. How do you feel — now? The anterior insula and human awareness. Nat. Rev. Neurosci. 10, 59–70 (2009).
Tsakiris, M., Tajadura-Jiménez, A. & Costantini, M. Just a heartbeat away from one’s body: interoceptive sensitivity predicts malleability of body-representations. Proc. Biol. Sci. 278, 2470–2476 (2011).
Botvinick, M. & Cohen, J. Rubber hand feels touch that eyes see. Nature 391, 756 (1998).
Ehrsson, H. H. The experimental induction of out-of-body experiences. Science (80-.). 317, 1048 (2007).
Lenggenhager, B., Tadi, T., Metzinger, T. & Blanke, O. Video ergo sum: manipulating bodily self-consciousness. Science 317, 1096–9 (2007).
Petkova, V. I. & Ehrsson, H. H. If I were you: perceptual illusion of body swapping. PLoS One 3, e3832 (2008).
Slater, M., Spanlang, B., Sanchez-Vives, M. V. & Blanke, O. First person experience of body transfer in virtual reality. PLoS One 5, 1–9 (2010).
Tsakiris, M. & Haggard, P. The rubber hand illusion revisited: visuotactile integration and self-attribution. J Exp Psychol Hum Percept Perform 31, 80–91 (2005).
Maselli, A. & Slater, M. The building blocks of the full body ownership illusion. Front. Hum. Neurosci. 7, 83 (2013).
Petkova, V. I., Khoshnevis, M. & Ehrsson, H. H. The Perspective Matters! Multisensory Integration in Ego-Centric Reference Frames Determines Full-Body Ownership. Front. Psychol. 2, 1–7 (2011).
Preston, C., Kuper-Smith, B. J. & Ehrsson, H. H. Owning the body in the mirror: The effect of visual perspective and mirror view on the full-body illusion. Sci. Rep. 5, 18345 (2015).
Haans, A., IJsselsteijn, W. A. & de Kort, Y. A. W. The effect of similarities in skin texture and hand shape on perceived ownership of a fake limb. Body Image 5, 389–394 (2008).
Tsakiris, M., Carpenter, L., James, D. & Fotopoulou, A. Hands only illusion: Multisensory integration elicits sense of ownership for body parts but not for non-corporeal objects. Exp. Brain Res. 204, 343–352 (2010).
Bergström, I., Kilteni, K. & Slater, M. First-Person Perspective Virtual Body Posture Influences Stress: A Virtual Reality Body Ownership Study. PLoS One 11, 1–21 (2016).
Lloyd, D. M. Spatial limits on referred touch to an alien limb may reflect boundaries of visuo-tactile peripersonal space surrounding the hand. Brain Cogn. 64, 104–109 (2007).
Preston, C. The role of distance from the body and distance from the real hand in ownership and disownership during the rubber hand illusion. Acta Psychol. (Amst). 142, 177–183 (2013).
Kilteni, K., Maselli, A., Kording, K. P. & Slater, M. Over my fake body: body ownership illusions for studying the multisensory basis of own-body perception. Front. Hum. Neurosci. 9, 141 (2015).
Samad, M., Chung, A. J. & Shams, L. Perception of body ownership is driven by Bayesian sensory inference. PLoS One 10, 1–23 (2015).
Longo, M. R., Cardozo, S. & Haggard, P. Visual enhancement of touch and the bodily self. Conscious. Cogn. 17, 1181–1191 (2008).
Pavani, F., Spence, C. & Driver, J. Visual capture of touch: Out-of-the-body experiences with rubber gloves. Psychol. Sci. 11, 353–359 (2000).
Costantini, M. et al. Temporal limits on rubber hand illusion reflect individuals’ temporal resolution in multisensory perception. Cognition 157, 39–48 (2016).
Rohde, M., Luca, M. & Ernst, M. O. The rubber hand illusion: Feeling of ownership and proprioceptive drift Do not go hand in hand. PLoS One 6 (2011).
Zeller, D., Friston, K. J. & Classen, J. Dynamic causal modeling of touch-evoked potentials in the rubber hand illusion. Neuroimage 138, 266–273 (2016).
Giummarra, M. J., Georgiou-Karistianis, N., Nicholls, M. E. R., Gibson, S. J. & Bradshaw, J. L. The phantom in the mirror: A modified rubber-hand illusion in amputees and normals. Perception 39, 103–118 (2010).
Noel, J. P., Blanke, O. & Serino, A. From multisensory integration in peripersonal space to bodily self-consciousness: From statistical regularities to statistical inference. Ann. N. Y. Acad. Sci. 1426, 146–165 (2018).
Perez-Marcos, D. et al. Selective distortion of body image by asynchronous visuotactile stimulation. Body Image 24, 55–61 (2018).
Ponzo, S., Kirsch, L. P., Fotopoulou, A. & Jenkinson, P. M. Balancing body ownership: Visual capture of proprioception and affectivity during vestibular stimulation. Neuropsychologia 117, 311–321 (2018).
Crucianelli, L., Krahé, C., Jenkinson, P. M. & Fotopoulou, A. (Katerina). Interoceptive ingredients of body ownership: Affective touch and cardiac awareness in the rubber hand illusion. Cortex 1–13, https://doi.org/10.1016/j.cortex.2017.04.018 (2017).
Martinaud, O., Besharati, S., Jenkinson, P. M. & Fotopoulou, A. Ownership Illusions in Patients with Body Delusions: Different Neural Profiles of Visual Capture and Disownership. Cortex 174–185, https://doi.org/10.1016/j.cortex.2016.09.025 (2017).
Fotopoulou, A. et al. The role of motor intention in motor awareness: An experimental study on anosognosia for hemiplegia. Brain 131, 3432–3442 (2008).
Tidoni, E., Grisoni, L., Liuzza, M. T. & Aglioti, S. M. Rubber hand illusion highlights massive visual capture and sensorimotor face-hand remapping in a tetraplegic man. Restor. Neurol. Neurosci. 32, 611–622 (2014).
van Stralen, H. E., van Zandvoort, M. J. E., Kappelle, L. J. & Dijkerman, H. C. The Rubber Hand Illusion in a patient with hand disownership. Perception 42, 991–993 (2013).
Greenfield, K., Ropar, D., Smith, A. D., Carey, M. & Newport, R. Visuo-tactile integration in autism: Atypical temporal binding may underlie greater reliance on proprioceptive information. Mol. Autism 6 (2015).
Noel, J. P., Lytle, M., Cascio, C. & Wallace, M. T. Disrupted integration of exteroceptive and interoceptive signaling in autism spectrum disorder. Autism Res. 11, 194–205 (2018).
Wynn, J. K., Jahshan, C. & Green, M. F. Multisensory integration in schizophrenia: A behavioural and event-related potential study. Cogn. Neuropsychiatry 19, 319–336 (2014).
Tschacher, W. & Bergomi, C. Cognitive binding in schizophrenia: Weakened integration of temporal intersensory information. Schizophr. Bull. 37 (2011).
Stevenson, R. A. et al. The associations between multisensory temporal processing and symptoms of schizophrenia. Schizophr. Res. 179, 97–103 (2017).
Eshkevari, E., Rieger, E., Longo, M. R., Haggard, P. & Treasure, J. Increased plasticity of the bodily self in eating disorders. Psychol. Med. 42, 819–28 (2012).
Keizer, A., Smeets, M. A. M., Postma, A., van Elburg, A. & Dijkerman, H. C. Does the experience of ownership over a rubber hand change body size perception in anorexia nervosa patients? Neuropsychologia 62, 26–37 (2014).
Serino, S. & Dakanalis, A. Bodily illusions and weight-related disorders: Clinical insights from experimental research. Ann. Phys. Rehabil. Med. 60, 217–219 (2017).
Noel, J. P., Stevenson, R. A. & Wallace, M. T. Atypical audiovisual temporal function in autism and schizophrenia: similar phenotype, different cause. Eur. J. Neurosci. 47, 1230–1241 (2018).
Eshkevari, E., Rieger, E., Longo, M. R., Haggard, P. & Treasure, J. Persistent body image disturbance following recovery from eating disorders. Int. J. Eat. Disord. 47, 400–409 (2014).
Keizer, A., van Elburg, A., Helms, R. & Dijkerman, H. C. A Virtual Reality Full Body Illusion Improves Body Image Disturbance in Anorexia Nervosa. PLoS One 11, e0163921 (2016).
Preston, C. & Ehrsson, H. H. Illusory Changes in Body Size Modulate Body Satisfaction in a Way That Is Related to Non-Clinical Eating Disorder Psychopathology. PLoS One 9, e85773 (2014).
Preston, C. & Ehrsson, H. H. Illusory obesity triggers body dissatisfaction responses in the insula and anterior cingulate cortex. Cereb. Cortex 1–11, https://doi.org/10.1093/cercor/bhw313 (2016).
Preston, C. & Ehrsson, H. H. Implicit and explicit changes in body satisfaction evoked by body size illusions: Implications for eating disorder vulnerability in women. PLoS One 13, 1–31 (2018).
Aspell, J. E. et al. Turning Body and Self Inside Out: Visualized Heartbeats Alter Bodily Self-Consciousness and Tactile Perception. Psychol. Sci. 24, 2445–2453 (2013).
Suzuki, K., Garfinkel, S. N., Critchley, H. D. & Seth, A. K. Multisensory integration across exteroceptive and interoceptive domains modulates self-experience in the rubber-hand illusion. Neuropsychologia 51, 2909–2917 (2013).
Löken, L. S., Wessberg, J., Morrison, I., McGlone, F. & Olausson, H. Coding of pleasant touch by unmyelinated afferents in humans. Nat. Neurosci. 12, 547–548 (2009).
Olausson, H., Wessberg, J., Morrison, I., McGlone, F. & Vallbo, Å. The neurophysiology of unmyelinated tactile afferents. Neurosci. Biobehav. Rev. 34, 185–191 (2010).
Olausson, H. et al. Unmyelinated tactile afferents signal touch and project to insular cortex. Nat. Neurosci. 5, 900–904 (2002).
Björnsdotter, M., Morrison, I. & Olausson, H. Feeling good: On the role of C fiber mediated touch in interoception. Exp. Brain Res. 207, 149–155 (2010).
Crucianelli, L., Cardi, V., Treasure, J., Jenkinson, P. M. & Fotopoulou, A. The perception of affective touch in anorexia nervosa. Psychiatry Res. 239, 72–78 (2016).
Morrison, I., Björnsdotter, M. & Olausson, H. Vicarious Responses to Social Touch in Posterior Insular Cortex Are Tuned to Pleasant Caressing Speeds. J. Neurosci. 31, 9554–9562 (2011).
Crucianelli, L., Metcalf, N. K., Fotopoulou, A. & Jenkinson, P. M. Bodily pleasure matters: Velocity of touch modulates body ownership during the rubber hand illusion. Front. Psychol. 4, 1–7 (2013).
Lloyd, D. M., Gillis, V., Lewis, E., Farrell, M. J. & Morrison, I. Pleasant touch moderates the subjective but not objective aspects of body perception. Front. Behav. Neurosci. 7, 207 (2013).
van Stralen, H. E. et al. Affective touch modulates the rubber hand illusion. Cognition 131, 147–158 (2014).
de Jong, J. R., Keizer, A., Engel, M. M. & Dijkerman, H. C. Does affective touch influence the virtual reality full body illusion? Exp. Brain Res. 0, 0 (2017).
Moseley, G. L. Distorted body image in complex regional pain syndrome. Neurology 65, 773 (2005).
Moseley, G. L. I can’t find it! Distorted body image and tactile dysfunction in patients with chronic back pain. Pain 140, 239–243 (2008).
Moseley, G. L., Parsons, T. J. & Spence, C. Visual distortion of a limb modulates the pain and swelling evoked by movement. Curr. Biol. 18, 1047–1048 (2008).
Kammers, M. P. M., Rose, K. & Haggard, P. Feeling numb: Temperature, but not thermal pain, modulates feeling of body ownership. Neuropsychologia 49, 1316–1321 (2011).
Ainley, V., Apps, M. A. J., Fotopoulou, A. & Tsakiris, M. ‘Bodily precision’: A predictive coding account of individual differences in interoceptive accuracy. Philos. Trans. R. Soc. B Biol. Sci. 371 (2016).
Faul, F., Erdfelder, E., Lang, A. G. & Buchner, A. G* Power 3: A flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav. Res. Methods 39, 175 (2007).
Longo, M. R., Schüür, F., Kammers, M. P. M., Tsakiris, M. & Haggard, P. What is embodiment? A psychometric approach. Cognition 107(3), 978–998, https://doi.org/10.1016/j.cognition.2007.12.004 (2008).
Fairburn, C. G. & Beglin, S. Assessment of eating disorders: interview or self- report questionnaire? Int J Eat Disord 16, 363–370 (1994).
Berg, K. C., Peterson, C. B., Frazier, P. & Crow, S. J. Psychometric evaluation of the eating disorder examination and eating disorder examination-questionnaire: A systematic review of the literature. Int. J. Eat. Disord. 45, 428–438 (2012).
Peirce, J. W. PsychoPy—Psychophysics software in Python. J. Neurosci. Methods 162, 8–13 (2007).
McGlone, F. et al. Touching and feeling: Differences in pleasant touch processing between glabrous and hairy skin in humans. Eur. J. Neurosci. 35, 1782–1788 (2012).
Pallant, J. SPSS Survival Manual: A Step by Step Guide to Data Analysis Using SPSS for Windows Version 15. (2007).
Kalckert, A. & Ehrsson, H. H. Moving a Rubber Hand that Feels Like Your Own: A Dissociation of Ownership and Agency. Front. Hum. Neurosci. 6, 1–14 (2012).
Caola, B., Montalti, M., Zanini, A., Leadbetter, A. & Martini, M. The Bodily Illusion in Adverse Conditions: Virtual Arm Ownership During Visuomotor Mismatch. Perception https://doi.org/10.1177/0301006618758211 (2018).
Panagiotopoulou, E., Filippetti, M. L., Tsakiris, M. & Fotopoulou, A. Affective Touch Enhances Self-Face Recognition During Multisensory Integration. Sci. Rep. 7, 12883 (2017).
Zeller, D., Litvak, V., Friston, K. J. & Classen, J. Sensory Processing and the Rubber Hand Illusion—An Evoked Potentials Study. J. Cogn. Neurosci. 27, 573–582 (2015).
Limanowski, J. & Blankenburg, F. Network activity underlying the illusory self-attribution of a dummy arm. Hum. Brain Mapp. 36, 2284–2304 (2015).
Gentsch, A., Crucianelli, L., Jenkinson, P. & Fotopoulou, A. The Touched Self: Affective Touch and Body Awareness in Health and Disease. In Affective Touch and the Neurophysiology of CT Afferents 355–384, https://doi.org/10.1007/978-1-4939-6418-5 (2016).
Gentsch, A., Panagiotopoulou, E. & Fotopoulou, A. Active Interpersonal Touch Gives Rise to the Social Softness Illusion. Curr. Biol. 25, 2392–2397 (2015).
Apps, M. A. J. & Tsakiris, M. The free-energy self: A predictive coding account of self-recognition. Neurosci. Biobehav. Rev. 41, 85–97 (2014).
Makin, T. R., Holmes, N. P. & Ehrsson, H. H. On the other hand: Dummy hands and peripersonal space. Behav. Brain Res. 191, 1–10 (2008).
Petkova, V. I. et al. From part- to whole-body ownership in the multisensory brain. Curr. Biol. 21, 1118–1122 (2011).
Mussap, A. J. & Salton, N. A ‘Rubber-hand’ Illusion Reveals a Relationship between Perceptual Body Image and Unhealthy Body Change. J. Health Psychol. 11, 627–639 (2006).
Frank, G. K. W. Altered Brain Reward Circuits in Eating Disorders: Chicken or Egg? Curr. Psychiatry Rep. 15, 396 (2013).
Hay, P. J. & Sachdev, P. Brain dysfunction in anorexia nervosa: cause or consequence of under-nutrition? Curr. Opin. Psychiatry 24, 251–256 (2011).
Kilteni, K. & Ehrsson, H. H. Body ownership determines the attenuation of self-generated tactile sensations. Proc. Natl. Acad. Sci. 201703347, https://doi.org/10.1073/PNAS.1703347114 (2017).
Acknowledgements
We are grateful to Chloe Robinson for her help and feedback in piloting the present study. The study was supported by a European Research Council Starting Investigator Award [ERC-2012-STG GA313755] (to AF), and a University of York studentship (to MC). Funding for the time of AF and LC has been partially provided by the Fund for Psychoanalytic Research through the American Psychoanalytic Association.
Author information
Authors and Affiliations
Contributions
M.C., L.C., C.P., and A.F. designed the experiment. M.C. performed data collection and data analysis, under the supervision of C.P., L.C., and A.F. M.C. drafted the manuscript, and C.P., L.C., and A.F. provided critical revisions. All authors approved the manuscript before submission.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Carey, M., Crucianelli, L., Preston, C. et al. The Effect of Visual Capture Towards Subjective Embodiment Within the Full Body Illusion. Sci Rep 9, 2889 (2019). https://doi.org/10.1038/s41598-019-39168-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-39168-4
This article is cited by
-
Using narrative 360° video as a tool to promote breast self-examination
Virtual Reality (2024)
-
Mirror, mirror, on the wall: During pandemics, how can self-perception research in people with eating disorders happen at all?
Journal of Eating Disorders (2022)
-
Body weight distortions in an auditory-driven body illusion in subclinical and clinical eating disorders
Scientific Reports (2022)
-
Vicarious ratings of self vs. other-directed social touch in women with and recovered from Anorexia Nervosa
Scientific Reports (2022)
-
Effects of virtual hands and feet on the onset time and duration of illusory body ownership
Scientific Reports (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.